Abstract
Mitochondrial dysfunctions and oxidative stress play important roles in the early pathogenesis of Alzheimer’s disease (AD), which also involves the aberrant expression levels of mitochondrial proteins. However, the molecular mechanisms underlying the aberrant expression levels of these proteins in the pathogenesis of AD are still not completely understood. Tid1 (DnaJA3/mtHsp40), a mammalian homolog of the Drosophila tumor suppressor Tid56, is reported to induce mitochondrial fragmentation associated with an increase in reactive oxygen species (ROS) levels, resulting in cell death in some cancer cells. However, the involvement of Tid1 in AD pathogenesis is as yet unknown. In this study, we found that the Tid1 protein levels were upregulated in the hippocampus of AD patients and Tg2576 mice. Our in vitro studies showed that Aβ42 increased the expression levels of Tid1 in primary rat cortical neurons. The knockdown of Tid1 protected against neuronal cell death induced by Aβ42, and Tid1-mediated neuronal cell death, was dependent on the increased ROS generation and caspase-3 activity. The overexpression of Tid1 in HEK293-APP cells increased the BACE1 levels, resulting in increased Aβ production. Conversely, Tid1 knockdown in HEK293-APP cells and primary cultured neurons decreased Aβ production through the reduction in the BACE1 levels. We also found that the overexpression of Tid1 activated c-Jun N-terminal kinase (JNK) leading to increased Aβ production. Taken together, our results suggest that upregulated Tid1 levels in the hippocampus of patients with AD and Tg2576 mice induce apoptosis and increase Aβ production, and Tid1 may therefore be a suitable target in therapeutic interventions for AD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD), a progressive neurodegenerative disorder, is associated with a chronic loss of synapses and neuronal cell death [1], and is characterized by the presence of amyloid-β (Aβ) plaques and neurofibrillary tangles (NFTs) [2]. Aβ is generated from the amyloid-β precursor protein (APP) by two-step proteolytic cleavage. APP is first cleaved by β-secretase, that is, β-site APP cleaving enzyme 1 (BACE1), to generate a membrane-bound C-terminal fragment of 99 amino acids (APP-C99), and then APP-C99 is further cleaved by γ-secretase to generate Aβ40 and Aβ42. Alternatively, APP can be cleaved by α-secretase to generate non-amyloidogenic soluble APPα, thereby preventing Aβ generation [3,4,5,6].
In mammals, mitochondria are unique organelles that are pivotal for various cellular functions including ATP synthesis, calcium homeostasis, and cell survival [7]. Aβ may directly disrupt mitochondrial functions and contribute to the deficiency of energy metabolism and neuronal cell death seen in AD. The presence of Aβ in mitochondria is associated with impaired mitochondrial metabolism and increased mitochondrial reactive oxygen species (ROS) production [8, 9]. Mitochondrial proteostasis has been found to be one of the main factors for neuronal cell death in AD [10, 11], and mitochondrial protein quality control is affected by mitochondrial chaperones [12]. Extensive studies have demonstrated that mitochondrial dysfunctions and oxidative stress play important roles in the early pathogenesis of AD [13,14,15]. Indeed, mitochondrial dysfunctions including decreased ATP generation and increased ROS generation induced by the aberrant expression levels of mitochondrial proteins are frequently observed in the brain of patients with AD [14, 16, 17] and Tg2576 mice [18]. Although mitochondrial dysfunctions are a prominent feature of AD, the molecular mechanisms underlying the aberrant expression levels of these proteins in the pathogenesis of AD are as yet unclarified.
Tumorous imaginal disc 1 (Tid1), also known as mitochondrial Hsp40 (mtHsp40), is a mitochondrial protein. Mammals express two alternatively spliced forms of Tid1: Tid1-Long (43 kDa) and Tid1-Short (41 kDa). Both isoforms have an amino-terminal mitochondrial targeting sequence, a DnaJ domain, a glycine/phenylalanine linker region, a central cysteine-rich region resembling a zinc finger repeat, and a carboxyl-terminal region. Via the DnaJ domain, Tid1 interacts with and activates the Hsp70/Hsc70 family of chaperones to induce protein folding and degradation [19]. Tid1 also regulates mitochondrial functions by maintaining mitochondrial membrane potential and mitochondrial DNA (mtDNA) integrity [20]. Since Tid1 is a homolog of the Drosophila tumor suppressor Tid56, the function of Tid1 in apoptosis has been investigated. Certain stress stimuli, including oxidative stress, DNA damage, and oncogene dysregulation, induce the stabilization and elevation of p53 protein levels and the subsequent activation of apoptotic programs. The loss of Tid1 expression abrogates p53 translocation to the mitochondria and inhibits apoptosis, whereas the overexpression of Tid1 promotes p53 mitochondrial localization and apoptosis [21]. In cancer cells, Tid1 induces Drp1-dependent mitochondrial fragmentation associated with increased ROS levels and decreased cell viability [22]. However, the role of Tid1 in neurodegenerative diseases such as AD, which are always accompanied by impaired mitochondrial functions as a causative factor, is as yet unknown.
In this study, we investigated whether Tid1 expression is altered in the hippocampus of AD patients and Tg2576 mice, and we found it to be upregulated compared with that in non-AD and age-matched wild-type mice, respectively. Moreover, our in vitro studies showed that Aβ42 increased the expression levels of Tid1 in primary rat cortical neurons, which in turn increased ROS generation and caspase-3 activity leading to neuronal cell death. Furthermore, the overexpression of Tid1-L in HEK293-APP cells increased the BACE1 levels, inducing increased Aβ production. Conversely, Tid1 knockdown in HEK293-APP cells and primary cultured neurons decreased Aβ production through the reduction in the BACE1 levels. We also found that the overexpression of Tid1 activated JNK leading to increased Aβ production.
Materials and Methods
Human Brains
Hippocampus postmortem tissue samples from AD patients (n = 10) and non-AD control (n = 10) were obtained with the approval of the Committees on Human Research of Nagoya City University and Choju Medical Institute of Fukushimura Hospital. The information of the brain specimens is shown in Table 1.
APP Transgenic Mice
Female Tg2576 mice overexpressing human APP695 with the Swedish mutation K660N/M671L were obtained from Taconic (Germantown, NY, USA). Tg2576 and age-matched wild-type (WT) mice were housed under a 12-h light/dark cycle and had access to food and water ad libitum. All the experiments were performed in accordance with the Guidelines for Animal Experiments of the Animal Experimentation Committee of Nagoya City University. Both Tg2576 and WT mice were sacrificed at 6, 11, and 18 months of age, and hippocampal tissues were dissected, removed, and placed immediately on dry ice, and stored at – 80 °C until processing.
Cell Cultures
Cerebral cortical neurons were obtained from embryonic day 17 (E17) Wistar rats (Japan SLC, Inc., Hamamatsu, Japan) and cultured as described previously [23]. Embryonic brains were dissected, stripped of meninges, and minced with forceps. The minced tissue was incubated in 0.125% trypsin and 2 mg/ml DNase I in PBS at 37 °C for 15 min. The resulting fragments were then dissociated into single cells by pipetting. The dissociated cells were suspended in neurobasal medium (Invitrogen, Carlsbad, CA, USA) containing MACS NeuroBrew-21 without vitamin A (Miltenyi Biotec, Auburn, CA, USA) and 0.1% bovine albumin fraction V solution (Invitrogen), and plated onto poly-D-lysine-coated 60 mm dishes (Iwaki, Japan) or poly-D-lysine-coated glass slides in 24-well plates at a density of 1 × 106 cells/ml. These cells were used on day 4 after plating for future experiments. HEK293-APP cells stably express human APP695, whereas HEK293-C99 cells stably express human APP-C99. The Neuro2A cell line stably expressing Myc-tagged human tau (P301L) was kindly provided by Dr. Akihiko Takashima (Gakushuin University, Japan) [24]. These cells were cultured in Dulbecco’s modified Eagle’s medium (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan) supplemented with 10% fetal bovine serum.
Aβ42 Treatment
Human amyloid β-peptide (1–42) was obtained from Peptide Institute Inc., Japan. Aβ42 peptide (0.54 mg) was dissolved in 120 μl of DMSO to prepare a 1 mM amyloid β-peptide solution. The solution was kept at – 20 °C until use. To test the effect of Aβ42 on the Tid1 expression levels, primary cultured neurons were treated with 5 μM Aβ42 for 0, 1, 2, 3, or 6 h followed by total RNA isolation and protein extraction for real-time PCR and Western blot analysis, respectively.
Plasmid Constructs and Transfection
A Tid1-L expression construct containing a 1488 bp mouse cDNA was inserted into the pCMV vector with a Myc-tag sequence at the 5′ terminus of the insert (Myc-Tid1-L). The plasmid was purchased from Sino Biological Inc. (Beijing, China). HEK293-APP, HEK293-C99, and Neuro2A-P301L cells were transfected with 5 μg of the Myc-tagged Tid1-L (Myc-Tid1-L) or mock control (Myc) vector using Lipofectamine 3000 reagent (Invitrogen) in accordance with the manufacturer’s instructions. The effects of transfection were examined 48 h after the transfection.
RNA Interference
Endogenous Tid1 was knocked down using predesigned Stealth siRNA against Tid1 (Tid1 siRNA) and Stealth siRNA negative control (control siRNA) from Invitrogen. The Tid1 siRNA sequences are as follows: Tid1 siRNA sense, 5′-CCCGAGGAGCUAUUCAGGAAGAUCU-3′; Tid1 siRNA antisense, 5′-AGAUCUUCCUGAAUAGCUCCUCGGG-3′. The cells were transiently transfected with 30 nM Tid1 or control siRNA using Lipofectamine RNAiMAX (Invitrogen) in accordance with the manufacturer’s instructions. The effects of knockdown were examined 72 h after transfection. Conditioned medium was collected and used for ELISA, and then the cells were processed for ROS detection, TUNEL assay, or Western blot analysis.
RNA Extraction and Real-Time PCR
Total RNA was isolated from primary cultured neurons using TRIzol reagent (Invitrogen). Reverse transcription was performed using 1 μg of total RNA and an iScript select cDNA synthesis kit (Bio-Rad, California, USA) in accordance with the manufacturer’s instructions. Real-time PCR was performed with GeneAce SYBR qPCR Mix (Nippon Gene, Tokyo, Japan) in MicroAmp Optical 96-well reaction plates using a 7500 Real-Time PCR System (Applied Biosystems). The expression levels of the Tid1 gene were normalized with the corresponding amount of actin mRNA using the comparative threshold cycle method. Amplification was performed using the following primers: Tid1 (sense, 5′-TAGGCAGCAGAACCTGATCC-3′; anti-sense, 5′-GGAATCCCTCATTGTCCT-CCC-3′) and actin (sense, 5′-CATCCGTAAAGACCTCTATGCCAAC-3′; anti-sense, 5′-ATGGA-GCCACCGATC-CACA-3′).
Western Blot Analysis
Cells and hippocampal tissues from human and mice brains were homogenized in RIPA buffer containing a protease inhibitor cocktail (Roche, Mannheim, Germany) and a phosphatase inhibitor cocktail solution I (FUJIFILM Wako Pure Chemical Corporation, Japan). The resulting homogenates were incubated on ice for 30 min and centrifuged for 30 min at 13,000×g at 4 °C. The protein concentrations in the supernatants were determined using a BCA protein assay kit (Thermo Fisher Scientific, USA). Equal amounts of proteins were separated by SDS polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, Billerica, MA, USA). These membranes were then blocked with 5% skim milk in TBS-T for 1 h at room temperature, and then incubated overnight at 4 °C with the following primary antibodies: anti-Tid1 (Santa Cruz Biotechnology, USA), anti-Myc (MBL Life Science, Japan), anti-caspase-3 (Cell Signaling Technology, USA), anti-phospho (p)-tau (AT180, Invitrogen), anti-tau (tau5, BioLegend, USA), anti-p-AKT (p-AKT (S473), Cell Signaling Technology), anti-AKT (Cell Signaling Technology), anti-p-GSK3α/β (S21/9, Cell Signaling Technology), anti-GSK3α/β (Cell Signaling Technology), anti-APP (22C11, Millipore), anti-BACE1 (Cell Signaling Technology), anti-PS1 (Millipore), anti-ADAM10 (Millipore), anti-actin (Proteintech Group, USA), anti-p-JNK (p-SAPK/JNK (T183/Y185) (G9), Cell Signaling Technology), and anti-JNK (SAPK/JNK, Cell Signaling Technology) antibodies. The membranes were washed with TBS-T and then incubated with the appropriate secondary antibodies conjugated to horseradish peroxidase. Immunoreactive bands were visualized using Immunostar Zeta or Immunostar LD (FUJIFILM Wako Pure Chemical Corporation) and analyzed using Amersham Imager 680 (GE Health Care, Japan).
Aβ ELISA
Primary cultured neurons, HEK293-APP cells, and HEK293-C99 cells were transfected with Tid1 or control siRNA for 72 h, and the conditioned medium was collected and centrifuged at 12,500 rpm for 15 min at 4 °C to remove cell debris. The levels of Aβ40 and Aβ42 secreted in the medium were measured using sandwich enzyme-linked immunosorbent assay (ELISA) kits (Wako Pure Chemical Industries, Japan) in accordance with the manufacturer’s instructions. For HEK293-APP cells transfected with the Myc-tagged Tid1-L or mock control vector, the culture medium was replaced with a fresh one 24 h after transfection, and then the cells were treated with 10 μM JNK inhibitor (SP600125) or vehicle control (0.1% DMSO) for 24 h. The levels of Aβ40 and Aβ42 secreted in the medium were measured using ELISA kits. The obtained Aβ levels were normalized to the amount of total protein.
TUNEL Assay
Apoptosis was assessed by the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay using an ApopTag Fluorescein Direct In Situ Apoptosis Detection Kit (Chemicon, Temecula, USA). Primary cultured neurons transfected with control or Tid1 siRNA were treated with 0, 5, or 10 μM Aβ42 for 24 h. The cells were fixed with 1% paraformaldehyde in PBS for 10 min at room temperature and permeabilized in EtOH: acetic acid (2:1) for 5 min at – 20 °C, and then the cells were washed with PBS. Fluorescein-conjugated nucleotide and TdT enzyme were added to the cells, which were then incubated for 1 h at 37 °C. Nuclei were stained with DAPI. Images were obtained using an AX70 fluorescence microscope (Olympus, Japan). The percentage of apoptotic cells was determined as the ratio of the number of DAPI-TUNEL-double-positive cells with respect to the total number of DAPI-positive cells.
ROS Detection
Intercellular ROS was assessed using a DCFDA probe with a ROS Detection Cell-Based Assay Kit (Cayman Chemical, USA). HEK293-APP cells transfected with the Tid1-L vector or Tid1 siRNA were incubated with 20 μM DCFDA in the dark for 30 min at 37 °C. Twenty millimolar N-acetyl cysteine (NAC) was added as a negative control. Intracellular hydrogen peroxide levels were assessed using a microplate reader (SpectraMax Gemini EM).
Statistical Analysis
Statistical analysis was performed using GraphPad prism software (GraphPad Software, San Diego, CA, USA). All values are presented as the mean ± SEM of at least three independent experiments.
Results
Tid1 Is Upregulated in the Hippocampus of AD Patients and Tg2576 Mice
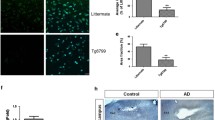
First, we investigated whether the Tid1 protein levels are altered in the hippocampus of AD patients. Total protein was extracted from the hippocampus, one of the most affected brain regions in AD, of AD patients (n = 10), and non-AD control (n = 10), and subjected to Western blot analysis. In mammals, two alternatively spliced forms of Tid1 are expressed, Tid1-L and Tid1-S, which differ only at their carboxyl-terminal tails [19]. We found that the protein levels of both Tid1-L and Tid1-S in the hippocampus of AD patients were higher than those in the hippocampus of non-AD control (Fig. 1a–c). We also determined the Tid1 protein levels in the hippocampus of Tg2576 mice overexpressing human APP with the Swedish mutation, which is linked to early-onset familial AD. These mice showed that the levels of Aβ begin to increase at 6–7 months and subsequent Aβ deposition is observed at approximately 9 months of age [25]. Total protein was extracted from the hippocampus of Tg2576 mice and WT mice at 6, 11, or 18 months of age (n = 6 per each age). We found that both Tid1-L and Tid1-S protein levels were significantly upregulated in the 6- and 11-month-old Tg2576 mice compared with the age-matched WT mice. In the hippocampus of 18-month-old Tg2576 mice, the Tid1-L levels were also significantly upregulated compared with those in the age-matched WT mice. At 18 months of age, although the Tid1-S levels did not significantly change, they tended to be higher than those in the age-matched WT mice (Fig. 1d–f). These results suggest that Tid1 induction occurred at an early stage of age, and Aβ may increase Tid1 levels because Aβ production in Tg2576 mice is higher than that in WT mice.
Tid1 protein levels are upregulated in the hippocampus of AD patients and Tg2576 mice. a–c Tid1 protein levels in the hippocampus of AD patients (n = 10) and non-AD control (n = 10) were determined by Western blot analysis using the anti-Tid1 antibody. a A representative immunoblot is shown. b and c The protein levels of Tid1-Long (Tid1-L) and Tid1-Short (Tid1-S) were quantified by densitometry, and their amounts were normalized to that of actin, and expressed as a value relative to that of non-AD as a control. All the values are presented as the mean ± SEM, *p < 0.05 vs non-AD, as determined by Student’s t test. d–f Tid1 protein levels in the hippocampus of Tg2576 mice and age-matched wild-type (WT) mice. Lysates were prepared from the hippocampus of 6-, 11-, and 18-month-old Tg2576 (Tg) mice (n = 6 per each age) and age-matched wild-type (WT) mice (n = 6 per each age). The protein levels of Tid1 were determined by Western blot analysis using the anti-Tid1 antibody. d A representative immunoblot is shown. e and f Tid1-Long (Tid1-L) and Tid1-Short (Tid1-S) were quantified by densitometry, and their amounts were normalized to that of actin, and expressed as a value relative to that in the 6-month-old WT mice as a control. All the values are presented as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001 vs age-matched WT mice, N.S., no significant difference, as determined by two-way ANOVA
Aβ42 Increases Tid1 Expression Levels in Cultured Rat Cortical Neurons
Aβ induces mitochondrial impairment at an early stage of AD [26, 27]. Therefore, we hypothesized that the upregulated Tid1 protein levels in the hippocampus of AD patients and Tg2576 mice are due to the increased Aβ level. To test this hypothesis, we treated primary rat cortical neurons with 5 μM Aβ42 for 0, 1, 2, 3, or 6 h, and determined the Tid1 protein levels by Western blot analysis. We observed that Aβ42 significantly increased both Tid1-L and Tid1-S levels 1 and 2 h after the treatment and then gradually decreased until 6 h (Fig. 2a–c). Next, we measured the mRNA expression levels of Tid1 in these cells after Aβ42 treatment by semiquantitative real-time PCR analysis. As shown in Fig. 2d, the mRNA expression levels of Tid1 significantly increased 1 h after Aβ42 treatment, reached a peak at 2 h, and then slightly decreased. These findings indicate that the increase in Tid1 protein levels induced by Aβ42 is caused by Tid1 gene expression. Taken together, these results suggest that the upregulated Tid1 protein levels found in the hippocampus of AD patients and Tg2576 mice may be caused by Aβ, and Tid1 induction appeared at an early stage in response to Aβ.
Aβ42 increases Tid1 protein and mRNA expression levels in primary rat cortical neurons. a–c Primary cultured neurons were incubated with 5 μM Aβ42 for 0, 1, 2, 3, or 6 h and then lysed. The protein levels of Tid1 were determined by Western blot analysis using the anti-Tid1 antibody. a A representative immunoblot is shown. b and c The protein levels of Tid1-Long (Tid1-L) and Tid1-Short (Tid1-S) were quantified by densitometry, and their amounts were normalized to that of actin, and expressed as a value relative to that of without Aβ42 treatment. d Primary cultured neurons were incubated with 5 μM Aβ42 for 0, 1, 2, 3, or 6 h. The Tid1 mRNA expression levels were determined by real-time PCR analysis. The expression levels of Tid1 mRNA were normalized to the corresponding amount of actin mRNA and expressed as a value relative to that of without Aβ42 treatment. All the values are presented as the mean ± SEM of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 vs without Aβ42 treatment, N.S., no significant difference, as determined by Student’s t test
Knockdown of Tid1 in Primary Cultured Neurons Protects Against Aβ42-Induced Neurotoxicity
Tid1 is involved in apoptosis and cell survival, and it is negatively associated with the proliferation of tumor cells [19, 28]. Therefore, we examined whether Tid1 is involved in Aβ42-induced neuronal cell death. For this purpose, we first decreased the Tid1 levels in primary cultured neurons by Tid1 siRNA transfection. The cells were transfected with control or Tid1 siRNA, as described in Materials and methods. Seventy-two hours after transfection, the cells were treated with 0, 5, or 10 μM Aβ42 for 24 h. Apoptotic cells were analyzed by TUNEL assay. In control siRNA-transfected cells, the percentage of TUNEL-positive cells increased after Aβ42 treatment in an Aβ42 dose-dependent manner. However, the percentage of TUNEL-positive cells was reduced in Tid1 siRNA-transfected cells compared with control siRNA-transfected cells after Aβ42 treatment (Fig. 3a and b). These findings suggest that Tid1 mediates neuronal cell death in response to Aβ42 treatment.
Tid1 regulates caspase-3-dependent apoptosis. a and b Primary cultured neurons were transfected with control or Tid1 siRNA for 72 h and then incubated with 0, 5, or 10 μM Aβ42 for 24 h, and apoptosis was analyzed by TUNEL assay. Cell nuclei were also stained with DAPI. a Representative images showing DAPI-stained and TUNEL-positive cells. b Quantification of TUNEL-positive cells among DAPI-stained cells. TUNEL-positive cells were counted from twelve randomly selected fields in three independent experiments. All the values are presented as the mean ± SEM of three independent experiments. **p < 0.01, ***p < 0.001 vs control siRNA, Aβ42 0 μM. ###p < 0.001, N.S., no significant difference, as determined by two-way ANOVA. c and d Primary cultured neurons were transfected with control or Tid1 siRNA for 72 h and then incubated with 0 or 5 μM Aβ42 for 24 h. The protein levels of Tid1 and cleaved caspase-3 were determined by Western blot analysis using the anti-Tid1 and anti-caspase-3 antibodies respectively. c A representative immunoblot is shown, and bands were quantified by densitometry, and their amounts were normalized to that of actin, and expressed as a value relative to that of control (d). All the values are presented as the mean ± SEM of three independent experiments. *p < 0.05, ***p < 0.001 vs control siRNA, Aβ42 – . #p < 0.05, N.S., no significant difference, as determined by one-way ANOVA. e and f HEK293-APP cells were transfected with the mock (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h. The protein levels of Tid1 and cleaved caspase-3 were determined by Western blot analysis using the anti-Tid1 and anti-caspase-3 antibodies respectively. e A representative immunoblot is shown, and bands were quantified by densitometry, normalized to actin, and expressed as a value relative to that of the mock vector (f). All the values are presented as the mean ± SEM of three independent experiments. **p < 0.01 vs mock vector, as determined by Student’s t test
Tid1-Mediated Apoptosis Is Caspase-3-Dependent
Next, we examined whether Tid1-mediated neuronal cell death after Aβ42 treatment is dependent on caspase-3 activity. Primary cultured neurons were transfected with control or Tid1 siRNA for 72 h, and then the cells were treated with 0 or 5 μM Aβ42 for 24 h. The cleaved caspase-3 levels were determined by Western blot analysis. Under non-Aβ42-treatment condition, the knockdown of Tid1 did not change the cleaved caspase-3 levels compared with control siRNA transfection. The cleaved caspase-3 levels in Tid1 siRNA-transfected cells were lower than those in control siRNA-transfected cells after Aβ42 treatment (Fig. 3c and d). These findings indicate that caspase-3 activity was involved in Tid1-mediated neuronal cell death induced by Aβ42. Since the Tid1 protein levels were upregulated in the hippocampus of AD patients and Tg2576 mice, we further investigated whether the overexpression of Tid1 can induce apoptosis. For this purpose, we used HEK293-APP cells stably expressing human APP695 because the transfection efficiency of primary cultured neurons is too low. It has been reported that the Tid1-L and Tid1-S isoforms have opposing effects on apoptosis induced by exogenous stimuli, and elevated levels of Tid1-L promote apoptosis, whereas elevated levels of Tid1-S inhibit apoptosis [19]. Thus, we used the Tid1-L plasmid vector for overexpression experiments in HEK293-APP cells. HEK293-APP cells were transiently transfected with the Myc-tagged Tid1-L (Myc-Tid1-L) or mock control (Myc) vector. We found that the overexpression of Tid1-L significantly increased the cleaved caspase-3 levels compared with those of the mock control vector (Fig. 3e and f). This finding indicates that Tid1-L overexpression can induce apoptosis in HEK293-APP cells in the absence of stimuli.
Tid1 Regulates ROS Generation
The overexpression of Tid1-L enhances the generation of ROS in normal fibroblast cells and cancer cell lines [22], which trigger caspase-3-mediated apoptosis. Thus, we investigated the effect of Tid1-L on ROS generation after Tid1-L overexpression or Tid1 knockdown in HEK293-APP cells. ROS levels were measured using DCFH-DA, a membrane-permeable non-fluorescent dye. The membrane-permeable dye was cleaved to DCFH with intracellular esterase, and then the cleaved dye was oxidized with ROS to produce a green fluorescence. HEK293-APP cells were transfected with the Myc-tagged Tid1-L or mock control vector for 48 h, and then the cells were labeled with DCFH-DA and analyzed using a microplate reader to detect ROS generation. The overexpression of Tid1-L increased the DCF fluorescence intensity relative to the mock control vector transfection, indicating ROS generation, which was reversed by the simultaneous presence of 20 mM N-acetyl-cysteine (NAC) (Fig. 4a). Conversely, the knockdown of Tid1 in these cells decreased ROS levels relative to control siRNA transfection (Fig. 4b). Therefore, these results suggest that Tid1-mediated cell death is caused in part by the generation of ROS.
Tid1 regulates ROS generation. a HEK293-APP cells were transfected with the mock control (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h. b HEK293-APP cells were transfected with control or Tid1 siRNA for 72 h. ROS levels were determined using a DCFDA probe. 20 mM N-acetyl-cysteine (NAC) was used as a ROS inhibitor. Quantification results were expressed as a value relative to that of control. All the values are presented as the mean ± SEM of three to six independent experiments. *p < 0.05, **p < 0.01 vs mock vector, NAC – , ***p < 0.001 vs control siRNA, NAC – , ###p < 0.001, as determined by one-way ANOVA
Tid1 Increases Aβ Production
It was suggested previously that ROS positively contribute to Aβ production [29]. Therefore, we further investigated the effect of Tid1 on Aβ production using HEK293-APP cells and primary cultured neurons. HEK293-APP cells were transfected with the Tid1-L or mock control vector for 48 h, and then the levels of Aβ40 and Aβ42 in the medium of the cells were measured by ELISA. We found that both Aβ40 and Aβ42 levels in the medium of the cells transfected with the Tid1-L vector were significantly increased compared with those of cells transfected with the mock control vector (Fig. 5a and b). Conversely, the knockdown of Tid1 in HEK293-APP cells reduced the production of both Aβ40 and Aβ42 (Fig. 5c and d). These results were further confirmed in primary cultured neurons, that is, the knockdown of Tid1 reduced the production of both Aβ40 and Aβ42 compared with control siRNA transfection (Fig. 5e and f). These results indicate that the overexpression of Tid1 promotes Aβ production.
Tid1 regulates Aβ level in HEK293-APP cells and primary cultured neurons. a and b HEK293-APP cells were transfected with the mock control (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h. c and d HEK293-APP cells were transfected with the control or Tid1 siRNA for 72 h. e and f Primary cultured neurons were transfected with control or Tid1 siRNA for 72 h. The levels of Aβ40 and Aβ42 secreted in the medium were measured by sandwich ELISA. The Aβ levels were normalized to the amount of total protein in the cells. All the values are presented as the mean ± SEM of three to six independent experiments. *p < 0.05 vs control siRNA, ***p < 0.001 vs mock vector, as determined by Student’s t test
Tid1-Induced Aβ Production Is Mediated by BACE1
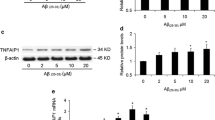
Altered levels of APP processing enzymes, such as ADAM10 (α-secretase), BACE1 (β-secretase), and PS1 (γ-secretase component), affect the production of Aβ. To study the mechanism underlying Tid1-induced Aβ production, we investigated the effect of Tid1 on the levels of these proteins in HEK293-APP cells and primary cultured neurons. Western blot analysis showed that Tid1-L vector-transfected cells expressed higher levels of BACE1 than the cells transfected with the mock control vector, but there were no significant differences in APP, PS1, and ADAM10 levels between the mock control vector- and Tid1-L vector-transfected cells (Fig. 6a and b). Conversely, the knockdown of Tid1 in HEK293-APP cells reduced the BACE1 levels, but not the APP, PS1, and ADAM10 levels (Fig. 6c and d). Similar results were obtained in primary cultured neurons, that is, the knockdown of Tid1 decreased the BACE1 levels without altering the levels of APP, PS1, and ADAM10 (Fig. 6e and f). These results indicate that Tid1-induced Aβ production is mediated by the BACE1 levels. APP-C99 is the product of β-secretase and harbors α- and γ-cleavage sites, but not β-cleavage sites. Therefore, to further clarify whether Tid1-induced Aβ production is dependent on the activation of β-secretase but not that of α- or γ-secretase, HEK293-C99 cells were transfected with the Tid1-L or mock control vector for 48 h, and then the levels of Aβ40 and Aβ42 in the medium of the cells were measured using an ELISA kit. We did not find any significant difference in neither Aβ40 nor Aβ42 level between the mock control vector- and Tid1-L vector-transfected cells (Supplementary Figs. A and B). Furthermore, Tid1 knockdown in HEK293-C99 cells did not change the Aβ40 and Aβ42 levels (Supplementary Figs. C and D). Taken together, these results indicate that Tid1-mediated increases in Aβ40 and Aβ42 levels are caused by the increased BACE1 levels.
Tid1 regulates BACE1 level. a and b HEK293-APP cells were transfected with the mock control (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h. c and d HEK293-APP cells were transfected with control or Tid1 siRNA for 72 h. e and f Primary cultured neurons were transfected with control or Tid1 siRNA for 72 h. The protein levels of Tid1, APP, BACE1, PS1, and ADAM10 were determined by Western blot analysis using the anti-Tid1, anti-22C11, anti-BACE1, anti-PS1, and anti-ADAM10 antibodies respectively. a, c, and e Representative immunoblots are shown, and bands were quantified by densitometry, normalized to actin, and expressed as a value relative to that of control b, d, and f. All the values are presented as the mean ± SEM of three to five independent experiments. *p < 0.05, ***p < 0.001 vs control, N.S., no significant difference, as determined by Student’s t test
Tid1 Increases Aβ Production via JNK Activation
Several lines of evidence suggest that oxidative stress induces JNK activation, which further regulates apoptosis and induces Aβ production [30,31,32,33]. We therefore investigated whether JNK activation is involved in Tid1-mediated Aβ production. For this purpose, HEK293-APP cells were transfected with the Tid1-L or mock control vector for 48 h, and then the cells were treated with the vehicle (control) or 10 μM JNK inhibitor (SP600125) for 24 h. We found that the overexpression of Tid1-L increased the phosphorylated (p-) JNK levels compared with mock control vector transfection, and the Tid1-mediated increases in the p-JNK levels were decreased by the treatment with the JNK inhibitor (Fig. 7a and b). The Tid1-mediated increases in the BACE1 levels were also decreased by the treatment with the JNK inhibitor (Fig. 7a and c). Next, we further investigated Aβ production and found that the JNK inhibitor reduced the Tid1-mediated increases in Aβ40 and Aβ42 levels (Fig. 7d and e). These results suggest that the Tid1-mediated activation of JNK contributes to the production of Aβ.
JNK inhibitor rescues Tid1-mediated Aβ production. HEK293-APP cells were transfected with the mock control (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h, and the cells were treated with 10 μM SP600125 (JNK inhibitor) for 24 h. a–c The protein levels of Tid1, p-JNK, JNK, and BACE1 were determined by Western blot analysis using the anti-Tid1, anti-p-JNK, anti-JNK, and anti-BACE1 antibodies respectively. a A representative immunoblot is shown, and the p-JNK levels were quantified by densitometry, and normalized to JNK levels, and expressed as a value relative to that of control (b). The BACE1 levels were quantified by densitometry, normalized to actin levels, and expressed as a value relative to that of control (c). All the values are presented as the mean ± SEM of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 vs mock vector, SP600125 – , #p < 0.05, N.S., no significant difference, as determined by one-way ANOVA. d and e The levels of Aβ40 and Aβ42 secreted in the medium were measured by sandwich ELISA. Quantification results were expressed as a value relative to that of the mock control vector, SP600125. All the values are presented as the mean ± SEM of three independent experiments. *p < 0.05, **p < 0.01 vs mock vector, SP600125, #p < 0.05, ##p < 0.01, N.S., no significant difference, as determined by one-way ANOVA
Tid1 Has No Effect on Tau Phosphorylation
The hyperphosphorylation of tau is a hallmark of synaptic dysfunction and neurodegeneration in the brain of patients with AD. Therefore, we also determined whether Tid1-L can affect tau phosphorylation. The phosphorylated (p-) tau levels after Tid1-L overexpression in Neuro2A-P301L cells, which stably express the human 2N4R tau isoform and the repeat domain fragment with the P301L mutation [24], were determined by Western blot analysis. We found that the overexpression of Tid1-L did not affect the p-tau levels (Fig. 8a and b). Next, we also examined whether the knockdown of Tid1 in primary cultured neurons can affect the p-tau levels. The phosphorylation of AKT (p-AKT) and GSK3α/β (p-GSK3α/β), which are essential components of PI3K signaling to reduce tau phosphorylation, was also examined. We did not find any significant difference in neither p-tau, p-AKT, nor p-GSK3α/β levels between Tid1 siRNA- and control siRNA-transfected primary cultured neurons (Fig. 8c and d). These results indicate that Tid1 is not involved in tau phosphorylation.
Tid1 has no effect on the phosphorylation of tau. a and b Neuro2A-P301L cells were transfected with the mock control (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h. c and d Primary cultured neurons were transfected with control or Tid1 siRNA for 72 h. The protein levels of Tid1, p-tau, total tau, p-AKT, AKT, p-GSK3α/β, and GSK3α/β were determined by Western blot analysis using the anti-Tid1, anti-AT180, anti-Tau5, anti-p-AKT, anti-AKT, anti-p-GSK3α/β, and anti-GSK3α/β antibodies respectively. a and c Representative immunoblots are shown, and bands were quantified by densitometry, and expressed as a value relative to that of control (b and d). All the values are presented as the mean ± SEM of three independent experiments. ***p < 0.001 vs control, N.S., no significant difference, as determined by Student’s t test
Discussion
Several lines of evidence suggest that mitochondrial dysfunction is an important factor in the pathogenesis of neurodegenerative diseases including AD, Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS) [34,35,36]. Several mitochondrial and metabolic abnormalities have been identified in the brain of AD patients compared with the age-matched control. Furthermore, mitochondrial oxidative changes and synaptic abnormalities have been reported as early events in AD progression [37,38,39]. Moreover, oxidative stress has been proposed to be an important factor in the pathogenesis of AD and contributes to Aβ generation and NFT formation, resulting in neuronal cell death [40,41,42]. However, the causal factors for mitochondrial oxidative damage in AD pathogenesis are still unclear.
Tid1 is proposed to play a role in multiple signal transduction processes including neurite outgrowth, apoptosis, and mitochondrial protein translocation. However, the involvement of Tid1 in AD pathogenesis, as well as its expression in the brains of patients with AD, is as yet unclarified. In this study, we first investigated whether the Tid1 protein levels are altered in the hippocampus of AD patients and Tg2576 mice, and also determined the effect of Tid1 on neuronal cell death and Aβ generation in primary cultured neurons and HEK293-APP cells. As a result, we found that the Tid1 protein levels were upregulated in the hippocampus of AD patients and Tg2576 mice, and the treatment of primary cultured neurons with Aβ42 significantly increased the Tid1 levels at the early stage, which suggests that the upregulated Tid1 protein levels found in the brains of AD patients and Tg2576 mice may be caused by Aβ. Moreover, the overexpression of Tid1-L increased ROS generation, apoptosis, and Aβ production, and these effects were reversed by Tid1 knockdown.
Recently, a time-course global gene expression study of Tg2576 mice and age-matched non-transgenic littermates has revealed that the genes related to mitochondrial energy metabolism and apoptosis were upregulated in Tg2576 mice before Aβ plaques were evident [18], which suggest that mitochondrial impairment is an early event in AD progression. Tg2576 mice showed that the levels of Aβ begin to increase at 6–7 months and subsequent Aβ deposition is observed at approximately 9 months of age [25]. In our study, we found that elevated Tid1 protein levels in the hippocampus of Tg2576 mice appeared at 6 months of age compared with age-matched WT mice. This finding suggests that the induction of Tid1 at an early stage may contribute to mitochondrial impairment. Treatment of N2a cells and neurons with Aβ induced abnormal mitochondrial dynamics through decreased mRNA levels of mitochondrial fusion genes and increased the expression levels of fission genes [43, 44]. Recent studies suggest that Aβ interacts with a mitochondrial fission protein, dynamic-related protein 1 (Drp1), which causes excessive fragmentation of mitochondria, leading to abnormal mitochondrial dynamics and synaptic degeneration in AD neurons and Aβ increases its mRNA expression levels in mouse neuroblastoma (N2a) cells [45]. It has been reported that elevated Tid1 protein levels induced Drp1-dependent mitochondrial fragmentation, which suggests that Tid1 acts as an inducer of mitochondrial fragmentation [22]. Therefore, we considered that the increase in Tid1 protein levels was due to Aβ, and we found that Aβ42 treatment significantly increased the expression levels of Tid1 mRNA and its protein in primary cultured neurons. This result suggests that the increase in Tid1 protein levels in the hippocampus of 6-month-old Tg2576 mice could be triggered by Aβ similarly to the Aβ-mediated increase in Tid1 expression levels observed in cultured cortical neurons. Although the mechanism by which the Tid1 levels increased in Aβ42-stimulated primary neurons is as yet unclarified, one possibility is that Aβ42 induces the impairment of mitochondria, consequently affecting mitochondrial protein Tid1 levels.
The reduction in physiologic levels of Tid1 in cancer cell lines causes resistance to apoptosis induced by multiple exogenous stimuli, including tumor necrosis factor α [28, 46], suggesting that Tid1 has its tumor suppressor activity. In this study, we also examined the effect of Tid1 on neuronal cell death induced by Aβ42 in primary cultured neurons, and we found that the knockdown of Tid1 by Tid1 siRNA transfection significantly decreased both the percentage of TUNEL-positive cells and cleaved caspase-3 levels after Aβ42 treatment. These findings suggest that the increased Tid1 expression levels may mediate the apoptotic function in primary cultured neurons against Aβ42-induced neurotoxicity. It has been reported that Aβ induces the expression and activation of p53, which plays an important role in promoting apoptosis in cultured neurons [47, 48]. Therefore, the increased Tid1 expression levels might simultaneously activate p53 to promote neuronal cell death, because Tid1 interacts with p53 leading to the promotion of apoptosis [21, 49]. Regarding apoptosis, the two Tid1 protein isoforms exhibit opposing effects on apoptosis in response to exogenous stimuli, as previously observed in protein overexpression studies. The expression of Tid1-L was found to promote apoptosis induced by the DNA-damaging agent mitomycin C, whereas Tid1-S expression suppressed it [19]. Here, we report that the ectopic expression of Tid1-L in HEK293-APP cells increased cleaved caspase-3 levels even in the absence of any stimuli. These findings are consistent with a previous finding that elevated Tid1-L levels induce apoptosis in the absence of any stimuli [22, 28]. Furthermore, the depletion of both endogenous Tid1-L and Tid1-S strongly protects primary cultured neurons from Aβ-induced apoptosis, which is in agreement with a previous finding that the depletion of both endogenous Tid1-L and Tid1-S isoforms resistance to apoptosis mediated by multiple exogenous stimuli [46].
Impaired mitochondrial function results in excessive ROS generation and the subsequent oxidative damage to neurons; oxidative stress is characteristic of AD brains [15]. In this study, we found that the overexpression of Tid1-L increased ROS generation, whereas the depletion of Tid1 reduced ROS generation in HEK293-APP cells. It has been reported that Tid1 induce Drp1-mediated mitochondrial fragmentation associating with increased ROS level in cancer cells [22]. The increased ROS generation observed in Tid1-L overexpression cells might be induced by Tid1-mediated mitochondrial fragmentation. These findings indicate the involvement of ROS generation and caspase-3 activation in Tid1-induced cytotoxicity.
Aβ is generated from APP by a two-step proteolytic cleavage by β-secretase and γ-secretase. Oxidative stress is enhanced during normal aging and is believed to be an early event in AD pathogenesis [44, 48], and oxidative stress has been known to increase the expression level and enzyme activity of BACE1 [50,51,52]. Moreover, the expression level and activity of BACE1 are increased in the brain of late-onset sporadic AD patients [53,54,55,56]. In this study, we also determined the effect of Tid1 on Aβ production in primary cultured neurons and HEK293-APP cells. Our study provides for the first time the finding that the overexpression of Tid1-L increased the production of both Aβ40 and Aβ42 in HEK293-APP cells. Conversely, the depletion of Tid1 in HEK293-APP cells and in primary cultured neurons decreased their production. Furthermore, the overexpression of Tid1-L significantly increased BACE1 levels without altering APP, ADAM10, and PS1 levels, which suggest that the Tid1-mediated increase in ROS levels increased BACE1 levels, resulting in Aβ production.
JNK is a member of the mitogen-activated protein kinase family and phosphorylates and activates transcription factors of the activator protein-1 family in response to various stresses, including oxidative stress [57]. Furthermore, JNK expression is upregulated in the brains of AD patients and Tg2576 mice [58, 59]. Additionally, the loss of JNK activity prevents the amyloidogenic cleavage of APP and the formation of amyloid plaques in vivo. Furthermore, JNK may contribute to the increase in Aβ production by promoting β-secretase-mediated APP cleavage. We also found that the overexpression of Tid1 activated JNK leading to increased Aβ production, whereas the inhibition of JNK by the JNK inhibitor prevented Tid1-mediated Aβ production. However, whether Tid1 is directly involved in JNK activation is unclear; one possible mechanism is that the overexpression of Tid1-L increases ROS generation which may in turn activate JNK.
In the present study, Tid1 increased Aβ production and conversely Aβ42 induced in increase of Tid1 expression levels. These two results implicate to that they are reciprocally regulated to synergistically facilitate neuronal cell death.
Although it has been shown that Tid1-L and Tid1-S exhibit opposing activities in regulating apoptosis, in general, other studies indicate that both isoforms of Tid1 display similar functions. For instance, both Tid1-L and Tid1-S promote autophagy flux by interacting with Beclin1-containing autophagy protein complex [60]. Also, the ratio of Tid1-L/S and/or modulation of relative expression levels of the two isoforms may affect the biological function [19]. In this study, the expression level of Tid1-S is higher in the brains compared with that of Tid1-L and is also increased in AD and Tg2576 brains along with Tid1L. Therefore, we cannot rule out the possibility that the elevated Tid1-S levels found in the hippocampus of AD patients and Tg2576 mice are also involved in the apoptosis induced by Aβ and Aβ production. Further investigation is necessary on whether the overexpression of Tid1-S can affect Aβ-induced neuronal cell death and Aβ production.
Conclusion
In summary, our findings suggest that the elevated levels of Tid1 found in the hippocampus of AD patients and Tg2576 mice are caused by Aβ, and the increased Tid1 levels promoted neuronal cell death through increased ROS generation and caspase-3 activity. Moreover, elevated Tid1 levels also induced Aβ production via increased BACE1 levels and JNK activity. Thus, Tid1 may be a suitable target for therapeutic intervention for AD.
References
Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297(5580):353–356. https://doi.org/10.1126/science.1072994
Karran E, Mercken M, De Strooper B (2011) The amyloid cascade hypothesis for Alzheimer’s disease: an appraisal for the development of therapeutics. Nat Rev Drug Discov 10(9):698–712. https://doi.org/10.1038/nrd3505
Buxbaum JD, Liu KN, Luo Y, Slack JL, Stocking KL, Peschon JJ, Johnson RS, Castner BJ et al (1998) Evidence that tumor necrosis factor alpha converting enzyme is involved in regulated alpha-secretase cleavage of the Alzheimer amyloid protein precursor. J Biol Chem 273(43):27765–27767. https://doi.org/10.1074/jbc.273.43.27765
Hussain I, Powell D, Howlett DR, Tew DG, Meek TD, Chapman C, Gloger IS, Murphy KE et al (1999) Identification of a novel aspartic protease (Asp 2) as beta-secretase. Mol Cell Neurosci 14(6):419–427. https://doi.org/10.1006/mcne.1999.0811
Lammich S, Kojro E, Postina R, Gilbert S, Pfeiffer R, Jasionowski M, Haass C, Fahrenholz F (1999) Constitutive and regulated alpha-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc Natl Acad Sci U S A 96(7):3922–3927. https://doi.org/10.1073/pnas.96.7.3922
Lin X, Koelsch G, Wu S, Downs D, Dashti A, Tang J (2000) Human aspartic protease memapsin 2 cleaves the beta-secretase site of beta-amyloid precursor protein. Proc Natl Acad Sci U S A 97(4):1456–1460. https://doi.org/10.1073/pnas.97.4.1456
Benard G, Bellance N, James D, Parrone P, Fernandez H, Letellier T, Rossignol R (2007) Mitochondrial bioenergetics and structural network organization. J Cell Sci 120(Pt 5):838–848. https://doi.org/10.1242/jcs.03381
Caspersen C, Wang N, Yao J, Sosunov A, Chen X, Lustbader JW, Xu HW, Stern D et al (2005) Mitochondrial Abeta: a potential focal point for neuronal metabolic dysfunction in Alzheimer’s disease. FASEB J 19(14):2040–2041. https://doi.org/10.1096/fj.05-3735fje
Manczak M, Anekonda TS, Henson E, Park BS, Quinn J, Reddy PH (2006) Mitochondria are a direct site of A beta accumulation in Alzheimer’s disease neurons: implications for free radical generation and oxidative damage in disease progression. Hum Mol Genet 15(9):1437–1449. https://doi.org/10.1093/hmg/ddl066
Sorrentino V, Romani M, Mouchiroud L, Beck JS, Zhang H, D’Amico D, Moullan N, Potenza F et al (2017) Enhancing mitochondrial proteostasis reduces amyloid-beta proteotoxicity. Nature 552(7684):187–193. https://doi.org/10.1038/nature25143
Moehle EA, Shen K, Dillin A (2019) Mitochondrial proteostasis in the context of cellular and organismal health and aging. J Biol Chem 294(14):5396–5407. https://doi.org/10.1074/jbc.TM117.000893
Baker MJ, Tatsuta T, Langer T (2011) Quality control of mitochondrial proteostasis. Cold Spring Harb Perspect Biol 3(7). https://doi.org/10.1101/cshperspect.a007559
Moreira PI, Cardoso SM, Santos MS, Oliveira CR (2006) The key role of mitochondria in Alzheimer’s disease. J Alzheimers Dis 9(2):101–110
Moreira PI, Carvalho C, Zhu X, Smith MA, Perry G (2010) Mitochondrial dysfunction is a trigger of Alzheimer’s disease pathophysiology. Biochim Biophys Acta 1802(1):2–10. https://doi.org/10.1016/j.bbadis.2009.10.006
Wang X, Wang W, Li L, Perry G, Lee HG, Zhu X (2014) Oxidative stress and mitochondrial dysfunction in Alzheimer’s disease. Biochim Biophys Acta 1842(8):1240–1247. https://doi.org/10.1016/j.bbadis.2013.10.015
Onyango IG, Dennis J, Khan SM (2016) Mitochondrial dysfunction in Alzheimer’s disease and the rationale for bioenergetics based therapies. Aging Dis 7(2):201–214. https://doi.org/10.14336/AD.2015.1007
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443(7113):787–795. https://doi.org/10.1038/nature05292
Reddy PH, McWeeney S, Park BS, Manczak M, Gutala RV, Partovi D, Jung Y, Yau V et al (2004) Gene expression profiles of transcripts in amyloid precursor protein transgenic mice: up-regulation of mitochondrial metabolism and apoptotic genes is an early cellular change in Alzheimer’s disease. Hum Mol Genet 13(12):1225–1240. https://doi.org/10.1093/hmg/ddh140
Syken J, De-Medina T, Munger K (1999) TID1, a human homolog of the Drosophila tumor suppressor l(2)tid, encodes two mitochondrial modulators of apoptosis with opposing functions. Proc Natl Acad Sci U S A 96(15):8499–8504. https://doi.org/10.1073/pnas.96.15.8499
Ng AC, Baird SD, Screaton RA (2014) Essential role of TID1 in maintaining mitochondrial membrane potential homogeneity and mitochondrial DNA integrity. Mol Cell Biol 34(8):1427–1437. https://doi.org/10.1128/MCB.01021-13
Ahn BY, Trinh DL, Zajchowski LD, Lee B, Elwi AN, Kim SW (2010) Tid1 is a new regulator of p53 mitochondrial translocation and apoptosis in cancer. Oncogene 29(8):1155–1166. https://doi.org/10.1038/onc.2009.413
Elwi AN, Lee B, Meijndert HC, Braun JE, Kim SW (2012) Mitochondrial chaperone DnaJA3 induces Drp1-dependent mitochondrial fragmentation. Int J Biochem Cell Biol 44(8):1366–1376. https://doi.org/10.1016/j.biocel.2012.05.004
Jung CG, Uhm KO, Horike H, Kim MJ, Misumi S, Ishida A, Ueda Y, Choi EK et al (2015) Auraptene increases the production of amyloid-beta via c-Jun N-terminal kinase-dependent activation of gamma-secretase. J Alzheimers Dis 43(4):1215–1228. https://doi.org/10.3233/JAD-141692
Hatakeyama S, Matsumoto M, Kamura T, Murayama M, Chui DH, Planel E, Takahashi R, Nakayama KI et al (2004) U-box protein carboxyl terminus of Hsc70-interacting protein (CHIP) mediates poly-ubiquitylation preferentially on four-repeat Tau and is involved in neurodegeneration of tauopathy. J Neurochem 91(2):299–307. https://doi.org/10.1111/j.1471-4159.2004.02713.x
Kawarabayashi T, Younkin LH, Saido TC, Shoji M, Ashe KH, Younkin SG (2001) Age-dependent changes in brain, CSF, and plasma amyloid (beta) protein in the Tg2576 transgenic mouse model of Alzheimer’s disease. J Neurosci 21(2):372–381
Hoekstra JG, Hipp MJ, Montine TJ, Kennedy SR (2016) Mitochondrial DNA mutations increase in early stage Alzheimer disease and are inconsistent with oxidative damage. Ann Neurol 80(2):301–306. https://doi.org/10.1002/ana.24709
Pagani L, Eckert A (2011) Amyloid-Beta interaction with mitochondria. Int J Alzheimers Dis 2011:925050. https://doi.org/10.4061/2011/925050
Chen CY, Chiou SH, Huang CY, Jan CI, Lin SC, Hu WY, Chou SH, Liu CJ et al (2009) Tid1 functions as a tumour suppressor in head and neck squamous cell carcinoma. J Pathol 219(3):347–355. https://doi.org/10.1002/path.2604
Cheignon C, Tomas M, Bonnefont-Rousselot D, Faller P, Hureau C, Collin F (2018) Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biol 14:450–464. https://doi.org/10.1016/j.redox.2017.10.014
Son Y, Cheong YK, Kim NH, Chung HT, Kang DG, Pae HO (2011) Mitogen-activated protein kinases and reactive oxygen species: how can ROS activate MAPK pathways? J Signal Transduct 2011:792639. https://doi.org/10.1155/2011/792639
Santabarbara-Ruiz P, Lopez-Santillan M, Martinez-Rodriguez I, Binagui-Casas A, Perez L, Milan M, Corominas M, Serras F (2015) ROS-induced JNK and p38 signaling is required for unpaired cytokine activation during Drosophila regeneration. PLoS Genet 11(10):e1005595. https://doi.org/10.1371/journal.pgen.1005595
Oswald MCW, Garnham N, Sweeney ST, Landgraf M (2018) Regulation of neuronal development and function by ROS. FEBS Lett 592(5):679–691. https://doi.org/10.1002/1873-3468.12972
Shen C, Chen Y, Liu H, Zhang K, Zhang T, Lin A, Jing N (2008) Hydrogen peroxide promotes Abeta production through JNK-dependent activation of gamma-secretase. J Biol Chem 283(25):17721–17730. https://doi.org/10.1074/jbc.M800013200
Haun F, Nakamura T, Lipton SA (2013) Dysfunctional mitochondrial dynamics in the pathophysiology of neurodegenerative diseases. J Cell Death 6:27–35. https://doi.org/10.4137/JCD.S10847
Haun F, Nakamura T, Shiu AD, Cho DH, Tsunemi T, Holland EA, La Spada AR, Lipton SA (2013) S-nitrosylation of dynamin-related protein 1 mediates mutant huntingtin-induced mitochondrial fragmentation and neuronal injury in Huntington’s disease. Antioxid Redox Signal 19(11):1173–1184. https://doi.org/10.1089/ars.2012.4928
Mishra P, Chan DC (2014) Mitochondrial dynamics and inheritance during cell division, development and disease. Nat Rev Mol Cell Biol 15(10):634–646. https://doi.org/10.1038/nrm3877
Aluise CD, Robinson RA, Beckett TL, Murphy MP, Cai J, Pierce WM, Markesbery WR, Butterfield DA (2010) Preclinical Alzheimer disease: brain oxidative stress, Abeta peptide and proteomics. Neurobiol Dis 39(2):221–228. https://doi.org/10.1016/j.nbd.2010.04.011
Pallas M, Camins A, Smith MA, Perry G, Lee HG, Casadesus G (2008) From aging to Alzheimer’s disease: unveiling “the switch” with the senescence-accelerated mouse model (SAMP8). J Alzheimers Dis 15(4):615–624
Singh M, Dang TN, Arseneault M, Ramassamy C (2010) Role of by-products of lipid oxidation in Alzheimer’s disease brain: a focus on acrolein. J Alzheimers Dis 21(3):741–756. https://doi.org/10.3233/JAD-2010-100405
Aliev G (2011) Oxidative stress induced-metabolic imbalance, mitochondrial failure, and cellular hypoperfusion as primary pathogenetic factors for the development of Alzheimer disease which can be used as a alternate and successful drug treatment strategy: past, present and future. CNS Neurol Disord Drug Targets 10(2):147–148
Butterfield DA, Boyd-Kimball D (2004) Amyloid beta-peptide(1-42) contributes to the oxidative stress and neurodegeneration found in Alzheimer disease brain. Brain Pathol 14(4):426–432
Christen Y (2000) Oxidative stress and Alzheimer disease. Am J Clin Nutr 71(2):621S–629S. https://doi.org/10.1093/ajcn/71.2.621s
Manczak M, Mao P, Calkins MJ, Cornea A, Reddy AP, Murphy MP, Szeto HH, Park B et al (2010) Mitochondria-targeted antioxidants protect against amyloid-beta toxicity in Alzheimer’s disease neurons. J Alzheimers Dis 20(Suppl 2):S609–S631. https://doi.org/10.3233/JAD-2010-100564
Wang X, Su B, Siedlak SL, Moreira PI, Fujioka H, Wang Y, Casadesus G, Zhu X (2008) Amyloid-beta overproduction causes abnormal mitochondrial dynamics via differential modulation of mitochondrial fission/fusion proteins. Proc Natl Acad Sci U S A 105(49):19318–19323. https://doi.org/10.1073/pnas.0804871105
Manczak M, Calkins MJ, Reddy PH (2011) Impaired mitochondrial dynamics and abnormal interaction of amyloid beta with mitochondrial protein Drp1 in neurons from patients with Alzheimer’s disease: implications for neuronal damage. Hum Mol Genet 20(13):2495–2509. https://doi.org/10.1093/hmg/ddr139
Edwards KM, Munger K (2004) Depletion of physiological levels of the human TID1 protein renders cancer cell lines resistant to apoptosis mediated by multiple exogenous stimuli. Oncogene 23(52):8419–8431. https://doi.org/10.1038/sj.onc.1207732
Kruman II, Wersto RP, Cardozo-Pelaez F, Smilenov L, Chan SL, Chrest FJ, Emokpae R Jr, Gorospe M et al (2004) Cell cycle activation linked to neuronal cell death initiated by DNA damage. Neuron 41(4):549–561
Uberti D, Ferrari Toninelli G, Memo M (2003) Involvement of DNA damage and repair systems in neurodegenerative process. Toxicol Lett 139(2-3):99–105
Trinh DL, Elwi AN, Kim SW (2010) Direct interaction between p53 and Tid1 proteins affects p53 mitochondrial localization and apoptosis. Oncotarget 1(6):396–404. https://doi.org/10.18632/oncotarget.100902
Paola D, Domenicotti C, Nitti M, Vitali A, Borghi R, Cottalasso D, Zaccheo D, Odetti P et al (2000) Oxidative stress induces increase in intracellular amyloid beta-protein production and selective activation of betaI and betaII PKCs in NT2 cells. Biochem Biophys Res Commun 268(2):642–646. https://doi.org/10.1006/bbrc.2000.2164
Tamagno E, Parola M, Bardini P, Piccini A, Borghi R, Guglielmotto M, Santoro G, Davit A et al (2005) Beta-site APP cleaving enzyme up-regulation induced by 4-hydroxynonenal is mediated by stress-activated protein kinases pathways. J Neurochem 92(3):628–636. https://doi.org/10.1111/j.1471-4159.2004.02895.x
Tong Y, Zhou W, Fung V, Christensen MA, Qing H, Sun X, Song W (2005) Oxidative stress potentiates BACE1 gene expression and Abeta generation. J Neural Transm (Vienna) 112(3):455–469. https://doi.org/10.1007/s00702-004-0255-3
Fukumoto H, Cheung BS, Hyman BT, Irizarry MC (2002) Beta-secretase protein and activity are increased in the neocortex in Alzheimer disease. Arch Neurol 59(9):1381–1389
Holsinger RM, McLean CA, Beyreuther K, Masters CL, Evin G (2002) Increased expression of the amyloid precursor beta-secretase in Alzheimer’s disease. Ann Neurol 51(6):783–786. https://doi.org/10.1002/ana.10208
Matsui T, Ingelsson M, Fukumoto H, Ramasamy K, Kowa H, Frosch MP, Irizarry MC, Hyman BT (2007) Expression of APP pathway mRNAs and proteins in Alzheimer’s disease. Brain Res 1161:116–123. https://doi.org/10.1016/j.brainres.2007.05.050
Yang LB, Lindholm K, Yan R, Citron M, Xia W, Yang XL, Beach T, Sue L et al (2003) Elevated beta-secretase expression and enzymatic activity detected in sporadic Alzheimer disease. Nat Med 9(1):3–4. https://doi.org/10.1038/nm0103-3
Davis RJ (2000) Signal transduction by the JNK group of MAP kinases. Cell 103(2):239–252
Puig B, Gomez-Isla T, Ribe E, Cuadrado M, Torrejon-Escribano B, Dalfo E, Ferrer I (2004) Expression of stress-activated kinases c-Jun N-terminal kinase (SAPK/JNK-P) and p38 kinase (p38-P), and tau hyperphosphorylation in neurites surrounding betaA plaques in APP Tg2576 mice. Neuropathol Appl Neurobiol 30(5):491–502. https://doi.org/10.1111/j.1365-2990.2004.00569.x
Zhu X, Raina AK, Rottkamp CA, Aliev G, Perry G, Boux H, Smith MA (2001) Activation and redistribution of c-jun N-terminal kinase/stress activated protein kinase in degenerating neurons in Alzheimer’s disease. J Neurochem 76(2):435–441
Niu G, Zhang H, Liu D, Chen L, Belani C, Wang HG, Cheng H (2015) Tid1, the mammalian homologue of Drosophila tumor suppressor Tid56, mediates macroautophagy by interacting with Beclin1-containing autophagy protein complex. J Biol Chem 290(29):18102–18110. https://doi.org/10.1074/jbc.M115.665950
Funding
This work was supported by a Grant-in-Aid for Scientific Research B (16H05559) and a Grant-in-Aid for challenging Exploratory Research (15K15712) (to M.M.) from the Ministry of Education, Culture, Sports, Science and Technology, Japan. This work was also supported by the Project of translational and clinical research seed A from Japan Agency for Medical Research and Development (AMED, A-128) (to M.M).
Author information
Authors and Affiliations
Contributions
C.Y.Z., F.T., M.A., and C.G.J. performed the experiments. C.Y.Z., C.G.J., and M.M designed all the experiments, analyzed the data, and wrote the paper. S.W.K. and H.A. contributed to the discussion of the experiments. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
All animal experiments were performed in accordance with institutional guidelines and approved by the Nagoya City University and all participants signed informed consent.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Fig. 1
Tid1 does not affect Aβ level in HEK293-C99 cells. (A and B) HEK293-C99 cells were transfected with the mock control (Myc) or Myc-tagged Tid1-L (Myc-Tid1-L) vector for 48 h. (C and D) HEK293-C99 cells were transfected with the control or Tid1 siRNA for 72 h. The levels of Aβ40 and Aβ42 secreted in the medium were measured by sandwich ELISA. The Aβ levels were normalized to the amount of total protein in the cells. All the values are presented as the mean ± SEM of three independent experiments. N.S., no significant difference, as determined by Student’s t-test. (PNG 776 kb)
Rights and permissions
About this article
Cite this article
Zhou, C., Taslima, F., Abdelhamid, M. et al. Beta-Amyloid Increases the Expression Levels of Tid1 Responsible for Neuronal Cell Death and Amyloid Beta Production. Mol Neurobiol 57, 1099–1114 (2020). https://doi.org/10.1007/s12035-019-01807-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-01807-2