Abstract
Chronic relapsing experimental allergic encephalomyelitis (CR-EAE) exhibits neuropathological and immunological dysfunctions similar to those found in multiple sclerosis (MS) and has been used as an animal model of MS. Inflammatory infiltrates and oxidative stress have been linked to the development of both diseases. Ethanolamine plasmalogen derivates have been shown to be powerful antioxidants and immunomodulators. Therefore, the objective of this study was to analyse inflammatory infiltrates, the state of the oxidative defences and the possible protective effects of calcium, magnesium and phosphate ethanolamine (EAP) in the CR-EAE rat hippocampus. To this aim, we evaluated, by immunohistochemistry, T cell infiltrates, Iba-1+ (a marker of activated microglia) immunoreactivity and TUNEL (+) cells. We also measured the protein levels and activity of the antioxidant enzymes catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GP) and glutathione reductase (GR). In addition, reduced (GSH) and oxidized (GSSG) glutathione levels, lipid peroxidation and cholesterol as well as desmosterol content were determined. We found an increase in T cell infiltrates and Iba1+ immunoreactivity, lipid peroxidation, SOD, GP and GR activities as well as enhanced cholesterol levels and a decrease in CAT activity, GSH and desmosterol levels in the first and second attack in the CR-EAE rat hippocampus. Pretreatment of CR-EAE rats with EAP led to a delay in the onset of the clinical signs of the disease as well as a decrease in inflammatory infiltrates and alterations of the antioxidant defences in the hippocampus. Altogether, the present results suggest a protective role of EAP in the CR-EAE rat hippocampus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory and demyelinating disease of the central nervous system (CNS) that represents the most frequent cause of chronic disability in young adults [1]. The chronic relapsing form of experimental allergic encephalomyelitis (CR-EAE) is an autoimmune disease widely used as an animal model of MS due to the autoimmune, histopathological, genetic and clinical similarities [2,3,4]. MS is considered a two-stage disease that consists of an inflammatory phase and a neurodegenerative phase [5]. At the histological level, inflammatory infiltrates accumulate within the CNS. These consist mainly of T cells [6], macrophages [7], B cells and granulocytes [8]. Activated macrophages and microglial cells have both been shown to produce a number of factors that can contribute to tissue damage, such as cytokines (TNF-α, IFN-γ), chemokines, nitroxides and reactive oxygen species (ROS) [9,10,11,12,13,14]. Although ROS play a physiological role in numerous cellular regulatory processes [15], oxidative stress occurs when the free radical generation exceeds the capacity of the antioxidant defences, leading to subsequent damage of different macromolecules, like proteins, lipids and nucleic acids [16].
MS has been classically regarded as a white matter disease. Recent studies, however, have convincingly shown that grey matter regions are also heavily affected [17]. In fact, some abnormalities in the grey matter have been consistently found with in vivo magnetic resonance [17]. Moreover, approximately 50–65% of MS patients experience cognitive deficits; among these, memory dysfunction is especially common [18, 19]. The cause of memory dysfunction is currently unknown, but recent studies have revealed hippocampal atrophy and neurodegeneration in MS [20] as well as hippocampal CA1 atrophy and synaptic loss during EAE [21]. The hippocampus, a brain area that acts as a memory device, seems to be strongly affected by the deleterious effects of oxidative insult [22]. In rats with the acute form of EAE, our group has previously reported alterations of the hippocampal somatostatinergic system, which is involved in behaviour and memory [23, 24].
Despite progress in recent years, an effective therapy that heals MS is not yet available. Most treatments against MS used today are based on immunomodulation. There are treatments, for example, that reduce the transfer of immune cells into the CNS parenchyma by interfering with the α4β1-integrin receptor on the surface of T cells [25,26,27] whereas other treatments sequester lymphocytes in lymph nodes, preventing them from contributing to an autoimmune reaction [28, 29]. Unfortunately, some of these treatments pose serious problems. Treatment with natalizumab, for instance, increases the risk of a potentially serious viral infection of the brain called progressive multifocal leukoencephalopathy in people who are positive for antibodies to the causative agent, the JC virus [30]. In EAE, a series of studies have revealed a protective effect of exogenous antioxidants [16, 31, 32]. However, despite promising results in the treatment of EAE, evidence for successful antioxidant therapies in MS patients is very limited.
Ethanolamine is a precursor in the biosynthesis of anandamide, an endocannabinoid whose levels are decreased in MS patients [33]. A study of the sclerotic plaques found in white matter of MS patients has revealed alterations in the composition of ethanolamine phosphoglycerides as compared to normal tissue [34,35,36,37]. In rat brain, certain evidence indicates that the plasmalogen phosphatidylethanolamine protects against oxidative stress due to its antioxidant effects against lipid peroxidation [38, 39]. In addition, ethanolamine plasmalogens have been shown to reduce membrane oxidizability and to have anti-inflammatory effects [39, 40] whereas certain phosphoethanolamine derivatives are inmunomodulators that regulate macrophage activation [41, 42].
In view of the above, the aim of the present study was to analyse the inflammatory infiltrates, the state of the antioxidant defence mechanisms and the possible preventive effects of a calcium, magnesium and phosphate ethanolamine salt (EAP) in the hippocampus of rats with CR-EAE at three different phases along the progression of the disease: first attack, remission and second attack.
Materials and Methods
Materials
Complete Freund’s adjuvant (CFA) and Mycobacterium tuberculosis H37RA were obtained from Difco (Detroit, MI, USA). Thiobarbituric acid, nitro blue tetrazolium (NBT), cacodylate, glutathione reductase (GR), trichloroacetic acid (TCA), 5,5′- dithiobis (2-nitrobenzoic acid) (DTNB) and bovine serum albumin (BSA) were obtained from Sigma-Aldrich (Madrid, Spain). Specific antibodies against catalase (C-0979) and GAPDH (SAB2500451) were purchased from Sigma-Aldrich (Madrid, Spain). Specific antibodies against superoxide dismutase (sc-8637), glutathione reductase (sc-133245) and glutathione peroxidase (sc-74498) were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA). Anti-goat (401515), anti-rabbit (401315) and anti-mouse (401215) secondary antibodies were obtained from Calbiochem (Madrid, Spain). Amersham Hybond-p PVDF membranes were purchased from GE-Healthcare Life Science (Barcelona, Spain). Specific antibodies against CD3+ (Sigma-Aldrich (C7930)) and CD8+ (Millipore.), Iba1+ (Wako, Japan) and immunoglobulin G (Becton, Dickinson, USA) were used for immunohistochemistry. To determine cell death, the DeadEnd™ Fluorometric TUNEL System (Promega, Wisconsin USA) was used. To amplify the signal, we used MaxVision, a rabbit polymer-peroxidase system (Master Diagnostica, Granada, Spain).
Experimental Animals
The animal experiments performed in the present study conform to the guidelines set by the Animal Care Committee of Alcalá University as well as to the International guidelines on the ethical use of animals set by the Council of Europe (Protection of Animals Used for Experimentation, 1986 ETS No. 123), and all experimental protocols were previously approved. Extreme efforts were made to minimize the number of animals used and their suffering. Sixty 6-week-old female Lewis rats were purchased from Harlan Interfauna Ibérica, S.L. (Barcelona, Spain). Rats were maintained on a 12-h light/dark cycle (07:00–19:00 h) regime and were provided food and water ad libitum. Rats were divided into the following experimental groups: control rats; rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE); rats with CR-EAE pretreated with a calcium, magnesium and phosphate ethanolamine salt (EAP) and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1); rats with CR-EAE pretreated with EAP and sacrificed in grade 3 (CR-EAE + EAPgrade 3) and control rats pretreated with EAP. Rats slightly narcotized with halothane were immunized with an emulsion of guinea-pig spinal cord tissue (GPSC) at a final concentration of 290 mg/ml buffered in PBS with an equal volume of complete Freund’s adjuvant (CFA) containing 11 mg/ml of Mycobacterium tuberculosis (strain H37RA) in a final volume of 100 μl [43, 44]. Control rats were injected only with PBS/CFA. Animals were weighed and monitored daily for clinical signs of CR-EAE, assessed according to the following criteria: 0, no clinical signs; 1, limp tail; 2, hind limb weakness; 3, complete hind limb paralysis [45].
EAP was freshly prepared in distilled water and buffered to approximate neutrality using HCl. EAP was injected i.p. at a dose of 600 mg/kg/day to CR-EAE and control rats for 2 days prior to induction. Rats were decapitated at the first or third stage of the disease. Rats from each group were sacrificed on days 15–17 following induction (at the first attack) or on day 24 after induction (at the remission phase). The group of CR-EAE + EAPgrade 3 rats was sacrificed in grade 3 on days 18–20. On day 28–30, rats from each group were sacrificed (at the second attack). The brains of the sacrificed rats were rapidly removed, and the hippocampus was dissected on ice according to the method of Glowinski and Iversen [46].
Immunohistochemistry
The brains were quickly removed, fixed with 10% (v/v) formaldehyde in phosphate-buffered saline (PBS) and embedded in paraffin wax. In this study, we evaluated similar areas of the medial section of the hippocampus rostro-caudal areas (Supplementary Fig. 1). Sections (5 μm) were deparaffined with xylene and rehydrated using graded ethanol concentrations. Antigen retrieval was achieved by incubation with 10 mM citrate buffer for 2 min using an express cooker. Endogenous peroxidase was inhibited with 3% H2O2 in H2O for 10 min and 0.3% H2O2 in methanol for 10 min at room temperature. After rinsing with Tris-buffered saline (TBS), the sections were incubated with blocking solution (3% normal donkey serum (NDS) plus 0.05% Triton X-100 in TBS) for 60 min to prevent non-specific background. Afterwards, the sections were incubated overnight at room temperature with the primary antibodies diluted in 0.3% NDS–0.05% triton X-100 in TBS: monoclonal rabbit anti-Iba-1 (Wako Chemicals USA) diluted 1:1500, polyclonal rabbit anti-CD3 (Sigma) diluted 1:1000, monoclonal mouse anti-CD8 (Millipore) diluted 1:1000 and polyclonal anti-IgG (Millipore) diluted 1/200. After washing with TBS, samples were incubated under dark conditions for 60 min at room temperature with the corresponding secondary antibody. For the immunoenzymatic detection method, the MaxVision rabbit polymer-peroxidase system (Master Diagnostica, Granada, Spain) was used. Peroxidase activity was detected with diaminobenzidine (DAB kit, Master Diagnostica). For determination of apoptosis in situ, brains embedded in paraffin wax were cut into 5-μm slices. Colorimetric apoptosis detection was performed with the terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) reaction according to the manufacturer’s instructions (Promega). After the reaction, slides were counterstained with Harris’ haematoxylin, dehydrated with increasing concentrations of ethanol and xylene, mounted with DePeX and observed under an optical microscope.
Preparation of Membrane and Cytoplasmic Fractions
The hippocampus was homogenized in a solution of 10 mM HEPES–KOH buffer (10% w/v) (pH 7.6) with a Brinkmann Polytron homogenizer (setting 5, 15 s). The homogenate was spun at 600×g for 5 min at 4 °C, and the resultant supernatant was subsequently centrifuged at 48,000×g for 30 min at 4 °C. The cytoplasmic fraction was aliquoted and immediately stored at − 80 °C. For each experimental animal, one aliquot was used to determine protein concentration following the Lowry method [47]. The resulting pellet was resuspended in 10 mM HEPES–KOH buffer (pH 7.6) (10% w/v), centrifuged as before, and the pellet was resuspended in 50 mM Tris–HCl buffer (pH 7.5). Aliquoted samples were immediately stored at − 80 °C until the day of assay. For each experimental animal, one aliquot was used to determine protein concentration following the Lowry method [47].
Measurement of Lipid Peroxidation
Lipid peroxidation was measured by assessing the levels of reactive species to thiobarbituric acid according to a slightly modified method of Ottolenghi [48]. Briefly, 100 μl of hippocampal membranes was suspended in 200 μl of trichloroacetic acid (TCA), sonicated during 15 s, incubated on ice for 15 min and then centrifuged in a tabletop centrifuge at 2200×g at 4 °C for 15 min. A small volume of each sample (200 μl) was mixed with an equal volume of thiobarbituric acid (TBA). The mixture was then placed in a boiling water bath for 10 min. The absorbance of the solution was read at 535 nm. The results were expressed in mmol of malondialdehyde per total milligrams of protein.
Superoxide Dismutase Activity
To measure hippocampal superoxide dismutase (SOD) activity, the cytoplasmic fraction of the enzyme was first extracted by eluting samples of the homogenized hippocampus in a solvent mix of 500 μl cold ethanol and 250 μl chloroform during 2 min. The mixture was then centrifuged at 18,000×g during 1 h at 4 °C, and the supernatants were stored at − 20 °C. SOD activity was measured according to the method described by Minami and Yoshikawa [49]. Briefly, a small volume of the samples (150 μl) was mixed with Triton X-100 (16%), 0.98 mM nitro blue tetrazolium (NBT) and 1.8 mM pirogallol and buffered with Tris-cacodylate buffer (pH 8.2). The absorbance was then measured at 540 nm. The solution was incubated for another 5 min at 37 °C. A stop solution made of 80% 2 M formic acid and 16% Triton X-100 was added and the absorbance of the solution was measured again at 540 nm. SOD activity was then calculated following the method of McCord and Fridovich [50].
Catalase Activity
Catalase (CAT) activity was measured by a method based on the reduction of hydrogen peroxide (Aebi, 1984). In a quartz cuvette, 0.1 ml of each sample was added to 0.9 ml of 50 mM potassium phosphate buffer (pH 7.0) and 0.1 ml of 19 mM hydrogen peroxide, and the absorbance was measured at 240 nm for 5 min. The absorbance decay observed is called “k” and reflects CAT activity. CAT activity was expressed as “k” per milligram of protein, following the method described by Aebi [51].
Determination of GP and GR Activity
Glutathione peroxidase (GP) and glutathione reductase (GR) activities were measured using commercial kits from BioVision Company (Catalogue number: no. 762-100 for GP activity and no. k761-100-200 for GR activity). To measure GP activity, 100 μl of sample was mixed with GPX assay Buffer, NADPH solution, GR solution and GSSG solution, according to the manufacturer’s instructions. The absorbance of the mixture was measured during 5 min at 340 nm at 25 °C. The results are expressed as μmol NADPH oxidized/min/mg protein.
To determine GR activity, 100 μl of sample was mixed with 5 μl of 3% H2O2 and incubated 5 min at 25 °C. Subsequently, a GR assay buffer, DTNB solution, NADPH-GNERAT™ solution and GSSG were added to 100 μl of pretreated samples and the absorbance was measured during 10 min at 450 nm at room temperature. GR activity is expressed as μmol NADPH oxidized/min/mg protein.
Glutathione Levels
Glutathione levels were measured using the cyclic method of Griffith [52]. For this purpose, it was necessary to deproteinize the samples by mixing them with a volume of 20% TCA during 20 min at 4 °C and subsequently centrifuging at 11,000×g for 10 min at 4 °C. The supernatants were then transferred to Eppendorf tubes and stored at − 80 °C.
Determination of T-GSH, GSSG, and GSH Levels
To assess t-GSH levels, 50 μl of each deproteinized sample obtained above was mixed with a reaction mixture made up of 0.30 mM NADPH, 0.225 mM DTNB and 1.6 U/ml glutathione reductase (GR) and incubated for 5 min at 37 °C. Afterwards, the absorbance was measured at 405 nm during 5 min. To assess GSSG levels, the deproteinized samples were mixed with 4-vinylpuridine (4-VP) (at a concentration 10 times the GSX levels) and 0.2 N NaOH and incubated during 1 h at room temperature. Afterwards, 50 μl of the solution was mixed with a reaction mixture containing 0.30 mM NADPH, 0.225 mM DTNB and 1.6 U/ml GR and incubated for 5 min at 37 °C. The absorbance was then measured as described previously. GSH levels were calculated, on the one hand, by subtracting the oxidized glutathione from total glutathione and, on the other hand, by measuring GSH levels using a fluorometric method as described by Hissin and Hilf [53].
Protein Immunodetection
Membranes and cytosolic or tissue extracts (100 μg) were solubilized in sodium dodecyl sulphate (SDS)-sample buffer, and the proteins were then run on an 8–12% SDS-polyacrylamide gel. The transfer of proteins to PVDF membranes and the immunodetection of proteins using specific antibodies were carried out as described elsewhere [54, 55]. Briefly, after protein transfer, the PVDF membranes were blocked with blotting buffer (TTBS) [50 mM Tris–HCl, pH 7.5; 150 mM NaCl; 0.05% (v/v) Tween-20] and 5% (w/v) non-fat dry milk. PVDF membranes were incubated overnight at 4 °C with primary antibodies diluted 1:1000 in blotting buffer [50 mM Tris–HCl, pH 7.5; 150 mM NaCl; 0.05% (v/v) Tween-20]. Subsequently, excess antibody was removed and membranes rinsed with blotting buffer. After washing, secondary horseradish peroxidase-conjugated antibodies diluted 1:2000 in blotting buffer were added for 60 min at 4 °C for the immunodetection of the target protein. After washing unbound antibody, the proteins were detected by chemiluminescence using an ECL western blotting detection kit, according to the manufacturer’s instructions. Quantification of the bands was carried out by densitometric analysis using the Scion Image computer program (Scion Inc., Frederick, MD, USA).
HPLC Analysis of Sterol Content
Hippocampal membranes from each experimental animal were washed twice with ice-cold PBS and resuspended in 0.5 ml of 10% (w/v) KOH. Ergosterol was added as an internal standard. Samples were treated sequentially, as described by Sánchez-Wandelmer [56], with a solvent mixture of chloroform-methanol in a volumetric proportion of 2:1 and distilled water to obtain the lipid and hydrosoluble fractions. The lipid extract was further separated into a saponifiable and non-saponifiable fraction. Non-saponifiable lipids were resuspended in hexane and sterol separation was performed by HPLC. Sterol separation was accomplished by reverse-phase HPLC with a Luna-Pack 5 μm pore size C18 column (250 mm X 4.60 mm; Phenomenex). Lipids were eluted with acetonitrile-water (95:5, v/v) for the first 37 min and then with methanol at a flow rate of 1.2 ml/min. Eluents were monitored by ultraviolet-absorption spectroscopy (Beckman 168 variable-wavelength detector; Beckman Instruments). Eluted sterols were identified by comparing retention times and ultraviolet spectra with those of pure standards.
Statistical Analysis
Statistical comparisons of enzyme activity and protein levels were carried out by a nonparametric test, the U Mann Whitney test. Differences among groups were considered significantly different when the P value was less than 0.05. Each individual experiment was performed in duplicate.
Evaluation of Iba-1+ cells and the IgG signal was performed using a system of subjective grading, ranging from no labelling intensity (−) to the maximum intensity (+ + +). This evaluation was performed by two independent observers. Subsequently, the intensity levels obtained were grouped into different levels of expression for further analysis and interpretation, following the grading scale: − = 0; −/+, + = 1; +/++, ++ = 2; ++/+++, +++ = 3. For evaluating microglia activation, Iba-1+ cells were classified, according to its morphology, into 4 scores ranging from 1 (highly ramified microglia -“surveillance” state-) to 4 (amoeboid phenotype) and, then, grouped and analyzed. Differences among groups were evaluated by ANOVA test followed by Bonferroni’s test.
Results
Clinical Evolution
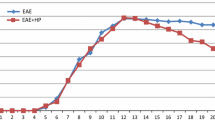
In the present study, motor impairments were used as indicators to assess disease progression, using the criteria described in materials and methods. In this animal model of MS, CR-EAE, 100% of the Lewis rats immunized with GPSC showed the first signs of the disease between days 10 and 12 post-immunization, while more severe signs were detected on day 17, as shown in Fig. 1, panel c. The appearance of the clinical signs of CR-EAE correlated with a decrease in both body weight (Fig. 1, panel a) and food intake (Fig. 1, panel b). Five percent of rats with CR-EAE pretreated with the EAP salt (CR-EAE + EAP) did not show any clinical signs of the disease whereas the remaining 95% showed a delayed onset of the clinical signs when compared to CR-EAE. CR-EAE + EAP rats manifested the most severe signs of the disease between days 19 to 20 following immunization (Fig. 1, panel d). Once again, the appearance of the first clinical signs correlated with a decrease in body weight and food intake (Fig. 1, panels a, b). Both rats with CR-EAE and CR-EAE + EAP rats recovered around day 24 post-immunization. The results show a shortening in the duration of the first attack in rats with CR-EAE + EAP as well as a delay in the onset of the clinical signs. The clinical signs of the second attack were observed around days 26–27 in CR-EAE rats, and around days 28–30 in rats with CR-EAE + EAP (Fig. 1, panel c). The analysis of the rats at different stages of the disease revealed that 3.5% of CR-EAE rats remained in grade 1, no rats remained in grade 2, and 96.5% of the rats reached grade 3. However, only 19.0% of the rats with CR-EAE + EAP reached grade 3, as shown in Fig. 1, panel d. Both control and control + EAP rats showed a progressive increase in body weight in correlation with increased food intake.
Temporal evolution of body weight (panel a), food intake (panel b), clinical signs (panel c) and percentage of rats in different stages of the disease (panel d) in chronic relapsing experimental allergic encephalomyelitis and effects of an EAP salt. Control rats (white square, n = 10), CR-EAE rats (black square, n = 10), CR-EAE rats pretreated with EAP (black inverted triangle, n = 10), control rats pretreated with EAP (white diamond, n = 10). Statistical significance: ***p < 0.001 for comparison between control rats and rats with CR-EAE; ###p < 0.001 for comparison between CR-EAE rats and CR-EAE rats pretreated with EAP
Autoimmune Response
The study of sections immunolabeled for CD3+T cells (including CD4+ and CD8+) revealed no presence of T-lymphocytes in the hippocampus of control animals (Fig. 2, panels a, b). In CR-EAE rats, scarce and scattered CD3+ cells were observed in the parenchyma (Fig. 2, panels a, b; Supplementary Fig. 2) in the first attack. In the second attack, however, there was a significant increase in CD3+ T and CD8+ cell infiltrates in the hippocampus, especially in the hippocampal commissure and molecular layer, whereas the rats pretreated with EAP showed a marked decrease of CD3+ T cells as compared with CR-EAE rats (Fig. 2, panels a, b).
CD3+ infiltrates (panel a), CD8+ infiltrates (panel b) and Iba1+ cells (panel c) in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. Panel d: Activated Iba1+ cells in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. The activity score was determined as described in Materials and Methods. Panel e: Ig G levels in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. For each treatment, values are presented as the mean ± S.E.M of five to ten fields per rat of five rats per group. Statistical significance: *p<0.05; **p<0.01; ***p<0.001 (Bonferroni’s test)
Microglial activation can occur as a result of immune activation, as has been reported for EAE and MS [57,58,59,60,61], but there is little data available regarding the early stages of the disease in the hippocampus [62]. Since microglia are the resident, immune cells in the brain and their morphology changes when activated, we first investigated if their morphology was altered in the hippocampus early on in the disease course, (first attack, remission and second attack). To this end, hippocampal sections were analyzed for Iba-1+ immunoreactivity (a marker of activated microglia) (Fig. 2, panel c). Activated microglia can be morphologically identified by their thick, retracted processes and hypertrophied soma (Supplementary Fig. 3). We did not find any evidence for microglial activation in the control group, whereas a high level of microglial activation was observed in both the first and second attack (Fig. 2, panel d). Interestingly, EAP pretreatment decreased microglial activation when compared with the CR-EAE group.
As shown in Fig. 2, panel e and Supplementary Fig. 4, there was no presence of or very scarce amount of immunoglobulin G (IgG) immunoreactivity in the hippocampus of the control group. However, IgG immunoreactivity was significantly increased in the hippocampus of CR-EAE rats and markedly decreased in those rats pretreated with EAP.
Superoxide Dismutase and Catalase Activity and Protein Levels
In view of the above results, we next evaluated total SOD activity, which represents the first line of antioxidant defence. A moderate increase in total SOD activity was detected during the first attack in the hippocampus of CR-EAE rats when compared to control animals (Fig. 3). This increase disappeared during the remission phase but was once again observed during the second attack (Fig. 3). CR-EAE + EAP rats did not exhibit any increase in SOD activity in any of the phases studied. CR-EAE rats pretreated with EAP (CR-EAE + EAPgrade 3) showed the same degree of SOD activity as CR-EAE rats in the three stages of the disease (Fig. 3). Next, SOD protein levels were measured. The results reveal an increase in SOD protein levels in the CR-EAE rat hippocampus in the first attack, which was not found in either the remission stage or in the second attack (Fig. 3).
Superoxide dismutase (SOD) activity and protein levels in the three stages of chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. Total SOD activity and SOD protein levels in the hippocampus of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate (EAP) and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1, speckled bars), rats with CR-EAE pretreated with EAP and sacrificed at grade 3 (CR-EAE + EAPgrade 3, solid grey bars) and control rats pretreated with EAP (hatched bars). For each treatment, values are presented as the means ± S.E.M of five rats. Statistical significance: *p < 0.05; **p < 0.01; ***p<0.001 (Mann-Whitney’s U-test)
H2O2 is a by-product of SOD activity and is involved in oxidative stress. Consequently, we measured the activity of catalase, an enzyme which catalyses the decomposition of H2O2 into oxygen and water. CAT activity was diminished during the first attack, remission stage and second attack in the hippocampus of rats with CR-EAE (Fig. 4). This decrease was less pronounced in the CR-EAE rats pretreated with EAP than in CR-EAE rats. CR-EAE + EAPgrade 3 rats, like CR-EAE rats and CR-EAE + EAPgrade 1 rats, showed a decrease in CAT activity in the first and second attack (Fig. 4). CAT protein levels were next measured by western blot. The results showed a decrease in CAT protein levels in CR-EAE rats as compared with control levels in the three stages of the disease (Fig. 4). This decrease was also found in CR-EAE + EAPgrade 1 rats (Fig. 4).
Catalase (CAT) activity and protein levels in the three stages of chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. CAT activity and CAT protein levels in the hippocampus of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1, speckled bars), rats with CR-EAE pretreated with EAP and sacrificed at grade 3 (CR-EAE + EAPgrade 3, solid grey bars) and control rats pretreated with EAP (hatched bars). For each treatment, values are presented as the mean ± S.E.M of five rats. Statistical significance: *p < 0.05; **p < 0.01; ***p < 0.001 (Mann-Whitney’s U-test)
Glutathione System
Another system involved in the decomposition of H2O2 is the glutathione system. Hence, we also measured glutathione peroxidase (GP) activity (Fig. 5) and glutathione reductase (GR) activity (Fig. 6). Both enzyme activities were found to be significantly increased in CR-EAE rats in the first attack as compared to the controls. In the second attack, GP was reduced in CR-EAE rats as compared to controls, although GR was increased. No significant changes in GP activity were detected in the remission stage whereas GR activity was reduced. CR-EAE + EAPgrade 1 rats showed values similar to controls in the three stages of the disease. CR-EAE + EAPgrade 3 rats showed an increase of GP and GR in the first attack, like CR-EAE rats, whereas a decrease in GP activity, concomitant with an increase in GR activity, was found in the second attack. GP and GR protein levels were also measured. The results showed an increase in GP and GR levels in CR-EAE and CR-EAE + EAPgrade 1 rats in the first attack when compared to controls (Figs. 5 and 6). In the second attack, however, CR-EAE rats showed a decrease in GP levels (Fig. 5, bottom panel).
Glutathione peroxidase (GP) activity and protein levels in the three stages of chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. Total GP activity and GP protein levels in the hippocampus of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1, specked bars), rats with CR-EAE pretreated with EAP and sacrificed at grade 3 (CR-EAE + EAPgrade 3, solid grey bars) and control rats pretreated with EAP (hatched bars). For each treatment, values are presented as the mean ± S.E.M of five rats. Statistical significance: *p < 0.05; **p < 0.01 (Mann-Whitney’s U-test)
Glutathione reductase (GR) activity and protein levels of in the three stages of chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. Total GR activity and GR protein levels in the hippocampus of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1,specked bars), rats with CR-EAE pretreated with EAP and sacrificed at grade 3 (CR-EAE + EAPgrade 3, solid grey bars) and control rats pretreated with EAP (hatched bars). For each treatment, values are presented as the mean ± S.E.M of five rats. Statistical significance: *p < 0.05; **p < 0.01; ***p<0.001 (Mann-Whitney’s U-test)
In view of the above results, we next measured the levels of total glutathione (t-GSH), reduced glutathione (GSH) and oxidized glutathione (GSSG). No changes in t-GSH levels were detected in the hippocampus of rats with CR-EAE throughout the progression of the disease when compared to control rats (Fig. 7, top panel). In the first attack, however, a decrease in GSH levels was observed in the hippocampus of CR-EAE rats when compared with control rats, whereas in the second attack, a high increase was detected (Fig. 7, middle panel). GSSG levels augmented in the hippocampus of rats with CR-EAE in the first attack when compared to control rats (Fig. 7, bottom panel), whereas CR-EAE + EAPgrade 3 rats showed a decrease in GSH levels and an increase of GSSG in this same stage of the disease. No changes in GSSG were found in any of the experimental groups in the second attack when compared to control values (Fig. 7, bottom panel).
Total glutathione (t-GSH), reduced glutathione (GSH) and oxidized glutathione (GSSG) levels in the three stages of chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. Hippocampal t-GSH levels (Top panel), GSH levels (middle panel) and GSSG levels (bottom panel), of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1, speckled bars), rats with CR-EAE pretreated with EAP and sacrificed at grade 3 (CR-EAE + EAPgrade 3, solid grey bars) and control rats pretreated with EAP (hatched bars) at different phases of the disease (first attack, remission and second attack). For each treatment, values are presented as the mean ± S.E.M of five rats. Statistical significance: *p < 0.05; **p < 0.01; ***p<0.001 (Mann-Whitney’s U-test)
Lipid Peroxidation
Our results showed significant alterations in the antioxidant defences, especially in the second attack. The next parameter assessed was lipid peroxidation, an oxidative stress indicator, which was determined by measuring the amount of hippocampal malondialdehyde produced. The results of the present study show a significant increase in lipid peroxidation in the hippocampus of rats with CR-EAE during the first attack, when compared to control rats (Fig. 8), which was not found during the remission phase. Lipid peroxidation increased significantly once again in the second attack of CR-EAE rats. These values were significantly greater than those obtained during either the first attack or the remission phase. In CR-EAE + EAP rats, no increase was registered at any of these stages. CR-EAE + EAPgrade 3 rats showed the same degree of lipid peroxidation as CR-EAE rats in the three stages of the disease (Fig. 8).
Lipid peroxidation levels in the rat hippocampus and effects of an EAP salt. Levels of reactive species to thiobarbituric acid in the hippocampus of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1, speckled bars), rats with CR-EAE pretreated with EAP and sacrificed at grade 3 (CR-EAE + EAPgrade 3, solid grey bars) and control rats pretreated with EAP (hatched bars) at different phases of the disease (first attack, remission and second attack). For each treatment, values are presented as the mean ± S.E.M of five rats. Statistical significance: *p < 0.05; **p<0.01; ***p<0.001 (Mann-Whitney’s U-test)
Sterol Levels
An increase of oxidative stress is known to induce cholesterol accumulation in membranes. Hence, we measured, by HPLC, sterol levels in hippocampal membranes of control rats, CR-EAE rats, CR-EAE rats pretreated with EAP and control rats pretreated with EAP. Cholesterol levels were increased in CR-EAE rats in the first and second attack, returning to the initial values in the remission phase (Fig. 9, top panel). These changes were accompanied by a decrease in desmosterol levels in CR-EAE rats in the first and second attack (Fig. 9, bottom panel).
Hippocampal sterols levels in chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. Cholesterol levels (top panel), and desmosterol levels (bottom panel) in the hippocampus of control rats (open bars), rats with CR-EAE (black bars), rats with CR-EAE pretreated with ethanolamine phosphate and sacrificed at the same time as the CR-EAE rats (CR-EAE + EAPgrade 1, speckled bars) and control rats pretreated with EAP (hatched bars) at different phases of the disease (first attack, remission and second attack). Values are presented as the mean ± S.E.M of five rats per each group. Statistical significance: *p < 0.05; **p < 0.01; ***p < 0.001 (Mann-Whitney’s U-test)
Cell Death
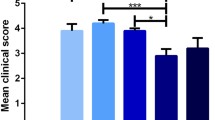
In view of the increase in T cell infiltrates and Iba1+ immunoreactivity, lipid peroxidation, SOD, GP and GR activities as well as enhanced cholesterol levels and the decrease in CAT activity, GSH and desmosterol levels in the first and second attack in the CR-EAE rat hippocampus, when compared to controls, we explored the possibility that this impairment might be affecting hippocampal cell survival. To evaluate this hypothesis, we analysed cell death by TUNEL staining. In situ cell detection showed an increase in the number of apoptotic nuclei in the hippocampus of rats with CR-EAE, especially in the molecular stratum and radiatum stratum in the second attack. The number of apoptotic cells in CR-EAE rats pretreated with EAP was lower than in CR-EAE rats in the three stages of the disease (Fig. 10, supplementary Fig. 5).
TUNEL (+) cells in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. For each treatment, values are presented as the mean ± S.E.M of five to ten fields per rat of five rats per group. Statistical significance: *p<0.05; **p<0.01; ***p<0.001 (Mann-Whitney’s U-test)
Discussion
Multiple sclerosis (MS) is a chronic inflammatory and demyelinating disease of the CNS that represents the most frequent cause of chronic disability in young adults. The present study shows changes in the hippocampus of an animal model of MS, chronic relapsing experimental allergic encephalomyelitis (CR-EAE). These changes include T lymphocyte infiltrates and increased Iba1+ cells, high IgG levels and alterations in the antioxidant defences, at three different phases along the progression of the disease: first attack, remission and second attack. Our results also show that EAP attenuates these changes.
At present, the antigen which induces activation of the immune system against the CNS is unknown. Several studies on T cell infiltrates in MS plaques have shown that these cells are activated against myelin oligodendrocyte glycoprotein (MOG) and myelin basic protein (MBP) [63, 64]. For this reason, we used an animal model in which EAE was induced with an emulsion of guinea-pig spinal cord tissue (GPSC) [43] since it has all the potential encephalitogenic peptides.
Our results showed that all the rats immunized with the GPSC emulsion developed the chronic form of the disease; in particular, 96.5% of them reached grade 3 of the clinical score. In these CR-EAE rats, the greatest weight loss and lowest food intake coincided with the days in which the rats presented the most severe clinical signs. In the case of CR-EAE rats pretreated with EAP, our results showed a delay in the onset of the clinical signs, a reduction in the duration of the attack and a decrease in the severity of the clinical signs: only 19.0% of the rats reached grade 3 of the clinical score. In these rats, the greatest weight loss and lowest food intake coincided with the days in which the rats presented the most severe clinical signs.
The immunohistochemical analysis of the hippocampus revealed the presence of T cell infiltrates and activated microglial cells in the three stages of the disease in the CR-EAE group, especially in the second attack. The increase in Iba1+ cells found in our study may be due to either migration of microglial cells to that area, to proliferation of pre-existing microglia in the hippocampus or to infiltration of peripheral macrophages. Microglial cells activated by T cells can secrete pro-inflammatory cytokines and free radicals, which could produce direct damage to oligodendrocytes, astrocytes and neurons [9,10,11,12,13,14]. Our results showed an increase in IgG levels in the hippocampus, which could increase the damage exerted on these cells. In the second attack, the infiltrates, Iba1+ cells and IgG levels are higher and are especially located in the molecular layer and girus dentatus of the hippocampus; interestingly, those hippocampal areas involved in memory processes. These results might partly explain the increase of lipid peroxidation seen in the first and second attack of CR-EAE, which is more abrupt during the second attack. This increase also correlates with a dysfunction in the antioxidant defences. In line with this, superoxide anion (O2.) production has been previously shown to increase during EAE [65,66,67] and O2− is known to inhibit CAT [68, 69].
In addition, we found a significant increase in total SOD activity in the hippocampus of rats with CR-EAE as compared with control animals (Supplementary Figs. 6 and 7). An increased O2.‑ production may favour SOD activity, since it is the major SOD allosteric activator [70]. Here, we have shown that CR-EAE induces a decrease in CAT activity in both the first and second attack. The concomitant increase in SOD activity and decrease in CAT activity might result in an increased H2O2 concentration, since the production of the latter from O2.‑ by SOD is increased and its elimination by CAT is decreased. In MS, a significantly enhanced expression of SOD1 has been observed in active demyelinating lesions [71,72,73]. In brain homogenates of rats suffering from EAE, a decreased peroxisomal function was demonstrated, which was accompanied by both a reduction in CAT gene expression and activity [74]. Recently, a decrease in CAT activity has been found in the hippocampus of rats with acute EAE induced with MOG [21] (Sajad et al., 2009). Several studies have shown beneficial effects of CAT treatment on the development of EAE [75,76,77].
The reduced CAT activity found in the present study might be associated with a compensatory increased activity of GP, which acts as a defence against H2O2 formation. GP plays a central role in the removal of toxic peroxides, and the increase of its activity in the EAE hippocampus affected by oxidation could possibly reflect the cells´ attempt to counteract increased lipid peroxide products (Supplementary Figs. 6 and 7).
On the other hand, GR has an important role in cellular antioxidant protection because it catalyses the regeneration of GSH from GSSG. The elevation of GR activity observed in the second attack of rats with CR-EAE can partly explain the increase in GSH in this episode, although the recovery of GSH seems to be insufficient for alleviating oxidative stress. In our study, GSH depletion was limited to the first attack. This finding is in agreement with that reported in mice [78] and in patients suffering from MS [79]. Actually, the GSH decrease is considered an early event of the disease [21]. Since GSH inactivates a number of oxidizing species, this data is in accordance with observations showing that the levels of ROS indicators are elevated in EAE [80, 81].
This study also demonstrates that the cholesterol content in hippocampal membranes increases in the first attack of CR-EAE, returning nearly to initial values in the remission phase and increasing, once again, in the second attack, although to a lesser extent (Supplementary Figs. 6 and 7). These results are in accordance with the fact that brain membrane-associated oxidative stress induces cholesterol accumulation [82, 83]. In fact, studies of non-neuronal cells have demonstrated that oxidative stress enhances cholesterol accumulation [84,85,86,87]. In addition, previous studies in brain tissue from patients suffering MS indicate that the cholesterol content increases when compared to control patients [88].
Coincident with the increase in brain cholesterol, we found a decrease in the desmosterol content. Conversion of desmosterol into cholesterol is catalysed by 3β-hydroxysterol-Δ24 reductase (DHCR24; alias hDiminuto/Seladin-1) [89,90,91]. Endogenous DHCR24/seladin-1 levels are highly upregulated upon acute oxidative stress [92, 93], while its expression declines to very low levels upon chronic exposure [92], suggesting that DHCR24/seladin-1 plays a role in integrating cellular responses to oxidative stress. Several studies seem to indicate that increased cholesterol synthesis potentially counteracts lipid peroxidation associated with oxidative stress, thus maintaining plasma membrane integrity [83].
In the first attack of CR-EAE rats, we did not observe a higher cell death, probably due to increased cholesterol content and a good functioning of the glutathione system. On the other hand, we observed an increase in TUNEL (+) cells in the second attack, which could be caused by the lymphocyte infiltrates and microglial activation as well as by the alterations in the antioxidant defences.
Interestingly, the corpus callosum of CR-EAE mice, which is in close proximity to the hippocampus, was reported by Mangiardi et al. [94] to contain many inflammatory demyelinating lesions, 30–40% of the infiltrating cells being CD3+ T cells.
Our results demonstrate a delay in the appearance of the clinical signs of CR-EAE in rats that were pretreated with EAP and less severity of the symptoms. The histological results of these rats show less inflammatory infiltrates and a decrease in the levels and activation of microglial cells and IgG. The antioxidant defence mechanisms in the CR-EAE + EAPgrade 1 rats were similar to control levels whereas the CR-EAE + EAgrade 3 rats showed alterations in both the activity and protein levels of the antioxidant enzymes, albeit to a lesser extent than CR-EAE rats. This might be attributed to modulation of the immune system by ethanolamine, which is part of anandamide, exerting immunomodulatory effects on T and B cells. It is known that activation of antigen-specific T cells requires a reorganization of the “lipid rafts” in the plasma membrane, the CD4 receptor [95], CD8 [96] and other proteins involved in the early stages of activation [97, 98]. These lipid rafts, where these receptors are located, are microdomains rich in cholesterol and different sphingolipids. Dipalmitoyl phosphatidylethanolamine and N-acylethanolamine are known to be involved in the rearrangement and compartmentalization of lipid rafts [98,99,100]. Variations in the ratios of these components could trigger the disintegration of the lipid rafts and, thereby, modify associated signalling pathways [101].
On the other hand, several studies have shown that ethanolamine derivates such as palmitoylethanolamine reduce the inflammation induced by PPAR-α [102]. Previous studies from our research group have shown that in rats with acute EAE, EAP treatment blunts the increase in TNF-α and IFN-γ [103]. Taken together, this might suggest that the delay in the onset of the disease is due, at least partly, to decreased T and B cell infiltration and a reduction of microglial cell activity. Alternatively, it might be due to a scavenger role of the EAP salt that scavenges the ROS, although this role is not sufficiently efficient to prevent the alterations in the antioxidant defences.
Ethanolamine plasmalogens have been demonstrated to possess antioxidant properties [38]. Our results reveal that EAP acts as an antioxidant in the hippocampus of rats with CR-EAE. It has been proposed that the plasmalogens serve as endogenous antioxidants to protect cells from oxidative stress, especially cholesterol-rich areas of the lipid membrane, by scavenging radicals at the vinyl ether linkage [39, 104, 105]. Ethanolamine plasmalogens also tend to form a hexagonal phase that contributes to stabilizing the structure of the lipid rafts [39]. These data may help explain the decrease in the cholesterol content in membranes from the CR-EAE + EAP rats.
In the hippocampus of CR-EAE rats pretreated with EAP, we observed less TUNEL(+) cells than in CR-EAE rats in the three stages of the disease. This result could be explained by a decrease in lymphocyte infiltrates and less impairment of the antioxidant defences.
In conclusion, the present study demonstrates a significant impairment in the antioxidant defence mechanisms in CR-EAE and suggests a protective role of EAP in the rat hippocampus.
Conclusion
In conclusion, the hippocampus, a brain area related with memory processes, is affected by lymphocyte infiltrates and alterations in the antioxidant defences in CR-EAE rats, especially in the second attack. Moreover, hippocampal membrane cholesterol levels are increased, concomitant with lipid peroxidation, at the three stages of the disease. Our results suggest that the EAP salt delays the onset and severity of the clinical signs of CR-EAE. This salt also decreased the lymphocyte infiltrates, possibly due to a decreased activation of lymphocyte subsets in peripheral blood, and maintained the activity of the antioxidant defences. Hence, the present results show that CR-EAE is an ideal model for the study of the chronic course of MS as well as for the evaluation of potential therapeutic treatments such as EAP. Moreover, this salt could be a potential candidate for the treatment of MS.
References
Polman CH, Uitdehaag BM (2000) Drug treatment of multiple sclerosis. BMJ 321:490–494
Conlon P, Oksenberg JR, Zhang J, Steinman L (1999) The immunobiology of multiple sclerosis: an autoimmune disease of the central nervous system. Neurobiol Dis 6:149–166
Gold R, Hartung HP, Toyka KV (2000) Animal models for autoimmune demyelinating disorders of the nervous system. Mol Med Today 6:88–91
Slavin A, Ewing C, Liu J, Ichikawa M, Slavin J, Bernard CC (1998) Induction of a multiple sclerosis-like disease in mice with an immunodominant epitope of myelin oligodendrocyte glycoprotein. Autoimmunity 28:109–120
Steinman L (2001) Multiple sclerosis: a two-stage disease. Nat Immunol 2:762–764
Sobel RA, Blanchette BW, Bhan AK, Colvin RB (1984) The immunopathology of experimental allergic encephalomyelitis. I. Quantitative analysis of inflammatory cells in situ. J Immunol 132:2393–2401
Polman CH, Dijkstra CD, Sminia T, Koetsier JC (1986) Immunohistological analysis of macrophages in the central nervous system of Lewis rats with acute experimental allergic encephalomyelitis. J Neuroimmunol 11:215–222
Cross AH, Raine CS (1991) Central nervous system endothelial cell-polymorphonuclear cell interactions during autoimmune demyelination. Am J Pathol 139:1401–1409
Babior BM (1984) Oxidants from phagocytes: agents of defense and destruction. Blood 64:959–966
Banati RB, Gehrmann J, Schubert P, Kreutzberg GW (1993) Cytotoxicity of microglia. Glia 7:111–118
Colton CA, Gilbert DL (1987) Production of superoxide anions by a CNS macrophage, the microglia. FEBS Lett 223:284–288
Matsumoto Y, Hara N, Tanaka R, Fujiwara M (1986) Immunohistochemical analysis of the rat central nervous system during experimental allergic encephalomyelitis, with special reference to Ia-positive cells with dendritic morphology. J Immunol 136:3668–3676
Matsumoto Y, Ohmori K, Fujiwara M (1992) Microglial and astroglial reactions to inflammatory lesions of experimental autoimmune encephalomyelitis in the rat central nervous system. J Neuroimmunol 37:23–33
Vass K, Lassmann H, Wekerle H, Wisniewski HM (1986) The distribution of Ia antigen in the lesions of rat acute experimental allergic encephalomyelitis. Acta Neuropathol 70:149–160
Fialkow L, Wang Y, Downey GP (2007) Reactive oxygen and nitrogen species as signaling molecules regulating neutrophil function. Free Radic Biol Med 42:153–164
Gilgun-Sherki Y, Melamed E, Offen D (2004) The role of oxidative stress in the pathogenesis of multiple sclerosis: the need for effective antioxidant therapy. J Neurol 251:261–268
Calabrese M, Favaretto A, Martini V, Gallo P (2012) Grey matter lesions in MS: From histology to clinical implications. Prion 7:20–27
Bobholz JA, Rao SM (2003) Cognitive dysfunction in multiple sclerosis: a review of recent developments. Curr Opin Neurol 16:283–288
Rao SM, Leo GJ, Bernardin L, Unverzagt F (1991) Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41:685–691
Sicotte NL, Kern KC, Giesser BS, Arshanapalli A, Schultz A, Montag M, Wang H, Bookheimer SY (2008) Regional hippocampal atrophy in multiple sclerosis. Brain 131:1134–1141
Sajad M, Zargan J, Chawla R, Umar S, Sadaqat M, Khan HA (2009) Hippocampal neurodegeneration in experimental autoimmune encephalomyelitis (EAE): potential role of inflammation activated myeloperoxidase. Mol Cell Biochem 328:183–188
Mann EO, Greenfield SA (2003) Novel modulatory mechanisms revealed by the sustained application of nicotine in the guinea-pig hippocampus in vitro. J Physiol 551:539–550
Aguado-Llera D, Puebla-Jimenez L, Yebenes-Gregorio L, Arilla-Ferreiro E (2007) Alteration of the somatostatinergic system in the striatum of rats with acute experimental autoimmune encephalomyelitis. Neuroscience 148:238–249
Nisticò R, Mango D, Mandolesi G, Piccinin S, Berretta N, Pignatelli M, Feligioni M, Musella A et al (2013) Inflammation subverts hippocampal synaptic plasticity in experimental multiple sclerosis. PLoS One 8:e54666
Chaudhuri A, Behan PO (2003) Natalizumab for relapsing multiple sclerosis. N Engl J Med 348:1598–1599
Doggrell SA (2003) Is natalizumab a breakthrough in the treatment of multiple sclerosis? Expert Opin Pharmacother 4:999–1001
Miller DM, Rudick RA, Baier M, Cutter G, Doughtery DS, Weinstock-Guttman B, Mass MK, Fisher E et al (2003) Factors that predict health-related quality of life in patients with relapsing-remitting multiple sclerosis. Mult Scler 9:1–5
Brinkmann V, Davis MD, Heise CE, Albert R, Cottens S, Hof R, Bruns C, Prieschl E et al (2002) The immune modulator FTY720 targets sphingosine 1-phosphate receptors. J Biol Chem 277:21453–21457
Fujino M, Funeshima N, Kitazawa Y, Kimura H, Amemiya H, Suzuki S, Li XK (2003) Amelioration of experimental autoimmune encephalomyelitis in Lewis rats by FTY720 treatment. J Pharmacol Exp Ther 305:70–77
Mazda ME, Brosch JR, Wiens AL, Bonnin J, Kamer AP, Mattson DH, Snook RJ (2013) A case of natalizumab-associated progressive multifocal leukoencephalopathy with repeated negative CSF JC virus testing. Int J Neurosci 123:353–357
Hendriks JJ, Teunissen CE, de Vries HE, Dijkstra CD (2005) Macrophages and neurodegeneration. Brain Res Brain Res Rev 48:185–195
van Meeteren ME, Teunissen CE, Dijkstra CD, van Tol EA (2005) Antioxidants and polyunsaturated fatty acids in multiple sclerosis. Eur J Clin Nutr 59:1347–1361
Di Filippo M, Pini LA, Pelliccioli GP, Calabresi P, Sarchielli P (2008) Abnormalities in the cerebrospinal fluid levels of endocannabinoids in multiple sclerosis. J Neurol Neurosurg Psychiatry 79:1224–1229
Andriamampandry C, Freysz L, Kanfer JN, Dreyfus H, Massarelli R (1989) Conversion of ethanolamine, monomethylethanolamine and dimethylethanolamine to choline-containing compounds by neurons in culture and by the rat brain. Biochem J 264:555–562
Buratta S, Hamberger A, Ryberg H, Nystrom B, Sandberg M, Mozzi R (1998) Effect of serine and ethanolamine administration on phospholipid-related compounds and neurotransmitter amino acids in the rabbit hippocampus. J Neurochem 71:2145–2150
Marshall DL, De Micheli E, Bogdanov MB, Wurtman RJ (1996) Effects of ethanolamine (Etn) administration on Etn and choline (Ch) levels in plasma, brain extracellular fluid (ECF) and brain tissue, and on brain phospholipid levels in rats: an in vivo study. Neurosci Res Commun 18:87–96
Wilson R, Tocher DR (1991) Lipid and fatty acid composition is altered in plaque tissue from multiple sclerosis brain compared with normal brain white matter. Lipids 26:9–15
Kuczynski B, Reo NV (2006) Evidence that plasmalogen is protective against oxidative stress in the rat brain. Neurochem Res 31:639–656
Maeba R, Ueta N (2004) A novel antioxidant action of ethanolamine plasmalogens in lowering the oxidizability of membranes. Biochem Soc Trans 32:141–143
Jagannatha HM, Sastry PS (1981) Ethanolamine plasmalogen & cholesterol ester metabolism in experimental allergic encephalomyelitis. Indian J Biochem Biophys 18:411–416
Guo L, Davies SS (2013) Bioactive aldehyde-modified phosphatidylethanolamines. Biochimie 95:74–78
Shiratsuchi A, Ichiki M, Okamoto Y, Ueda N, Sugimoto N, Takuwa Y, Nakanishi Y (2009) Inhibitory effect of N-palmitoylphosphatidylethanolamine on macrophage phagocytosis through inhibition of Rac1 and Cdc42. J Biochem 145:43–50
Feurer C, Prentice DE, Cammisuli S (1985) Chronic relapsing experimental allergic encephalomyelitis in the Lewis rat. J Neuroimmunol 10:159–166
Massella A, D'Intino G, Fernández M, Sivilia S, Lorenzini L, Giatti S, Melcangi RC, Calzà L et al (2012) Gender effect on neurodegeneration and myelin markers in an animal model for multiple sclerosis. BMC Neurosci 13:12
Gold DP, Schroder K, Powell HC, Kelly CJ (1997) Nitric oxide and the immunomodulation of experimental allergic encephalomyelitis. Eur J Immunol 27:2863–2869
Glowinski J, Iversen L (1966) Regional studies of catecholamines in the rat brain. Subcellullar distribution of endogenous and exogenous catecholamines in various brain regions. Biochem Pharmacol 15:977–987
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Ottolenghi CE (1959) Bol Trab Soc Cir B Aires 43:47–50
Minami M, Yoshikawa H (1979) A simplified assay method of superoxide dismutase activity for clinical use. Clin Chim Acta 92:337–342
McCord JM, Fridovich I (1969) The utility of superoxide dismutase in studying free radical reactions. I. Radicals generated by the interaction of sulfite, dimethyl sulfoxide, and oxygen. J Biol Chem 244:6056–6063
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Griffith OW (1980) Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal Biochem 106:207–212
Hissin PJ, Hilf R (1976) A fluorometric method for determination of oxidized and reduced glutathione in tissues. Anal Biochem 74:214–226
Mumby SM, Kahn RA, Manning DR, Gilman AG (1986) Antisera of designed specificity for subunits of guanine nucleotide-binding regulatory proteins. Proc Natl Acad Sci U S A 83:265–269
Penn RB, Pronin AN, Benovic JL (2000) Regulation of G protein-coupled receptor kinases. Trends Cardiovasc Med 10:81–89
Sánchez-Wandelmer J, Davalos A, Herrera E, Giera M, Cano S, de la Pena G, Lasuncion MA, Busto R (2009) Inhibition of cholesterol biosynthesis disrupts lipid raft/caveolae and affects insulin receptor activation in 3T3-L1 preadipocytes. Biochim Biophys Acta 1788:1731–1739
Abourbeh G, Thézé B, Maroy R, Dubois A, Brulon V, Fontyn Y, Dollé F, Tavitian B et al (2012) Imaging microglial/macrophage activation in spinal cords of experimental autoimmune encephalomyelitis rats by positron emission tomography using the mitochondrial 18 kDa translocator protein radioligand [18F]DPA-714. J Neurosci 32:5728–5736
Barnett MH, Prineas JW (2004) Relapsing and remitting multiple sclerosis: pathology of the newly forming lesion. Ann Neurol 55:458–468
Constantinescu CS, Farooqi N, O'Brien K, Gran B (2011) Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). Br J Pharmacol 164:1079–1106
Hampton DW, Anderson J, Pryce G, Irvine KA, Giovannoni G, Fawcett JW, Compston A, Franklin RJ et al (2008) An experimental model of secondary progressive multiple sclerosis that shows regional variation in gliosis, remyelination, axonal and neuronal loss. J Neuroimmunol 201-202:200–211
Howell OW, Rundle JL, Garg A, Komada M, Brophy PJ, Reynolds R (2010) Activated microglia mediate axoglial disruption that contributes to axonal injury in multiple sclerosis. J Neuropathol Exp Neurol 69:1017–1033
Acharjee S, Nayani N, Tsutsui M, Hill MN, Ousman SS, Pittman QJ (2013) Altered cognitive-emotional behavior in early experimental autoimmune encephalitis--cytokine and hormonal correlates. Brain Behav Immun 33:164–172
Elong Ngono A, Pettré S, Salou M, Bahbouhi B, Soulillou JP, Brouard S, Laplaud DA (2012) Frequency of circulating autoreactive T cells committed to myelin determinants in relapsing-remitting multiple sclerosis patients. Clin Immunol 144:117–126
Martin R, Jaraquemada D, Flerlage M, Richert J, Whitaker J, Long EO, McFarlin DE, McFarland HF (1990) Fine specificity and HLA restriction of myelin basic protein-specific cytotoxic T cell lines from multiple sclerosis patients and healthy individuals. J Immunol 145:540–548
MacMicking JD, Willenborg DO, Weidemann MJ, Rockett KA, Cowden WB (1992) Elevated secretion of reactive nitrogen and oxygen intermediates by inflammatory leukocytes in hyperacute experimental autoimmune encephalomyelitis: enhancement by the soluble products of encephalitogenic T cells. J Exp Med 176:303–307
Scott GS, Bolton C (2000) L-arginine modifies free radical production and the development of experimental allergic encephalomyelitis. Inflamm Res 49:720–726
Scott GS, Williams KI, Bolton C (1996) A pharmacological study on the role of nitric oxide in the pathogenesis of experimental allergic encephalomyelitis. Inflamm Res 45:524–529
Kono Y, Fridovich I (1982) Superoxide radical inhibits catalase. J Biol Chem 257:5751–5754
Shimizu N, Kobayashi K, Hayashi K (1984) The reaction of superoxide radical with catalase. Mechanism of the inhibition of catalase by superoxide radical. J Biol Chem 259:4414–4418
Halliwell B, Gutteridge JM (1992) Biologically relevant metal ion-dependent hydroxyl radical generation. An update. FEBS Lett 307:108–112
Liu XH, Kato H, Nakata N, Kogure K, Kato K (1993) An immunohistochemical study of copper/zinc superoxide dismutase and manganese superoxide dismutase in rat hippocampus after transient cerebral ischemia. Brain Res 625:29–37
Matsuyama T, Michishita H, Nakamura H, Tsuchiyama M, Shimizu S, Watanabe K, Sugita M (1993) Induction of copper-zinc superoxide dismutase in gerbil hippocampus after ischemia. J Cereb Blood Flow Metab 13:135–144
Tajouri L, Mellick AS, Ashton KJ, Tannenberg AE, Nagra RM, Tourtellotte WW, Griffiths LR (2003) Quantitative and qualitative changes in gene expression patterns characterize the activity of plaques in multiple sclerosis. Brain Res Mol Brain Res 119:170–183
Singh I, Paintlia AS, Khan M, Stanislaus R, Paintlia MK, Haq E, Singh AK, Contreras MA (2004) Impaired peroxisomal function in the central nervous system with inflammatory disease of experimental autoimmune encephalomyelitis animals and protection by lovastatin treatment. Brain Res 1022:1–11
Guy J, Qi X, Hauswirth WW (1998) Adeno-associated viral-mediated catalase expression suppresses optic neuritis in experimental allergic encephalomyelitis. Proc Natl Acad Sci U S A 95:13847–13852
Liu Y, Teige I, Birnir B, Issazadeh-Navikas S (2006) Neuron-mediated generation of regulatory T cells from encephalitogenic T cells suppresses EAE. Nat Med 12:518–525
Ruuls SR, Bauer J, Sontrop K, Huitinga I, Hart BA, Dijkstra CD (1995) Reactive oxygen species are involved in the pathogenesis of experimental allergic encephalomyelitis in Lewis rats. J Neuroimmunol 56:207–217
Zargari M, Allameh A, Sanati MH, Tiraihi T, Lavasani S, Emadyan O (2007) Relationship between the clinical scoring and demyelination in central nervous system with total antioxidant capacity of plasma during experimental autoimmune encephalomyelitis development in mice. Neurosci Lett 412:24–28
Calabrese V, Scapagnini G, Ravagna A, Bella R, Butterfield DA, Calvani M, Pennisi G, Giuffrida Stella AM (2003) Disruption of thiol homeostasis and nitrosative stress in the cerebrospinal fluid of patients with active multiple sclerosis: evidence for a protective role of acetylcarnitine. Neurochem Res 28:1321–1328
Ilhan A, Akyol O, Gurel A, Armutcu F, Iraz M, Oztas E (2004) Protective effects of caffeic acid phenethyl ester against experimental allergic encephalomyelitis-induced oxidative stress in rats. Free Radic Biol Med 37:386–394
Liu Y, Zhu B, Wang X, Luo L, Li P, Paty DW, Cynader MS (2003) Bilirubin as a potent antioxidant suppresses experimental autoimmune encephalomyelitis: implications for the role of oxidative stress in the development of multiple sclerosis. J Neuroimmunol 139:27–35
Cutler RG, Kelly J, Storie K, Pedersen WA, Tammara A, Hatanpaa K, Troncoso JC, Mattson MP (2004) Involvement of oxidative stress-induced abnormalities in ceramide and cholesterol metabolism in brain aging and Alzheimer’s disease. Proc Natl Acad Sci U S A 101:2070–2075
Kuehnle K, Crameri A, Kalin RE, Luciani P, Benvenuti S, Peri A, Ratti F, Rodolfo M et al (2008) Prosurvival effect of DHCR24/Seladin-1 in acute and chronic responses to oxidative stress. Mol Cell Biol 28:539–550
Gesquiere L, Loreau N, Minnich A, Davignon J, Blache D (1999) Oxidative stress leads to cholesterol accumulation in vascular smooth muscle cells. Free Radic Biol Med 27:134–145
Stein O, Oette K, Dabach Y, Hollander G, Ben Naim M, Stein Y (1992) Persistence of increased cholesteryl ester in human skin fibroblasts is caused by residual exogenous sphingomyelinase and is reversed by phospholipid liposomes. Biochim Biophys Acta 1165:153–159
Suzukawa M, Abbey M, Clifton P, Nestel PJ (1994) Effects of supplementing with vitamin E on the uptake of low density lipoprotein and the stimulation of cholesteryl ester formation in macrophages. Atherosclerosis 110:77–86
Zager RA, Kalhorn TF (2000) Changes in free and esterified cholesterol: hallmarks of acute renal tubular injury and acquired cytoresistance. Am J Pathol 157:1007–1016
Pitkanen AS, Halonen TO, Kilpelainen HO, Riekkinen PJ (1986) Cholesterol esterase activity in cerebrospinal fluid of multiple sclerosis patients. J Neurol Sci 74:45–53
Crameri A, Biondi E, Kuehnle K, Lutjohann D, Thelen KM, Perga S, Dotti CG, Nitsch RM et al (2006) The role of seladin-1/DHCR24 in cholesterol biosynthesis, APP processing and Abeta generation in vivo. EMBO J 25:432–443
Waterham HR, Koster J, Romeijn GJ, Hennekam RC, Vreken P, Andersson HC, FitzPatrick DR, Kelley RI et al (2001) Mutations in the 3beta-hydroxysterol Delta24-reductase gene cause desmosterolosis, an autosomal recessive disorder of cholesterol biosynthesis. Am J Hum Genet 69:685–694
Wechsler A, Brafman A, Shafir M, Heverin M, Gottlieb H, Damari G, Gozlan-Kelner S, Spivak I et al (2003) Generation of viable cholesterol-free mice. Science 302:2087
Benvenuti S, Saccardi R, Luciani P, Urbani S, Deledda C, Cellai I, Francini F, Squecco R et al (2006) Neuronal differentiation of human mesenchymal stem cells: changes in the expression of the Alzheimer’s disease-related gene seladin-1. Exp Cell Res 312:2592–2604
Wu C, Miloslavskaya I, Demontis S, Maestro R, Galaktionov K (2004) Regulation of cellular response to oncogenic and oxidative stress by Seladin-1. Nature 432:640–645
Mangiardi M, Crawford D et al (2011) An animal model of cortical and callosal pathology in multiple sclerosis. Brain Pathol 21:263–278
Crise B, Rose JK (1992) Human immunodeficiency virus type 1 glycoprotein precursor retains a CD4-p56lck complex in the endoplasmic reticulum. J Virol 66:2296–2301
Arcaro A, Grégoire C, Boucheron N, Stotz S, Palmer E, Malissen B, Luescher IF (2000) Essential role of CD8 palmitoylation in CD8 coreceptor function. J Immunol 165:2068–2076
Lee WT, Watson AR (2006) Single-cell analysis of lipid rafts in lymphocytes and in T cell-containing immunoconjugates. Curr Protoc Toxicol Chapter 2:Unit2.11
Legler DF, Doucey MA, Cerottini JC, Bron C, Luescher IF (2001) Selective inhibition of CTL activation by a dipalmitoyl-phospholipid that prevents the recruitment of signaling molecules to lipid rafts. FASEB J 15:1601–1603
Bosch B, Heipertz EL, Drake JR, Roche PA (2013) Major histocompatibility complex (MHC) class II-peptide complexes arrive at the plasma membrane in cholesterol-rich microclusters. JBC 288:13236–13242
Rimmerman N, Bradshaw HB, Kozela E, Levy R, Juknat A, Vogel Z (2012) Compartmentalization of endocannabinoids into lipid rafts in a microglial cell line devoid of caveolin-1. Br J Pharmacol 165:2436–2449
Hattersley KJ, Hein LK, Fuller M (2013) Lipid composition of membrane rafts, isolated with and without detergent, from the spleen of a mouse model of Gaucher disease. Biochem Biophys Res Commun 442:62–67
Lo Verme J, Fu J, Astarita G, La Rana G, Russo R, Calignano A, Piomelli D (2005) The nuclear receptor peroxisome proliferator-activated receptor-alpha mediates the anti-inflammatory actions of palmitoylethanolamide. Mol Pharmacol 67:15–19
Aguado-Llera D, Puebla-Jimenez L, Barrios V, Hernandez-Pinto A, Arilla-Ferreiro E (2011) Role of ethanolamine phosphate in the hippocampus of rats with acute experimental autoimmune encephalomyelitis. Neurochem Int 58:22–34
Maeba R, Ueta N (2003) Ethanolamine plasmalogens prevent the oxidation of cholesterol by reducing the oxidizability of cholesterol in phospholipid bilayers. J Lipid Res 44:164–171
Zoeller RA, Lake AC, Nagan N, Gaposchkin DP, Legner MA, Lieberthal W (1999) Plasmalogens as endogenous antioxidants: somatic cell mutants reveal the importance of the vinyl ether. Biochem J 338:769–776
Acknowledgments
We wish to thank the staff of the Animal Center of the Universidad de Alcalá for their support with the animal care and handling. CIBEROBN is an initiative of the Instituto de Salud Carlos III (ISCIII), Spain. MOIR Mechanisms of Insulin Resistance.
Funding
This work was supported by the Ministerio de Ciencia e Innovación (SAF2010-22277), (SAF2011-29951) and (S2010/BMD-2423).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The animal experiments performed in the present study conform to the guidelines set by the Animal Care Committee of Alcalá University as well as to the International guidelines on the ethical use of animals set by the Council of Europe (Protection of Animals Used for Experimentation, 1986 ETS No. 123), and all experimental protocols were previously approved.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Supplementary Fig. 1
The image shows the cutting plane of the brain of control rats, rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE), rats with CR-EAE pre-treated with a calcium, magnesium and phosphate ethanolamine salt (EAP) and control rats pre-treated with EAP used to evaluate the infiltrates and activation of T cells, Iba1+ cells, IgG levels and TUNEL(+) cells in the hippocampus. This study has been carried out in similar areas of the medial section of the hippocampus rostro-caudal axis. (PPTX 5810 kb)
Supplementary Fig. 2
CD3+ and CD8+ cells. Immunohistochemical labelling of CD3+ and CD8+ cell infiltrates in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) at three phases of the disease: first relapse, remission and second relapse, and effects of an EAP salt. The scale bar: 500 μm and 100 μm. The figure shows the most representative images of five independent stains of the hippocampal sagittal section of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. (PPTX 650 kb)
Supplementary Fig. 3
Iba1+ cells. Panel A: Immunohistochemical labelling of Iba1+ cells in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) at three phases of the disease: first relapse, remission and second relapse, and effects of an EAP salt. The scale bar: 100 μm. Panel B: Immunohistochemical labelling of Iba1+ cells and the score used to determine the degree of activation in the hippocampus. The scale bar: 50 μm. The figure shows the most representative images of five independent stains of the hippocampal sagittal section of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. (PPTX 2012 kb)
Supplementary Fig. 4
Ig G levels. in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) at three phases of the disease, and effects of EAP salt. Immunohistochemical labelling of IgG levels in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) at three phases of the disease: first relapse, remission and second relapse, and effects of an EAP salt. The scale bar: 500 μm. The figure shows the most representative images of five independent stains of the sagittal section of the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. (PPTX 455 kb)
Supplementary Fig. 5
TUNEL (+) cells. Immunohistochemical labelling of TUNEL (+) cells in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) at three phases of the disease; first relapse, remission and second relapse, and effects of an EAP salt. The scale bar: 500 μm and 50 μm. The figure shows the most representative images of five independent stains of the sagittal section of the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) and effects of an EAP salt. (PPTX 546 kb)
Supplementary Fig. 6
The diagram depicts the alterations in the antioxidant defences and the possible mechanism of action of an ethanolamine phosphate salt (EAP) in the hippocampus of rats with chronic relapsing experimental allergic encephalomyelitis (CR-EAE) at the first attack. Panel A: In rats with CR-EAE, the increase of CD3+ cells, TNF-α and IFN-γ levels in the rat hippocampus enhances the amount and activation of resident Iba- 1+ cells. These cells begin to produce reactive oxygen species in the hippocampus. The alterations in the activity and quantity of enzymes involved in antioxidant defence also contribute to cell dysfunction. Our results showed an increase in the amount and activity of superoxide dismutase (SOD),a decrease in the amount and activity of catalase (CAT) and an impairment in the glutathione system (glutathione peroxidase (GP) and glutathione reductase (GR)). These alterations might contribute to the increase of lipid peroxidation and a dysfunction in the cellular activities of the hippocampus. Panel B: The CR-EAE + EAP rats show a decrease in the number of CD3+ cells in the hippocampus and less amount and activation of microglial cells, which involves a decrease in the number of oxidative radicals and lower levels of TNF-α. We have shown that the amount and activity of the antioxidant enzymes SOD, CAT, and the glutathione system (GR and GP) remain stable. All these findings could explain that in the CR-EAE + EAP rats, there is less lipid peroxidation and an improvement in the cellular activities of the hippocampus when compared with EAE-CR at the first attack. (PPTX 2123 kb)
Supplementary Fig. 7
The diagram depicts the alterations in the antioxidant defences and the possible mechanism of action of an ethanolamine phosphate salt (EAP) in the hippocampus of rats with CR-EAE at the second attack. Panel A: In the second relapse of CR-EAE, the amount of CD3+ cells and the amount and activity of Iba1+ cells are higher than in the first relapse. The increase in TNF-α levels in these rats could induce alterations in mitochondrial function that involve an increase of ROS and RNS. In addition, the alterations in the activity and amount of antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT) and the impairment of the glutathione system (glutathione peroxidase (GP) and glutathione reductase (GR)) could contribute to the increase of lipid peroxidation, DNA damage and a dysfunction in the cellular activities of the hippocampus. Panel B: The EAP salt, in rats with CR-EAE, could reduce the alterations of cellular activities in the hippocampus by different mechanisms: these rats show a decrease in the number of CD3+ cells and less reduction in the amount and activation of microglial cells, which involves a decrease in the number of oxidative radicals and lower TNF-α levels. The present study also shows that the amount and activity of the antioxidant enzymes SOD, CAT, and the glutathione system (GR and GP) remain stable. (PPTX 2322 kb)
Rights and permissions
About this article
Cite this article
Perianes-Cachero, A., Lobo, M.V.T., Hernández-Pinto, A.M. et al. Oxidative Stress and Lymphocyte Alterations in Chronic Relapsing Experimental Allergic Encephalomyelitis in the Rat Hippocampus and Protective Effects of an Ethanolamine Phosphate Salt. Mol Neurobiol 57, 860–878 (2020). https://doi.org/10.1007/s12035-019-01774-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-01774-8