Abstract
Nardostachys jatamansi has long been used to prepare Medhya Rasayana in traditional Indian Ayurveda medicine to treat neurological disorders and enhance memory. Jatamansinol from the N. jatamansi against Alzheimer’s disease (AD) showed that it could be a multitargeted drug against AD. Drosophila is an ideal model organism for studying a progressive age-related neurodegenerative disease such as AD since its neuronal organizations and functioning are highly similar to that of humans. The current study investigates the neuroprotective properties of jatamansinol against Tau-induced neurotoxicity in the AD Drosophila model. Results indicate jatamansinol is not an antifeedant for larva and adult Drosophila. Lifespan, locomotor activity, learning and memory, Tau protein expression level, eye degeneration, oxidative stress level, and cholinesterase activities were analyzed in 10, 20, and 30-day-old control (wild type), and tauopathy flies reared on jatamansinol supplemented food or regular food without jatamansinol supplementation. Jatamansinol treatment significantly extends the lifespan, improves locomotor activity, enhances learning and memory, and reduces Tau protein levels in tauopathy flies. It boosts the antioxidant enzyme activities, prevents Tau-induced oxidative stress, ameliorates eye degeneration, and inhibits cholinesterase activities in Tau-induced AD model. This study provides the first evidence that jatamansinol protects against Tau’s neurotoxic effect in the AD Drosophila model, and it can be a potential therapeutic drug candidate for AD.
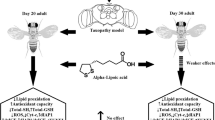
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD) is a significant age-related human neurodegenerative disease that causes the most common form of dementia [1]. In 2021, about 58 million people with AD may increase to 88 million in 2050 [2]. Major pathophysiological features of AD are the amyloid-β (Aβ) plaque accumulation in the extracellular region of neurons and deposition of neurofibrillary tangles in the intracellular area of neurons [3]. Impaired cellular antioxidant systems, bioenergetics, and neurotransmitters also contribute to the onset of AD [4]. Oxidative stress in the pathogenesis of AD is associated with Aβ, and Tau protein linked neural damage. It has been evident that cognitive impairment in AD patients is associated with increased oxidative stress in the neuronal cells [5]. Human AD patients’ brains have increased oxidative stress by forming senile plaques and neurofibrillary tangles [6]. It has been evidenced that the pan-neuronal overexpression of human tau protein in Drosophila leads to increased oxidative stress and reduced antioxidant enzymes [7]. Hence, oxidative stress is considered one of the significant cellular factors in AD pathogenesis.
No therapeutic drugs prevent AD’s onset, cure or reverse AD progression, treat AD-related cognitive symptoms, and prevent further cognitive decline. The available FDA-approved rivastigmine, galantamine, and donepezil drugs for AD treatments relieve symptoms by inhibiting cholinesterase enzymes. Several lines of research are under investigation to find the best therapeutic medicines to treat AD. One such approach is developing an antioxidant-based therapeutic agent against oxidative stress-mediated AD [8]. Since AD is a multifactorial disease [9], it is essential to develop multitargeted therapeutic drugs for its treatment. Medicinal plants and their metabolites used in traditional medicine systems are the most prospective candidates for developing multitargeted therapeutics against AD.
Ayurveda is an ancient Indian medicine system in which medicinal plants and plant compositions treat many neurological disorders and enhance memory [10]. Nardostachys jatamansi is one of the important memory-enhancing plants (Medhya) mentioned in the compendium of Ayurveda. It is a perennial herb with a hairy rhizome and grows in the Himalayas at an altitude of 3000–5000 m. Studies have demonstrated that root extract of N. jatamansi improves learning and memory in mice [11], anticonvulsant and antioxidant potential in rats [12, 13], and it can decrease the Aβ42-induced toxicity in Drosophila [14]. Our in silico studies showed a pyranocoumarin, jatamansinol, from N. jatamansi can be a multitargeted therapeutic agent for multifactorial AD [15]. Accordingly, supplementation of isolated jatamansinol from the N. jatamansi root to the Aβ42 protein-induced AD Drosophila model evidenced the neuroprotective efficacy of jatamansinol [16]. However, there is no study on the antitauopathic activity of jatamansinol in an in vivo model organism.
Drosophila melanogaster is popular in vivo model organism in biomedical research, including neurotoxicity studies [17, 18]. Its ease of maintenance, short generation time (9–12 days), the maximum lifespan of 55–80 days, genetic tools, fully sequenced genome, and sequence similarity with a human make it more acceptable for this study model. Approximately 75% of human disease-related genes have homologs in fly, suggesting molecular mechanisms of human disease conserved in the Drosophila. The Drosophila brain has about 300,000 neurons organized into a separate area with specialized functions such as memory, learning, olfaction, and vision, similar to humans, making Drosophila an ideal model organism for studying a progressive age-related neurodegenerative disease such as AD. Tissue-specific expression of microtubule-associated Tau/Aβ42 protein is possible by the UAS-Gal4 system of Drosophila [19]. In our previous study, the Aβ42 protein-induced AD Drosophila model evidenced the expression of AD characters [16]. Flies overexpressed with Tau protein in neuronal cells have specific characteristics of reduced lifespan, behavioral alteration, increased oxidative stress, and locomotor defects [7]. The Tau protein overexpressed in eye cells induces progressive eye cell degeneration [20]. Hence, the well-characterized AD Drosophila model was employed to screen novel drug candidates for developing the best therapeutic agents for treating various human diseases. This study evaluates the multitargeted therapeutic effect of jatamansinol isolated from the plant N. jatamansi, on the Tau-induced AD Drosophila model.
Materials and Methods
Drosophila Husbandry
Oregon K (OK) of Drosophila melanogaster and GMR-Gal4 driver strains were obtained from the Drosophila Stock Center, University of Mysore, Karnataka, India. Transgenic UAS-Tau LacZ and Elav-Gal4 driver strains were a generous gift from Dr. S. Srikrishna, Department of Biochemistry, Banaras Hindu University, Varanasi, India. All the strains were maintained at 22 ± 2 °C and 60–70% relative humidity in the Drosophila Laboratory, Department of Zoology, Central University of Kerala. OK strain was used as a control in all the assays. UAS-Tau LacZ strain was used to express human microtubule-associated Tau protein. GMR-Gal4 and Elav-Gal4 driver strains were used for expressing Tau protein in the eye and neuronal cells to generate transgenic AD model flies. The generated AD model flies (Elav-Gal4, UAS-Tau lacZ or GMR-Gal4, UAS-Tau lacZ) were used for all the in vivo experiments. The Tau protein-induced AD model generation scheme is provided in the supplementary (Fig. SF1). The progenies (Elav-Gal4, UAS-Tau lacZ) of UAS-Tau lacZ crossed with Elav-Gal4 flies were used to analyze Tau protein level, memory and climbing performance, oxidative stress markers, level of antioxidant enzymes, and choline esterase activities in 10, 20, and 30-day-old adult flies with appropriate replicates. Tau-induced eye degeneration was analyzed in 20-day-old GMR-Gal4, UAS-Tau lacZ flies.
Preparation of Jatamansinol Supplemented Drosophila Food Media
Jatamansinol isolated from the root of N. jatamansi [16] was used in this study. In brief, 100 g of root powder was soaked in the diethyl ether and extracted the coumarins using a Soxhlet extraction apparatus. After extraction, the solvent was removed using a rotatory evaporator. The yield of the extract was 2.5 g (0.25%). The yield of the crystallized jatamansinol from the 1 g of ether extract was 0.12 g. The oral diet supplementation method was employed by mixing jatamansinol with wheat cream agar media of Drosophila food [10]. Desired concentration (weight/volume) of jatamansinol dissolved in 0.1% acetone was added to the freshly prepared 5 mL media in the experimental vials, thoroughly mixed, and allowed to solidify before use. The preliminary toxicity experiment showed that 0.1% acetone in the food media (solvent control) had no significant toxic effect on the Drosophila than in control (without the 0.1% acetone) food media. Hence, the solvent (0.1% acetone) in the food media was considered the control in all experiments. Flies were reared in the jatamansinol supplemented food media or without jatamansinol supplemented food media as required.
Toxicity Analysis and Selection of Jatamansinol Concentrations for the Assays
LC50, or the concentration at which half of the eggs failed to hatch to adults, of jatamansinol was determined to select the effective concentrations for the experiments. It was conducted by following the method established in our laboratory [18]. First, different concentrations of jatamansinol (0–120 μM) were mixed with food media and allowed to hatch eggs and grow to adults. From the first to the last day of emergence, the number of flies that emerged was noted. Then, the viability percentage in treated and control groups was calculated and determined LC50 by the Probit method [21]. The calculated LC50 was 55.25 μM for OK, 54.59 μM for Elav-Gal4, UAS-Tau lacZ, and 54.83 μM for GMR-Gal4, UAS-Tau lacZ. So, sublethal concentrations of 10 μM and 20 μM of jatamansinol were selected to study its neuroprotective effect. Depending on the experimental type, flies were reared in a control food, and 10 μM or 20 μM jatamansinol was supplemented.
Tau Protein Quantification by Enzyme-Linked Immunosorbent Assay
Tau protein levels in the flies of jatamansinol supplemented and control groups were quantified following the enzyme-linked immunosorbent assay (ELISA) kit manufacturer’s instructions (Elabscience, UK). The samples were prepared by homogenizing the heads of 30 flies of respective age and experimental groups in 200 μL of ice-cold PBS (0.01 M; pH 7.4) and centrifuged at 3000 g for 10 min at 4 °C, and the supernatant was filtered. Standard or samples were added to the respective micro-ELISA plate precoated with an antibody hMAPτ (human microtubule-associated protein Tau). A biotinylated detection antibody specific for hMAPτ and avidin horseradish peroxidase (HRP) conjugate was added to each well and incubated for 1 h at 37 °C. The substrate solution was added to each well after washing the free components. The blue color in the wells contains hMAPτ, biotinylated detection antibody, and avidin HRP conjugate. The enzyme–substrate reaction was terminated by adding an H2SO4 solution. A spectrophotometer measured the reaction mixtures’ optical density (OD) at 450 nm. The concentration of hMAPτ (pg/mL) in the experimental group samples was obtained by comparing the standard curve.
Tau Protein Expression by Western Blot Analysis
The western blot analysis confirmed the Tau protein expression in Drosophila reared in control and jatamansinol supplemented food. For this, 20 mg of Drosophila heads was lysed in an extraction buffer (sodium phosphate buffer, pH 7.4) containing a protease inhibitor cocktail (Bio-Rad). The protein concentrations were determined using the Bradford assay. Tau protein in the samples was separated on 12% SDS polyacrylamide gels and transferred onto PVDF (polyvinylidene difluoride) membranes. The membranes were blocked with non-fat milk and incubated with primary antibody against Tau protein (1:2000; Elabscience, UK) at 4 °C overnight. The reaction was stopped by adding wash buffer and incubated for 2 h at room temperature with anti-HRP-conjugated secondary antibody (1:1000; Elabscience). After washing, protein bands were detected with a chemiluminescent HRP substrate (Bio-Rad, USA). The membrane image was taken using Versa Doc (Bio-Rad, USA). Band intensity was analyzed to measure protein concentration using the gel analyzer option from ImageJ 1.52a software.
Tau Gene Expression
The Tau gene expression in the experimental groups was analyzed using quantitative real-time polymerase chain reaction (qRT-PCR). Total RNA from the flies was extracted using TRIzol reagent (Invitrogen, USA), and cDNA was synthesized using RT2 first-strand synthesis kit (Qiagen, USA). qRT-PCR was carried out by the protocol established in our laboratory [22]. The ribosomal protein 49 (RP49) gene was used for normalization. Tau gene primers were manually designed using Integrated DNA Technologies (IDT). These primer sequences were used for Tau (forward) 5′-GCTCTGAAACCTCTGATG-3′ and (reverse) 5′-CCTGGCTTATGATGGATG-3′ and RP49 (forward) 5′-GCTAAGCTGTCGCACAATG-3′ and (reverse) 5′-TGTGCACCAGGAACTTCTTG-3′. The SYBR Green qPCR master mix (Applied Biosystems, USA) was used to quantify the relative gene expression.
Lifespan Assay
Lifespan assay was carried out with freshly emerged flies from the regular and jatamansinol supplemented food as per the protocol given in our previous publication [16].
Behavioral Assays
All the behavioral assays were performed with a double-blinded approach. A climbing assay or negative geotaxis assay was conducted to assess the effect of jatamansinol on the motor activity of OK or Elav-Gal4, UAS-Tau LacZ flies using our established method [16]. The climbing behavior assay video is provided as supplementary data (SV1). The classical olfactory conditioning assay was conducted to measure short-term memory and learning in Drosophila following the established protocol [16]. The supplementary figure (SF2) illustrates the protocol.
Eye Degeneration Analysis
A phenotypic marker study was conducted to analyze the therapeutic effect of jatamansinol on Tau protein-induced eye degeneration in Drosophila. GMR-Gal4, UAS-Tau lacZ progenies obtained from the UAS-Tau lacZ transgenic Drosophila crossed with GMR Gal4 driver strains were used. Twenty-day-old adult flies from each experimental group were observed under a stereomicroscope (Carl Zeiss, Germany) and photographed. Eye degeneration analysis was made using an open-source package of ImageJ plugins, Flynotyper (http://flynotyper.sourceforge.net/). This method uses eye images to spot the individual ommatidium and calculates the phenotypic score to measure its degeneration. Normal ommatidia with the symmetrical arrangement and degenerated ommatidia with asymmetrical arrangements were quantified using phenotypic (P) scores [23]. The eyes of experimental groups (10 replicates) were studied, and P scores were compared. The scanning electron microscope (SEM) analysis was performed to validate the therapeutic effect of jatamansinol on Tau protein-induced eye degeneration in Drosophila. The eye samples from respective experimental groups were collected and fixed in 70% alcohol. The samples were further dehydrated in a series of alcohol grades (70%, 80%, 90%, 95%, and 100% alcohol) for 20 min each, followed by washing in acetone twice. The samples were dried and mounted on carbon tape on an aluminum stub. The samples were gold-coated by sputter deposition for 5 min. Samples were imaged in the Zeiss field emission scanning electron microscope (FESEM) facility at DST-PURSE, Mangalore University, India.
Oxidative Stress Markers
Dihydrofluorescein diacetate (DCF-DA) was used to measure the ROS generation in fly heads following the Driver et al. [24] method. The thiobarbituric acid reactive substance (TBARS) was used to measure LPO in the fly head homogenate by following the Gajare et al. [25] method. H2O2 in the Drosophila head samples was quantified based on the ferrous oxidation in the presence of the xylenol orange (FOX) method [26]. Dintrophenylhydrazine (DNPH) was used to quantify the PC in the Drosophila head by following the Levine et al. [27] method. The NO level in the Drosophila head was determined using the Griess reagent [28]. The detailed assay methods used to analyze oxidative stress markers are provided in our previous publication [16].
Antioxidant Analysis
SOD activity was measured using the inhibition of nitroblue tetrazolium (NBT) auto-oxidation method [29]. CAT activity was determined using the method of Aebi [30]. GST activity was measured using 1-chloro-2,4-dinitrobenzene (CDNB) [31]. The detailed assay methods are provided in our previous publication [16].
Acetylcholine Esterase and Butrylcholine Esterase
Neurotransmitter enzymes acetylcholine esterase (AChE) and butrylcholine esterase (BuChE) were analyzed following the biochemical method of Ellman et al. [32]. The methods used to analyze neurotransmitters are provided in our previous publication [16].
Quantification of Jatamansinol Consumption by Larvae and Adult Fly
Intake of supplemented jatamansinol through food by the individual larva or adult fly was quantified using the methods given in our previous publication [16].
Gustatory Assay
The feeding assay was performed following the Anet et al. [33] method with minor modifications to confirm the intake of jatamansinol-containing food. The details of the experimental protocol are given in our publication [16].
Data Analysis
All the experiments were conducted in replicates. The obtained data were tested for normality using the homogeneity variance test. The difference between survival curves was analyzed using the Kaplan–Meier log-rank test followed by the Tukey HSD post hoc test. The mean lifespan, CI, PI, and eye degeneration P scores among experimental groups were shown as box and whisker plots. All error bars in the whisker plots represent a 95% CI. The 95% CI was resampled 5000 times, bias-corrected, and accelerated using bootstrap methods for behavioral assays. All the biochemical assays were conducted in triplicate, and independent experiments were repeated thrice. The qRT-PCR experiment was conducted in triplicate. A significant difference between the experimental groups in all the assays was analyzed by one-way ANOVA (analysis of variance) followed by the Tukey HSD post hoc test. Two levels of probability, p < 0.05 and p < 0.01, were adopted for the comparisons. The statistical analyses were performed using SPSS (version 16.0) (SPSS Inc., Chicago, IL, USA).
Results
Jatamansinol Reduces the Tau Protein Level
Tau protein expression was significantly increased in 20-day-old Elav-Gal4, UAS-Tau LacZ flies than control flies when both were reared on regular food (Fig. 1A, B; p < 0.01). On the other hand, Tau protein expression was reduced in 20-day-old Elav-Gal4, UAS-Tau LacZ flies reared on 10 μM or 20 μM jatamansinol supplemented food compared with the Elav-Gal4, UAS-Tau LacZ flies reared on regular food (Fig. 1A, B; p < 0.01). The quantity of Tau protein was significantly increased in all the age groups of Elav-Gal4, UAS-Tau LacZ flies reared on regular food than the control flies reared on the same type of food (Fig. 1C; p < 0.01). Conversely, there was a decrease in the quantity of Tau protein in all the age groups of Elav-Gal4, UAS-Tau LacZ flies reared on 10 μM or 20 μM jatamansinol supplemented food. But the reduction in the quantity of Tau protein was significant in 10 or 20-day-old Elav-Gal4, UAS-Tau LacZ flies only (Fig. 1C).
Quantification of Tau protein. A Tau protein expression in western blot and B band intensity of expressed Tau protein. C Tau protein level by ELISA method in 20-day-old control, Elav Gal4, UAS-Tau LacZ flies reared in regular food media, and 10 and 20 μM jatamansinol supplemented food media. Values are mean ± SE Tau protein level by ELISA or western blot band intensity. Significant against OK control at **p < 0.01. Significant against Elav-Gal4, UAS-Tau LacZ at ††p < 0.01. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against Elav-Gal4, UAS-Tau LacZ
Jatamansinol Does Not Change the Tau Gene Expression
There was endogenous Tau gene expression in 10 and 20-day-old OK flies reared on regular and 10 μM or 20 μM jatamansinol supplemented food. But a comparatively higher Tau gene expression is evident in the Elav-Gal4, UAS-Tau LacZ flies when reared on regular food. Also, there were no noticeable changes in Tau gene expression in 10, 20, and 30-day-old Elav-Gal4, UAS-Tau LacZ flies reared on 10 μM or 20 μM jatamansinol supplemented food compared to the same type of flies reared on regular food (Fig. 2; p > 0.05).
Tau gene expression in OK and Tau protein-induced AD Drosophila was reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. Values are mean ± SE. Significant against OK control at **p < 0.01. Without any symbol in the error bars is not significant against OK control or Elav-Gal4, UAS-Tau LacZ
Jatamansinol Prolongs the Lifespan
The Drosophila was reared on regular food media; the lifespan of tauopathy (Elav-Gal4, UAS-Tau lacZ) flies was 38.27 ± 0.621 days and control (OK) flies was 69.14 ± 0.94 days (Fig. 3A). The supplementation of jatamansinol with food media improved OK and Elav-Gal4, UAS-Tau lacZ flies’ survival rate in a dose-dependent manner (Fig. 3B). The OK flies reared in 10 μM (72.10 ± 0.87 days) or 20 μM (73.46 ± 0.84 days) and Elav-Gal4, UAS-Tau lacZ flies reared in 10 μM (40.73 ± 0.758 days) or 20 μM (41.85 ± 0.774 days) jatamansinol supplementation extended their lifespan than of Tau-expressed AD flies reared in regular food.
Survival rate (A) and mean lifespan (B) of OK and Tau protein-induced AD Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. Values are mean ± SE mean lifespan (n = 20/assay, assay repeated 20 times/experimental group). Significant against OK control at **p < 0.01. Significant against Elav-Gal4, UAS-Tau LacZ at ††p < 0.01. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against Elav-Gal4, UAS-Tau LacZ
Jatamansinol Increases the Locomotor Activity
There was a significant reduction in the number of flies with regular locomotor activity in Elav-Gal4, UAS-Tau lacZ flies reared on regular food in 10, 20 and 30-day-aged groups compared to control (p < 0.01; Fig. 4A). There was no significant change in the locomotor activity of OK flies reared in regular and jatamansinol supplemented food media (p > 0.05; Fig. 4A). The supplementation of 10 μM or 20 μM of jatamansinol in food-fed Elav-Gal4, UAS-Tau lacZ flies improved locomotor activity. A significant improvement in locomotor activity was observed in all the age groups of Elav-Gal4, UAS-Tau lacZ flies reared in 20 μM jatamansinol supplemented food. But significantly improved locomotor activity was observed only in 10-day-old Elav-Gal4, UAS-Tau lacZ flies reared in 10 μM of jatamansinol supplemented food compared to Elav-Gal4, UAS-Tau lacZ flies reared on regular food (Fig. 4A; p < 0.05).
Climbing index (CI) (A) and memory performance index (PI) (B) of OK and Tau protein-induced AD Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. The box represents the 1st quartile (Q1–25th percent) and 3rd quartile (Q3–75th percent), and the line inside the box represents the data set’s median. All error bars represent a 95% confidence interval. Values are mean ± SE CI/PI (n = 20 or 50/assay, assay repeated 20 times/experimental group). Significant against OK control at **p < 0.01. Significant against Elav-Gal4, UAS-Tau LacZ at †p < 0.05, ††p < 0.01. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against Elav-Gal4, UAS-Tau LacZ
Jatamansinol Enhances the Learning and Memory
There was a significant decrease in memory performance index in Elav-Gal4, UAS-Tau lacZ flies reared on regular food compared to control flies with their respective age groups (Fig. 4B). The memory performance index was increased in 10, 20, and 30-day-old tauopathy, and OK flies reared in 10 μM or 20 μM jatamansinol supplemented food. A significant increase in the memory performance index was recorded in all the age groups of OK and Elav-Gal4, UAS-Tau lacZ flies reared in 20 μM jatamansinol supplemented food compared to Elav-Gal4, UAS-Tau lacZ flies reared without jatamansinol food (Fig. 4B).
Jatamansinol Ameliorates the Eye Degeneration
The eye images of control and GMR-Gal4, UAS-Tau lacZ flies evidence the eye degeneration in GMR-Gal4, UAS-Tau lacZ flies reared in regular food (Fig. 5A, B). Tau-induced eye degeneration was significant compared to control flies reared in regular food (p < 0.01; Fig. 5C). A significant reduction in the phenotypic score of eye degeneration was recorded in GMR-Gal4, UAS-Tau lacZ flies reared in 10 μM or 20 μM jatamansinol supplemented food to GMR-Gal4, UAS-Tau lacZ flies reared in regular food (p < 0.01; Fig. 5C).
Eye degeneration in 20-day-old OK and Tau protein-induced AD Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. A Scanning electron microscopic (SEM) fly eye pictures. B Flynotyper analysis eye pictures. C Eye phenotypic score in respective experimental groups. The box represents the 1st quartile (Q1–25th percent) and 3rd quartile (Q3–75th percent), and the line inside the box represents the data set’s median. All error bars represent a 95% confidence interval. n = 10/assay/experimental group. Significant against OK control at **p < 0.01. Significant against GMR-Gal4, UAS-Tau LacZ at ††p < 0.01. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against GMR-Gal4, UAS-Tau LacZ
Jatamansinol Prevents the Oxidative Stress Generation
Ten or 20 μM jatamansinol treatment to the OK flies reduced the ROS, LPO, H2O2, PC, and NO oxidative stress markers (Fig. 6). In all the age groups of Elav-Gal4, UAS-Tau LacZ flies, oxidative stress marker levels increased significantly compared to OK flies reared on regular food (p < 0.01). There was a significant reduction of ROS in 10-day-old Elav-Gal4, UAS-Tau LacZ flies reared in 10 μM (p < 0.05) and 20 μM (p < 0.01) jatamansinol supplemented food, whereas 20- and 30-day-old Elav-Gal4, UAS-Tau LacZ flies were significantly reduced in the 20 μM jatamansinol supplemented group (Fig. 6A). A significant reduction of LPO was noticed in 20 μM jatamansinol fed 20- and 30-day-old Elav-Gal4, UAS-Tau LacZ flies (Fig. 6B; p < 0.01) compared to the same set of flies reared on regular food. Also, it reduced significantly in 20-day-old Elav-Gal4, UAS-Tau LacZ flies reared on 10 μM jatamansinol supplemented food (Fig. 6B; p < 0.01). Irrespective of age groups, H2O2, PC, and NO levels were significantly reduced in Elav-Gal4, UAS-Tau LacZ flies reared on 10 μM or 20 μM jatamansinol supplemented food (Fig. 6C, D, E).
Oxidative stress analysis. A ROS, B LPO, C HP, D PC, and E NO in OK and Tau protein-induced AD Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. Values are mean ± SE. Significant against OK control at *p < 0.05, **p < 0.01. Significant against Elav-Gal4, UAS-Tau LacZ at †p < 0.05, ††p < 0.01. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against Elav-Gal4, UAS-Tau LacZ
Jatamansinol Improves Antioxidant Defense Level
The SOD, CAT, and GST enzyme activities were enhanced in OK flies reared on 10 μM or 20 μM jatamansinol supplemented food compared to the same type of flies reared on regular food (Fig. 7). Significant reduction is evidenced in the SOD, CAT, and GST enzyme activities in Elav-Gal4, UAS-Tau LacZ flies than in the OK flies reared on regular food (Fig. 7; p < 0.01). Significantly increased SOD activity is shown in the 20 and 30-day-old Elav-Gal4, UAS-Tau LacZ flies reared on 10 μM or 20 μM jatamansinol supplemented food than Elav-Gal4, UAS-Tau LacZ flies reared on regular food (Fig. 7A; p < 0.01). But significantly increased SOD activity was noticed in 10-day-old Elav-Gal4, UAS-Tau LacZ flies reared on 20 μM jatamansinol supplemented food (p < 0.01). CAT activity significantly increased in 10 μM or 20 μM jatamansinol supplemented 20-day-old Elav-Gal4, UAS-Tau LacZ flies and 20 μM jatamansinol supplemented 10-day-old Elav-Gal4, UAS-Tau LacZ flies than the same type of flies reared on regular food (Fig. 7B; p < 0.01). GST activity was significantly increased in 10-day-old Elav-Gal4, UAS-Tau LacZ flies fed with 10 μM or 20 μM jatamansinol supplemented food compared to Elav-Gal4, UAS-Tau LacZ flies reared on regular food (Fig. 7C; p < 0.01).
Antioxidant enzymes: SOD (A), CAT (B) and GST (C) activities in OK and Tau protein-induced AD Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. Values are mean ± SE. Significant against OK control at **p < 0.01. Significant against Elav-Gal4, UAS-Tau LacZ at ††p < 0.05. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against Elav-Gal4, UAS-Tau LacZ
Jatamansinol Inhibits the Cholinesterase Activity
An age-dependent decrease in AChE and BuChE activity was recorded in OK flies reared in jatamansinol supplemented regular food (Fig. 8). But a significant age-dependent increase in AChE and BuChE activity was recorded in Elav-Gal4, UAS-Tau lacZ flies compared to control flies when both flies were reared in regular food media (Fig. 8A, B; p < 0.01). The reduced AChE and BuChE activity was recorded in Elav-Gal4, UAS-Tau lacZ flies reared in 10 μM or 20 μM jatamansinol supplemented with regular food (Fig. 8A, B). The significant inhibition of AChE activity was recorded in 10 or 20-day-old Elav-Gal4, UAS-Tau lacZ flies reared in 10 μM or 20 μM jatamansinol supplemented food (Fig. 8A), whereas significant BuChE inhibitory activity was recorded in Elav-Gal4, UAS-Tau lacZ flies reared on 20 μM jatamansinol supplemented food compared to the Elav-Gal4, UAS-Tau lacZ flies reared on regular food (without the jatamansinol supplementation) (Fig. 8B).
AChE (A) and BuChE (B) activities in OK and Tau protein-induced AD Drosophila were reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. Values are mean ± SE. Significant against OK control at **p < 0.01. Significant against Elav-Gal4, UAS-Tau LacZ at †p < 0.05, ††p < 0.01. Without the asterisk symbol (*) is not significant against OK control; without the dagger symbol (†) is not significant against Elav-Gal4, UAS-Tau LacZ
Jatamansinol Is Not an Antifeedant
No significant difference is evidenced (p > 0.05) in the OD values of OK and Elav-Gal4, UAS-Tau LacZ larvae or adults reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media (Fig. 9A). Also, insignificant differences (p > 0.05) were seen in the red color intensity plot of larvae or adults of OK and Elav-Gal4, UAS-Tau LacZ Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media (Fig. 9B, C).
Jatamansinol food intake analysis: A optical density (OD) values as a guesstimate of food consumption, B representative of larval and adult fly pictures, and C red color intensity plot of third instar larvae and adult OK and Tau protein-induced AD Drosophila reared on regular and 10 μM or 20 μM concentrations of jatamansinol supplemented food media. Values are mean ± SE. Without any symbol in error bars are not significant against OK control or Elav-Gal4, UAS-Tau LacZ
Quantification of Jatamansinol Consumption by Drosophila
The HPLC chromatogram of the adults or larvae of OK and Elav-Gal4, UAS-Tau LacZ showed the consumption of jatamansinol (SF3). It is clear that the consumption of jatamansinol increases with the increased concentrations of jatamansinol (Table 1).
Discussion
The jatamansinol isolated from the plant N. jatamansi is an angular pyranocoumarin [34]. It contains a six-membered pyran ring angularly fused with coumarin moiety and has neuroprotective properties [35, 36]. In our earlier study, jatamansinol showed its multitarget therapeutic nature against AD in Aβ42-induced AD Drosophila model [16]. But its neuroprotective properties against Tau-induced Drosophila are not yet validated. Hence, the current study investigated the neuroprotective effect of jatamansinol in the Tau-induced Drosophila AD model.
The Tau protein hyperphosphorylation in the brain leads to neurofibrillary tangle formation and is one of AD’s leading pathological markers. It has been evidenced that pan-neuronal overexpression of Tau protein in Drosophila increases its expression and level in Elav-Gal4, UAS-Tau lacZ flies (Fig. 1A, B), causing AD-like symptoms. The reduced Tau protein expression level in Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 8) is evidence of the antitauopathic nature of jatamansinol. It is also evidenced that medicinal plants such as N. jatamansi root extract ameliorate Aβ42-expressed protein in Drosophila [14]; Convolvulus pluricaulis root extract reduces the Tau protein in tauopathy flies [7]. The reduction of Tau protein level in Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food indicates that jatamansinol could have targeted Tau-based therapies.
Ten-, 20-, and 30-day-old OK flies show Tau gene expression (Fig. 2), confirming the Tau gene in OK flies. Elav-Gal4, UAS-Tau LacZ flies express Tau gene indicates transgenic Tau expression in the Drosophila AD model (Fig. 2). Jatamansinol supplemented Elav-Gal4, UAS-Tau LacZ flies show no change in the Tau gene expression compared to Elav-Gal4, UAS-Tau LacZ flies reared on regular food; it is the indication that jatamansinol is not transcriptionally regulating Tau gene expression. Tau-based therapies include targeting Tau phosphorylation inhibition, microtubule stabilization, blocking Tau oligomerization, and enhancing Tau degradation [37]. This result proposes that (1) Jatamansinol might be translationally modifying the Tau protein expression or (2) Jatamansinol might be inducing Tau protein degradation. Further molecular-level studies are required about the mechanism of neuroprotective activity of jatamansinol against Tau protein-induced AD.
The improved survival rate and extended lifespan of OK and Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 3) confirm that jatamansinol has an antiaging property. Phytochemicals isolated from medicinal plants have demonstrated the model organisms’ antiaging properties. The antiaging property of jatamansinol in the Aβ42-induced AD Drosophila model [16]; these results give strong evidence for the same. The chlorogenic acid isolated from the N. jatamansi showed a better survival rate in the Aβ42-expressed transgenic Drosophila [14]. The curcumin isolated from Curcuma longa acts against aging in Drosophila [38] and Caenorhabditis elegans [39].
Tauopathies are the complex clinicopathological assemblages of symptoms, including disorders in locomotor activity [40]. Tauopathic flies showed age-related locomotor deficits and a decline in their climbing ability [7]. The current study also showed a significantly reduced climbing ability in Elav-Gal4, UAS-Tau lacZ flies compared to control flies, while both were reared on regular food (Fig. 4A; p < 0.01). The consumption of jatamansinol food-fed Elav-Gal4, UAS-Tau lacZ flies improved locomotor activity (Fig. 4A). Also, jatamansinol improved locomotor ability in Aβ42-induced AD Drosophila model [16], suggesting that jatamansinol can improve AD’s motor or physiological disability symptoms.
Tau protein accumulation in the Drosophila brain mushroom body cells causes learning and memory deficits [41]. The increased memory performance index in all the age groups of tauopathic flies reared in jatamansinol supplemented food (Fig. 4B) confirms jatamansinol enhances memory. In line with this study, Joshi and Parle [11] have experimentally proved that the extract of N. jatamansi improves memory in mice. Our previous study on the Aβ42-induced AD Drosophila model has demonstrated the memory-enhancing property of isolated jatamansinol from N. jatamansi [16]. In line with the earlier observations, the current study suggests that jatamansinol can improve learning and memory in Tau-induced AD fly model.
Neurodegeneration is not restricted to human AD patients’ brain area; eventually, it leads to extensive ganglion cell loss in the eye retina [42]. Using the UAS-Gal4 system human Tau gene expressed in the fly eye evidenced the Tau protein-induced eye degeneration (Fig. 5A, B); it has been quantified by the Flynotyper computational method (Fig. 5C). GMR-Gal4, UAS-Tau lacZ flies’ phenotypic scores reared on regular food media are comparatively higher than control flies (p < 0.01); it indicated Tau-induced neurodegeneration in fly eyes. The significantly reduced eye phenotypic score in GMR-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food media (p < 0.01) is evidence of its ameliorative role on eye degeneration. Also, our earlier study has proven to lessen the property of jatamansinol on the eye degeneration of Aβ42-induced AD Drosophila model [16]. This research work is another sign of the neuroprotective role of jatamansinol in the Tau protein-induced AD Drosophila model.
The brains of human AD patients show a high level of oxidative stress with the formation of senile plaques and neurofibrillary tangles [43]. It was shown that the pan-neuronal overexpression of human Tau protein in rats and Drosophila leads to an increased level of ROS and a reduced level of antioxidant enzymes [6, 7]. Age-dependent increase in ROS level in Elav-Gal4, UAS-Tau lacZ flies reared in regular food (Fig. 6A; p < 0.01) confirms that Tau protein expression in flies induces oxidative stress by generating reactive free radicals. A dose-dependent decrease in the ROS level in Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 6A) suggests that jatamansinol scavenges the reactive free radicals generated by Tau protein. In line with this study, ethanolic extract of N. jatamansi reduces the in vitro cells’ ROS level [44].
Increased malondialdehyde (MDA), an LPO product in human AD patients’ brains, indicated brain cells under oxidative stress conditions [45]. An increased MDA level in Elav-Gal4, UAS Tau-lacZ flies compared to control flies (Fig. 6B; p < 0.01) suggests that the overexpression of human Tau protein in the Drosophila brain induces oxidative stress and, in turn, initiates the LPO process [7]. The reduction in the MDA level in Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 6B) evidences the anti-LPO activity of jatamansinol. Previous studies have demonstrated that N. jatamansi extract protects cardiac tissues from LPO in Wistar rats [46], attenuating lipid peroxidative potential in 6-hydroxydopamine-induced Parkinsonism in rats [47].
H2O2 is a stable, uncharged, and freely diffusible ROS; its level increases in human AD patients’ brains [48]. Tau protein forms the neurofibrillary tangles, binds and reduces copper ions, and serves as a metalloenzyme to catalyze the generation of neurotoxic H2O2 from O2 through Cu2+ reduction [49]. Highly toxic H2O2 generated by tau protein is directly causing mitochondria permeability transition and cytochrome release. It activates caspase-3, which is pivotal event responsible for the dysfunction or death of neuronal cells (neurodegeneration) that contribute to AD pathogenesis [50]. An increased H2O2 level in Elav-Gal4, UAS-Tau lacZ flies compared to control flies reared in regular food media (Fig. 6C; p < 0.01) confirms that the overexpression of Tau protein generates the H2O2, by which Tau protein exerts its neuronal toxicity. Decreased level of H2O2 in Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 6C) confirms that jatamansinol has the property of scavenging the H2O2 generated by human Tau protein. N. jatamansi rhizome extract protects the H2O2-induced oxidative stress in C6 glioma cells [51]. Since the Tau protein causes a neurotoxicant (H2O2), it plays a significant role in AD pathogenesis; this study empowers the development of novel therapeutic strategies to treat AD patients using jatamansinol.
Oxidative stress can cause oxidative damage to the protein, and oxidatively modified proteins accumulate as protein carbonyls [52, 53]. Neurofibrillary tangles are associated with carbonylated protein accumulation in the AD brain [54]. The increased PC in the Elav-Gal4, UAS-Tau lacZ flies compared to control flies reared on regular food (Fig. 6D; p < 0.01) indicates that Tau protein overexpression causes protein oxidation. Conversely, the decreased PC in the Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 6D) evidences its oxidative stress inhibitory potential. An in vitro study also demonstrated that ethanolic root extract of N. jatamansi reduces protein oxidation and, in turn, the PC [44].
RNS are derivatives of nitric oxide compounds such as nitric oxide radical (NO or NO•), nitrogen dioxide radical (NO2•), nitrite (NO2−), and peroxynitrite (ONOO−). Excess RNS results in cell injury and cell death by inducing nitrosative stress [28]. Higher RNS damages the protein and nucleic acids involved in AD’s pathogenesis [55]. The higher NO level in Elav-Gal4, UAS-Tau lacZ flies reared on regular food (Fig. 6E; p < 0.01) showed the involvement of Tau protein in the generation of RNS. The significant NO reduction in the Elav-Gal4, UAS-Tau lacZ flies reared on jatamansinol supplemented food (Fig. 6E) evidences its RNS scavenging activity. A previous study also demonstrated the N. jatamansi inhibiting NO activity in Aβ42-expressed flies [56].
SOD and CAT are essential antioxidants safeguarding oxidative stress-induced damages [18]. In AD patients, central and peripheral nerve tissues show reduced SOD and CAT activity [57]. It is also evident that pan-neuronal Tau protein expression in Drosophila reduces the SOD and CAT activity [7, 58]. The significant SOD and CAT activity reduction in Elav-Gal4, UAS-Tau lacZ flies reared on regular food (Fig. 7A, B; p < 0.01) confirms that Tau protein generates overwhelming ROS production; the depletion of SOD and CAT antioxidants might be for scavenging generated ROS. The gradual increase in SOD and CAT activities in Elav-Gal4, UAS-Tau lacZ flies fed jatamansinol supplemented food (Fig. 7A, B) confirms that jatamansinol can potentially influence the activity of SOD and CAT. An aqueous extract of N. jatamansi containing coumarin compounds has antioxidant potential and can reverse haloperidol-induced catalepsy in rats with increased SOD and CAT activities [13].
GST is an essential mediator in ROS response in all eukaryotes [59]. The significant reduction of GST level in Elav-Gal4, UAS-Tau lacZ flies reared on regular food (Fig. 7C; p < 0.01) indicates GST’s involvement in the metabolism of ROS generated by Tau protein. It has been evident a declined GST level in the frontal cortex of AD patients [60]. Hydro-ethanolic extract of N. jatamansi increases the GST and other antioxidant enzymes in the cold-restraint stress rat model [61]. The increased GST level in Elav-Gal4, UAS-Tau lacZ flies reared in jatamansinol supplemented food (Fig. 7C) suggests that jatamansinol mitigates oxidative stress by increasing the GST level.
It is evident from the biochemical assays that jatamansinol increases the antioxidant level and reduces the oxidative stress in the Tau protein-expressed AD Drosophila model. Our earlier in silico work demonstrated that jatamansinol inhibits Keap1 protein, activating Nrf2 to detoxify protein, increasing the antioxidant level, and reducing oxidative stress [15]. Also, jatamansinol suppresses oxidative stress and induces antioxidant enzyme activity in Aβ42-induced neurotoxicity in the AD Drosophila model [16]. The current work provides additional evidence for the antioxidant capacity of jatamansinol against the Tau protein-induced neurotoxicity in the AD Drosophila model.
The cholinergic system, composed of organized nerve cells that use the neurotransmitters during neuronal signal transmission, plays a crucial role in AD’s pathogenesis [62, 63]. The clinical symptoms associated with AD result from deterioration of cholinergic transmission in the human cerebral cortex region with the increased neurotransmitter enzymes such as AChE and BuChE [64]. One of the therapeutic strategies against AD is to increase the cholinergic level by inhibiting AChE and BuChE activities [65]. The increased AChE and BuChE in the Elav-Gal4, UAS-Tau lacZ flies fed with regular food (Fig. 8) indicated that the Tau-induced neurofibrillary tangles might be involved in disrupting the cholinergic system. A gradual decrease of AChE and BuChE activities in Elav-Gal4, UAS-Tau lacZ flies reared on jatamansinol supplemented food media (Fig. 8) evidenced jatamansinol has the cholinesterase inhibition property. The previous in silico work indicates the dual cholinesterase inhibitory activity of jatamansinol [15]; in the Aβ42 protein-induced AD Drosophila model, dual cholinesterase inhibitory activity of jatamansinol was proved [16]. The current study supports such properties in the Tau-induced AD Drosophila model.
Our recent study [16] confirmed that the intake of jatamansinol by the Tau-induced Drosophila AD model through food (Fig. 9, Table 1) affirms that jatamansinol is not an antifeedant for larva and adult Drosophila.
Conclusion
This study demonstrates that jatamansinol from the N. jatamansi is a multitargeted neuroprotective agent against human Tau-induced neurotoxicity in the AD Drosophila model. Jatamansinol suppresses ROS generation and increases the antioxidant enzyme activities in Tau protein-expressed flies. It inhibits cholinesterase activities and improves locomotory, learning, and memory in the Tau protein-induced AD flies. Jatamansinol ameliorates Tau protein-induced eye degeneration and decreases the level of Tau protein expression in tauopathy flies. The pyrone ring and dihydropyran moiety in the jatamansinol might have been responsible for its antioxidant nature.
Data Availability
All data generated or analyzed that support our findings of this study are included in this manuscript and presented as tables, figures, images, and supplementary files.
Abbreviations
- AChE:
-
Acetylcholine esterase
- AD:
-
Alzheimer’s disease
- ANOVA:
-
Analysis of variance
- Aβ42 :
-
Amyloid beta-42
- BuChE:
-
Butrylcholine esterase
- CAT:
-
Catalase
- CDNB:
-
1-Chloro-2,4-dinitrobenzene
- CI:
-
Climbing index
- 95% CI:
-
95% Confidence interval
- DCF-DA:
-
2′,7′-Dichlorodihydrofluorescein diacetate
- DNPH:
-
Dinitrophenyl hydrazine
- ELISA:
-
Enzyme-linked immunosorbent assay
- FDA:
-
Food and Drug Administration
- GMR:
-
Glass multimer reporter
- GST:
-
Glutathione S-transferase
- H2O2 :
-
Hydrogen peroxide
- HRP:
-
Horseradish peroxidase
- Keap1:
-
Kelch-like ECH-associating protein 1
- LC50 :
-
Lethal concentration 50
- LPO:
-
Lipid peroxidation
- MAPτ:
-
Microtubule-associated protein tau
- NBT:
-
Nitroblue tetrazolium
- NFTs:
-
Neurofibrillary tangles
- NO:
-
Nitric oxide
- Nrf2:
-
Nuclear receptor factor 2
- P scores:
-
Eye phenotypic score
- PC:
-
Protein carbonyls
- PI:
-
Performance index
- PVDF:
-
Polyvinylidene difluoride
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- RNS:
-
Reactive nitrogen species
- ROS:
-
Reactive oxygen species
- RP49:
-
Ribosomal protein 49
- SDS:
-
Sodium dodecyl sulfate
- SOD:
-
Superoxide dismutase
- UAS:
-
Upstream activation sequence
- μM:
-
Micromolar
References
Fan L, Mao C, Hu X, Zhang S, Yang Z, Hu Z, Sun H, Fan Y, Dong Y et al (2020) New insights into the pathogenesis of Alzheimer’s disease. Front Neurol 10:1312. https://doi.org/10.3389/fneur.2019.01312
Alzheimer's Association (2021) Alzheimer’s disease facts and figures. Alzheimer's Dement 17(3). https://doi.org/10.1002/alz.12328
Cubinkova V, Valachova B, Uhrinova I, Brezovakova V, Smolek T, Jadhav S, Zilka N (2018) Alternative hypotheses related to Alzheimer’s disease. Bratis Med J 119:210–216. https://doi.org/10.4149/BLL_2018_039
Guan ZZ (2008) Cross-talk between oxidative stress and modifications of cholinergic and glutaminergic receptors in the pathogenesis of Alzheimer’s disease. Acta Pharmacol Sin 29:773–780. https://doi.org/10.1111/j.1745-7254.2008.00819.x
Mangialasche F, Polidori MC, Monastero R, Ercolani S, Camarda C, Cecchetti R, Mecocci P (2009) Biomarkers of oxidative and nitrosative damage in Alzheimer’s disease and mild cognitive impairment aging. Res Rev 8:285–305. https://doi.org/10.1016/j.arr.2009.04.002
Cente M, Filipcik P, Pevalova M, Novak M (2006) Expression of a truncated tau protein induces oxidative stress in a rodent model of tauopathy. Eur J Neurosci 24:1085–1090. https://doi.org/10.1111/j.1460-9568.2006.04986.x
Anupama KP, Shilpa O, Anet A, Siddhanna TK, Gurushankara HP (2019) Convolvulus pluricaulis (Shankhapushpi) ameliorates human microtubule-associated protein tau (hMAPτ) induced neurotoxicity in Alzheimer’s disease Drosophila model. J Chem Neuroanat 95:115–122. https://doi.org/10.1016/j.jchemneu.2017.10.002
Feng Y, Wang X (2012) Antioxidant therapies for Alzheimer’s disease. Oxid Med Cell Longev 472932. https://doi.org/10.1155/2012/472932
Maramai S, Benchekroun M, Gabr MT, Yahiaoui S (2020) Multitarget therapeutic strategies for Alzheimer’s disease: review on emerging target combinations. BioMed Research Int 5120230.https://doi.org/10.1155/2020/5120230
Tiwari RS, Tripathi JS (2013) A critical appraisal of dementia with special reference to Smritibuddhihrass. Ayu 34:235–242. https://doi.org/10.4103/0974-8520.123102
Joshi H, Parle M (2006) Nardostachys jatamansi improves learning and memory in mice. J Med Food 9(1):113–118. https://doi.org/10.1089/jmf.2006.9.113
Rao VS, Rao A, Karanth KS (2005) Anticonvulsant and neurotoxicity profile of Nardostachys jatamansi in rats. J Ethnopharmacol 102(3):351–356. https://doi.org/10.1089/jmf.2006.9.11310.1016/j.jep.2005.06.031
Rasheed AS, Venkataraman S, Jayaveera KN, Fazil AM, Yasodha KJ, Aleem MA, Mohammed M, Khaja Z et al (2010) Evaluation of toxicological and antioxidant potential of Nardostachys jatamansi in reversing haloperidol-induced catalepsy in rats. Int J Gen Med 3:127–136. https://doi.org/10.2147/ijgm.s9156
Liu QF, Jeon Y, Sung YW, Lee JH, Jeong H, Kim YM, Yun HS, Chin YW et al (2018) Nardostachys jatamansi ethanol extract ameliorates Aβ42 cytotoxicity. Biol Pharm Bull 41(4):470–477. https://doi.org/10.1248/bpb.b17-00750
Anupama KP, Shilpa O, Anet A, Gurushankara HP (2021) Jatamansinol from Nardostachys jatamansi: a multi-targeted neuroprotective agent for Alzheimer’s disease. J Biomol Struct Dyn 2:1–21. https://doi.org/10.1080/07391102.2021.2005681
Anupama KP, Shilpa O, Anet A, Raghu SV, Gurushankara HP (2022) Jatamansinol from Nardostachys jatamansi (D.Don) DC protects Aβ42-induced neurotoxicity in Alzheimer’s disease Drosophila model. Neurotoxicol 90:62–78. https://doi.org/10.1016/j.neuro.2022.02.011
Vasudev V, Gurushankara HP (2016) Understandings in cytogenetic techniques. Today and Tomorrow Publishers, New Delhi., p 320
Shilpa O, Anet A, Anupama KP, Siddanna TK, Gurushankara HP (2018) Lead modulated heme synthesis inducing oxidative stress-mediated genotoxicity in Drosophila melanogaster. Sci Total Environ 634:628–639. https://doi.org/10.1016/j.scitotenv.2018.04.004
Raghu SV, Mohammad F, Chua JY, Lam J, Loberas M, Sahani S, Barros CS, Claridge-Chang A (2018) A zinc-finger fusion protein refines Gal4-defined neural circuits. Mol brain 11(1):46. https://doi.org/10.1186/s13041-018-0390-7
Dourlen P (2017) Identification of Tau toxicity modifiers in the Drosophila eye. Methods Mol Biol 1523:375–389. https://doi.org/10.1007/978-1-4939-6598-4_26
Finney DT (1971) Probit analysis. 3rd ed. Cambridge University Press, New York, xv + 333 pp.
Shilpa O, Anupama KP, Antony A, Gurushankara HP (2021) Lead (Pb)-induced oxidative stress mediates sex-specific autistic-like behaviour in Drosophila melanogaster. Mol Neurobiol 58(12):6378–6393. https://doi.org/10.1007/s12035-021-02546-z
Iyer J, Wang Q, Le T, Pizzo L, Grönke S, AmbegaOKar SS, Imai Y, Srivastava A et al (2016) Quantitative assessment of eye phenotypes for functional genetic studies using Drosophila melanogaster. G3 (Bethesda) 6(5):1427–1437. https://doi.org/10.1534/g3.116.027060
Driver AS, Kodavanti PS, Mundy YR (2000) Age-related changes in reactive oxygen species production in rat brain homogenates. Neurotoxicol Teratol 22:175–181. https://doi.org/10.1016/S0892-0362(99)00069-0
Gajare KA, Deshmukh AA, Mehwish S, Parveen S (2005) Effect of ashwagandha (Withania somnifera) root powder on lipid peroxidation in Drosophila melanogaster. Ind J of Gerontol 19:265–272
Rhee AG, Tong-Shin C, Woojin J, Dongmin K (2010) Methods for detection and measurement of hydrogen peroxide inside and outside of cells. Mol Cells 29:539–549. https://doi.org/10.1007/s10059-010-0082-3
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz A, Ahn B, Shalteil S et al (1990) Determination of carbonyl content of oxidatively modified proteins. Methods Enzymol 186:464–478. https://doi.org/10.1016/0076-6879(90)86141-h
Bryan NS, Grisham MB (2007) Methods to detect nitric oxide and its metabolites in biological samples. Free Radic Biol Med 43:645–657. https://doi.org/10.1016/j.freeradbiomed.2007.04.026
Rawal S, Singh P, Gupta A, Mohanty S (2014) Dietary intake of Curcuma longa and Emblica officinalis increases life span in Drosophila melanogaster. Bio Med Res Inter 910290. https://doi.org/10.1155/2014/910290
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Habig WH, Jakoby WB (1981) Assays for differentiation of glutathione-S-transferases. Methods Enzymol 77:398–405. https://doi.org/10.1016/s0076-6879(81)77053-8
Ellman GI, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Anet A, Shilpa O, Anupama KP, Gurushankara HP (2019) Bisphenol A-induced oxidative stress-mediated genotoxicity in Drosophila melanogaster. J Hazard Mater 370:42–53. https://doi.org/10.1016/j.jhazmat.2018.07.050
Feunang YD, Roman E, Craig K, Leonid C, Janna H, Gareth O, Eoin F, Christoph S et al (2016) ClassyFire: automated chemical classification with a comprehensive, computable taxonomy. J Chem Inf 61. https://doi.org/10.1186/s13321-016-0174-y
Murray RDH, Mendez J, Brown SA (1982) The natural coumarins: occurrence, chemistry, and biochemistry. John Wiley & Sons Ltd., Chichester, pp 702
Sharma A (2016) Neuroprotective agents. In: Penta S (ed) Advances in structure and activity relationship of coumarin derivatives. Academic Press, pp 77–99
Kumar A, Singh E (2015) A review on Alzheimer’s disease pathophysiology and its management: an update. Pharmacol Rep 67:195–203. https://doi.org/10.1016/j.pharep.2014.09.004
Shen LR, Xiao F, Yuan P, Chen Y, Gao QK, Parnell LD, Meydani M, Ordovas JM et al (2013) Curcumin-supplemented diets increase superoxide dismutase activity and mean lifespan in Drosophila. Age (Dordr) 35:1133–1142. https://doi.org/10.1007/s11357-012-9438-2
Liao VH, Yu CW, Chu YJ, Li WH, Hsieh YC, Wang TT (2011) Curcumin-mediated lifespan extension in Caenorhabditis elegans. Mech Ageing Dev 132:480–487. https://doi.org/10.1016/j.mad.2011.07.008
Ganguly J, Jog M (2020) Tauopathy and movement disorders unveiling the chameleons and mimics. Front Neurol 11:599384. https://doi.org/10.3389/fneur.2020.599384
Mershin A, Pavlopoulos E, Fitch O, Braden BC, Nanopoulos DV, Skoulakis EM (2004) Learning and memory deficits upon TAU accumulation in Drosophila mushroom body neurons. Learn Mem 3:277–287. https://doi.org/10.1101/lm.70804
Blanks JC, Torigoe Y, Hinton DR, Blanks RH (1996) Retinal pathology in Alzheimer’s disease. I. Ganglion cell loss in foveal/parafoveal retina. Neurobiol Aging 17(3):377–384. https://doi.org/10.1016/0197-4580(96)00010-3
Haque MM, Murale DP, Kim YK, Lee JS (2019) Cross-talk between oxidative stress and tauopathy. Int J Mol Sci 20:1959. https://doi.org/10.3390/ijms20081959
Razack S, Kumar KH, Nallamuthu I, Naika M, Khanum F (2015) Antioxidant, biomolecule oxidation protective activities of Nardostachys jatamansi DC. and its phytochemical analysis by RP-HPLC and GC-MS. Antioxidants 4:185–203. https://doi.org/10.3390/antiox4010185
Praticò D, Sung S (2004) Lipid peroxidation and oxidative imbalance: early functional events in Alzheimer’s disease. J Alzheimer’s Dis 6:171–175. https://doi.org/10.3233/jad-2004-6209
Singh M, Khan MA, Kamal YT, Ahmad J, Fahmy UA, Kotta S, Alhakamy, Ahmad NS (2020) Effect of Nardostachys jatamansi DC. on apoptosis, inflammation and oxidative stress induced by doxorubicin in Wistar rats, Plants 9: 1579. https://doi.org/10.3390/plants9111579
Ahmad M, Yousuf S, Khan MB, Hoda MN, Ahmad AS, Ansari MA, Ishrat T, Agrawal AK et al (2006) Attenuation by Nardostachys jatamansi of 6-hydroxydopamine-induced Parkinsonism in rats: behavioral, neurochemical, and immunohistochemical studies. Pharmacol Biochem Behav 83:50–60. https://doi.org/10.1016/j.pbb.2006.01.005
Yang J, Yang J, Liang SH, Xu Y, Moore A, Ran C (2016) Imaging hydrogen peroxide in Alzheimer’s disease via cascade signal amplification. Sci Rep 6:35613. https://doi.org/10.1038/srep35613
Su XY, Wu WH, Huang ZP, Hu J, Lei P, Yu CH, Zhao YF, Li YM (2007) Hydrogen peroxide can be generated by Tau in the presence of Cu(II). Biochem Biophys Res Commun 358(2):661–665. https://doi.org/10.1016/j.bbrc.2007.04.191
DiPietrantonio AM, Hsieh T, Wu JM (1999) Activation of caspase 3 in HL-60 cells exposed to hydrogen peroxide. Biochem Biophys Res Commun 255:477–482. https://doi.org/10.1006/bbrc.1999.0208
Dhuna K, Dhuna V, Bhatia B, Singh J, Kamboj SS (2013) Cytoprotective effect of methanolic extract of Nardostachys jatamansi against hydrogen peroxide-induced oxidative damage in C6 glioma cells. Acta Biochim Pol 60:21–31
Butterfield DA, Kanski J (2001) Brain protein oxidation in age-related neurodegenerative disorders that are associated with aggregated proteins. Mech Ageing Dev 122:945–962. https://doi.org/10.1016/s0047-6374(01)00249-4
Beal MF (2002) Oxidatively modified proteins in aging and disease. Free Radic Bio Med 32:797–803. https://doi.org/10.1016/s0891-5849(02)00780-3
Smith MA, Sayre LM, Anderson V, Harris PL, Beal MF, Kowall N, Perry G (1998) Cytochemical demonstration of oxidative damage in Alzheimer disease by immunochemical enhancement of the carbonyl reaction with 2,4-dinitrophenylhydrazine. J Histochem Cytochem 46:731–735. https://doi.org/10.1177/002215549804600605
Reddy VP, Zhu X, Perry G, Smith MA (2009) Oxidative stress in diabetes and Alzheimer’s disease. J Alzheimers Dis 16:763–774. https://doi.org/10.3233/JAD-2009-1013
Berghe WM (2012) Epigenetic impact of dietary polyphenols in cancer chemoprevention: life long remodelling of our epigenomes. Pharmacol Res 65:565–576. https://doi.org/10.1016/j.phrs.2012.03.007
Marcus DL, Thomas C, Rodriguez C, Simberkoff K, Tsai JS, Strafaci JA, Freedman ML (1998) Increased peroxidation and reduced antioxidant enzyme activity in Alzheimer’s disease. Exp Neurol 150:40–44. https://doi.org/10.1006/exnr.1997.6750
Haddadi M, Nongthomba U, Ramesh SR (2016) Biochemical and behavioral evaluation of human MAPTau mutations in transgenic Drosophila melanogaster. Biochem Genet 54:61–72. https://doi.org/10.1007/s10528-015-9701-1
Enayati AA, Ranson H, Hemingway J (2005) Insect glutathione transferases and insecticide resistance. Insect Mol Biol 14:3–8. https://doi.org/10.1111/j.13652583.2004.00529.x
Ansari MA, Scheff SW (2010) Oxidative stress in the progression of Alzheimer’s disease in the frontal cortex. Neuropathol Exp Neurol 69:155–167. https://doi.org/10.1097/NEN.0b013e3181cb5af4
Lyle N, Chakrabarti S, Sur T, Gomes A, Bhattacharyya A (2012) Nardostachys jatamansi protects against cold restraint stress-induced central monoaminergic and oxidative changes in rats. Neurochem Res 37:2748–2757. https://doi.org/10.1007/s11064-012-0867-1
Chen XQ, Mobley WC (2019) Exploring the pathogenesis of Alzheimer disease in basal forebrain cholinergic neurons: converging insights from alternative hypotheses. Front Neurosci 13:446. https://doi.org/10.3389/fnins.2019.00446
Hampel H, Mesulam MM, Cuello AC, Farlow MR, Giacobini E, Grossberg GT, Khachaturian AS, Vergallo A, Cavedo E et al (2018) The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain 141:1917–1933. https://doi.org/10.1093/brain/awy132
Perry EK, Perry RH, Blessed G, Tomlinson BE (1978) Changes in brain cholinesterase in senile dementia of Alzheimer’s type. Neuro Pathol Appl Neurobiol 4:273–277. https://doi.org/10.1111/j.1365-2990.1978.tb00545.x
Lane RM, Kivipelto M, Greig NH (2004) Acetylcholinesterase and its inhibition in Alzheimer’s disease. Clin Neuro Pharmacol 27:141–149. https://doi.org/10.1097/00002826-200405000-00011
Acknowledgements
The authors are thankful to the Central University of Kerala authorities for providing facilities and the Kerala State Council for Science, Technology, and Environment (KSCSTE), CSIR-UGC, and CSIR-SRF for the research fellowships. SVR is grateful to DBT-Ramalingaswami Fellowship.
Author information
Authors and Affiliations
Contributions
KPA: conducted experiments, formal analysis, statistical analysis, writing—original draft.
AA: formal analysis, statistical analysis, review of the original draft
OS: formal analysis, statistical analysis, review of the original draft
SVR: formal analysis, review of the original draft
HPG: original conceptualization, resources, writing original draft, review and editing, and continuous supervision
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable. This study uses the model organism “Drosophila melanogaster,” which does not need ethical approval.
Consent for Publication
All authors have consented to publish in the journal Molecular Neurobiology.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Jatamansinol is a pyranocoumarin present in Nardostachys jatamansi.
• It increases lifespan and improves memory and climbing ability in tauopathy flies.
• Jatamansiol protects the eye degeneration.
• It prevents oxidative stress and boosts antioxidant enzymes.
• Jatamansinol inhibits the choline esterase enzymes.
• Jatamansinol reduces tau protein level.
• Jatamasninol does not change the tau gene expression.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 23.3 MB)
Rights and permissions
About this article
Cite this article
Anupama, K.P., Antony, A., Shilpa, O. et al. Jatamansinol from Nardostachys jatamansi Ameliorates Tau-Induced Neurotoxicity in Drosophila Alzheimer’s Disease Model. Mol Neurobiol 59, 6091–6106 (2022). https://doi.org/10.1007/s12035-022-02964-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-02964-7