Abstract
The cholinergic anti-inflammatory pathway controls the inflammatory response and nonreflexive consciousness through bidirectional communication between the brain and immune system. Moreover, brain acetylcholinesterase activity may have a role in regulating the vagus nerve in this pathway. Thus, we analyzed the role of acetylcholine (ACh) in the inflammatory response 15 days after induction of sepsis by cecal ligation and puncture (CLP). Balb/c mice were pretreated with or without donepezil (5 mg/kg/day, orally) 7 days before CLP, and mice homozygous for vesicular ACh transporter (VAChT) knockdown (KD) were subjected to CLP. All animals were sacrificed 15 days after CLP, and the plasma, spleen, and hippocampus were collected. Characterization of splenic lymphocytes and cytokine levels in the plasma, spleen, and hippocampus was determined. Our results showed a splenomegaly in group CLP. The numbers of cytotoxic T cells, helper T cells, regulatory T cells, B cells, and Th17 cells differed between mice subjected to CLP and to sham operation in both untreated and donepezil-treated groups. In VAChT-KD mice, CLP resulted in decreased cytotoxic and helper T cells and increased in Th17 cells compared with the sham. Additionally, in VAChT-KD mice, the levels of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF-α, were increased following CLP. Thus, we concluded that ACh affected the inflammatory response at 15 days after CLP since stimulation of cholinergic transmission increased the proliferation of lymphocytes, including regulatory T cells, in association with a lower inflammatory profile and VAChT-KD decreased the number of lymphocytes and increased inflammation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis is a systemic inflammatory response to infection [1–4] characterized by an imbalance between pro- and anti-inflammatory responses to pathogens [5, 6]. Reciprocal interactions between the immune system and central nervous system (CNS) are one of the major components of the host response in septic shock.
Tracey [7] proposed an important role of parasympathetic cholinergic pathways in the bidirectional communication between the brain and immune system; this pathway is called the cholinergic anti-inflammatory pathway and has been shown to control the inflammatory response and nonreflexive consciousness. Activation of this pathway, either by electrical stimulation of the vagus nerve or through pharmacological approaches, has shown to significantly ameliorate cytokine-mediated disease models, including endotoxemia [8, 9] and sepsis [10]. Moreover, this pathway is functionally hardwired to the spleen via the common coeliac branch of the vagus nerve, and the spleen is required for the anti-inflammatory potential of the vagus nerve [11].
Previous studies have indicated that brain acetylcholinesterase activity and muscarinic receptors, which regulate cholinergic network signaling, modulate vagus nerve outflow [12, 13]. Accordingly, brain acetylcholinesterase activity may have a role in regulating the vagus-nerve-based cholinergic anti-inflammatory pathway [14]. Thus, in this study, we analyzed the role of acetylcholine (ACh) in the inflammatory response in mice 15 days after induction of sepsis by cecal ligation and puncture (CLP).
Methods
Mice
Adult male Balb/c and homozygous mutant vesicular ACh transporter-knockdown (VAChT-KD) mice (8 weeks old, weighing 20–25 g) were used for experiments. The animals provided from the School Facility were specific pathogen-free (SPF). Animals were maintained in a climate-controlled facility with an automatic light/dark cycle (http://www.biot.fm.usp.br/), with food and water available ad libitum. All procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. The study protocol was approved by the Research Ethics Committee of the São Paulo School of Medicine (#0417/13).
Mutant VAChT-KD used in these experiments was generated by intercrossed heterozygous mice. These mice showed a reduction in approximately 65 % in VAChT levels in the nervous system and also in other organs [15] which is correlated to a reduction in ACh levels [16, 17].
Balb/c mice were pretreated with or without donepezil (5 mg/kg/day, orally) 7 days before CLP (n = 10 mice per group). Alternatively, mice homozygous for VAChT-KD were subjected to CLP. Sham operations were performed as a control. All animals were sacrificed 15 days after CLP.
CLP
Mice were anesthetized with a mixture of ketamine (80 mg/kg) and xylazine (10 mg/kg), given intraperitoneally (ip). CLP was induced as previously described [18]. Under aseptic conditions, a 2-cm midline laparotomy was performed to allow exposure of the cecum with the adjoining intestine. Subsequently, 50 % of the cecum was ligated and then perforated by a single puncture with a 21-gauge needle at the least vascularized area. The cecum was then gently squeezed to extrude a small amount of feces from the perforation site. The cecum was returned to the peritoneal cavity, and the laparotomy was closed with 4.0 silk sutures. Sham-operated mice were subjected to all surgical procedures, but the cecum was neither ligated nor perforated. The animals were returned to their cages with free access to food and water. Animals were monitored thereafter, and survival rates were determined.
Tissue Isolation
After 15 days, the animals were sacrificed, and blood was collected by cardiac puncture and transferred to EDTA-coated tubes. The blood was centrifuged for 15 min at 3200×g, and plasma samples were collected. The spleens and brains were harvested under aseptic conditions. Spleens were weighed and kept on ice. Spleens were then divided into two parts; one was used for isolation of splenocytes, and the other was used for measurement of cytokines. The brain was removed, and the hippocampus was isolated for measurement of cytokines.
Lymphocyte Isolation
Spleens were placed between two frosted autoclaved slides that were gently pressed together in opposing directions to dissociate the tissue. As the tissue began to dissociate, the slides were dipped into 8 mL phosphate-buffered saline (PBS) in a Petri dish until only the connective tissue remained. The cell suspension was aseptically transferred into a sterile 15-mL centrifuge tube. Spleen cell suspensions were centrifuged at 400×g for 10 min and then resuspended in PBS. Erythrocytes were removed from splenic cell suspensions by a brief (5–10 min) exposure to 1 mL of Tris-buffered NH4Cl on ice, and cells were then centrifuged at 400×g for 10 min and resuspended in PBS.
Characterization of Lymphocytes by Flow Cytometry
Using flow cytometry, we characterized five types of lymphocytes: cytotoxic T lymphocytes (CD3+, CD4−, CD8+), helper T lymphocytes (CD3+, CD4+, CD8−), B lymphocytes (CD3+, CD19+), regulatory T cells (CD3+, CD4+, CD25+), and T helper 17 (Th17) cells (CD3+, interleukin (IL)-17+). Lymphocytes were incubated for 30 min at 4 °C with the proper antibody set diluted in PBS containing 2 % inactivated fetal bovine serum or permeabilization buffer. All antibodies were supplied by BioLegend (anti-CD3-FITC, anti-CD4-PE, anti-CD8-PE/Cy5, anti-CD19-PE/Cy7, anti-CD25-APC/Cy7, and anti-IL-17A-APC). All analyses were carried out on an EasyCyte 8HT flow cytometer (Guava, Hayward, CA, USA) for a minimum of 5000 events. The analyses were performed using Guava Express Pro software.
Cytokines
For determination of cytokines levels, frozen tissues (100 mg), including the spleen and hippocampus, were pulverized in liquid nitrogen. Samples were homogenized in Triton-X100, 150 mM NaCl, 10 mM Tris HCl (pH 7.5), 1 % NP40, 1 % sodium oxybate, 0.1 % SDS, and proteolytic enzyme inhibitors (40 μg/mL phenylmethylsulfonyl fluoride [1 mM]; Sigma, St, Louis, MO, USA). After debris separation through centrifugation for 40 min at 10,000 rpm, the supernatants were collected, and protein concentrations were determined by the Bradford method (Bio-Rad, Hercules, CA, USA). Samples were kept at −80 °C until assayed.
The concentrations of IL-1α, IL-1β, IL-4, IL-6, IL-10, IL-12p40, IL-12p70, macrophage inflammatory protein (MIP)-1α (MIP-1α), MIP-1β, and tumor necrosis factor α (TNF-α) in the spleen, hippocampus, and plasma were determined by MilliPlex technology, (# MCYTOMAG-70K, Merck KGaA, Darmstadt, Germany). The assays were performed according to the manufacturer’s instructions. The samples were analyzed on a MagPix system, and the data were collected by Luminex xPONENT software.
Statistical Analysis
All values were expressed as means ± standard errors of the mean (SEMs). Analyses were performed using InStat Statistical Software (GraphPad, La Jolla, CA, USA). Comparisons between experimental groups were performed using ANOVA and Bonferroni’s post-test. Log-rank tests were used to analyze survival. Differences with p values of less than 0.05 were considered significant.
Results
Survival Rates
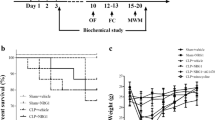
The results of the survival experiments are shown in Fig. 1. The survival rate was 100 % in all sham groups. In normal mice subjected to CLP, the survival rate was 90 % at 15 days after CLP. In contrast, that after donepezil treatment was 70 %, and a further reduction to 60 % survival was observed in VAChT-KD mice subjected to CLP; this latter reduction was statistically significant compared with the sham group.
Spleen Weights
Spleens were weighed after mice were sacrificed at 15 days after CLP. Significant increases in spleen weights and the spleen weight to body weight ratio were observed in all mice subjected to CLP as compared with all sham-operated mice (Fig. 2a, b). Representative images of spleens are shown in Fig. 2c.
Weights of spleens 15 days after induction of sepsis by CLP in mice. a Spleen weight, b ratio of spleen weight to body weight, and c representative image of spleens from sham and CLP groups. Data are presented as the mean ± SEM (n = 10 mice per group). ANOVA and Bonferroni’s post-test, a p < 0.05 compared with the sham untreated group; c p < 0.05 compared with the sham donepezil group; e p < 0.05 compared with the sham VAChT-KD group
Lymphocytes
The numbers of different lymphocyte subtypes are presented in Fig. 3. For untreated normal mice, increases in cytotoxic T cells, helper T cells, regulatory T cells, B cells, and Th17 cells were observed following CLP as compared with those in the sham group. We also observed a significant increase in this group (CLP untreated) compared with groups sham and CLP VAChT-KD of cytotoxic T cells and helper T cells and a significant decrease compared to the CLP donepezil group in regulatory T cells, B cells, and Th17 cells.
Characterization of splenic lymphocytes 15 days after induction of sepsis by CLP in mice. Five types of lymphocytes were characterized by flow cytometry. Cytotoxic T cells (a), helper T cells (b), B lymphocytes (c), regulatory T cells (d), and T helper 17 cells (e). Data are presented as the mean ± SEM (n = 10 mice per group). ANOVA and Bonferroni’s post-test, a p < 0.05 compared with the sham untreated group; c p < 0.05 compared with the sham donepezil group; d p < 0.05 compared with the CLP donepezil group; e p < 0.05 compared with the sham VAChT-KD group; f p < 0.05 compared with the CLP VAChT-KD group
Similarly, in mice treated with donepezil, increases in cytotoxic T cells, regulatory T cells, B cells, and Th17 cells were observed in the CLP group compared with all sham groups (untreated, donepezil, and VAChT-KD) and CLP VAChT-KD groups.
In the sham VAChT-KD group, the cytotoxic T cells are less than the sham donepezil group. However, CLP VAChT-KD mice caused decreases in cytotoxic and helper T cells compared to all other groups and an increase in Th17 cells as compared with those in the sham group.
Cytokines
The results of cytokine expression analysis in spleens, hippocampus tissues, and plasma samples are shown in Tables 1, 2, and 3, respectively. In spleen tissues, CLP induced significant upregulation of IL-6 and IL-12p40 content as compared to all sham groups and CLP donepezil group. An increase was also observed in IL-12p70 compared to all sham groups, in MIP-1α and TNF compared to all other groups and MIP-1β compared to sham and CLP VAChT-KD groups.
In mice treated with donepezil, CLP caused increases in IL-4 in the spleen as compared to groups CLP untreated, sham donepezil, and sham VAChT-KD. Finally, in VAChT-KD mice, IL-6, IL-10, and IL-12p40 levels were increased in the spleen following CLP compared to all sham groups and CLP donepezil group.
In the hippocampus, the CLP VAChT-KD group caused a significant increase in IL-6 content compared with the sham untreated group and increased in IL-10 content compared with the sham and CLP untreated groups and CLP donepezil group.
In plasma samples from untreated normal mice, CLP caused a significant increase in IL-6 levels compared with all sham groups and a decrease in MIP-1β levels after 15 days compared with the sham untreated group. In mice sham VAChT-KD, IL-4 levels were increased compared with CLP untreated group, sham, and CLP donepezil groups. Additionally, in VAChT-KD mice, CLP resulted in increased levels of IL-6 compared with all sham groups and CLP donepezil group, and IL-10 increased compared with the sham donepezil group, and MIP-1α increased compared with sham and CLP untreated groups, CLP donepezil group and sham VAChT-KD group. However, CLP VAChT-KD group decreased in IL-12p40 compared with sham untreated and sham donepezil groups.
Discussion
In the present study, we examined the role of ACh in the inflammatory response 15 days after induction of sepsis by CLP. Our results showed that stimulation of cholinergic transmission was elevated by ACh and that donepezil increased the proliferation of lymphocytes and decreased pro-inflammatory cytokine expression. However, knockdown of VAChT resulted in decreased numbers of cytotoxic and helper T cells and increased numbers of Th17 cells. Thus, these data provided important insights into the mechanisms through which ACh mediates the inflammatory response after induction of sepsis.
Parasympathetic cholinergic pathways are involved in bidirectional communication between the brain and immune system, providing both immunosensing and immunosuppressive functions. The CNS controls the function of the peripheral vagus nerve through the efferent arm (cholinergic anti-inflammatory pathway), and the primary released neurotransmitter ACh regulates cytokine production specifically via α7nAChR-dependent signaling [8, 19, 20]. Stimulation of the cholinergic anti-inflammatory pathway by electrical or pharmacological methods significantly suppresses systemic levels of TNF and other pro-inflammatory cytokines during endotoxemia [8, 21, 22]. However, little is known about the role of ACh in the inflammatory response in survivors of sepsis.
In order to study the role of the efferent arm of the CNS in the spleen, brain, and systemic inflammation, we used two strategies: increase the levels of ACh or decrease the levels of ACh. Donepezil [diethyl(3,5-di-ter-butyl-4-hydroxybenzyl)phosphonate] is a long-acting, reversible cholinesterase inhibitor known to improve memory and cognitive function in patients with Alzheimer’s disease [23]. This compound inhibits the breakdown of ACh and compensates for ACh deficiency in the brain [24, 25]. Thus, in this study, we used donepezil to facilitate cholinergic transmission in septic animals. On the other hand, to reduce cholinergic transmission, we used animals genetically modified to have a reduced expression of VAChT [26–29]. VAChT-KC mice exhibit a 65 % reduction in VAChT expression [16]. This transporter controls the storage of ACh, representing an ideal target to block cholinergic function.
In this study, we hypothesized that septic brain damage impairs control of the cholinergic parasympathetic efferent arm, thereby aggravating immune dysfunction and creating a vicious circle. Late stages of sepsis have been shown to be associated with a state of immunosuppression. However, it is still unknown how changes in immune function found in late sepsis are related to brain damage. Sepsis involves dysregulated production of cytokines, a pathological state that causes tissue injury and organ dysfunction, including septic encephalopathy, renal failure, respiratory dysfunction, and death. In sepsis, immune cellular and humoral responses are hyperactivated [30, 31], manifested mainly as enhanced production and release of pro-inflammatory mediators [32]. In this context, we verified the effects of ACh on the inflammatory responses of mice 15 days after induction of sepsis by CLP through activation or suppression of cholinergic transmission.
The anatomical basis of the cholinergic anti-inflammatory pathway includes the preganglionic neurons, which originate in the dorsal motor nucleus of the vagus, and the postganglionic neurons, located in ganglia of the celiac-superior mesenteric plexus; this pathway then reaches the spleen through the splenic nerve [33], supporting the functional hardwiring of this pathway to the spleen. The spleen is a vascular organ, and one of its main functions is to initiate adaptive immune responses to antigens captured in the blood [34]. After infection, splenocytes are activated and proliferate rapidly, leading to splenomegaly, an important complication of acute and chronic infection first described more than 100 years ago [35, 36]. In order to verify whether the CLP model was able to induce splenomegaly, we weighed the spleens of the mice in the sham and CLP groups. Spleens from mice subjected to CLP were larger and weighed more than those from sham-operated mice, regardless of treatment with donepezil or VAChT-KD. Consistent with this, Valdés-Ferrer and colleagues [37] showed that sepsis survivors developed significant splenomegaly from the first week to the fourth week after CLP.
Studies have shown that sepsis induces alterations in spleen cell populations, with increased apoptosis in lymphocytes and dendritic cells [38]. Apoptosis of immune cells is followed by generalized immune dysfunction in T/B cells [39, 40]. These changes, along with changes in the phagocytic arm of the immune response, have been shown to be associated with increased morbidity and mortality in patients with sepsis [41]. In a study by Gomez et al. [42], patients with sepsis exhibited higher percentages of apoptotic CD4+ T cells and CD8+ T cells and higher levels of both pro- and anti-inflammatory cytokines at admission. Ono et al. [43] found a dramatic increase in the percentage of regulatory T cells and circulating CD4+ T cells in the peripheral blood in patients with septic shock. Consistent with these observations, we observed significant increases in the numbers of cytotoxic T lymphocytes, helper T lymphocytes, B lymphocytes, regulatory T cells, and Th17 cells in the spleens of mice subjected to CLP. Moreover, our data demonstrated that ACh regulated the numbers of lymphocytes in the spleen.
T cells from the spleen play an important role in the inflammatory reflex efferent arc, and T cells expressing ChAT+ release ACh in the spleen. ACh then acts on the α7 nicotinic acetylcholine receptors (α7) on macrophages and other cells and suppresses endotoxin-induced release of TNF [44, 45]. Regarding cytokines, we observed that CLP increased the expression of a variety of pro- and anti-inflammatory cytokines in all experimental groups. Moreover, ACh played an important role in mediating cytokine production in tissues and plasma. In particular, VAChT-KD promoted a pro-inflammatory profile, while donepezil reduced the levels of cytokines or maintained their low expression, thereby blocking the inflammatory response. In terms of the anti-inflammatory effects of ACh, administration of acetylcholinesterase inhibitors has been shown to attenuate disease severity and decrease pro-inflammatory cytokine levels in various experimental models of inflammation [45]. A study by Pavlov and collaborators [14] showed that treatment of an endotoxemic animal with an acetylcholinesterase inhibitor (galantamine) caused a sharp decrease in TNF levels in the serum. In another experiment, animals received the acetylcholinesterase inhibitor neostigmine, and reduced levels of TNF-α, IL-1β, and IL-6 were observed at 24 h after CLP [46]. In the current study, we also found that donepezil, an acetylcholinesterase inhibitor, caused reductions in IL-6 levels in the spleen, hippocampus, and plasma and in IL-1β and TNF-α in the plasma at 15 days after CLP, showing that the increase of ACh in addition to increasing regulatory lymphocytes, decreases inflammatory cytokines, decreasing inflammation.
ACh significantly attenuates the release of TNF-α, IL-1β, IL-6, and IL-18 but not that of the anti-inflammatory cytokine IL-10 in lipopolysaccharide-stimulated human macrophage cultures [8]. In our study, we found that IL-10 expression was higher in the spleen, hippocampus, and plasma at 15 days after CLP following suppression of cholinergic transmission by VAChT-KD in mice. IL-10 plasma levels function to counteract the existing inflammatory process, and we observed an increase in inflammation in VAChT-KD mice, concurrent with reduced numbers of regulatory T cells. These results supported the survival analysis, in which VAChT-KD animals exhibited the highest mortality rates, potentially due to the induction of pro-inflammatory pathways. On the other hand, animals treated with donepezil also exhibited increased mortality concurrent with a predominantly anti-inflammatory profile; this may have resulted in reduced antibacterial capacity of the host immune system. The CAN mediates the balance of pro- and anti-inflammatory responses through the function of ACh in the spleen; therefore, it is necessary to avoid changes or impairment in the brain, which may affect the brain-spleen interaction.
Taken together, our data supported that donepezil, i.e., elevated ACh levels, caused an increased number of lymphocytes, particularly regulatory T cells, and decreased levels of pro-inflammatory cytokines, thereby lowering systemic inflammation and inflammation in the brain and spleen. Conversely, reduced levels of ACh increased inflammation and mortality rates following CLP-induced sepsis; however, the increase in ACH resulted in reduced organ damage. Intense anti-inflammatory activity may contribute to lower bacterial clearance and consequently increased mortality rates. ACh exhibited systemic and local anti-inflammatory activities in the spleen, although changes in ACh prevented the immune system from functioning properly.
In conclusion, Ach has an important role in the inflammatory profile 15 days after CLP, since the stimulation of cholinergic transmission, the donepezil experiment, increases the proliferation of lymphocytes, including regulatory T cells, where we observe a lower profile inflammation. And unlike the decrease in cholinergic transmission, the VAChT-KD experiment, we observed a decrease lymphocytes and increased inflammation.
References
Balk RA, Bone RC (1989) The septic syndrome. Definition and clinical implications. Crit Care Clin 5:1–8
Barbeiro DF, Barbeiro HV, Faintuchi J, Ariga SK, Mariano M, Popi AF, De Souza HP, Velasco IT, Soriano FG (2011) B-1 cells temper endotoxemic inflammatory responses. Immunobiology 216:302–308. doi:10.1016/j.imbio.2010.08.002
Lorigados CB, Soriano FG, Szabo C (2011) Pathomechanisms of myocardial dysfunction in sepsis. Endocr Metab Immune Disord Drug Targets 10:274–284. doi:10.2174/187153010791936856
Melo ES, Goloubkova T, Barbeiro DF, Gorjao R, Vasconcelos D, Szabo C, Curi R, De Lima-Salgado TM, Velasco IT, Soriano FG (2011) Endotoxin tolerance: selective alterations in gene expression and protection against lymphocyte death. Immunobiology 215:435–442. doi:10.1016/j.imbio.2009.09.002
Vandijck D, Decruyenaere JM, Blot SI (2006) The value of sepsis definitions in daily ICU-practice. Acta Clin Belg 5:220–226, http://dx.doi.org/10.1179/acb.2006.037
Hotchkiss RS, Karl IK (2003) The pathophysiology and treatment of sepsis. New Engl J Med 348:138–150. doi:10.1056/NEJMra021333
Tracey KJ (2002) The inflammatory reflex. Nature 420:853–859. doi:10.1038/nature01321
Borovikova LV, Ivanova S, Zhang M, Yang H, Botchkina GI, Watkins LR, Wang H, Abumrad N, Eaton JW, Tracey KJ (2000) Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405:458–462. doi:10.1038/35013070
Pavlov VA, Ochani M, Yang LH, Gallowitsch-Puerta M, Ochani K, Lin X, Levi J, Parrish WR, Rosas-Ballina M, Czura CJ, Larosa GJ, Miller EJ, Tracey KJ, Al-Abed Y (2007) Selective alpha7-nicotinic acetylcholine receptor agonist GTS-21 improves survival in murine endotoxemia and severe sepsis. Crit Care Med 35:1139–1144. doi:10.1097/01.CCM.0000259381.56526.96
Van Westerloo DJ, Giebelen IA, Florquin S, Daalhuisen J, Bruno MJ, De Vos AF, Tracey KJ, Van Der Poll T (2005) The cholinergic anti-inflammatory pathway regulates the host response during septic peritonitis. J Infect Dis 191:2138–2148. doi:10.1086/430323
Huston JM, Ochani M, Rosas-Ballina M, Liao H, Ochani K, Pavlov VA, Gallowitsch-Puerta M, Ashok M, Czura CJ, Foxwell B, Tracey KJ, Ulloa L (2006) Splenectomy inactivates the cholinergic antiinflammatory pathway during lethal endotoxemia and polymicrobial sepsis. J Exp Med 203:1623–1628. doi:10.1084/jem.20052362
Gotoh M, Iguchi A, Yatomi A, Uemura K, Miura H, Futenma A, Kato K, Sakamoto N (1989) Vagally mediated insulin secretion by stimulation of brain cholinergic neurons with neostigmine in bilateral adrenalectomized rats. Brain Res 493(1):97–102. doi:10.1016/0006-8993(89)91003-2
Pavlov VA, Ochani M, Gallowitsch-Puerta M, Ochani K, Huston JM, Czura CJ, Al-Abed Y, Tracey KJ (2006) Central muscarinic cholinergic regulation of the systemic inflammatory response during endotoxemia. Proc Natl Acad Sci U S A 103(13):5219–5223. doi:10.1073/pnas.0600506103
Pavlov VA, Parrish WR, Rosas-Ballina M, Ochani M, Puerta M, Ochani K, Chavan S, Al-Abed Y, Tracey KJ (2009) Brain acetylcholinesterase activity controls systemic cytokine levels through the cholinergic anti-inflammatory pathway. Brain Behav Immun 23(1):41–45. doi:10.1016/j.bbi.2008.06.011
Pinheiro NM, Miranda CJ, Perini A, Câmara NO, Costa SK, Alonso-Vale MI, Caperuto LC, Tibério IF, Prado MA, Martins MA, Prado VF, Prado CM (2015) Pulmonary inflammation is regulated by the levels of the vesicular acetylcholine transporter. PLoS One 10(3):e0120441. doi:10.1371/journal.pone.0120441
Prado VF, Martins-Silva C, De Castro BM, Lima RF, Barros DM, Amaral E, Ramsey AJ, Sotnikova TD, Ramirez MR, Kim HG, Rossato JI, Koenen J, Quan H, Cota VR, Morales MF, Gomez MV, Guatimosim C, Wetsel WC, Kushmerick C, Pereira GS, Gainetdinova RR, Izquierdo I, Caron MG, Prado MA (2006) Mice deficient for the vesicular acetylcholine transporter are myasthenic and have deficits in object and social recognition. Neuron 51:601–612, http://dx.doi.org/10.1016/j.neuron.2006.08.005
Lima RF, Prado VF, Prado MA, Kushmerick C (2010) Quantal release of acetylcholine in mice with reduced levels of the vesicular acetylcholine transporter. J Neurochem 113(4):943–951. doi:10.1111/j.1471-4159.2010.06657.x
Soriano FG, Liaudet L, Szabo E, Virag L, Mabley JG, Pacher P, Szabo C (2002) Resistance to acute septic peritonitis in poly(ADP-ribose) polymerase-1-deficient mice. Shock 17:286–292
Tracey KJ (2005) Fatal sequence: the killer within. J Clin Invest 115(12):3304. doi:10.1172/JCI27259
Tracey KJ (2007) Physiology and immunology of the cholinergic antiinflammatory pathway. J Clin Invest 117(2):289–296. doi:10.1172/JCI30555
Pavlov VA, Wang H, Czura CJ, Friedman SG, Tracey KJ (2003) The cholinergic anti-inflammatory pathway: a missing link in neuroimmunomodulation. Mol Med 9(5–8):125–134
Gallowitsch-Puerta M, Pavlov VA (2007) Neuro-immune interactions via the cholinergic anti-inflammatory pathway. Life Sci 80(24–25):2325–2329. doi:10.1016/j.lfs.2007.01.002
Hansen RA, Gartlehner G, Webb AP, Morgan LC, Moore CG, Jonas DE (2008) Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: a systematic review and meta-analysis. Clin Interv Aging 3:211–225, http://ukpmc.ac.uk/abstract/MED/18686744
Francis PT, Palmer AM, Snape M, Wilcock GK (1999) The cholinergic hypothesis of Alzheimer’s disease: a review of progress. J Neurol Neurosurg Psychiatry 66:137–147. doi:10.1136/jnnp.66.2.137
Giacobini E (1993) Pharmacotherapy of Alzheimer disease: new drugs and novel strategies. Prog Brain Res 98:447–454. doi:10.1016/S0079-6123(08)62431-0
Barbosa JJR, Massensini AR, Santos MS, Meireles SI, Gomez RS, Gomez MV, Romano-Silva MA, Prado VF, Prado MA (1999) Expression of the vesicular acetylcholine transporter, proteins involved in exocytosis, and functional calcium signaling in varicosities and soma of a murine septal cell line. J Neurochem 73:1881–1893. doi:10.1046/j.1471-4159.1999.01881.x
Santos MS, Barbosa J Jr, Veloso GS, Ribeiro F, Kushmerick C, Gomez MV, Ferguson SS, Prado VF, Prado MA (2001) Trafficking of green fluorescent protein tagged-vesicular acetylcholine transporter to varicosities in a cholinergic cell line. J Neurochem 78:1104–1113. doi:10.1046/j.1471-4159.2001.00494.x
Barbosa JJR, Ferreira LT, Martins-Silva C, Santos MS, Torres GE, Caron MG, Gomez MV, Ferguson SS, Prado MA, Prado VF (2002) Trafficking of the vesicular acetylcholine transporter in SN56 cells: a dynamin-sensitive step and interaction with the AP-2 adaptor complex. J Neurochem 82:1221–1228. doi:10.1046/j.1471-4159.2002.01068.x
Prado MAM, Reis RAM, Prado VF, De Mello MC, Gomez MV, De Mello FG (2002) Regulation of acetylcholine synthesis and storage. Neurochem Int 41:291–299. doi:10.1016/S0197-0186(02)00044-X
Karima R, Matsumoto S, Higashi H, Matsushima K (1999) The molecular pathogenesis of endotoxic shock and organ failure. Mol Med Today 5:123–132. doi:10.1016/S1357-4310(98)01430-0
Kunze K (2002) Metabolic encephalopathies. J Neurol 249:1150–1159. doi:10.1007/s00415-002-0869-z
Hoesel LM, Gao H, Ward PA (2006) New insights into cellular mechanisms during sepsis. Immunol Res 34:133–141. doi:10.1385/IR:34:2:133
Rosas-Ballina M, Ochani M, Parrish WR, Ochani K, Harris YT, Huston JM, Chavan S, Tracey KJ (2008) Splenic nerveis required for cholinergic antiinflammatory pathway control of TNF in endotoxemia. Proc Natl Acad Sci U S A 105:11008–11013. doi:10.1073/pnas.0803237105
Abbas AK (2011) Imunologia celular e molecular. Elsevier, Rio de Janeiro
Osler W (1908) Remarks on the functions of an out-patient department: made at the opening of the new out - patient department, Cardiff Infirmary, May 20th,1908. Br Med J 1:1470–1471
Pozo AL, Godrey EM, Bowles KM (2009) Splenomegaly: investigation, diagnosis and management. Blood Rev 23:105–111. doi:10.1016/j.blre.2008.10.001
Valdés-Ferrer SI, Rosas-Ballina M, Olofsson PS, Dancho ME, Ochani M, Li JH, Scheinerman JA, Katz DA, Levine YA, Hudson LK, Yang H, Pavlov VA, Roth J, Blanc L, Antoine DJ, Chavan SS, Andersson U, Diamond B, Tracey KJ (2013) HMGB1 mediates splenomegaly and expansion of splenic CD11b+ Ly-6C (high) inflammatory monocytes in murine sepsis survivors. J Intern Med 274:381–390. doi:10.1111/joim.12104
Hotchkiss RS, Monneret G, Payen D (2013) Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat Rev Immunol 13:862–874. doi:10.1038/nri3552
Ayala A, Chung CS, Song GY. (1999) Lymphocyte activation, anergy, and apoptosis in polymicrobial sepsis, in: Marshall, J.C., Cohen, J. (Eds.), Immune Response in the Critically Ill. Springer, pp. 227–245. doi: 10.1007/978-3-642-57210-4_16
Mannick JÁ (1993) Trauma, sepsis, and immune defects. In: Faist E, Meakins JL, Schildberg FW (eds) Host defense dysfunction in trauma, shock and sepsis. Springer, Berlin, pp 15–21
Ayala A, Lomas JL, Grutkoski PS, Chung CS (2003) Pathological aspects of apoptosis in severe sepsis and shock? Int J Biochem Cell Biol 35:7–15. doi:10.1016/S1357-2725(02)00099-7
Gomez HG, Gonzalez SM, Londoño JM, Hoyos NA, Niño CD, Leon AL, Velilla PA, Rugeles MT, Jaimes FA (2014) Immunological characterization of compensatory anti-inflammatory response syndrome in patients with severe sepsis: a longitudinal study*. Crit Care Med 42:771–780. doi:10.1097/CCM.0000000000000100
Ono S, Kimura A, Hiraki S, Takahata R, Tsujimoto H, Kinoshita M, Miyazaki H, Yamamoto J, Hase K, Saitoh D (2013) Removal of increased circulating CD4+CD25+Foxp3+ regulatory T cells in patients with septic shock using hemoperfusion with polymyxin B-immobilized fibers. Surgery 153:262–271. doi:10.1016/j.surg.2012.06.023
Rosas-Ballina M, Olofsson PS, Ochani M, Valdés-Ferrer SI, Levine YA, Reardon C, Tusche MW, Pavlov VA, Andersson U, Chavan S, Mak TW, Tracey KJ (2011) Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science 334:98–101. doi:10.1126/science.1209985
Olofsson PS, Rosas-Ballina M, Levine YA, Tracey KJ (2012) Rethinking inflammation: neural circuits in the regulation of immunity. Immunol Rev 248:188–204. doi:10.1111/j.1600-065X.2012.01138.x
Hofer S, Eisenbach C, Lukic IK, Schneider L, Bode K, Brueckmann M, Mautner S, Wente MN, Encke J, Werner J, Dalpke AH, Stremmel W, Nawroth PP, Martin E, Krammer PH, Bierhaus A, Weigand MA (2008) Pharmacologic cholinesterase inhibition improves survival in experimental sepsis. Crit Care Med 36:404–408. doi:10.1097/01.CCM.0B013E31816208B3
Acknowledgments
The authors thank FAPESP for providing financial support. We would like to thank Editage (www.editage.com.br) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeremias, I.C., Victorino, V.J., Barbeiro, H.V. et al. The Role of Acetylcholine in the Inflammatory Response in Animals Surviving Sepsis Induced by Cecal Ligation and Puncture. Mol Neurobiol 53, 6635–6643 (2016). https://doi.org/10.1007/s12035-015-9538-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9538-y