Abstract
Müller glia originate from neuroepithelium and are the principal glial cells in the retina. During retinal development, Müller glia are one of the last cell types to be born. In lower vertebrates, such as zebrafish, Müller glia possess a remarkable capacity for retinal regeneration following various forms of injury through a reprogramming process in which endogenous Müller glia proliferate and differentiate into all types of retinal cells. In mammals, Müller glia become reactive in response to damage to protect or to further impair retinal function. Although mammalian Müller glia have regenerative potential, it is limited as far as repairing damaged retina. Lessons learned from zebrafish will help reveal the critical mechanisms involved in Müller glia reprogramming. Progress has been made in triggering Müller glia to reprogram and generate functional neurons to restore vision in mammals indicating that Müller glia reprogramming may be a promising therapeutic strategy for human retinal diseases. This review comprehensively summarizes the mechanisms related to retinal regeneration in model animals and the critical advanced progress made in Müller glia reprogramming in mammals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the World Health Organization, at least 2.2 billion people suffer from vision impairment. Retinal degenerative disease is one of the most common causes of blindness, especially in developed countries. Currently, there are no effective treatments for vision loss due to diseases, including glaucoma, diabetic retinopathy, retinitis pigmentosa, and age-related macular degeneration [1,2,3]. Regenerating retinal neurons and restoring visual function have remained an unsolved problem. The retina and retinal function can potentially be repaired/reconstructed by transplanting exogenous stem/progenitor cells or activating endogenous precursor cells. Activation of endogenous precursor cells to supplement retinal cells is a critical therapeutic strategy because it precludes risks, such as insufficient seed cell sources, oncogenesis, and immune rejection, as well as the ethical issues associated with exogenous stem/progenitor cell transplantation [4,5,6,7,8].
There is no doubt that the endogenous self-healing mechanism is a nearly perfect strategy for repairing injured retina and Müller glia offer such a possibility [9]. In lower vertebrate like zebrafish, Müller glia possess a strongly regenerative response and can completely repair the retina after various injuries [9, 10]. However, this ability is limited in chicks and even more so in mammals [11, 12]. The turnover efficiency of mammalian and human endogenous retinal precursor cells is extremely low under conditions of degeneration or injury and cannot meet the requirements for retinal self-repair. Fortunately, a further understanding of the mechanisms of Müller glia reprogramming has been acquired in lower vertebrate, which has facilitated the use of suitable tools and methods to induce Müller glia to convert into retinal neurons in mammals [9, 13, 14].
In this review, the physiological characteristics and functions of Müller glia in healthy and diseased retinas are briefly introduced, the molecular mechanisms associated with inducing Müller glia reprogramming in zebrafish and chicks are summarized, and major advancements in triggering Müller glia to reprogram in mammals are examined. Finally, several unsolved, but critical issues are described and the prospect of using Müller glia as endogenous stem cells as a future treatment for retinal diseases is discussed.
Müller Glia and Its Function
There are four types of glial cells in the retina: Müller glia, microglia, astrocytes, and oligodendrocytes (not in mammals). Müller glia are predominant, representing 90% of glial cells. Müller glia nuclei are located at the inner nuclear layer (INL) and Müller glia process insinuate themselves between the cell bodies of the neurons and envelope groups of neural processes in the plexiform layers in formation of the inner limiting membrane and outer limiting membrane [15,16,17,18].
Müller glia express special markers in the retina, including gamma-Aminobutyric acid (GABA)A receptors [19], epidermal growth factor receptors (EGFR) [20], the glial glutamate transporter GLAST [21], glutamine synthetase (GS) [22, 23], cellular retinaldehyde-binding protein (CRALBP) [24], AQP4 water channel, Kir4.1 channel, carboanhydrase, Vimentin, pyruvate carboxylase, α-crystallin, the small GTP-binding protein RhoB, glutamate carboxypeptidase II, carbonic anhydrase C, and microtubule-associated protein 4 [15, 16], which are the foundation of the distinctive morphology and function of Müller glia.
Müller glia not only act to maintain the integrity of the retinal structure, but they are essential for retinal homeostasis and physiological functions. Müller glia regulate the excitability of neurons, protecting neurons from excitotoxicity by taking in and recycling neurotransmitters like GABA and glutamine [19, 22, 25]. Müller glia participate in glucose metabolism to provide neurons with nutrition like pyruvate and transport metabolic waste, including CO2. Müller glia also help to maintain water-ion balance and participate in photopigment recycling, anti-oxidation, and hemodynamics. Interestingly, Müller glia play an important role in the retinal immune response by engulfing dead cell debris, releasing inflammatory mediators, and regulating microglia behaviors(Fig. 1) [15, 16, 26,27,28].
The morphology and functions of Müller glia in the retina. a Müller glia maintain the ion and water homeostasis of the retina [15, 16]. b Müller glia participate in glucose metabolism and provide nutrients for retinal neurons [15, 16]. c Müller glia are involved in phagocytosis of cell debris, secreting cytokines, and regulating the behavior of microglia [30, 200, 201]. d Müller glia modulate neuronal excitability by taking in glutamate and synthesizing glutamine [19, 23]. e Müller glia are involved in anti-oxidation and recycling of photopigments [15, 24]
Müller Glia Respond Differently to Injury in Different Species
As mentioned above, Müller glia undergo a series of changes in response to damage, but these changes are different in each species. Here, we focus on three classic animal models in the field of retinal regeneration: zebrafish, chicks, and murine [9, 10, 29] .
First, how do Müller glia receive injury signals? The first step in the Müller glia reaction is sensing the injury signal. In both zebrafish and mammals, Müller glia are actively involved in phagocytosis and contribute to the clearance of dead cells debris via the phosphatidylserine-rac1 pathway [30,31,32,33]. In addition to phagocytosis, Müller glia can sense injury signals by monitoring the level of neurotransmitters, like GABAA, produced by interneurons in the microenvironment, and thus start the regenerative program in species such as zebrafish [34]. Inflammatory signals and immune cells are also necessary for Müller glia proliferation in zebrafish and chicks [35,36,37,38]. However, most research into how Müller glia receive injury signals has focused on cytokines; growth factors such as heparin-binding epidermal-like growth factor (HB-EGF) [39, 40], adenosine diphosphate (ADP) [41, 42], tumor necrosis factor (TNF-α) [43,44,45], transforming growth factor-β (TGF-β) [46,47,48], Wnt [49,50,51], insulin, insulin-like growth factor 1 (IGF-1), interleukin 11 (IL-11) [50], leptin, and IL-6 family members, like ciliary neurotrophic factor (CNTF), which has been approved by the Food and Drug Administration (FDA) for the treatment of retinal degenerative diseases [52,53,54]. We will discuss in detail how these molecules perform signal transduction and initiate Müller glia reprogramming later.
How do Müller glia react after receiving injury signals? In zebrafish, when the retina endures injury as a result of such things as mechanical damage [39, 55, 56], chemical damage [42, 45, 57], or light-induced damage [31, 45, 58,59,60,61], Müller glia will de-differentiate from 0 to 2 days post injury (dpi) then go through asymmetric division to produce two daughters: one keeps glial properties, and another becomes a Müller glia-derived progenitor cell (MGPC) that can format a neurogenic cluster. After 2 dpi, MGPCs will quickly proliferate to produce more progenitors [59, 62, 63]. MGPCs migrate to different layers through interkinetic nuclear migration (INM) mediated by the actin-cytoskeleton and Rho-associated coiled-coil kinase (rock) [64]. N-cadherin is essential for the formation of the neurogenic cluster and migration of MGPCs [62]. The progenitors usually start to differentiate at 7 dpi to replenish lost neurons [56, 65] and help the retina fully recover [66, 67]. However, in the postnatal chicken retina, progenitors derived from Müller glia have restricted ability to differentiate into retinal neurons. Although Müller glia can re-enter the cell cycle and express progenitor markers, including paired box 6 (PAX6) and CHX10, and the basic helix-loop-helix transcription factor, chicken achaete schute homologue1 (CASH1), only a small percentage of progenitors can differentiate into specific neurons, such as amacrine and bipolar cells, but they cannot differentiate into photoreceptors [11, 12]. However, this spontaneous regeneration ability seems to be limited in mammals and in an age-dependent manner [68]. The regeneration of Müller glia in lower vertebrates may offer some clues for Müller glia reprogramming in mammals [26].
Like astrocytes in the brain, Müller glia demonstrate reactive gliosis upon the onset of injury. Reactive Müller glia usually have a hypertrophic morphology and the expressions of intermediate filament genes, such as glial fibrillary acidic protein (GFAP), nestin, and Vimentin are upregulated. At the initial stage of retinal injury, Müller glia can re-enter the cell cycle, de-differentiate into neuronal progenitor cells that are capable of regenerating retinal neurons (only limited in lower vertebrates), secrete neuroprotective factors, and remove degenerated cells by phagocytosis. Müller glia can downregulate the tumor suppressor protein, p27Kip1 and then re-enter the cell cycle in mice [69] and can express other neurons markers, like rhodopsin and protein kinase C (PKC), in rats [70], but they cannot spontaneously repair damaged retina as retinal stem cells [71]. In mammals, Müller glia mainly form glial scars, which further aggravates damage at the late stage of retinal injury. Based on the above phenomena, there are several issues that are worth considering. What caused the difference in regeneration ability of Müller glia? What is the significance of Müller glia similarities between different species and even different strains [72]? How can we learn from a successful regeneration model like zebrafish? How should we implement Müller glia reprogramming in mammals to regenerate retina? Fortunately, many excellent studies have recently provided useful information.
Mechanisms of Müller Glia Reprogramming in Zebrafish and Chicks
Important Signaling Transduction Pathway
ASCL1/LIN28/Let-7 Axis
In zebrafish, this is the core signaling pathway in the Müller glia reprogramming study. Achaete-scute complex-like 1a (ASCL1a), a proneural basic helix-loop-helix transcription factor, participates in cell cycle exit and differentiation of retinal neuron subtypes during retinal development. In the retinas of zebrafish, ASCL1a contributes to Müller glia reprogramming and acquisition of stem cell properties following injury. ASCL1a is rapidly upregulated within 4 h post injury (hpi), and reaches peak levels at 4 dpi [65, 73]. Fast induction of ASCL1a can regulate a lot of regeneration-associated genes (RAGs) including signal transducer and activator of transcription 3 (STAT3) [74], α1-tubulin [55, 65], Wnt signaling [75], Notch signaling [39], Hedgehog (Hh) signaling [76], forkhead box N4 (FOXN4), zic family member 2 (ZIC2) [76], insm1a [77], myelocytomatosis oncogene (MYC) [78], octamer-binding transcription factor 4 (OCT4) [79], RNA-binding protein LIN28 [65], and Apobec2b [80], which forms a signaling regulatory network and activates Müller glia from a quiescent state to multipotent progenitor state. ASCL1a can promote the expression of, LIN28, which is used as a critical factor to induce pluripotent stem cells [81], by directly binding to the promotor of LIN28. LIN28 suppresses the expression of microRNA Let-7 to release the repression of a variety of pluripotency genes including ASCL1a and LIN28 as positive feedback [65, 76].
Müller glia can also sense injury via growth factors and cytokines. The binding of these factors as ligands to their receptors activates downstream signal transduction cascades including Notch, Wnt/β-catenin, mitogen-activated protein kinase (MAPK), janus kinase (JAK)/STAT, Phosphatidylinositol 3-kinase (PI3K)/AKT, TGF-β, and Sonic Hedgehog (Shh) signaling pathways in autocrine and paracrine fashion to promote Müller glia-mediated retinal regeneration.
Notch Signaling Pathway
In zebrafish, Notch signaling negatively regulates Müller glia reprogramming. Notch signaling acts to maintain the Müller glia quiescent state in intact retina [44], restricting the proliferation of Müller glia to the injury site to avoid excessive proliferation and regulating progenitor differentiation at the late stage following injury [39, 78, 82, 83]. In developing and adult retinas, Notch signaling is activated in retinal stem cells [84], regulating expression of progenitor and glial related genes [85], promoting Müller gliogenesis [86, 87], and maintaining glial fate [88].
Suppressing the activation of Notch signaling induces the proliferation of Müller glia in the intact retina; however, these Müller glia fail to differentiate into retinal neurons. Conner et al. (2014) reported that repression of Notch signaling along with TNFα treatment was sufficient to imitate the regeneration response in uninjured retina [44]. However, Wan et al. (2017) [83] and Elsaeidi et al. (2018) [82] demonstrated that repression of Notch signaling did not stimulate Müller glia proliferation, but worked with ASCL1a and LIN28 to stimulate Müller glia reprogramming without injury. After the injury, Notch signaling is activated quickly and is mainly expressed on de-differentiated MGPCs, whereas the Notch ligand is expressed in the adjacent regions of MGPCs [39, 60, 84]. Suppression of Notch signaling following injury increases the number of MGPCs, but the proportion of MGPCs that differentiate into neurons is reduced, and vice versa [39, 89].
Notch signaling can be regulated by ASCL1a [39], OCT4 [79], and MYC [78], to suppress the expression of ASCL1a, HB-EGF [39], LIN28 [78, 89], JAK/STAT signaling [44] and stimulate the expression of insm1a [77] to drive a quiescence program of Müller glia in the uninjured retina and restrict proliferation in the injured retina. Notably, fibroblast growth factor 8a (FGF8a) plays an opposing role on Notch signaling to control injury-induced retinal regeneration. FGF8a can repress Notch signaling to enhance the regenerative response in the young retina, but induce Notch signaling to repress the regenerative response in mature retina [83]. Notch signaling is more like a monitor, compared to RAGs, for controlling the injury-induced regenerative response. However, Notch signaling has a dual role in that it can promote Müller glia to de-differentiate and proliferate, but impede differentiation into neurons in chicks [90]. Notch signaling is downstream of FGF/MAPK and inhibition of Notch signaling suppresses Müller glia proliferation [91], although inhibition of Notch signaling and gp130/JAK/STAT synergistically promotes MGPCs to differentiate into neurons [92]. It appears contradictory that MAPK signaling promotes the proliferation and transdifferentiation of Müller glia [93], but activates Notch signaling that impedes differentiation of MGPCs into neurons. Further study is required to reveal their relationship and role in Müller glia reprogramming.
Wnt/β-Catenin Signaling Pathway
In zebrafish, Wnt/β-catenin signaling is necessary and sufficient for the induction of Müller glia-mediated retinal regeneration. Wnt/β-catenin signaling can induce the expression of ASCL1, thus repressing the expression of dickkopf (dkk) [94], a Wnt inhibitor, and inducing activation of Wnt signaling within 6 hpi, which causes β-catenin to accumulate in proliferative MGPCs [39]. Inhibition of Wnt signaling can repress injury-dependent MGPC proliferation and activation of Wnt signaling via glycogen synthase kinase-3β (GSK-3β) inhibition without retinal injury induces a regeneration response like what occurs in the injured retina [49, 75, 95].
In the chick retina, β-catenin starts to accumulate in the nucleus of Müller glia at 4 hpi and reaches peak levels at 5 dpi following N-methyl-D-aspartic acid (NMDA) treatment. Inhibition of Wnt signaling suppresses injury-dependent formation of MGPCs. Compared with zebrafish, activation of Wnt signaling promotes injury-dependent Müller glia proliferation in chicks, but is not sufficient to induce Müller glia proliferation without injury [96]. Wnt/β-catenin can also be activated by FGF2/MAPK [93] signaling to stimulate the formation of MGPCs without injury [96]. However, Wnt/β-catenin is required, but not sufficient for Müller glia reprogramming in chicks.
JAK/STAT Signaling Pathway
STAT3 is a critical molecule in Müller glia reprogramming, which has been studied for an extensive period of time. In zebrafish, the expression of STAT3 increases significantly in all Müller glia and is required for Müller glia reprogramming [61, 74]. Knockdown of STAT3 suppresses the expression of RAGs, the proliferation of MGPCs [53] and Notch repression-independent promotion of Müller glia proliferation [44]. The expression of STAT3 requires EGFR/MAPK/PI3K signaling [50]. IL-6 family cytokines, like CNTF, can induce MGPC proliferation by activating JAK/STAT signaling via the gp130-coupled receptor [53, 61]. Other growth factors, like HB-EGF/insulin and IGF-1/FGF-2, also stimulate STAT3 expression [50] without injury.
In chicks, JAK/STAT signaling is upregulated and phosphorylated STAT3 (pSTAT3) accumulates in Müller glia following NMDA-induced injury. Insulin treatment or FGF/MAPK induces accumulation of pSTAT3 in the nuclei of MGPCs. Repressing the activity of JAK or STAT3 phosphorylation reduces Müller glia proliferation. Just like in zebrafish, binding of CNTF to gp130 stimulates the accumulation of pSTAT3, which promotes Müller glia proliferation without injury [92].
MAPK Signaling Pathway
In zebrafish, MAPK signaling is upregulated and necessary for Müller glia reprogramming following injury [97]. CNTF activates the MAPK signaling pathway, which mediates the role of neuroprotection in light-induced photoreceptor death [61]. HB-EGF activates MAPK signaling via EGFR in Müller glia and HB-EGF/EGFR/MAPK signaling regulates ASCL1a and PAX6, which initiates the regenerative response after mechanical injury [39]. Repressing EGFR or MAPK signaling decreases injury-dependent MGPC proliferation [39, 50].
In chicks, MAPK signaling is also quickly activated following NMDA-induced retinal damage to induce pERK1/2, c-fos, pCREB, and early growth response-1 (Egr-1) accumulation in Müller glia, which is required for Müller glia to de-differentiate and proliferate. FGF2 can selectively activate MAPK signaling components in Müller glia, which protects retinal neurons from neurotoxicity and stimulates the formation and proliferation of MGPCs [91, 93]. Inhibitors of MEK and FGF receptors repress the proliferation of MGPCs [98]. Activation of Glucocorticoid receptor (GCR) signaling represses MGPCs proliferation via repression of MAPK signaling [36]. MAPK signaling induces Hh signaling in the uninjured retina. FGF/MAPK signaling is also a part of the Hh signaling network and promotes the formation and proliferation of MGPCs [99].
TGF-β Signaling Pathway
In zebrafish, the regulation of TGF-β signaling is necessary for Müller glia-induced retinal regeneration, especially for the proliferation of MGPCs [48, 100]. TGF-β signaling components, like Smad2/3, are induced initially but are rapidly inhibited after acute light exposure. TGF-β signaling is a negative regulator for retinal regeneration. Pharmacological inhibition of TGF-β signaling enhances the retinal regeneration response [101]. Co-repressors, Tgif1 and Six3b, are necessary for inhibiting TGF-β signaling pathways [100]. However, Sharma et al. (2020) found that TGF-β signaling has a biphasic role during Müller glia reprogramming [48, 102]. TGF-β signaling upregulates genes, including RAGs (ASCL1a, LIN28, OCT4, SRY-box transcription factor 2 (SOX2), histone deacetylase 1 (HDAC1)), Snail family genes that inhibit cell reprogramming [79], and Zeb family genes that inhibit the proliferation of MGPCs [79]. TGF-β signaling can also repress the expression of Her4.1 (a Notch signaling component) [89], miRNA-200a/miRNA-200b, and miRNA-134/miRNA-145 by binding the 5GC element and TGF-β inhibitory element (TIE) located in the target genes during the process of Müller glia reprogramming. TGF-β signaling also depends on the activity of matrix metalloprotein (MMP) 2/MMP9 to regulate retinal regeneration [48]. In summary, TGF-β signaling is necessary for the expression of RAGs, the proliferation of MGPCs and the cell cycle exit in the later phase of regeneration.
In chicks, TGF-β/Smad2/3 and bone morphogenetic protein (BMP)/Smad1/5/8 play opposite roles in controlling Müller glia reprogramming. TGF-β/Smad2/3 signaling is repressed, but BMP/Smad1/5/8 signaling is transiently increased following NMDA-induced injury. TGF-β signaling represses the formation of proliferative MGPCs and the expressions of PAX6, Kruppel-like factor 4 (KLF4), and Egr-1. In retinas where FGF/MAPK signaling stimulates the formation of MGPCs, inhibition of Smad3 causes an increase of proliferative MGPCs, while inhibition of the BMP/Smad1/5/8 signal represses the formation of proliferative MGPCs [103]. Therefore, TGF-β/Smad2/3 signaling negatively regulates Müller glia reprogramming and BMP/Smad1/5/8 signaling positively regulates Müller glia reprogramming.
Hedgehog Signaling Pathway
In zebrafish, Shh signaling is important for retinal regeneration following injury. After mechanical, chemical, or light-induced damage, Shh signaling components, including Shha, Shhb, Ptch1/2, Smo, and Gli1/2, are rapidly upregulated in MGPCs [76, 104]. Pharmacologic or genetic inhibition of Shh signaling represses the formation, proliferation, and differentiation of MGPCs. Knockdown of Sufu and Gli3, two negative regulators of Shh signaling, promotes the formation of MGPCs [76, 105, 106]. MicroRNA Let-7 directly represses Shh signaling by binding to the promotors of Shha/b, Ptch, Smo, and Gli1. Shh signaling upregulates the expression of ASCL1a and LIN28 therefore, Shh signaling is recruited into the ASCL1a/LIN28/Let-7 axis to regulate multiple RAGs. In addition, there is a feedback between Shh signaling and MMP9. Shh signaling can directly induce the expression of ZIC2b and FOXN4, both of which promote the formation and proliferation of MGPCs [76]. In addition, the SCL/TAL1 interrupting Locus (Stil) causes an upregulation of Shh signaling by repressing the Shh signaling repressor Sufu, which is required for MGPCs proliferation [106]. In summary, injury-dependent Shh signaling, which is regulated by Let-7 and Stil, induces a complicated regulatory network involving ASCL1a/LIN28, ZIC2b, MMP9, and FOXN4, which is essential for Müller glia reprogramming.
In chicks, Hh signaling is a key component of the signaling regulatory network that promotes the formation and proliferation of MGPCs. Hh signaling is upregulated and reaches peak levels at 2 dpi when Müller glia enter the cell cycle after NMDA-induced injury. Repression of Shh, Smo, or Gli1/2 suppresses the number of MGPCs. Crosstalk occurs between Hh signaling and MAPK signaling, as well as the mammalian target of rapamycin (mTOR) signaling, which promotes the formation and proliferation of MGPCs [99].
Pluripotent factors
Pluripotent transcriptional factors like PAX6, MYC, SOX2, OCT4, and insm1a also play essential roles in Müller glia reprogramming in zebrafish. Insm1a is a crucial repressor that has a biphasic expression pattern during retinal regeneration in zebrafish. During the early stage of regeneration, insm1a activates Wnt signaling to induce the proliferation and proliferation of MGPCs. During MGPCs proliferation, insm1a suppresses HB-EGF to sculpt the area during the regeneration response. In the late state, insm1a promotes cell cycle exit by downregulating the expression of bcl11, which represses p57kip2. Although ASCL1a might positively upregulate insm1a via dkk suppression, insm1a represses the expression of ASCL1a [77]. PAX6a and PAX6b are necessary for the proliferation of MGPCs and formation of neurogenic clusters [63]. SOX2 is rapidly upregulated and necessary for the proliferation, formation, and regeneration of cones, but not rods, derived from MGPCs. The expressions of ASCL1a and LIN28 also require SOX2 [107]. OCT4 seems to have a broader regulatory role compared to SOX2. During retinal regeneration following injury, OCT4 directly regulates several RAGs, such as ASCL1a, LIN28a, E-cadherin, SOX2, Zeb, and many microRNAs (miRNA)s, including Let-7a, miRNA-143/miRNA-145, and miRNA-200a/miRNA-200b. The cell cycle exit requires the collaboration between OCT4 and members of the nucleosome remodeling and deacetylase complex (NuRD), such as HDAC1. Notably, OCT4 has a dual role in the regulation of TGF-β at different stages of Müller glia reprogramming [79].MYC is also necessary and upregulated during Müller glia reprogramming. There is a positive feedback between ASCL1a and MYC, but MYC also is repressed by ASCL1a through activation of insm1a. In the adjacent areas of MGPCs, MYC, in collaboration with HDAC1, represses the expressions of LIN28 and Her4.1 to control regenerative response. In summary, MYC is a dual regulator that precisely controls retinal regeneration [78].
The re-entry of Müller glia into the cell cycle is bound to be accompanied by changes in cell cycle-related genes. In zebrafish, Midkine-a (Mdka) is activated following injury to regulate cell cycle progression. Mdka induces the expression of pSTAT3 through the activation of its receptor, anaplastic lymphoma kinase (Alk), which is required for the proliferation of MGPCs. In addition, Mdka represses cell cycle inhibitor, p130 via upregulation of inhibitor of DNA-binding 2a (Id2a), which promotes cell division. Inhibition of Mdka causes only a small number of Müller glia to progress through cell division and permanently compromises the regeneration of cones [108, 109].
Epigenetics in Müller Glia Reprogramming
DNA methylation that occurs at the CpG island is highly associated with gene silencing and demethylation causes the gene to be open or accessible [110]. During the induction of iPSC, the methylation of pluripotent genes decreases [111, 112]. In zebrafish, the methylation pattern of Müller glia is changed during retinal regeneration. Methylation decreases at the early stage, but increases at the late stage, suggesting that genes become more accessible when Müller glia de-differentiate, but become more closed when MGPCs start to differentiate into retinal neurons [113]. The promoters of pluripotent genes, such as ASCL1a, LIN28, HB-EGF, insm1a, SOX2, and OCT4, maintain hypomethylation [113], which may indicate that Müller glia are ready for de-differentiation. Although Apobec2a/2b is involved in DNA demethylation, it is independent of DNA methylation in retinal regeneration in zebrafish [80, 113, 114].
Histone modification is an essential part of epigenetics and participates in the regulation of genes transcription in many ways including methylation, acetylation, phosphorylation, adenylation, and ubiquitination of histone [115,116,117]. The role of histone modification in Müller glia reprogramming was recently observed. In zebrafish, HDAC1 is quickly downregulated after injury to release the inhibition on RAGs, including ASCL1a, LIN28, and MYC [89]. Simultaneously, HDAC1 represses the expression of Her4.1, a repressor of Müller glia-induced retinal regeneration. Pharmacologic or genetic inhibition of HDAC1 reversibly represses the formation and proliferation of MGPCs [89]. Therefore, HDAC1 is a key regulator of retinal regeneration.
miRNA is a type of conserved non-coding RNA that regulates gene silencing after transcription through targeting the 3′ untranslated region (UTR) of mRNA, which is essential for nearly all kinds of biological processes [118, 119]. Recently, the role of miRNA in Müller glia reprogramming has been gradually revealed. Knockdown of DICER, which is necessary for the processing of miRNA, causes a significant reduction in MGPCs. Direct repression of miRNA-142b/miRNA-146a/miRNA-7a/miRNA-27c/miRNA-32 expression results in similar outcomes [120]. An extremely critical miRNA, Let-7, is a repressor for retinal generation that is negatively regulated by LIN28 both in zebrafish and rats and mediates inclusive gene suppression as mentioned above [65, 76, 121]. In zebrafish, downregulation of miRNA-216a activates Wnt/β-catenin through H3K79 methyltransferase Dot1l, which promotes Müller glia-mediated retinal regeneration [95].
Müller glia reprogramming involves changes in epigenetics, including DNA methylation, histone modification and miRNA mediated degradation of mRNA. However, this is still a new field with great research value since we know little about epigenetic mechanisms, such as phosphorylation, methylation, ubiquitination of histone, chromatin remodeling, and other non-coding RNAs. Exploration of how these mechanisms apply to Müller glia reprogramming in mammals is needed.
Influences of Microenvironment on Müller Glia Reprogramming
Microglia are recognized as the resident immune cells in the central nervous system (CNS). The immune response is crucial for maintaining the health of the retina and protecting it from disease. Microglia participate in synaptic pruning, phagocytosis of cell debris, monitoring the retinal environment, and releasing inflammatory and anti-inflammatory factors to maintain retinal homeostasis and physiological function. When the retina is injured, microglia are one of the earliest cells to respond and migrate to the injured area to perform phagocytosis, present antigens, and secrete inflammatory factors. Though the inflammatory reaction mediated by microglia in the retinal microenvironment during injury or degeneration may aggravate retinal injury, microglia also perform beneficial functions, including removal of apoptotic cells and cell debris. Quiescent microglia have ramified morphology and active microglia have amoeboid morphology [15, 122]. Activation of microglia plays an important role in initiating Müller glia reprogramming (Fig. 2).
Immune regulation of Müller glia reprogramming. a In zebrafish, microglia promote Müller glia reprogramming but excessive inflammatory response mediated by mmp9 dysfunction impedes normal cone regeneration [37, 38, 96, 122, 123]. b In chicks, microglia secrete multiple factors to promote Müller glia reprogramming, but sustained NF-κB signaling mediated microglia causes a decrease in MGPCs [35, 36, 125, 126]. c In mammals, microglia may affect the process of Müller glia reprogramming but how M1 and M2 microglia affect Müller glia reprogramming is unknown [200,201,202]
In zebrafish larva, macrophages and microglia are activated following injury. They transform their morphology from ramified to amoeboid, migrate to the outer nuclear layer (ONL) and outer plexiform layer (OPL), phagocytose apoptotic rod cell debris and proliferate. Inhibition of microglia/macrophages before injury reversibly represses the formation and proliferation of MGPCs and causes a delay in retinal regeneration [123]. In adult zebrafish, immune cells undergo similar changes following injury [124]. Microglia and infiltrating immune cells can respond to retinal injury by migration, phagocytosis, and release inflammatory factors that may act on Müller glia to initiate their reprogramming and affect the proliferation and differentiation of MGPCs. Ablation of all immune cells or pharmacologic immune suppressants reversibly repress the regeneration of the injured retina and reduce Müller glia reactivity; however, the selective ablation of macrophages does not affect Müller glia-induced retinal regeneration [37, 38, 97]. The regulation of the immune response on Müller glia reprogramming is involved in MMP9 and the mTOR signaling. MMP9 is upregulated, which limits the inflammatory response to avoid excessive activation and protects the newly generated cones [38].The macrophage/microglia-mediated inflammatory response activates mTOR signaling via PI3K and MAPK signaling, which plays a crucial role in the de-differentiation, proliferation and differentiation of Müller glia into retinal neurons(Fig. 2) [38].
In chicks, activation of macrophages/microglia is necessary for the formation and proliferation of MGPCs. In the retina where macrophages/microglia are ablated, the formation of MGPCs is diminished and the expressions of TNF, IL1β, ASCL1a, complement component 3 (C3) and C3a receptor are decreased significantly. The ablation of macrophages and microglia also blocks the induction of MGPCs in the retina where a combination of insulin and FGF2 induces Müller glia reprogramming without injury [35, 125]. Activation of GCR located in the nuclei of Müller glia, and usually associated with anti-inflammatory response, has a neuroprotective role and represses the formation and proliferation of MGPCs and inhibition of GCR stimulates this process [36, 101]. Therefore, the activation of the immune response contributes to the induction of MGPCs, which might be through inflammatory factors and the complement system. In addition, mTOR signaling is required for the induction of Müller glia reprogramming. mTOR signaling quickly increases in Müller glia following NMDA-induced injury. Inhibition of mTOR signaling with rapamycin represses the activation of microglia and the formation of MGPCs [126]. In addition to mTOR signaling, nuclear factor kappa B (NF-κB) signaling is an essential inflammatory regulator, which represses the reprogramming of Müller glia into MGPCs in a reactive microglia-independent fashion [127]. Further analysis of the contribution of immune cells to retinal regeneration is warranted. Comparing the injury response of immune cells in lower vertebrates with response of mammals may lead to the new discovery of strategies for promoting mammalian retinal regeneration(Fig. 2).
Recent Progression of Müller Glia Reprogramming in Mammal
Cell reprogramming technology has created new opportunities for regenerative medicine. In 2006, Yamanaka and Takahashi induced reprogramming of mouse fibroblasts in vitro to form induced pluripotent stem cells. Based on this, the new second generation in situ cellular reprogramming technology was developed by transfecting lineage restricted transcription factors into somatic cells to transdifferentiate into other specific somatic cells in vivo. This technique omits the in vitro steps and also reduces the risk of tumor formation caused by transplantation of stem cell-derived cells. Cellular reprogramming technology is expected to become a new method for treating retinal degenerative diseases. This technology has been applied to various types of cells in the pancreas, heart, liver, and CNS. In the CNS, astrocytes and NG2 glia have been successfully converted into functional neurons to mitigate damage [128, 129]. A lot of transcription factors and cytokines, such as NeuroD1 [130], NeuroD4 [131], ASCL1 [132], Brn2, Myt1l [133], Neurogenin2, FGF2, EGF [134], Bcl [135], SOX2 [136,137,138], and polypyrimidine tract-binding protein 1 (Ptbp1) [139] are associated with glial cell reprogramming in vivo. Some of these also play important roles in Müller glia reprogramming. Reprogramming in the CNS may provide vital information and a strong reference for Müller glia reprogramming in the retina due to similarities between the two.
Cell Biology Mechanisms
Cell-Cell Fusion-Mediated Müller Glia Reprogramming
Cell-cell fusion is an important mechanism during developmental processes, including muscle and bone development and fertilization [140]. Cell-cell fusion usually occurs after stem cell transplantation to repair damaged tissues. Although cell-cell fusion-mediated reprogramming has a limitation of security and effectiveness, it is still a prospective therapeutic strategy in the regenerative field.
In damaged retinas of mice, cell-cell fusion-mediated Müller glia reprogramming induces the formation of hybrids with retinal progenitor characteristics that can replace the loss of retinal neurons and rescue visual function. Upon activation of Wnt/β-catenin signaling, transplanting hematopoietic stem and progenitor cells (HSPCs) into NMDA-treated retinas induces transplanted cells to fuse with Müller glia, amacrine cells, and retinal ganglion cells (RGCs). These hybrids can de-differentiate, proliferate and generate amacrine cells and RGCs. The axons of newly generated RGCs are projected in the optic nerve and newly generated neurons improve the ability to respond to light stimulus [141]. In a photoreceptor degeneration model, transplanted HSPCs mainly fuse with Müller glia. Not amacrine cells or RGCs, inducing hybrids with retinal progenitor characteristics that differentiate into functional photoreceptors. In addition, these hybrids have a protective role of residual retinal neurons [142]. Without introducing exogenous cells, cell-cell mediated Müller glia reprogramming occurs. In rodents, endogenous bone-marrow cells (BMCs) are recruited into NMDA-treated retinas and mainly fuse with Müller glia. The BMCs-Müller glia hybrids act as endogenous stem cells to differentiate into amacrine cells and RGCs to repair damaged retina. This process of recruitment is regulated by the stromal cell-derived factor 1 (SDF1)/C-X-C motif chemokine receptor type 4 (CXCR4) pathway [143].
There is no doubt that cell-cell fusion induced Müller glia reprogramming is an effective and very promising strategy for retinal regeneration. However, the molecular mechanisms behind this are unknown. It is unclear how cell-cell fusion initiates Müller glia reprogramming. Is cell-cell fusion-mediated Müller glia reprogramming associated with critical molecular events like ASCL1, Notch, and Hippo signaling? How do these foreign cells change the retinal microenvironment? Recent studies have shown that metabolic reprogramming may be involved in cell-cell fusion-mediated Müller glia reprogramming.
The Potential Role of Metabolic Reprogramming
Cell metabolism not only produces energy for cell function, but also directly regulates cell division and the epigenome by post-translational modification of DNA, histone and transcription factors. These findings suggest the importance of cellular metabolism for regulating cell fate decisions. Changes in cellular metabolism regulating cell fate decisions is termed “metabolic reprogramming” [144]. Stem cells mainly rely on glycolysis to obtain energy in a pluripotent state. During differentiation, the mitochondrial oxidative phosphorylation metabolic ability gradually increases, and the transformation of energy metabolism from glycolysis to oxidative phosphorylation happens; while during the process of somatic cell reprogramming, the main energy source of somatic cells shifts from oxidative phosphorylation to glycolysis [145, 146]. Mitochondria are major players in cell metabolism and are the critical mechanism affecting stem cell fate decisions. A lot of evidence indicates that mitochondria are central regulators that induce transcription, translation, and post-translational regulation. The reactive oxygen species (ROS) level, products from the tricarboxylic acid (TCA) cycle, and the shape of mitochondria play important roles in stem cell fate [147]. Recent studies have found that mitochondria are involved in the exchange of substances during cell fusion-mediated reprogramming and they are one of the critical mechanisms that mediate the transformation of recipient cells into stem cells. When transplanted photoreceptor precursor cells are integrated into a host retina, a lot of materials including mitochondria are transferred from the donor cells to the host, thus rescuing the terminal function of the degenerated retina [148,149,150]. Injecting induced pluripotent stem cell-derived MSCs (iPSC-MSC) into the vitreous cavity of Ndufs4 KO mice, a mitochondrial complex I deficiency mouse model, results in mitochondrial transfer from iPSC-MSCs to host cells and decreases excessive inflammation and rescues degenerated retinal function caused by RGCs damage [151]. Transcription factor Yin Yang1 (YY1)-mediated metabolic reprogramming by modulating both mitochondrial and glycolytic pathways is necessary for skeletal muscle regeneration [152]. The destruction of stem cell mitochondria abolishes the heterologous cell fusion-mediated cardiomyocyte reprogramming back to a progenitor-like state [153]. Inhibitors of Oxphos and small molecule activators of glycolysis cause a significant increase in reprogramming efficiencies; in contrast, pharmacological inhibition of glycolysis or mitochondrial division greatly decreases reprogramming ability to induce pluripotent stem cells with Yamanaka factors [144, 154,155,156,157]. These findings indicate that metabolic reprogramming also plays a critical role in the induction of Müller glia reprogramming.
Signaling Pathways and Molecules Induce Müller Glia Reprogramming
ASCL1-Mediated Müller Glia Reprogramming
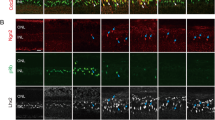
ASCL1, a crucial transcription factor in the Müller glia reprogramming of zebrafish, is highly upregulated after injury. However, ASCL1 fails to upregulate in mammalian Müller glia following injury. Even when combined treatment with NMDA and EGF stimulates an extensive proliferation response and MGPCs differentiate into amacrine cells, ASCL1 is not induced [71]. Overexpression of ASCL1 causes Müller glia to re-enter the cell cycle, express progenitor genes, such as hes5 and dll, and neuronal markers, such as TUJ1 and MAP2, and have morphology of neurons in dissociated P12 mouse Müller glia. In intact retinal explants, a similar phenomenon is observed in that de-differentiated Müller glia express the bipolar marker, PKCα [158]. NMDA treatment after forced expression of ASCL1 in young mice induces Müller glia to acquire neuronal morphology, express retinal neuronal markers, such as HuC/D, PKC, orthodenticle homeobox 2 (OTX2), and recover and integrate into the circuit. Although ASCL1 can reprogram Müller glia into neurons in vitro and in young mice in vivo, forced expression of ASCL1 alone or with NMDA/light-induced injury fails to induce Müller glia to reprogram in the retinas of adult mice. Although reprogramed Müller glia can express OTX2, a photoreceptor/bipolar marker, they cannot further differentiate into functional neurons [159], indicating that as mice age, the regeneration potential of Müller glia gradually decreases [68]. This may be because chromatin accessibility and the progenitor-associated genes are more open in the Müller glia of young mice, as shown by DNaseI-seq [159]. Epigenetic modifications may be an effective way to overcome the limitation of chromatin accessibility. Surprisingly, overexpression of ASCL1, together with the HDAC inhibitor TSA, induces Müller glia reprogramming and generates functional interneurons, such as amacrine and bipolar cells, in adult mice after retinal injury. This represents major scientific progress in the field of Müller glia reprogramming, though only a small portion of Müller glia seem to directly experience transdifferentiation, not proliferation, and only generate interneurons and not photoreceptors or ganglions [13]. The cause for the inefficiency of Müller glia reprogramming is not clear. STAT is one candidate transcription factor. Single-cell RNA-seq shows that progenitor-like cells, derived from Müller glia following treatment with ANT treatment (ASCL1+NMDA+a histone deacetylase inhibitor TSA) have a high level of STAT signaling [159] and gp130 signaling, which can activate JAK/STAT signaling, inhibits the neurogenic potential of MGPCs in chicks [92]. Jorstad et al. (2020) [160] revealed that the STAT signaling inhibits the capacity of ASCL1 to reprogram Müller glia in mammals. Activation of STAT signaling directs ASCL1 to unsuitable targets, which limits the reprogrammed Müller glia to differentiate into other retinal neurons. ANT treatment, together with a STAT inhibitor, causes an increase in newly generated retinal neurons from Müller glia compared to ANT treatment alone [159]. In addition to the HDAC inhibitor and STAT inhibitor, ASCL1 can also work with LIN28 to stimulate a conspicuous proliferative response and a small number of proliferative Müller glia express photoreceptor marker CRX, amacrine cell marker AP2, bipolar cell marker CHX10 and RGCs marker RBPMS at the late stage in NMDA-treated mice retinas [82]. These results suggest that ASCL1 is a critical transcription factor for reprogramming Müller glia in mammals (Fig. 3).
Recent progression of Müller glia reprogramming in adult mammals. a The ANT treatment induces Müller glia reprogramming into functional retinal interneurons and STAT inhibitor improves this efficiency [13]. b β-catenin triggers Müller glia to proliferate and the combination of OTX2, CRX, and NRL induces Müller glia to differentiate into functional rod photoreceptors [14]. c Ptbp1 knockdown mediated by CRISPR-CasRx triggers Müller glia to generate functional RGCs [139]. d Transcriptional factors Math5 and Brn3 convert Müller glia into functional RGCs [209]
Wnt Signaling-Induced Müller Glia Reprogramming
In the adult mammalian retina, Wnt/β-catenin signaling triggers neuroprotective response via Müller glia in the mouse model of retinal degeneration [161] and promotes axonal regeneration after optic nerve damage [162]. Wnt signaling is rapidly induced following neurotoxic injury but returns to the base level at round 36hpi [51] and the number of MGPCs is severely limited after injury [70]. Activation Wnt/β-catenin signaling with GSK-3β inhibitor or Wnt3a, a Wnt signaling ligand, results in accumulation of β-catenin in the nucleus, which greatly promotes the number of MGPCs following light-induced injury [163], and vice versa [51]. Interestingly, without injury, activation of Wnt signaling via adenovirus transfection or pharmacological methods stimulates Müller glia to re-enter the cell cycle and proliferate. Further study has demonstrated that the regulation of Wnt signaling on Müller glia is dependent on the LIN28/Let-7 pathway. β-catenin directly stimulates the expression of LIN28 by binding the promotor of LIN28, which represses the Let-7 to relieve the inhibition on Müller glia reprogramming. Both Wnt signaling and LIN28 are sufficient to stimulate Müller glia to re-enter the cell cycle. Unfortunately, only a small portion of proliferative cells express amacrine cell markers and most die by the late stage [51]. Yao et al. (2018) induced Müller glia reprogramming in two steps. First, they activated Wnt/β-catenin signaling to stimulate Müller glia proliferation and then promoted differentiation of the proliferative cells into photoreceptors via the photoreceptor differentiation-associated genes OTX2, CRX, and NRL. Remarkably, they successfully induced Müller glia to differentiate into functional rods, thus rescuing vision in Gnat1(rd17)/Gnat2(cpfl3) mutant mice, a mouse model of congenital blindness [14]. In addition, Wnt and Notch signaling synergistically promotes the formation and proliferation of MGPCs, which increases the number of MGPCs that take on the rod photoreceptor phenotype and respond to light in the S334ter rat [164]. Wnt signaling also protects photoreceptors in a mouse model of retinal degeneration via Müller glia [161] and promotes axonal regeneration after optic nerve injury(Fig. 3) [162].
Ptbp1/miRNA-124/REST Axis
Recently, Ptbp1, a single RNA-binding protein, was successfully used to convert from Müller glia into retinal neurons via high efficiency CRISPR-CasRx. In healthy and NMDA-injured retinas of mature mice, CasRx-mediated Ptbp1 knockdown triggers Müller glia to transdifferentiate into RGCs and amacrine cells. In NMDA-injured retina, the RGCs converted from Müller glia project their axons to optic nerve, the dorsal lateral geniculate nucleus (dLGN) and superior colliculus (SC) in the brain. The RGCs can respond to light and restore visual function in a permanent visual impairment model (Fig. 3). This approach also converts astrocytes into dopaminergic neurons in the striatum and basically eliminates the symptoms of the Parkinson’s disease [139]. This is a very valuable and important study and provides a promising therapeutic strategy for retinal degenerative diseases.
According to previous studies, Ptbp1-mediated conversion of cell identity may involve miRNA-124, miRNA-9/9*, and repressor element1-silencing transcription factor (REST) complex. REST mediates epigenetic remodeling and is an important regulator during the differentiation of pluripotent stem cells into neural progenitor cells and then mature neurons, resulting in repression of expression of neuron-specific genes in non-neuron cells [165,166,167]. REST also blocks the Neurog2 mediated reprogramming of astrocytes into neurons by preventing Neurog2 from binding to the NeuroD4 promotor and REST deletion promotes Neurog2 mediated reprogramming [131]. miRNA-124 is a noted inhibitor of REST and is capable of reprogramming fibroblasts into neurons in vitro. Enhancer of zeste homolog 2 (EZH2) can methylate REST and maintain its stability, while miRNA-9/9*-124 represses the EZH2-REST axis through targeting USP14, an ubiquitin-specific peptidase [168]. Ptbp strongly negatively regulates miRNA-124 and Ptbp blocks miRNA-124 mediated reprogramming into neurons. Further study has revealed that Ptbp regulates the function of miRNA by competitive binding on target mRNA or altering local RNA secondary structure. Relief of Ptbp-mediated blockage of miRNA by downregulation of Ptbp also induces the conversion from fibroblasts into neurons [169].
Consistent with the data above, overexpression of miRNA-124-9-9* induces expression of ASCL1 and markers of neurons, like TUJ1 and MAP2, in cultured Müller glia. miRNA-124-9-9* also potentiates ASCL1-mediated reprogramming of dissociated Müller glia by repressing the REST pathway, including Ctdsp1 and Ptbp1, thus causing an increase in generated retinal neurons from Müller glia [170]. Whether miRNA-124 can induce MG reprogramming in vivo is unknown and whether a regulatory relationship exists between miRNA-124, REST and Ptbp in Müller glia reprogramming needs to be further studied in vivo. In addition, REST is regulated by Wnt/β-catenin during embryonic development and progression of various diseases and REST also affects the Wnt/β-catenin [166]. However, the relationship between REST and Wnt/β-catenin is unknown in Müller glia reprogramming.
Hippo Signaling Blocks Müller Glia Reprogramming
Recently, the role of Hippo signaling in Müller glia reprogramming was revealed. The Hippo signaling pathway is a highly conserved signaling pathway that controls organ size through the regulation of cell proliferation and apoptosis, and also maintains stem cell characteristics and contributes to cancer development. Activation of Hippo signaling causes a series of kinase cascades, which phosphorylates co-activators, the Yes-associated protein (YAP) and the PDZ-binding motif (TAZ). Phosphorylated YAP and TAZ are sequestrated in the cytoplasm and degraded. In the nucleus, YAP/TAZ interacts with TEAD to regulate genes transcription [171, 172].
In the healthy mouse retina, both YAP and TEAD are co-localized in the nucleus of MG, which are upregulated in the degenerated retina [173, 174]. In mice treated with NMDA, Hippo signaling represses the activation of YAP, which may further repress cyclinD1 and, therefore repressing proliferation directly. Repression of Hippo signaling via Last1/Last2 knockout or activation of YAP via YAP5SA, which blocks the phosphorylation of YAP, triggers the proliferation of Müller glia following NMDA treatment. The latter induces the de-differentiation, proliferation, and differentiation of Müller glia into retinal neurons [175]. YAP conditional knockout prevents the induction of cell cycle-related genes and enhances the gliosis response after N-methyl-N-nitrosourea (MNU)-induced photoreceptor death in the mouse retina. Overexpression of YAP promotes Müller glia reprogramming and proliferation. Interestingly, ASCL1 can be induced by YAP and the YAP-EGFR axis [173]. Therefore, YAP promotes Müller glia reprogramming into a progenitor state, but Hippo signaling prevents Müller glia reprogramming by repressing YAP in mice.
Other Signaling Pathways that Affect Müller Glia Reprogramming
In mammals, Shh signaling can induce the proliferation and stem cells characteristics of Müller glia. In the presence of Shh, Shh signaling promotes rat Müller glia to proliferate, de-differentiate and express rhodopsin, a rod photoreceptor marker in vitro, and also in MNU-induced retinal degeneration in vivo. The inhibitor of Shh signaling, cyclopamine blocks this phenomenon [176]. The Shh signaling agonist, purmorphamine, induces Müller glia to proliferate, express progenitor-specific markers, and directionally transdifferentiate into rod-like photoreceptors [177]. Stimulation of Hh signaling on Müller glia is also regulated by other signals, such as Notch signaling [178] and multi-subunit infraglabellar transport protein complex IFT20 [179].
In mammals, Müller glia reprogramming also involves Notch signaling. Inhibition of Notch signaling induces the expression of ASCL1 in cultured Müller glia [68]. Activation of Notch signaling with jag1, a Notch ligand, alone or together with activation of Wnt signaling by Wnt3a stimulates Müller glia to acquire stem cell properties, and differentiate into the rod lineage. The Notch/Skp2/p27kip1 axis is involved in the activation of Müller glia in rats. Notch signaling directly represses the p27kip1 transcript, a cyclin-dependent kinase inhibitor, and upregulates the expression of Skp2, which further inhibits p27kip1 proteins, which promotes Müller glia proliferation [164, 180]. In the NaIO3-injuried retina, Notch signaling and p27kip1 are transiently downregulated, which might promote Müller glia to proliferate and differentiate into neurons and also repress gliosis [181]. Repression of Notch signaling without injury promotes the ASCL1 and LIN28-induced regenerative response in zebrafish, but not in mice [82].
In mammals, IL-6 family cytokines, such as CNTF and leukemia inhibitory factor (LIF), can bind the gp130 receptor to activate JAK/STAT3 signaling in Müller glia [182, 183], which is required for Müller glia to initiate the astrogliosis-like reaction and induction of neuroprotection in an optic nerve damage and retinal degeneration model [54, 184]. HB-EGF also activates JAK/STAT signaling in mice [40]. Jorstad et al (2020) [160] further revealed the role of JAK/STAT in mammalian Müller glia reprogramming.
TGF-β signaling also plays a major role to promote Müller glia reprogramming. TGF-β signaling represses the proliferation of Müller glia. Repression of TGF-β signaling with TGFβRII-Fc fusion protein restores the activation of Müller glia. TGF-β signaling stimulates the expression of p27kip1, which represses the proliferation of Müller glia and repression of TGF-β signaling can also suppress p27kip1, which promotes Müller glia proliferation in the P10 rat retina [185]. In 3-week-old juvenile mice, activation of TGF-β signaling via Smad7 deficiency stimulates Müller glia proliferation [47], however the regulation of Müller glia proliferation mediated by TGF-β signaling seems to disappear in adult mice that are 6 weeks of age [46]. This suggests that TGF-β signaling plays different regulatory roles during different development stages. In dissociated human Müller glial cell lines [186], TGF-β1 downregulates canonical Wnt signaling and represses differentiation into photoreceptors in cultured Müller cells [187]. BMP/Smad1/5/8 signaling also participates in the signaling regulatory network of EGF treatment-induced Müller glia proliferation [188].
SOX2 May Promote Müller Glia Reprogramming in Mammals
It has been proven that SOX2 is a critical transcriptional factor in Müller glia reprogramming in zebrafish [107], however its role in Müller glia reprogramming in mammals is not clear. In the retinas of mice, SOX2 is expressed in all Müller glia and maintains the quiescent state of Müller glia to prevent Müller glia from abnormal division [189, 190]. SOX2 is a critical factor that is used to induce reprogramming in vivo. SOX2 is sufficient for reprogramming astrocytes into neuroblasts in the adult brain and spinal cord. These neuroblasts can further differentiate into mature neurons and integrate into local neural circuits. It is worth mentioning that this in vivo reprogramming passes through ASCL1+ proliferative progenitors before differentiating into mature neurons [136,137,138]. Therefore, whether the overexpression or inhibition of SOX2 in Müller glia affects reprogramming is worth exploring.
Factors Influencing Müller Glia Reprogramming
Epigenetic Modification
In addition to miRNA-124, REST, and HDAC inhibition, other epigenetic modifications are also used to induce Müller glia reprogramming in mammals. The promoters of pluripotent genes, such as ASCL1a, LIN28, HB-EGF, insm1a, SOX2, and OCT4, maintain hypomethylated states as in zebrafish, indicating that Müller glia are potential endogenous stem cells [113]. However, other research has demonstrated that methylation of OCT4 initially decreases then returns to a fully methylated state, which is consistent with the expression pattern of OCT4, DNA methyltransferase Dnmt3b and Gadd proteins that associated with DNA demethylation. This analysis of DNA methylation after injury reveals that DNA methylation blocks the transcription of pluripotent genes, such as OCT4, which may be an important reason why Müller glia cannot regenerate damaged retina in mammals [191]. DNA demethylation mediated by Apobec protein, a member of AID/Apobec family of cytidine deaminases, promotes the de-differentiation of Müller glia in mice [192], although Apobec2a/2b is required for retinal regeneration independent of DNA methylation in zebrafish [80, 113, 114]. miRNA-25 has a similar effect on cultured Müller glia, such as miRNA-124, and can combine with inhibition of Let-7 to promote the transdifferentiation from Müller glia into neurons [193]. Single-cell RNA-sequence analysis reveals that miRNA-124, miRNA-25, and Let-7 may regulate many signaling pathways, as well as pluripotent genes such as Wnt/β-catenin, REST, and KLF4 [193]. Inhibition of miRNA-28 upregulates the expression of CRX, a photoreceptor-specific marker, which promotes cultured MGPCs to differentiate into the photoreceptor lineage [194].
The Regulation of the Immune Microenvironment
Microglia play an important role in the regeneration of zebrafish retina and brain and macrophages are essential in limb regeneration of salamanders. In mammals, ramified microglia are essential for modulating neurogenesis in the subventricular zone (SVZ) and the subgranular zone (SGZ) of the adult hippocampus by releasing soluble factors or by phagocytosis [195, 196]. LPS-induced M1 microglia (pro-inflammatory phenotype) negatively regulate the survival of neural stem cells (NSCs) and promote NSCs to differentiate into astrocytes; IL-4 induced M2 microglia (anti-inflammatory) support neurogenesis. However, both M1 and M2 microglia repress NSCs proliferation and accelerate differentiation [197, 198]. Recently, it has been reported that repopulating microglia mediates neuroprotection and neurogenesis via IL-6 trans-signaling to repair brain injury and alleviate cognitive dysfunction [199].
After retinal injury, microglia become activated and migrate to the injury site, indicating that they have a role in retinal regeneration. Under injury, ramified microglia stimulate Müller glia to initiate reprogramming, including downregulating glial genes, like GLAST and Vimentin, as well as secreting trophic and inflammatory factors in vitro [200]. In vivo, microglia secrete inflammatory factors, such as IL-1β and TNF-α, to trigger activation of Müller glia, while Müller glia expressing IL-1R1, Tnfrsfr1a, Tnfsfr12a, and Toll-like receptors (TLRs) make themselves more responsive to microglia. Nerve growth factor (NGF), CNTF, and brain-derived neurotrophic factor (BDNF) derived from microglia indirectly influence photoreceptor survival and apoptosis by regulating production and release of basic fibroblast growth factor (bFGF) and glial cell-derived neurotrophic factor (GDNF) from Müller glia [201, 202]. These data strongly suggest that microglia have an important influence on the reprogramming of Müller glia. How microglia and the immune microenvironment influence Müller glia de-differentiation and transdifferentiation, and how neuroimmunology principles or regenerative neuroimmunology [203] can be utilized to achieve retinal regeneration needs further exploration.
Different Subtypes in Müller Glia Affect the Ability to Reprogram
Although there are many strategies to induce Müller glia reprogramming, the efficiency was not as high as expected, since only a portion of the Müller glia participate in reprogramming and generating retinal neurons. This suggests that there may be different Müller glia cell subtypes with differences in reprogramming capabilities. Similar phenomenon and strong evidence for this have also been observed in zebrafish and chicks. One study defined three populations of Müller glia by the expression of STAT3 and ASCL1a: (1) ASCL1a-nonexpressing STAT3-expressing quiescent Müller glia that were not proliferative; (2) ASCL1a-dependent STAT3-dependent proliferating Müller glia; and (3) ASCL1a-dependent STAT3-independent proliferating Müller glia in zebrafish following light-induced injury [74]. Another study distinguished two types of Müller glia based on the opposite role of FGF8a on Notch signaling [83]. In chicks, Müller glia have different regenerative capacities in the central and peripheral retina in response to damage. Even if regenerative capacities of Müller glia in the same region are heterogeneous, the Müller glia that increase the expression of GFAP do not re-enter the cell cycle and others can proliferate [204]. In mammals, some progenitor-associated transcription factors, like CHX10 and PAX6, are expressed only in a portion of Müller glia cells [205]. The Müller glia in the central and peripheral regions of the primate and human retina have different morphologies, structures, and functions [206, 207]. The proliferative potentials of Müller glia in different sites during the development of the human fetal retina are also different [208]. Therefore, there may be different subtypes of Müller glia in the mammalian retina. High-throughput sequencing may enable us to discover differences between the transcriptomes and epigenomes of subtypes of Müller glia and MGPCs, which can help to define signals that stimulate their multipotency.
Factors Limiting Müller Glia Reprogramming in Mammals
It is widely accepted that non-mammalian animals maintain strong regenerative capacity with their tissues or organs when they are injured, while mammals lose this regenerative capacity gradually in the process of evolution especially when it comes to human beings. There is no doubt what causes this difference between species is a crucial issue to give the mammals regenerative ability, however, no satisfying mechanisms have been clarified so far. We summarize the differences in regenerative mechanisms among species regarding to Müller glia reprogramming, put forward some assumptions and hope these works about Müller glia reprogramming contribute to this question.
Upon damage, the physiological structure of retina gets destroyed, the dying retinal neurons and activating microglia make retinal microenvironment change dramatically. In non-mammalian animals, this “injured microenvironment” activates Müller glia to initiate regenerative program and is beneficial for Müller glia reprogramming [37, 38], when it comes to mammals, this “injured microenvironment” over-activates microglia which suppresses the regenerative potential of Müller glia. This relationship between glia reprogramming and microglia activation also takes place in the brain [198]. A series of signaling pathways have been identified to be involved the reprogramming of Müller cells in the non-mammalian and mammalian [213], including the key event of re-expression of gene ASCL1. After retina of zebrafish receiving damage, ASCL1 upregulated significantly and induces Lin28 to suppress let-7, which otherwise inhibits the proliferation of Müller cell. Suppressing the expression of ASCL1 prevents Müller cells from re-entering cell cycle and regeneration [82]. However, ASCL1 fails to be upregulated after retinal injury in adult mammals [159]. Recently, it demonstrated that overexpression of ASCL1 in Müller cells of degenerative mice of 16 days fail to regenerate the retina [13, 159], which implies that there are factors in the retinal microenvironment of degenerative retina which hinders the reprogramming of Müller cells [109, 210, 211]. It shows that the expression of ASCL1 is regulated by Notch pathway [87]. In zebrafish, inhibition of Notch pathway remarkedly promotes the upregulation of ASCL1 in the retina, upregulation of Notch pathway suppresses the expression of ASCL1 in the Müller cells in mammals, which maintains the glial fate of Müller cells [88].
Besides, there are significant differences in epigenetic characteristics of Müller glia between non-mammalian and mammalian animals, including DNA methylation and histone modification [113]. In zebrafish, HDAC1 modifies plenty of RAGs at different stages which achieves Müller glia reprogramming, while in mammals, inhibition of HDAC and upregulation of ASCL1 expression together converts Müller glia into retinal neurons in NMDA-injured retina [89]. Epigenetics might be the “switch” of Müller glia reprogramming.
Recently, it infers that the loss of regenerative capacity in mammals results from the activator protein 1 (AP-1) complex dependent regeneration-responsive enhancer (RRE) in response to damage. During the evolution and speciation, the function of RRE diminished gradually as the enhancer was repurposed to adapt the pressure of evolution, which may result in the fault of Müller glia reprogramming in mammals including human beings [212]. Recently, it demonstrated that the critical nuclear factor I (NFI) repressed the regeneration of retina; knockout of NFI caused the reprogramming of Müller glia which differentiated into the retinal neurons in mice [213]. It indicates that the key transcriptional factors in the gene regulatory networks of Müller glia reprogramming will be a promising target to regenerate the retina of mammals.
Conclusions and Prospects
Müller glia reprogramming involves multiple molecular mechanisms and requires a complicated and precise regulatory network in zebrafish. Understanding these mechanisms contributes to Müller glia-mediated retinal regeneration and a variety of discoveries have been made utilizing numerous disease models and advanced tools in mammals. There are still many issues that have not been resolved. A clearer understanding of the endogenous and exogenous regulatory mechanisms of Müller glia reprogramming is needed, especially what causes the differences in Müller glia-mediated retinal regeneration between different species. It is not clear whether the methods of inducing Müller glia reprogramming will be effective in human beings and/or cause serious side effects. There is still a long way to achieve retinal regeneration using Müller glia reprogramming in human beings. However, there are various advantages to Müller glia reprogramming and it exhibits great potential as a treatment or cure for human retinal diseases that result in loss of retinal neurons and reactive gliosis. We believe that this will bring the treatment to a large number of patients in the future.
References
VEGF Inhibitors for AMD and diabetic macular edema (2015). Jama 314 (20):2184–2185
Ambati J, Fowler Benjamin J (2012) Mechanisms of age-related macular degeneration. Neuron 75(1):26–39
Roska B, Sahel J-A (2018) Restoring vision. Nature 557(7705):359–367
Weinreb RN, Aung T, Medeiros FA (2014) The pathophysiology and treatment of glaucoma: a review. Jama 311(18):1901–1911
Mead B, Berry M, Logan A, Scott RAH, Leadbeater W, Scheven BA (2015) Stem cell treatment of degenerative eye disease. Stem Cell Res 14(3):243–257
Gasparini SJ, Llonch S, Borsch O, Ader M (2019) Transplantation of photoreceptors into the degenerative retina: current state and future perspectives. Prog Retin Eye Res 69:1–37
Rakoczy EP (2017) Gene therapy for the long term treatment of wet AMD. Lancet 390(10089):6–7
Luo YH, da Cruz L (2016) The Argus(®) II retinal prosthesis system. Prog Retin Eye Res 50:89–107
Goldman D (2014) Müller glial cell reprogramming and retina regeneration. Nat Rev Neurosci 15(7):431–442
Lenkowski JR, Raymond PA (2014) Müller glia: stem cells for generation and regeneration of retinal neurons in teleost fish. Prog Retin Eye Res 40:94–123
Fischer AJ, Reh TA (2001) Muller glia are a potential source of neural regeneration in the postnatal chicken retina. Nat Neurosci 4(3):247–252
Fischer AJ (2005) Neural regeneration in the chick retina. Prog Retin Eye Res 24(2):161–182
Jorstad NL, Wilken MS, Grimes WN, Wohl SG, VandenBosch LS, Yoshimatsu T, Wong RO, Rieke F et al (2017) Stimulation of functional neuronal regeneration from Müller glia in adult mice. Nature 548(7665):103–107
Yao K, Qiu S, Wang YV, Park SJH, Mohns EJ, Mehta B, Liu X, Chang B et al (2018) Restoration of vision after de novo genesis of rod photoreceptors in mammalian retinas. Nature 560(7719):484–488
Vecino E, Rodriguez FD, Ruzafa N, Pereiro X, Sharma SC (2016) Glia–neuron interactions in the mammalian retina. Prog Retin Eye Res 51:1–40
Bringmann A, Pannicke T, Grosche J, Francke M, Wiedemann P, Skatchkov SN, Osborne NN, Reichenbach A (2006) Müller cells in the healthy and diseased retina. Prog Retin Eye Res 25(4):397–424
Reichenbach A, Schneider H, Leibnitz L, Reichelt W, Schaaf P, Schümann R (1989) The structure of rabbit retinal Müller (glial) cells is adapted to the surrounding retinal layers. Anat Embryol (Berl) 180(1):71–79
Reichenbach A, Siegel A, Rickmann M, Wolff JR, Noone D, Robinson SR (1995) Distribution of Bergmann glial somata and processes: implications for function. J Hirnforsch 36(4):509–517
Malchow RP, Qian HH, Ripps H (1989) gamma-Aminobutyric acid (GABA)-induced currents of skate Muller (glial) cells are mediated by neuronal-like GABAA receptors. Proc Natl Acad Sci U S A 86(11):4326–4330
Lillien L (1995) Changes in retinal cell fate induced by overexpression of EGF receptor. Nature 377(6545):158–162
Barnett NL, Pow DV (2000) Antisense knockdown of GLAST, a glial glutamate transporter, compromises retinal function. Invest Ophthalmol Vis Sci 41(2):585–591
Linser P, Moscona AA (1979) Induction of glutamine synthetase in embryonic neural retina: localization in Müller fibers and dependence on cell interactions. Proc Natl Acad Sci U S A 76(12):6476–6480
Pow DV, Robinson SR (1994) Glutamate in some retinal neurons is derived solely from glia. Neuroscience 60(2):355–366
Das SR, Bhardwaj N, Kjeldbye H, Gouras P (1992) Muller cells of chicken retina synthesize 11-cis-retinol. Biochem J 285 (Pt3)(Pt3):907-913
Kawasaki A, Otori Y, Barnstable CJ (2000) Müller cell protection of rat retinal ganglion cells from glutamate and nitric oxide neurotoxicity. Invest Ophthalmol Vis Sci 41(11):3444–3450
Bringmann A, Iandiev I, Pannicke T, Wurm A, Hollborn M, Wiedemann P, Osborne NN, Reichenbach A (2009) Cellular signaling and factors involved in Müller cell gliosis: neuroprotective and detrimental effects. Prog Retin Eye Res 28(6):423–451
García M, Vecino E (2003) Role of Müller glia in neuroprotection and regeneration in the retina. Histol Histopathol 18(4):1205–1218
de Melo Reis RA, Ventura ALM, Schitine CS, de Mello MCF, de Mello FG (2008) Müller glia as an active compartment modulating nervous activity in the vertebrate retina: neurotransmitters and trophic factors. Neurochem Res 33(8):1466–1474
Wan J, Goldman D (2016) Retina regeneration in zebrafish. Curr Opin Genet Dev 40:41–47
Sakami S, Imanishi Y, Palczewski K (2019) Müller glia phagocytose dead photoreceptor cells in a mouse model of retinal degenerative disease. FASEB J 33(3):3680–3692
Bailey TJ, Fossum SL, Fimbel SM, Montgomery JE, Hyde DR (2010) The inhibitor of phagocytosis, O-phospho-L-serine, suppresses Müller glia proliferation and cone cell regeneration in the light-damaged zebrafish retina. Exp Eye Res 91(5):601–612
Bejarano-Escobar R, Sánchez-Calderón H, Otero-Arenas J, Martín-Partido G, Francisco-Morcillo J (2017) Müller glia and phagocytosis of cell debris in retinal tissue. J Anat 231(4):471–483
Nomura-Komoike K, Saitoh F, Fujieda H (2020) Phosphatidylserine recognition and Rac1 activation are required for Müller glia proliferation, gliosis and phagocytosis after retinal injury. Sci Rep 10(1):1488
Rao MB, Didiano D, Patton JG (2017) Neurotransmitter-regulated regeneration in the zebrafish retina. Stem Cell Reports 8(4):831–842
Fischer AJ, Zelinka C, Gallina D, Scott MA, Todd L (2014) Reactive microglia and macrophage facilitate the formation of Müller glia-derived retinal progenitors. Glia 62(10):1608–1628
Gallina D, Zelinka C, Fischer AJ (2014) Glucocorticoid receptors in the retina, Müller glia and the formation of Müller glia-derived progenitors. Development 141(17):3340–3351
Zhang Z, Hou H, Yu S, Zhou C, Zhang X, Li N, Zhang S, Song K et al (2020) Inflammation-induced mammalian target of rapamycin signaling is essential for retina regeneration. Glia 68(1):111–127
Silva NJ, Nagashima M, Li J, Kakuk-Atkins L, Ashrafzadeh M, Hyde DR, Hitchcock PF (2020) Inflammation and matrix metalloproteinase 9 (Mmp-9) regulate photoreceptor regeneration in adult zebrafish. Glia 68(7):1445–1465
Wan J, Ramachandran R, Goldman D (2012) HB-EGF is necessary and sufficient for Müller glia dedifferentiation and retina regeneration. Dev Cell 22(2):334–347
Todd L, Volkov LI, Zelinka C, Squires N, Fischer AJ (2015) Heparin-binding EGF-like growth factor (HB-EGF) stimulates the proliferation of Müller glia-derived progenitor cells in avian and murine retinas. Mol Cell Neurosci 69:54–64
Medrano MP, Bejarano CA, Battista AG, Venera GD, Bernabeu RO, Faillace MP (2017) Injury-induced purinergic signalling molecules upregulate pluripotency gene expression and mitotic activity of progenitor cells in the zebrafish retina. Purinergic Signal 13(4):443–465
Battista AG, Ricatti MJ, Pafundo DE, Gautier MA, Faillace MP (2009) Extracellular ADP regulates lesion-induced in vivo cell proliferation and death in the zebrafish retina. J Neurochem 111(2):600–613
Iribarne M, Hyde DR, Masai I (2019) TNFα induces Müller glia to transition from non-proliferative gliosis to a regenerative response in mutant zebrafish presenting chronic photoreceptor degeneration. Front Cell Dev Biol 7:296
Conner C, Ackerman KM, Lahne M, Hobgood JS, Hyde DR (2014) Repressing notch signaling and expressing TNFα are sufficient to mimic retinal regeneration by inducing Müller glial proliferation to generate committed progenitor cells. J Neurosci 34(43):14403–14419
Nelson CM, Ackerman KM, O’Hayer P, Bailey TJ, Gorsuch RA, Hyde DR (2013) Tumor necrosis factor-alpha is produced by dying retinal neurons and is required for Muller glia proliferation during zebrafish retinal regeneration. J Neurosci 33(15):6524–6539
Kugler M, Schlecht A, Fuchshofer R, Kleiter I, Aigner L, Tamm ER, Braunger BM (2015) Heterozygous modulation of TGF-β signaling does not influence Müller glia cell reactivity or proliferation following NMDA-induced damage. Histochem Cell Biol 144(5):443–455
Kugler M, Schlecht A, Fuchshofer R, Schmitt SI, Kleiter I, Aigner L, Tamm ER, Braunger BM (2017) SMAD7 deficiency stimulates Müller progenitor cell proliferation during the development of the mammalian retina. Histochem Cell Biol 148(1):21–32
Sharma P, Gupta S, Chaudhary M, Mitra S, Chawla B, Khursheed MA, Saran NK, Ramachandran R (2020) Biphasic role of Tgf-β signaling during Müller glia reprogramming and retinal regeneration in zebrafish. iScience 23 (2):100817
Meyers JR, Hu L, Moses A, Kaboli K, Papandrea A, Raymond PA (2012) β-Catenin/Wnt signaling controls progenitor fate in the developing and regenerating zebrafish retina. Neural Dev 7:30
Wan J, Zhao X-F, Vojtek A, Goldman D (2014) Retinal injury, growth factors, and cytokines converge on β-catenin and pStat3 signaling to stimulate retina regeneration. Cell Rep 9(1):285–297
Yao K, Qiu S, Tian L, Snider William D, Flannery John G, Schaffer David V, Chen B (2016) Wnt regulates proliferation and neurogenic potential of Muller glial cells via a LIN28/Let-7 miRNA-dependent pathway in adult mammalian retinas. Cell Rep 17(1):165–178
Wen R, Tao W, Li Y, Sieving PA (2012) CNTF and retina. Prog Retin Eye Res 31(2):136–151
Zhao XF, Wan J, Powell C, Ramachandran R, Myers MG, Goldman D (2014) Leptin and IL-6 family cytokines synergize to stimulate Müller glia reprogramming and retina regeneration. Cell Rep 9(1):272–284
Rhee KD, Nusinowitz S, Chao K, Yu F, Bok D, Yang X-J (2013) CNTF-mediated protection of photoreceptors requires initial activation of the cytokine receptor gp130 in Müller glial cells. Proc Natl Acad Sci U S A 110(47):E4520–E4529
Senut MC, Gulati-Leekha A, Goldman D (2004) An element in the alpha1-tubulin promoter is necessary for retinal expression during optic nerve regeneration but not after eye injury in the adult zebrafish. J Neurosci 24(35):7663–7673
Fausett BV, Goldman D (2006) A role for alpha1 tubulin-expressing Müller glia in regeneration of the injured zebrafish retina. J Neurosci 26(23):6303–6313
Fimbel SM, Montgomery JE, Burket CT, Hyde DR (2007) Regeneration of inner retinal neurons after intravitreal injection of ouabain in zebrafish. J Neurosci 27(7):1712–1724
Vihtelic TS, Hyde DR (2000) Light-induced rod and cone cell death and regeneration in the adult albino zebrafish (Danio rerio) retina. J Neurobiol 44(3):289–307
Bernardos RL, Barthel LK, Meyers JR, Raymond PA (2007) Late-stage neuronal progenitors in the retina are radial Müller glia that function as retinal stem cells. J Neurosci 27(26):7028–7040
Kassen SC, Ramanan V, Montgomery JE, T Burket C, Liu C-G, Vihtelic TS, Hyde DR (2007) Time course analysis of gene expression during light-induced photoreceptor cell death and regeneration in albino zebrafish. Dev Neurobiol 67 (8):1009–1031
Kassen SC, Thummel R, Campochiaro LA, Harding MJ, Bennett NA, Hyde DR (2009) CNTF induces photoreceptor neuroprotection and Müller glial cell proliferation through two different signaling pathways in the adult zebrafish retina. Exp Eye Res 88(6):1051–1064
Nagashima M, Barthel LK, Raymond PA (2013) A self-renewing division of zebrafish Muller glial cells generates neuronal progenitors that require N-cadherin to regenerate retinal neurons. Development 140(22):4510–4521
Thummel R, Enright JM, Kassen SC, Montgomery JE, Bailey TJ, Hyde DR (2010) Pax6a and Pax6b are required at different points in neuronal progenitor cell proliferation during zebrafish photoreceptor regeneration. Exp Eye Res 90(5):572–582
Lahne M, Li J, Marton RM, Hyde DR (2015) Actin-cytoskeleton- and rock-mediated INM are required for photoreceptor regeneration in the adult Zebrafish retina. J Neurosci 35(47):15612–15634
Ramachandran R, Fausett BV, Goldman D (2010) ASCL1a regulates Müller glia dedifferentiation and retinal regeneration through a Lin-28-dependent, Let-7 microRNA signalling pathway. Nat Cell Biol 12(11):1101–1107
Powell C, Cornblath E, Elsaeidi F, Wan J, Goldman D (2016) Zebrafish Müller glia-derived progenitors are multipotent, exhibit proliferative biases and regenerate excess neurons. Sci Rep 6:24851
Thomas JL, Ranski AH, Morgan GW, Thummel R (2016) Reactive gliosis in the adult zebrafish retina. Exp Eye Res 143:98–109
Löffler K, Schäfer P, Völkner M, Holdt T, Karl MO (2015) Age-dependent Müller glia neurogenic competence in the mouse retina. Glia 63(10):1809–1824
Dyer MA, Cepko CL (2000) Control of Müller glial cell proliferation and activation following retinal injury. Nat Neurosci 3(9):873–880
Ooto S, Akagi T, Kageyama R, Akita J, Mandai M, Honda Y, Takahashi M (2004) Potential for neural regeneration after neurotoxic injury in the adult mammalian retina. Proc Natl Acad Sci U S A 101(37):13654–13659
Karl MO, Hayes S, Nelson BR, Tan K, Buckingham B, Reh TA (2008) Stimulation of neural regeneration in the mouse retina. Proc Natl Acad Sci U S A 105(49):19508–19513
Oswald M, Robison BD (2008) Strain-specific alteration of zebrafish feeding behavior in response to aversive stimuli. Can J Zool 86(10):1085–1094
Fausett BV, Gumerson JD, Goldman D (2008) The proneural basic helix-loop-helix gene ASCL1a is required for retina regeneration. J Neurosci 28(5):1109–1117
Nelson CM, Gorsuch RA, Bailey TJ, Ackerman KM, Kassen SC, Hyde DR (2012) Stat3 defines three populations of Müller glia and is required for initiating maximal müller glia proliferation in the regenerating zebrafish retina. J Comp Neurol 520(18):4294–4311
Ramachandran R, Zhao XF, Goldman D (2011) ASCL1a/Dkk/beta-catenin signaling pathway is necessary and glycogen synthase kinase-3beta inhibition is sufficient for zebrafish retina regeneration. Proc Natl Acad Sci USA 108(38):15858–15863
Kaur S, Gupta S, Chaudhary M, Khursheed MA, Mitra S, Kurup AJ, Ramachandran R (2018) Let-7 microRNA-mediated regulation of Shh signaling and the gene regulatory network is essential for retina regeneration. Cell Rep 23(5):1409–1423
Ramachandran R, Zhao XF, Goldman D (2012) Insm1a-mediated gene repression is essential for the formation and differentiation of Muller glia-derived progenitors in the injured retina. Nat Cell Biol 14(10):1013–1023
Mitra S, Sharma P, Kaur S, Khursheed MA, Gupta S, Chaudhary M, Kurup AJ, Ramachandran R (2019) Dual regulation of by Myc is necessary during zebrafish retina regeneration. J Cell Biol 218(2):489–507
Sharma P, Gupta S, Chaudhary M, Mitra S, Chawla B, Khursheed MA, Ramachandran R (2019) OCT4 mediates Müller glia reprogramming and cell cycle exit during retina regeneration in zebrafish. Life Sci Alliance 2(5):e201900548
Powell C, Elsaeidi F, Goldman D (2012) Injury-dependent Müller glia and ganglion cell reprogramming during tissue regeneration requires Apobec2a and Apobec2b. J Neurosci 32(3):1096–1109
Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA et al (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318(5858):1917–1920
Elsaeidi F, Macpherson P, Mills EA, Jui J, Flannery JG, Goldman D (2018) Notch suppression collaborates with ASCL1 and LIN28 to unleash a regenerative response in fish retina, but not in mice. J Neurosci 38(9):2246–2261
Wan J, Goldman D (2017) Opposing actions of Fgf8a on notch signaling distinguish two Muller glial cell populations that contribute to retina growth and regeneration. Cell Rep 19(4):849–862
Raymond PA, Barthel LK, Bernardos RL, Perkowski JJ (2006) Molecular characterization of retinal stem cells and their niches in adult zebrafish. BMC Dev Biol 6:36
Jadhav AP, Cho S-H, Cepko CL (2006) Notch activity permits retinal cells to progress through multiple progenitor states and acquire a stem cell property. Proc Natl Acad Sci U S A 103(50):18998–19003
de Melo J, Zibetti C, Clark BS, Hwang W, Miranda-Angulo AL, Qian J, Blackshaw S (2016) Lhx2 is an essential factor for retinal gliogenesis and notch signaling. J Neurosci 36(8):2391–2405
Mills EA, Goldman D (2017) The regulation of notch signaling in retinal development and regeneration. Curr Pathobiol Rep 5(4):323–331
Nelson BR, Ueki Y, Reardon S, Karl MO, Georgi S, Hartman BH, Lamba DA, Reh TA (2011) Genome-wide analysis of Müller glial differentiation reveals a requirement for Notch signaling in postmitotic cells to maintain the glial fate. PLoS One 6(8):e22817
Mitra S, Sharma P, Kaur S, Khursheed MA, Gupta S, Ahuja R, Kurup AJ, Chaudhary M, Ramachandran R (2018) Histone deacetylase-mediated Müller glia reprogramming through Her4.1-LIN28a axis is essential for retina regeneration in zebrafish. iScience 7:68-84
Hayes S, Nelson BR, Buckingham B, Reh TA (2007) Notch signaling regulates regeneration in the avian retina. Dev Biol 312(1):300–311
Ghai K, Zelinka C, Fischer AJ (2010) Notch signaling influences neuroprotective and proliferative properties of mature Müller glia. J Neurosci 30(8):3101–3112
Todd L, Squires N, Suarez L, Fischer AJ (2016) Jak/Stat signaling regulates the proliferation and neurogenic potential of Müller glia-derived progenitor cells in the avian retina. Sci Rep 6:35703
Fischer AJ, Scott MA, Ritchey ER, Sherwood P (2009) Mitogen-activated protein kinase-signaling regulates the ability of Müller glia to proliferate and protect retinal neurons against excitotoxicity. Glia 57(14):1538–1552
Niehrs C (2006) Function and biological roles of the Dickkopf family of Wnt modulators. Oncogene 25 (57):7469–7481
Kara N, Kent MR, Didiano D, Rajaram K, Zhao A, Summerbell ER, Patton JG (2019) The miR-216a-Dot1l regulatory axis is necessary and sufficient for Müller glia reprogramming during retina regeneration. Cell Rep 28 (8):2037-2047.e4
Gallina D, Palazzo I, Steffenson L, Todd L, Fischer AJ (2016) Wnt/β-catenin-signaling and the formation of Müller glia-derived progenitors in the chick retina. Dev Neurobiol 76(9):983–1002
Conedera FM, Pousa AMQ, Mercader N, Tschopp M, Enzmann V (2019) Retinal microglia signaling affects Müller cell behavior in the zebrafish following laser injury induction. Glia 67(6):1150–1166
Fischer AJ, Scott MA, Tuten W (2009) Mitogen-activated protein kinase-signaling stimulates Müller glia to proliferate in acutely damaged chicken retina. Glia 57(2):166–181
Todd L, Fischer AJ (2015) Hedgehog signaling stimulates the formation of proliferating Müller glia-derived progenitor cells in the chick retina. Development 142(15):2610–2622
Lenkowski JR, Qin Z, Sifuentes CJ, Thummel R, Soto CM, Moens CB, Raymond PA (2013) Retinal regeneration in adult zebrafish requires regulation of TGFβ signaling. Glia 61(10):1687–1697
Gallina D, Zelinka CP, Cebulla CM, Fischer AJ (2015) Activation of glucocorticoid receptors in Müller glia is protective to retinal neurons and suppresses microglial reactivity. Exp Neurol 273:114–125
Tappeiner C, Maurer E, Sallin P, Bise T, Enzmann V, Tschopp M (2016) Inhibition of the TGFβ pathway enhances retinal regeneration in adult zebrafish. PLoS One 11(11):e0167073
Todd L, Palazzo I, Squires N, Mendonca N, Fischer AJ (2017) BMP- and TGFβ-signaling regulate the formation of Müller glia-derived progenitor cells in the avian retina. Glia 65(10):1640–1655
Thomas JL, Morgan GW, Dolinski KM, Thummel R (2018) Characterization of the pleiotropic roles of Sonic Hedgehog during retinal regeneration in adult zebrafish. Exp Eye Res 166:106–115
Sherpa T, Lankford T, McGinn TE, Hunter SS, Frey RA, Sun C, Ryan M, Robison BD et al (2014) Retinal regeneration is facilitated by the presence of surviving neurons. Dev Neurobiol 74(9):851–876
Sun L, Li P, Carr AL, Gorsuch R, Yarka C, Li J, Bartlett M, Pfister D et al (2014) Transcription of the SCL/TAL1 interrupting locus (Stil) is required for cell proliferation in adult Zebrafish retinas. J Biol Chem 289(10):6934–6940
Gorsuch RA, Lahne M, Yarka CE, Petravick ME, Li J, Hyde DR (2017) SOX2 regulates Müller glia reprogramming and proliferation in the regenerating zebrafish retina via LIN28 and ASCL1a. Exp Eye Res 161:174–192
Gramage E, D’Cruz T, Taylor S, Thummel R, Hitchcock PF (2015) Midkine-a protein localization in the developing and adult retina of the zebrafish and its function during photoreceptor regeneration. PLoS One 10(3):e0121789
Nagashima M, D’Cruz TS, Danku AE, Hesse D, Sifuentes C, Raymond PA, Hitchcock PF (2020) Midkine-a is required for cell cycle progression of Müller glia during neuronal regeneration in the vertebrate retina. J Neurosci 40(6):1232–1247
Herman JG, Baylin SB (2003) Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med 349(21):2042–2054
Mikkelsen TS, Hanna J, Zhang X, Ku M, Wernig M, Schorderet P, Bernstein BE, Jaenisch R et al (2008) Dissecting direct reprogramming through integrative genomic analysis. Nature 454(7200):49–55
Polo JM, Anderssen E, Walsh RM et al (2012) A molecular roadmap of reprogramming somatic cells into iPS cells. Cell 151(7):1617–1632
Powell C, Grant AR, Cornblath E, Goldman D (2013) Analysis of DNA methylation reveals a partial reprogramming of the Muller glia genome during retina regeneration. Proc Natl Acad Sci U S A 110(49):19814–19819
Powell C, Cornblath E, Goldman D (2014) Zinc-binding domain-dependent, deaminase-independent actions of apolipoprotein B mRNA-editing enzyme, catalytic polypeptide 2 (Apobec2), mediate its effect on zebrafish retina regeneration. J Biol Chem 289(42):28924–28941
Audia JE, Campbell RM (2016) Histone modifications and cancer. Cold Spring Harb Perspect Biol 8(4):a019521
Karlić R, Chung HR, Lasserre J, Vlahovicek K, Vingron M (2010) Histone modification levels are predictive for gene expression. Proc Natl Acad Sci U S A 107(7):2926–2931
Lawrence M, Daujat S, Schneider R (2016) Lateral thinking: how histone modifications regulate gene expression. Trends Genet 32(1):42–56
Jonas S, Izaurralde E (2015) Towards a molecular understanding of microRNA-mediated gene silencing. Nat Rev Genet 16(7):421–433
Lu TX, Rothenberg ME (2018) MicroRNA. J Allergy Clin Immunol 141(4):1202–1207
Rajaram K, Harding RL, Bailey T, Patton JG, Hyde DR (2014) Dynamic miRNA expression patterns during retinal regeneration in zebrafish: reduced dicer or miRNA expression suppresses proliferation of Müller glia-derived neuronal progenitor cells. Dev Dyn 243(12):1591–1605
Zhao C, Tao Z, Xue L, Zeng Y, Wang Y, Xu H, Yin ZQ (2017) LIN28b stimulates the reprogramming of rat Müller glia to retinal progenitors. Exp Cell Res 352(1):164–174
Murakami Y, Ishikawa K, Nakao S, Sonoda K-H (2020) Innate immune response in retinal homeostasis and inflammatory disorders. Prog Retin Eye Res 74:100778
White DT, Sengupta S, Saxena MT, Xu Q, Hanes J, Ding D, Ji H, Mumm JS (2017) Immunomodulation-accelerated neuronal regeneration following selective rod photoreceptor cell ablation in the zebrafish retina. Proc Natl Acad Sci U S A 114(18):E3719–E3728
Mitchell DM, Lovel AG, Stenkamp DL (2018) Dynamic changes in microglial and macrophage characteristics during degeneration and regeneration of the zebrafish retina. J Neuroinflammation 15(1):163
Fischer AJ, McGuire CR, Dierks BD, Reh TA (2002) Insulin and fibroblast growth factor 2 activate a neurogenic program in Müller glia of the chicken retina. J Neurosci 22(21):9387–9398
Zelinka CP, Volkov L, Goodman ZA, Todd L, Palazzo I, Bishop WA, Fischer AJ (2016) mTor signaling is required for the formation of proliferating Müller glia-derived progenitor cells in the chick retina. Development 143(11):1859–1873
Palazzo I, Deistler K, Hoang TV, Blackshaw S, Fischer AJ (2020) NF-κB signaling regulates the formation of proliferating Müller glia-derived progenitor cells in the avian retina. Development 147(10): dev183418
Srivastava D, DeWitt N (2016) In vivo cellular reprogramming: the next generation. Cell 166(6):1386–1396
Li H, Chen G (2016) In vivo reprogramming for CNS repair: regenerating neurons from endogenous glial cells. Neuron 91(4):728–738
Guo Z, Zhang L, Wu Z, Chen Y, Wang F, Chen G (2014) In vivo direct reprogramming of reactive glial cells into functional neurons after brain injury and in an Alzheimer’s disease model. Cell Stem Cell 14(2):188–202
Masserdotti G, Gillotin S, Sutor B, Drechsel D, Irmler M, Jørgensen HF, Sass S, Theis FJ et al (2015) Transcriptional mechanisms of proneural factors and REST in regulating neuronal reprogramming of astrocytes. Cell Stem Cell 17(1):74–88
Liu Y, Miao Q, Yuan J, Han S, Zhang P, Li S, Rao Z, Zhao W et al (2015) ASCL1 converts dorsal midbrain astrocytes into functional neurons in vivo. J Neurosci 35(25):9336–9355
Torper O, Pfisterer U, Wolf DA, Pereira M, Lau S, Jakobsson J, Björklund A, Grealish S et al (2013) Generation of induced neurons via direct conversion in vivo. Proc Natl Acad Sci U S A 110(17):7038–7043
Grande A, Sumiyoshi K, López-Juárez A, Howard J, Sakthivel B, Aronow B, Campbell K, Nakafuku M (2013) Environmental impact on direct neuronal reprogramming in vivo in the adult brain. Nat Commun 4:2373
Gascón S, Murenu E, Masserdotti G et al (2016) Identification and successful negotiation of a metabolic checkpoint in direct neuronal reprogramming. Cell Stem Cell 18(3):396–409
Niu W, Zang T, Zou Y, Fang S, Smith DK, Bachoo R, Zhang CL (2013) In vivo reprogramming of astrocytes to neuroblasts in the adult brain. Nat Cell Biol 15(10):1164–1175
Niu W, Zang T, Smith DK, Vue TY, Zou Y, Bachoo R, Johnson JE, Zhang CL (2015) SOX2 reprograms resident astrocytes into neural progenitors in the adult brain. Stem Cell Reports 4(5):780–794
Su Z, Niu W, Liu ML, Zou Y, Zhang CL (2014) In vivo conversion of astrocytes to neurons in the injured adult spinal cord. Nat Commun 5:3338
Zhou H, Su J, Hu X et al (2020) Glia-to-neuron conversion by CRISPR-CasRx alleviates symptoms of neurological disease in mice. Cell 181(3): 590–603.e16
Vassilopoulos G, Russell DW (2003) Cell fusion: an alternative to stem cell plasticity and its therapeutic implications. Curr Opin Genet Dev 13(5):480–485
Sanges D, Romo N, Simonte G, Di Vicino U, Tahoces AD, Fernández E, Cosma MP (2013) Wnt/β-catenin signaling triggers neuron reprogramming and regeneration in the mouse retina. Cell Rep 4(2):271–286
Sanges D, Simonte G, Di Vicino U, Romo N, Pinilla I, Nicolás M, Cosma MP (2016) Reprogramming Müller glia via in vivo cell fusion regenerates murine photoreceptors. J Clin Invest 126(8):3104–3116
Pesaresi M, Bonilla-Pons SA, Simonte G, Sanges D, Di Vicino U, Cosma MP (2018) Endogenous mobilization of bone-marrow cells into the murine retina induces fusion-mediated reprogramming of Müller glia cells. EBioMedicine 30:38–51
Ryall JG, Cliff T, Dalton S, Sartorelli V (2015) Metabolic reprogramming of stem cell epigenetics. Cell Stem Cell 17(6):651–662
Nishimura K, Fukuda A, Hisatake K (2019) Mechanisms of the metabolic shift during somatic cell reprogramming. Int J Mol Sci 20(9):2254
Naik PP, Birbrair A, Bhutia SK (2019) Mitophagy-driven metabolic switch reprograms stem cell fate. Cell Mol Life Sci 76(1):27–43
Khacho M, Harris R, Slack RS (2019) Mitochondria as central regulators of neural stem cell fate and cognitive function. Nat Rev Neurosci 20(1):34–48
Dyer MA (2016) Biomedicine: an eye on retinal recovery. Nature 540(7633):350–351
Pearson RA, Gonzalez-Cordero A, West EL et al (2016) Donor and host photoreceptors engage in material transfer following transplantation of post-mitotic photoreceptor precursors. Nat Commun 7:13029
Singh MS, Balmer J, Barnard AR, Aslam SA, Moralli D, Green CM, Barnea-Cramer A, Duncan I et al (2016) Transplanted photoreceptor precursors transfer proteins to host photoreceptors by a mechanism of cytoplasmic fusion. Nat Commun 7:13537
Jiang D, Xiong G, Feng H et al (2019) Donation of mitochondria by iPSC-derived mesenchymal stem cells protects retinal ganglion cells against mitochondrial complex I defect-induced degeneration. Theranostics 9(8):2395–2410
Chen F, Zhou J, Li Y, Zhao Y, Yuan J, Cao Y, Wang L, Zhang Z et al (2019) YY1 regulates skeletal muscle regeneration through controlling metabolic reprogramming of satellite cells. EMBO J 38(10):e99727
Acquistapace A, Bru T, Lesault PF, Figeac F, Coudert AE, le Coz O, Christov C, Baudin X et al (2011) Human mesenchymal stem cells reprogram adult cardiomyocytes toward a progenitor-like state through partial cell fusion and mitochondria transfer. Stem Cells 29(5):812–824
Vazquez-Martin A, Cufi S, Corominas-Faja B, Oliveras-Ferraros C, Vellon L, Menendez JA (2012) Mitochondrial fusion by pharmacological manipulation impedes somatic cell reprogramming to pluripotency: new insight into the role of mitophagy in cell stemness. Aging 4(6):393–401
Zhu S, Li W, Zhou H, Wei W, Ambasudhan R, Lin T, Kim J, Zhang K et al (2010) Reprogramming of human primary somatic cells by OCT4 and chemical compounds. Cell Stem Cell 7(6):651–655
Folmes CD, Nelson TJ, Martinez-Fernandez A, Arrell DK, Lindor JZ, Dzeja PP, Ikeda Y, Perez-Terzic C et al (2011) Somatic oxidative bioenergetics transitions into pluripotency-dependent glycolysis to facilitate nuclear reprogramming. Cell Metab 14(2):264–271
Prigione A, Rohwer N, Hoffmann S, Mlody B, Drews K, Bukowiecki R, Blümlein K, Wanker EE et al (2014) HIF1α modulates cell fate reprogramming through early glycolytic shift and upregulation of PDK1-3 and PKM2. Stem Cells 32(2):364–376
Pollak J, Wilken MS, Ueki Y, Cox KE, Sullivan JM, Taylor RJ, Levine EM, Reh TA (2013) ASCL1 reprograms mouse Muller glia into neurogenic retinal progenitors. Development 140(12):2619–2631
Ueki Y, Wilken MS, Cox KE, Chipman L, Jorstad N, Sternhagen K, Simic M, Ullom K et al (2015) Transgenic expression of the proneural transcription factor ASCL1 in Müller glia stimulates retinal regeneration in young mice. Proc Natl Acad Sci U S A 112(44):13717–13722
Jorstad NL, Wilken MS, Todd L, Finkbeiner C, Nakamura P, Radulovich N, Hooper MJ, Chitsazan A, Wilkerson BA, Rieke F, Reh TA (2020) STAT signaling modifies ASCL1 chromatin binding and limits neural regeneration from Muller glia in adult mouse retina. Cell Rep 30 (7):2195–2208.e5
Patel AK, Surapaneni K, Yi H, Nakamura REI, Karli SZ, Syeda S, Lee T, Hackam AS (2015) Activation of Wnt/β-catenin signaling in Muller glia protects photoreceptors in a mouse model of inherited retinal degeneration. Neuropharmacology 91:1–12
Patel AK, Park KK, Hackam AS (2017) Wnt signaling promotes axonal regeneration following optic nerve injury in the mouse. Neuroscience 343:372–383
Osakada F, Ooto S, Akagi T, Mandai M, Akaike A, Takahashi M (2007) Wnt signaling promotes regeneration in the retina of adult mammals. J Neurosci 27(15):4210–4219
Del Debbio CB, Balasubramanian S, Parameswaran S, Chaudhuri A, Qiu F, Ahmad I (2010) Notch and Wnt signaling mediated rod photoreceptor regeneration by Müller cells in adult mammalian retina. PLoS One 5(8):e12425
Ballas N, Grunseich C, Lu DD, Speh JC, Mandel G (2005) REST and its corepressors mediate plasticity of neuronal gene chromatin throughout neurogenesis. Cell 121(4):645–657
Mampay M, Sheridan GK (2019) REST: an epigenetic regulator of neuronal stress responses in the young and ageing brain. Front Neuroendocrinol 53:100744
Yeo M, Lee SK, Lee B, Ruiz EC, Pfaff SL, Gill GN (2005) Small CTD phosphatases function in silencing neuronal gene expression. Science 307(5709):596–600
Lee SW, Oh YM, Lu Y-L, Kim WK, Yoo AS (2018) MicroRNAs overcome cell fate barrier by reducing EZH2-controlled REST stability during neuronal conversion of human adult fibroblasts. Dev cell 46 (1):73-84.e7
Xue Y, Ouyang K, Huang J, Zhou Y, Ouyang H, Li H, Wang G, Wu Q et al (2013) Direct conversion of fibroblasts to neurons by reprogramming PTB-regulated microRNA circuits. Cell 152(1–2):82–96
Wohl SG, Reh TA (2016) miR-124-9-9* potentiates ASCL1-induced reprogramming of cultured Muller glia. Glia 64(5):743–762
Moya IM, Halder G (2019) Hippo–YAP/TAZ signalling in organ regeneration and regenerative medicine. Nat Rev Mol Cell Biol 20(4):211–226
Maugeri-Saccà M, De Maria R (2018) The hippo pathway in normal development and cancer. Pharmacol Ther 186:60–72
Hamon A, García-García D, Ail D, Bitard J, Chesneau A, Dalkara D, Locker M, Roger JE, Perron M (2019) Linking YAP to Muller glia quiescence exit in the degenerative retina. Cell rep 27 (6):1712-1725.e6
Hamon A, Masson C, Bitard J, Gieser L, Roger JE, Perron M (2017) Retinal degeneration triggers the activation of YAP/TEAD in reactive Müller cells. Invest Ophthalmol Vis Sci 58(4):1941–1953
Rueda EM, Hall BM, Hill MC, Swinton PG, Tong X, Martin JF, Poché RA (2019) The hippo pathway blocks mammalian retinal Muller glial cell reprogramming. Cell rep 27 (6):1637-1649.e6
Wan J, Zheng H, Xiao HL, She ZJ, Zhou GM (2007) Sonic hedgehog promotes stem-cell potential of Müller glia in the mammalian retina. Biochem Biophys Res Commun 363(2):347–354
Gu D, Wang S, Zhang S, Zhang P, Zhou G (2017) Directed transdifferentiation of Müller glial cells to photoreceptors using the sonic hedgehog signaling pathway agonist purmorphamine. Mol Med Rep 16(6):7993–8002
Ringuette R, Atkins M, Lagali PS, Bassett EA, Campbell C, Mazerolle C, Mears AJ, Picketts DJ et al (2016) A notch-Gli2 axis sustains hedgehog responsiveness of neural progenitors and Müller glia. Dev Biol 411(1):85–100
Ferraro S, Gomez-Montalvo AI, Olmos R, Ramirez M, Lamas M (2015) Primary cilia in rat mature Müller glia: downregulation of IFT20 expression reduces sonic hedgehog-mediated proliferation and dedifferentiation potential of Müller glia primary cultures. Cell Mol Neurobiol 35(4):533–542
Del Debbio CB, Mir Q, Parameswaran S, Mathews S, Xia X, Zheng L, Neville AJ, Ahmad I (2016) Notch signaling activates stem cell properties of Müller glia through transcriptional regulation and Skp2-mediated degradation of p27Kip1. PLoS One 11(3):e0152025
Jian Q, Tao Z, Li Y, Yin ZQ (2015) Acute retinal injury and the relationship between nerve growth factor, Notch1 transcription and short-lived dedifferentiation transient changes of mammalian Müller cells. Vision Res 110:107–117
Wang Y, Smith SB, Ogilvie JM, McCool DJ, Sarthy V (2002) Ciliary neurotrophic factor induces glial fibrillary acidic protein in retinal Müller cells through the JAK/STAT signal transduction pathway. Curr Eye Res 24(4):305–312
Peterson WM, Wang Q, Tzekova R, Wiegand SJ (2000) Ciliary neurotrophic factor and stress stimuli activate the Jak-STAT pathway in retinal neurons and glia. J Neurosci 20(11):4081–4090
Kirsch M, Trautmann N, Ernst M, Hofmann HD (2010) Involvement of gp130-associated cytokine signaling in Müller cell activation following optic nerve lesion. Glia 58(7):768–779
Close JL, Gumuscu B, Reh TA (2005) Retinal neurons regulate proliferation of postnatal progenitors and Müller glia in the rat retina via TGF beta signaling. Development 132(13):3015–3026
Lawrence JM, Singhal S, Bhatia B, Keegan DJ, Reh TA, Luthert PJ, Khaw PT, Limb GA (2007) MIO-M1 cells and similar muller glial cell lines derived from adult human retina exhibit neural stem cell characteristics. Stem Cells 25(8):2033–2043
Angbohang A, Wu N, Charalambous T, Eastlake K, Lei Y, Kim YS, Sun XH, Limb GA (2016) Downregulation of the canonical WNT signaling pathway by TGFβ1 inhibits photoreceptor differentiation of adult human Müller glia with stem cell characteristics. Stem Cells Dev 25(1):1–12
Ueki Y, Reh TA (2013) EGF stimulates Müller glial proliferation via a BMP-dependent mechanism. Glia 61(5):778–789
Surzenko N, Crowl T, Bachleda A, Langer L, Pevny L (2013) SOX2 maintains the quiescent progenitor cell state of postnatal retinal Muller glia. Development 140(7):1445–1456
Whitney IE, Keeley PW, St. John AJ, Kautzman AG, Kay JN, Reese BE (2014) SOX2 regulates cholinergic amacrine cell positioning and dendritic stratification in the retina. J Neurosci 34(30):10109–10121
Reyes-Aguirre LI, Lamas M (2016) OCT4 methylation-mediated silencing as an epigenetic barrier preventing Müller glia dedifferentiation in a murine model of retinal injury. Front Neurosci 10:523
Xiao J, Li X, Chen L, Han X, Zhao W, Li L, Chen JG (2017) Apobec1 promotes neurotoxicity-induced dedifferentiation of Müller glial cells. Neurochem Res 42(4):1151–1164
Wohl SG, Hooper MJ, Reh TA (2019) MicroRNAs miR-25, Let-7 and miR-124 regulate the neurogenic potential of Müller glia in mice. Development 146 (17): dev179556
Ji H-P, Xiong Y, Song W-T, Zhang E-D, Gao Z-L, Yao F, Su T, Zhou R-R et al (2017) MicroRNA-28 potentially regulates the photoreceptor lineage commitment of Müller glia-derived progenitors. Sci Rep 7(1):11374
Diaz-Aparicio I, Paris I, Sierra-Torre V et al (2020) Microglia actively remodel adult hippocampal neurogenesis through the phagocytosis secretome. J Neurosci 40(7):1453–1482
Aarum J, Sandberg K, Haeberlein SL, Persson MA (2003) Migration and differentiation of neural precursor cells can be directed by microglia. Proc Natl Acad Sci U S A 100(26):15983–15988
Vay SU, Flitsch LJ, Rabenstein M, Rogall R, Blaschke S, Kleinhaus J, Reinert N, Bach A et al (2018) The plasticity of primary microglia and their multifaceted effects on endogenous neural stem cells in vitro and in vivo. J Neuroinflammation 15(1):226
Osman AM, Rodhe J, Shen X, Dominguez CA, Joseph B, Blomgren K (2019) The secretome of microglia regulate neural stem cell function. Neuroscience 405:92–102
Willis EF, MacDonald KPA, Nguyen QH, Garrido AL, Gillespie ER, Harley SBR, Bartlett PF, Schroder WA, Yates AG, Anthony DC, Rose-John S, Ruitenberg MJ, Vukovic J (2020) Repopulating microglia promote brain repair in an IL-6-dependent manner. Cell 180 (5):833-846.e16
Wang M, Ma W, Zhao L, Fariss RN, Wong WT (2011) Adaptive Müller cell responses to microglial activation mediate neuroprotection and coordinate inflammation in the retina. J Neuroinflammation 8:173
Harada T, Harada C, Nakayama N, Okuyama S, Yoshida K, Kohsaka S, Matsuda H, Wada K (2000) Modification of glia-neuronal cell interactions prevents photoreceptor apoptosis during light-induced retinal degeneration. Neuron 26(2):533–541
Harada T, Harada C, Kohsaka S, Wada E, Yoshida K, Ohno S, Mamada H, Tanaka K et al (2002) Microglia–Müller glia cell interactions control neurotrophic factor production during light-induced retinal degeneration. J Neurosci 22(21):9228–9236
Imitola J (2019) Regenerative neuroimmunology: the impact of immune and neural stem cell interactions for translation in neurodegeneration and repair. J Neuroimmunol 331:1–3
Fischer AJ, Reh TA (2003) Potential of Müller glia to become neurogenic retinal progenitor cells. Glia 43(1):70–76
Roesch K, Jadhav AP, Trimarchi JM, Stadler MB, Roska B, Sun BB, Cepko CL (2008) The transcriptome of retinal Müller glial cells. J Comp Neurol 509(2):225–238
Reichenbach A, Bringmann A (2020) Glia of the human retina. Glia 68(4):768–796
Bringmann A, Syrbe S, Görner K, Kacza J, Francke M, Wiedemann P, Reichenbach A (2018) The primate fovea: structure, function and development. Prog Retin Eye Res 66:49–84
Lu Y, Yi W, Wu Q, Zhong S, Zuo Z, Zhao F, Zhang M, Tsai N, Zhuo Y, He S, Zhang J, Duan X, Wang X, Xue T (2018) Single-cell RNA-seq analysis maps the development of human fetal retina. bioRxiv:423830
Xiao D, Qiu S, Huang X, Zhang R, Lei Q, Huang W, Chen H, Gou B, Tie X, Liu S, Liu Y, Jin K, Xiang M (2019) Directed robust generation of functional retinal ganglion cells from Müller glia. bioRxiv:735357
Lu Y, Zhang K (2018) Cellular reprogramming in the retina-seeing the light. N Engl J Med 378(11):1059–1060
Lin S, Guo J, Chen S (2017) Transcriptome and DNA methylome signatures associated with retinal Müller glia development, injury response, and aging. Invest Ophthalmol Vis Sci 60(13):4436–4450
Wang W, Hu K, Zeng A, Alegre D, Hu D, Gotting K, Ortega Granillo A, Wang Y, et al (2020). Changes in regeneration-responsive enhancers shape regenerative capacities in vertebrates. Science 369(6508): eaaz3090
Hoang T, Wang J, Boyd P, Wang F, Santiago C, Jiang L, Yoo S, Lahne M, et al (2020). Gene regulatory networks controlling vertebrate retinal regeneration. Science 370(6519): eabb8598
Funding
This study was supported by the National Key R&D Program of China (2018YFA0107302), the National Natural Science Foundation of China (No. 31930068, 81873688).
Author information
Authors and Affiliations
Contributions
Hui Gao, Luodan A, and Haiwei Xu write the review. Xiaona Huang and Xi Chen revise the review.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
Not applicable
Code Availability
Not applicable
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, H., A, L., Huang, X. et al. Müller Glia-Mediated Retinal Regeneration. Mol Neurobiol 58, 2342–2361 (2021). https://doi.org/10.1007/s12035-020-02274-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-020-02274-w