Abstract
Long non-coding RNA LINC01106 is an lncRNA aberrantly expressed in gastric cancer (GC). However, the accurate function remains unclear. The objective of this investigation is to explore detailed regulatory mechanism of lncRNA LINC01106 in GC. The expression of lncRNA LINC01106, MYCN, and miR-34a-5p was determined by quantitative real-time polymerase chain reaction (qRT-PCR). Cell viability was examined using MTT assay. Migratory and invasive abilities of GC cells were evaluated by transwell assay. The targeting relation among lncRNA LINC01106, MYCN, and miR-34a-5p was tested by dual-luciferase reporter (DLR) assay. Relative protein expression of MYCN was assessed via western blot. Besides, a xenograft mouse model was established to assess the role of LINC01106 in GC in vivo. LncRNA LINC01106 and MYCN expression were boosted and miR-34a-5p expression was reduced in GC cells and tissues compared to their controls. Functionally, decreased lncRNA LINC01106 or increased miR-34a-5p restrained GC cells in viability, invasion, and migration in vitro. LINC01106 down-regulation suppressed tumor growth of mice in vivo. In terms of mechanism, lncRNA LINC01106 directly targeted miR-34a-5p and was inversely correlated with miR-34a-5p. MYCN was targeted by miR-34a-5p and was inversely correlated with miR-34a-5p. There was a positive correlation between LINC01106 and MYCN. LINC01106 knockdown led to the suppression of cell invasion, migration, and viability, whereas these effects caused by LINC01106 knockdown were reversed by miR-34a-5p down-regulation or MYCN up-regulation in GC cells. Silencing of lncRNA LINC01106 attenuated cell viability, invasion, and migration by sponging miR-34a-5p to target MYCN in GC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC) refers to a common tumor of the digestive tract, and lifestyle and environmental factors are main contributors to GC etiology [1]. It is considered as the fifth most common cancer with high mortality worldwide [2]. According to statistics, over 1,000,000 new cases of GC are diagnosed globally, causing about 783,000 deaths in 2018 [3]. At present, surgery, radiation, and chemotherapy are major strategies for GC treatment [4]. Although great progress has been achieved in GC treatment, the therapeutic effect is far from satisfaction [5]. Hence, there is an urgent need to discover new options to improve GC therapy.
Long non-coding RNAs (lncRNAs) are catalogued as RNA molecules longer than 200 nucleotides, which are incapable of encoding proteins [6, 7]. Abundant reports have documented the critical roles of lncRNAs in the control of GC progression [8,9,10]. Huang et al. have proved that silencing of lncRNA AK023391 represses cell invasion and migration of GC cells and suppresses tumor growth in mice [8]. Luo et al. have revealed that down-regulation of lncRNA LINC00483 attenuates the viability, invasion, and migration of GC cells and restrains tumor growth in mice [9]. Pan et al. have uncovered that silencing of lncRNA LIFR-AS1 restrains GC cells from proliferating, invading, and migrating in vitro [10]. Among multiple lncRNAs, lncRNA LINC01106 has drawn our attention due to its association with GC [11, 12]. For instance, the expression of lncRNA LINC01106 is elevated in GC tissues in comparison with adjacent normal tissues [11]. LncRNA LINC01106 is identified as key prognostic factor in GC based on data of survival analyses [12]. However, the specific role and potential mechanisms of lncRNA LINC01106 in GC have not been elaborated.
It is well known that lncRNAs fulfill their functions via sponging microRNAs (miRNAs) [13]. MiRNAs, non-coding RNAs with 20–24 nucleotides, are reported to participate in post-transcriptional modulation of genes [14]. Ample evidence has revealed that miRNAs participate in regulating the pathological process of GC [15,16,17]. For example, miR-93-5p up-regulation accelerates GC cells to migrate, invade, and proliferate [15]. MiR-339-5p overexpression suppresses migration and proliferation of GC cells [16]. MiR-532 up-regulation enhances migratory and invasive abilities of GC cells [17]. MiR-34a-5p, belonging to the has-miR-34 family, has also shown potential as a biomarker and a regulator in GC [18, 19]. One study from Hu et al. has reported that miR-34a expression in GC tissues is lower than that in adjacent non-cancer tissues [18]. Another study from Wei et al. has indicated that miR-34a represses the proliferation and invasion of GC cells [19]. On the other hand, the regulatory interplay between miR-34a-5p and lncRNA HOTAIR/lncRNA DANCR/lncRNA XIST has been unveiled in varied cancers [20,21,22]. Nevertheless, the regulatory mechanism between lncRNA LINC01106 and miR-34a-5p in GC remains unclear.
To our knowledge, a common feature of miRNAs is that they can exert extensive roles by binding to their target genes [23]. Previously, miR-34a-5p is unveiled to directly target programmed cell death 1 ligand 1 (PD-L1) [24], the transcription factor YY1 [25], and sphingosine-1-phosphate phosphatase 1 (SGPP1) [26] in diverse cancers. MYCN, a member of the MYC family of proto-oncogenes, is usually expressed in the migrating neural crest [27]. It is uncovered to implicate in several cancers [28, 29]. Liu et al. have reported that MYCN contributes to cell proliferation in non-small cell lung cancer (NSCLC) cells [28]. Yasukawa et al. have indicated that down-regulation of MYCN shows the anti-cancer potential by repressing invasion and growth of hepatic cancer cells (HCC) [29]. However, whether lncRNA LINC01106 is involved in GC progression by targeting miR-34a-5p/MYCN axis has not been explored.
In this investigation, we explored the biological role and underlying mechanism of lncRNA LINC01106 in GC cells and then validated the role of lncRNA LINC01106 in xenograft mice.
Methods
Collection of clinical samples
From June 2019 to March 2020, 52 pairs of GC tissues and adjacent non-cancer tissues were obtained from patients of our hospital. These patients were diagnosed with GC according to histopathological evaluation and none of these patients received any surgery operation, chemical, or radiation treatment. The current study was conducted with approval of the ethics committee of our hospital and conformed to the Declaration of Helsinki. Written informed consent was obtained from all patients.
Cell Culture
GC cell lines (MKN45, MGC803, and AGS) and a normal gastric cell line (GES1) were bought from the American Type Culture Collection (ATCC, Manassas, VA, USA). All cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, Rockville, MD, USA) and 2.5% streptomycin and penicillin. These cells were incubated at 37 °C under a humidified atmosphere with 5% CO2.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
Total RNAs were extracted via TRIzol reagent (Invitrogen, Carlsbad, CA, USA) and were purified through DNase I treatment. Then purified RNAs were reversely transcribed into complementary DNAs (cDNAs) using PrimeScript RT Reagent (TaKaRa, Dalian, China). Next, cDNAs were used for performing qRT-PCR according to the instructions of SYBR® Premix Ex Taq™ (TaKaRa). The amplification procedures were as follows: 95 °C for 2 min, 40 cycles of 95 °C for 15 s, 60 °C for 30 s, and 70 °C for 15 s. Primer sequences are shown in Table 1. Expression levels of lncRNA LINC01106, miR-34a-5p, and MYCN were calculated based on \(2^{{ - \Delta \Delta C_{{\text{t}}} }}\) method and were normalized to GAPDH or U6.
Cell Transfection
The short hairpin (sh)-negative control (NC, 5′-CCGG ACATAGATATAATAAAGAAAGCTCGAGCTTTCTTTATTATATCTATGTTTTTTG-3′), shRNA against lncRNA LINC01106 (sh-LINC01106-1, 5′-CCGGACTGTATAAAAATAAAGAGAACTCGAGTTCTCTTTATTTTTATACAGTTTTTG-3′ and sh-LINC01106-2, 5′-CCGG TCTATTTTGGCTTGTATTGACCTCGAGGTCAATACAAGCCAAAATAGATTTTTG-3′), inhibitor NC (5′-CAGUACUUUUGUGUAGUACAA-3′), miR-34a-5p inhibitor (5′-CUACCUGCACCAACAGCACUU-3′), mimics NC (5′-UUCUCCGAACGUGUCACGUTT-3′), miR-34a-5p mimics (5′-GAUGGACGUGCUUGUCGUGAAAC-3′), pcDNA3.1-NC, and pcDNA3.1-MYCN were bought from RiboBio Company (Beijing, China). All above plasmids were constructed into the pLKO.1 vector, followed by lentiviral particle preparation as previously described. Then, lentiviral particles containing above plasmids were transfected into MKN45 and AGS cells using Lipofectamine 3000 (Invitrogen) for 48 h. Finally, stably transfected cells were screened out via 2 μg/ml of puromycin treatment for additional 48 h. The stably transfected cells were collected to implement further assays.
3-(4, 5-Dimethyl-2-Thiazolyl)-2, 5-Diphenyl-2-H-Tetrazolium Bromide (MTT) Assay
The transfected cells were plated into 96-well plates (5 × 103 cells/well). After incubation for 24, 48, and 72 h, 20 μL MTT (Sigma-Aldrich, St. Louis, MO, USA) was pipetted into each well to incubate at 37 °C for 4 h. To dissolve formazan crystals, each well was filled with 150 μL dimethyl sulfoxide (Sigma-Aldrich). Finally, a SpectraMax 360 pc microplate reader (Molecular Devices, LLC, Sunnyvale, CA, USA) was employed to measure the optical density (OD) at 490 nm.
Transwell Assays
To assess cell migration, the 24-well transwell chamber with 8 µm pore size (Corning Costar, Cambridge, MA, USA) was used. Cells (1 × 105) re-suspended in FBS-free medium (200 μL) were plated into the upper chamber. Meantime, the lower chamber was supplemented with medium (600 μL) containing 10% FBS. Following 48 h of incubation, the cells migrated to the lower chamber were fixed by 4% formaldehyde and then stained with crystal violet. Stained cells were counted under an inverted light microscope (Olympus, Tokyo, Japan). To examine cell invasion, the same operation was conducted with the 24-well transwell chamber coated with Matrigel (BD Biosciences, Sparks, MD, USA).
Dual-Luciferase Reporter (DLR) Assay
The 3′-UTR fragment of lncRNA LINC01106 or MYCN holding the predicted binding sites for miR-34a-5p was inserted into a pmiRGLO vector (Promega, Madison, WI, USA), named as LINC01106 wide type (wt) or MYCN wt. Analogously, the 3′-UTR fragment of LINC01106 or MYCN holding the mutated sites for miR-34a-5p was introduced into a pmiRGLO vector (Promega), named as LINC01106 mutant type (mut) or MYCN mut. After that, above reporter vectors along with mimics NC/miR-34a-5p mimics were transfected into MKN45 and AGS cells via Lipofectamine 3000 (Invitrogen). Following 48 h of transfection, firefly luciferase and renilla luciferase were examined separately with the DLR System (Promega) and their ratio indicated relative luciferase activity.
Western Blot Analysis
Total proteins were extracted from the GC cells and tissues via RIPA lysis buffer (Beyotime, Shanghai, China), followed by separation with 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). Then protein samples were transferred to polyvinylidene fluoride (PVDF) membranes. After the membranes were blocked with 5% non-fat milk for 1 h, they were incubated with primary antibodies anti-MYCN (1:1000; ab227822, Abcam, MA, USA) and anti-GAPDH (1:10,000; ab181602, Abcam) overnight at 4 °C. Subsequently, tris-buffered saline tween-20 was applied to wash the membranes and the anti-rabbit secondary antibody (1:5000; ab205718, Abcam) was added to incubate at 37 °C for 1 h. Finally, an electrochemiluminescence kit (Pierce Biotechnology, Rockford, IL, USA) was employed to visualize protein bands. Relative protein expression of MYCN over the GAPDH was quantified through the Alpha Innotech imaging software (San Leandro, CA, USA).
Establishment of a Xenograft Tumor Model
Animal experiments were conducted according to the institutional guide for the care and use of laboratory animals (National Institutes of Health, USA). The BALB/c nude mice (female, 20–28 g, 4–5 weeks of age) were obtained from Eusebio (Shanghai, China). All mice were housed under a controlled environment (12-h light/dark cycle, 23–26 °C, and 40–60% humidity) and had free access to water and food. Lentiviral particles containing sh-LINC01106 (Lv-sh-LINC01106) or sh-NC (Lv-sh-NC) were bought from Sigma-Aldrich. Mice were assigned into two groups: the Lv-sh-NC group (n = 5) and the LV-sh-LINC01106 group (n = 5). MKN45 cells (5 × 106) carrying Lv-sh-NC or Lv-sh-LINC01106 were subcutaneously injected into right flanks of mice [30]. After injection, the tumor volume was monitored every 5 days and calculated via the following formula: (length × width2)/2 [31]. On the 25th day after injection, pentobarbital sodium (50 mg/kg) was used to anesthetize mice and then mice were sacrificed by cervical dislocation. The tumors in mice were isolated and weighed.
Statistical Analysis
Statistical analysis was carried out with SPSS 21.0 software (IBM Corp. Armonk, NY, USA). The data from three independent assays were displayed as the mean ± standard deviation. The comparisons of two groups were analyzed using Student’s t test. Following comparisons among multiple groups with one-way ANOVA, Tukey’s post hoc test was utilized for pairwise comparisons. P < 0.05 signified statistically significant.
Results
Silencing of lncRNA LINC01106 Represses the Viability, Migration, and Invasion of MKN45 and AGS Cells
Firstly, we determined relative expression of lncRNA LINC01106 in GC cells. Compared to GES1 cells, an evident up-regulation of lncRNA LINC01106 was found in MKN45 (P < 0.001), MGC803 (P < 0.01), and AGS cells (P < 0.001), especially in MKN45 and AGS cells (Fig. 1A). Thus, MKN45 and AGS cells were utilized for the further assays. In order to investigate the role of lncRNA LINC01106 in GC cells, lncRNA LINC01106 was silenced. The expression of LINC01106 was significantly repressed after transfection of sh-LINC01106-1/-2, suggesting that both sh-LINC01106-1 and sh-LINC01106-2 were transfected into GC cells successfully (all P < 0.01, Fig. 1B). Then, sh-LINC01106-1 was selected for following functional assays due to the relatively high transfection efficiency. MTT assay demonstrated that silencing of lncRNA LINC01106 caused the marked reduction of cell viability in MKN45 and AGS cells (all P < 0.01, Fig. 1C). Additionally, transwell assay displayed that relative migration and invasion were also restrained when lncRNA LINC01106 was silenced (all P < 0.01, Fig. 1D, E).
Silencing of lncRNA LINC01106 represses the viability, migration, and invasion of MKN45 and AGS cells. A Relative expression of LINC01106 in GES1, MKN45, MGC803, and AGS cells was determined by quantitative real-time polymerase chain reaction (qRT-PCR). **P < 0.01, ***P < 0.001, vs. GES1. B Relative expression of LINC01106 in MKN45 and AGS cells was determined by qRT-PCR after transfection of short hairpin (sh)-LINC01106-1 and sh-LINC01106-2. **P < 0.01, vs. sh-negative control (NC). C After transfection of sh-LINC01106-1 and sh-NC, the viability of MKN45 and AGS cells was detected by MTT assay. **P < 0.01, vs. sh-NC. (D–E) After transfection of sh-LINC01106-1 and sh-NC, relative migration and invasion of MKN45 and AGS cells were assessed by transwell assay. **P < 0.01, vs. sh-NC
LncRNA LINC01106 Directly Targets miR-34a-5p
To probe how lncRNA LINC01106 affected GC cells, bioinformatics analysis was conducted with starbase. As displayed in Fig. 2A, there was complementary sequence between LINC01106 and miR-34a-5p. To verify this cooperation, DLR assay was implemented. It turned out that transfection of miR-34a-5p mimics remarkably reduced relative luciferase activity of LINC01106 wt (P < 0.01) rather than LINC01106 mut in MKN45 and AGS cells, which implied that miR-34a-5p is a target of lncRNA LINC01106 (Fig. 2B). Then we explored whether miR-34a-5p was modulated by lncRNA LINC01106 and discovered that miR-34a-5p expression was markedly boosted by addition of sh-LINC01106-1 in MKN45 and AGS cells (all P < 0.01, Fig. 2C). The lower expression of miR-34a-5p was also observed in MGC803, MKN45, and AGS cells than that in GES1 cells (all P < 0.001, Fig. 2D).
LncRNA LINC01106 acts as a sponge for miR-34a-5p. A The binding sequence between LINC01106 and miR-34a-5p was predicted by starbase (http://starbase.sysu.edu.cn/agoClipRNA.php?). The dotted lines indicate the non-complementary bases and the solid lines indicate the complementary bases. B The interaction between LINC01106 and miR-34a-5p was tested by dual-luciferase reporter (DLR) assay in MKN45 and AGS cells. **P < 0.01, vs. mimics negative control (NC). C After transfection of short hairpin (sh)-LINC01106-1 and sh-NC, relative expression of miR-34a-5p was detected by quantitative real-time polymerase chain reaction (qRT-PCR) in MKN45 and AGS cells. **P < 0.01, vs. sh-NC. D Relative expression of miR-34a-5p in GES1, MKN45, MGC803, and AGS cells was detected by qRT-PCR. ***P < 0.001, vs. GES1
Up-regulation of miR-34a-5p Attenuates Cell Viability and Impedes Invasion and Migration of GC Cells
To further probe miR-34a-5p function in MKN45 and AGS cells, miR-34a-5p expression was altered by transfection of miR-34a-5p inhibitor and miR-34a-5p mimics. As showed in Fig. 3A, miR-34a-5p expression was decreased by miR-34a-5p inhibitor (all P < 0.01) and elevated by miR-34a-5p mimics (all P < 0.001) in MKN45 and AGS cells. Accordingly, the impacts of miR-34a-5p on MKN45 and AGS cells were assessed. It was proved that addition of miR-34a-5p mimics weakened cell viability and impaired relative invasion and migration of MKN45 and AGS cells compared with addition of mimics NC (all P < 0.01, Fig. 3B-3D).
Up-regulation of miR-34a-5p attenuates cell viability and impedes migration and invasion of GC cells. A Relative expression of miR-34a-5p in MKN45 and AGS cells was detected by quantitative real-time polymerase chain reaction (qRT-PCR). ***P < 0.001, vs. mimics negative control (NC). ##P < 0.01, vs. inhibitor NC. B After transfection of mimics NC and miR-34a-5p mimics, the viability of MKN45 and AGS cells was detected by MTT assay. **P < 0.01, vs. mimics NC. (C–D) After transfection of mimics NC and miR-34a-5p mimics, relative migration and invasion of MKN45 and AGS cells were assessed by transwell assay. **P < 0.01, vs. mimics NC
MiR-34a-5p Directly Targets MYCN
For illuminating the potential mechanism of miR-34a-5p in GC cells, we sought for target genes of miR-34a-5p via starbase. As exhibited in Fig. 4A, there were binding sites between miR-34a-5p and MYCN. Then DLR assay showed an obvious decline in relative luciferase activity of MKN45 and AGS cells after co-transfection of miR-34a-5p mimics and MYCN wt (P < 0.01), confirming the targeting relation between miR-34a-5p and MYCN (Fig. 4B). Likewise, we discovered that MYCN was negatively modulated by miR-34a-5p in MKN45 and AGS cells (all P < 0.01, Fig. 4C). MYCN expression was boosted in MKN45, MGC803, and AGS cells compared to GES1 cells (all P < 0.01, Fig. 4D).
MiR-34a-5p directly targets MYCN. A The binding sequence between MYCN and miR-34a-5p was predicted by starbase (http://starbase.sysu.edu.cn/agoClipRNA.php?). The dotted lines indicate the non-complementary bases and the solid lines indicate the complementary bases. B The interaction between MYCN and miR-34a-5p was tested by dual-luciferase reporter (DLR) assay in MKN45 and AGS cells. **P < 0.01, vs. mimics negative control (NC). C After transfection of mimics NC and miR-34a-5p mimics, relative protein expression of MYCN was detected by western blot in MKN45 and AGS cells. **P < 0.01, vs. mimics NC. D Relative expression of MYCN in GES1, MKN45, MGC803, and AGS cells was detected by quantitative real-time polymerase chain reaction (qRT-PCR). **P < 0.01, vs. GES1
LncRNA LINC01106 Knockdown Represses Cell Viability, Migration, and Invasion by Sponging miR-34a-5p to target MYCN
Next, to verify whether miR-34a-5p and MYCN were involved in LINC01106-mediated inhibition of GC progression in vitro, the following assays were conducted. It was found that relative expression of MYCN was augmented by transfection of pcDNA3.1-MYCN in MKN45 cells (P < 0.001, Fig. 5A). MYCN expression was reduced by sh-LINC01106-1 (P < 0.001) and was elevated by co-transfection of sh-LINC01106-1 and miR-34a-5p inhibitor in MKN45 cells (P < 0.01, Fig. 5B). Importantly, rescue experiments indicated that LINC01106 knockdown resulted in evident reduction in viability (P < 0.001), migration (P < 0.01), and invasion (P < 0.01) of MKN45 cells, whereas the suppressive effects of LINC01106 knockdown on cell viability (P < 0.01), migration (P < 0.05), and invasion (P < 0.05) were partially eliminated by miR-34a-5p suppression and MYCN overexpression in MKN45 cells (Fig. 5C–E).
Knockdown of lncRNA LINC01106 represses cell viability, migration, and invasion by sponging miR-34a-5p to target MYCN. A Relative expression of MYCN in MKN45 cells was determined by quantitative real-time polymerase chain reaction (qRT-PCR). ***P < 0.001, vs. pcDNA3.1-negative control (NC). B Relative expression of MYCN in MKN45 cells was determined by qRT-PCR. ***P < 0.001, vs. short hairpin (sh)-NC. ##P < 0.01, vs. sh-LINC01106-1. C The viability of MKN45 cells was detected by MTT assay. ***P < 0.001, vs. sh-NC. ##P < 0.01, vs. sh-LINC01106-1. D–E Relative migration and invasion of MKN45 cells were assessed by transwell assay. **P < 0.01, vs. sh-NC. #P < 0.05, vs. sh-LINC01106-1
Silencing of lncRNA LINC01106 Suppresses Tumorigenesis in Mice
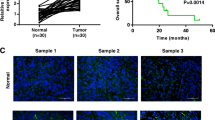
Then we validated the influence of lncRNA LINC01106 on tumorigenesis in xenograft mice. Following 25 days after injection, we observed evident decreases of the tumor volume (P < 0.001) and weight (P < 0.01) in the Lv-sh-LINC01106 group relative to the Lv-sh-NC group (Fig. 6A and B). Subsequently, we determined LINC01106 expression and verified the regulatory relation between lncRNA LINC01106 and miR-34a-5p/MYCN in tumor tissues of mice. We observed that lncRNA LINC01106 was down-regulated after Lv-sh-LINC01106 injection (P < 0.01, Fig. 6C). LncRNA LINC01106 inversely regulated miR-34a-5p and positively regulated MYCN in tumor tissues of mice (all P < 0.01, Fig. 6D and E).
Silencing of lncRNA LINC01106 suppresses tumorigenesis in mice. A Tumor volume was gauged in mice. ***P < 0.001, vs. Lv-short hairpin (sh)-negative control (NC). B Tumor weight was assessed in mice. **P < 0.01, vs. Lv-sh-NC. C Relative expression of LINC01106 in tumor tissues of mice was determined by quantitative real-time polymerase chain reaction (qRT-PCR). **P < 0.01, vs. Lv-sh-NC. D Relative expression of miR-34a-5p in tumor tissues of mice was determined by qRT-PCR. **P < 0.01, vs. Lv-sh-NC. E Relative protein expression of MYCN in tumor tissues of mice was determined by western blot. **P < 0.01, vs. Lv-sh-NC
LncRNA LINC01106 and MYCN are Highly Expressed and miR-34a-5p is Lowly Expressed in GC Tissues
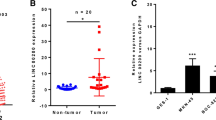
Finally, we validated relative expression of lncRNA LINC01106, miR-34a-5p, and MYCN and assessed their correlation in GC tissues. In line with the results of GC cells, we also found the up-regulation of lncRNA LINC01106 and MYCN as well as the down-regulation of miR-34a-5p in GC tissues compared to adjacent non-cancer tissues (all P < 0.001, Fig. 7A–C). There was an inverse correlation between miR-34a-5p and lncRNA LINC01106 (P = 0.0092) and between MYCN and miR-34a-5p (P = 0.0045) in GC tissues, and there was a positive correlation between lncRNA LINC01106 and MYCN (P = 0.0030) in GC tissues (Fig. 7D–F). Moreover, we analyzed the association between LINC01106 expression and pathological features of GC patients. As displayed in Table 2, LINC01106 expression was associated with lymph node metastasis, tumor node metastasis (TNM) stage, and distant metastasis of GC patients rather than tumor size, gender, and age.
LncRNA LINC01106 and MYCN are highly expressed and miR-34a-5p is lowly expressed in GC tissues. A Relative expression of LINC01106 in tumor tissues of GC patients was determined by quantitative real-time polymerase chain reaction (qRT-PCR). ***P < 0.001, vs. adjacent normal tissues. B Relative expression of miR-34a-5p in tumor tissues of GC patients was determined by qRT-PCR. ***P < 0.001, vs. adjacent normal tissues. C Relative expression of MYCN in tumor tissues of GC patients was determined by qRT-PCR. ***P < 0.001, vs. adjacent normal tissues. D The correlation between LINC01106 and miR-34a-5p in GC tissues was analyzed by Pearson's correlation analysis. P = 0.0092. r = − 0.3579. E The correlation between LINC01106 and MYCN in GC tissues was analyzed by Pearson's correlation analysis. P = 0.0030. r = 0.4041. F The correlation between MYCN and miR-34a-5p in GC tissues was analyzed by Pearson's correlation analysis. P = 0.0045. r = − 0.388
Discussion
For past few years, the incidence of GC has been on the rise, which seriously threatens human health and life [32]. Therefore, developing potential targets for GC treatment is imperative. Previously, available reports have revealed the up-regulation of lncRNA LINC01106 in colorectal cancer (CRC) tissues [33], endometrial cancer tissues [34], and GC tissues [11]. In this study, the higher expression of lncRNA LINC01106 was also observed in GC cells and tissues than their controls, which was consistent with the expression trend of above reports. We deduced that lncRNA LINC01106 was implicated in GC occurrence. Additionally, substantial evidence has suggested that LINC01106 expression is linked with the prognosis of several cancers [12, 35]. For example, up-regulation of lncRNA LINC01106 is negatively associated with the overall survival of colon adenocarcinoma (COAD) patients [35]. LncRNA LINC01106 is identified as a prognostic factor in GC according to data from survival analyses [12]. Here, we discovered that LINC01106 expression was associated with lymph node metastasis, TNM stage, and distant metastasis in GC patients, implying that lncRNA LINC01106 may serve as a prognostic biomarker of GC. Functionally, mounting studies have demonstrated that silencing of lncRNA LINC01106 exhibits an anti-tumor role in GC [33, 36]. Gu et al. have unveiled that silencing of lncRNA LINC01106 impedes the proliferation of CRC cells and restrains tumor growth of xenograft mice [33]. Meng et al. have uncovered that silencing of lncRNA LINC01106 reduces proliferative, invasive, and migratory abilities of bladder cancer (BC) cells [36]. Similar to prior studies, we also found that silencing of lncRNA LINC01106 repressed migration, invasion, and viability of GC cells as well as tumor growth of xenograft mice, which showed the anti-cancer potential in GC. Based on these findings, we inferred that knockdown of lncRNA LINC01106 may be a novel prognostic marker and therapeutic target in GC.
As we all known, lncRNAs commonly play their roles by targeting miRNAs [37]. As a well-explored miRNA, miR-34a-5p has been revealed to be the downstream target of lncRNA NEAT1 in prostate cancer [38], lncARSR in CRC [39], and lncRNA XIST in pancreatic cancer [22]. Similarly, miR-34a-5p was identified as the downstream target of lncRNA LINC01106 in this study. As previously reported, miR-34a-5p is lowly expressed in liver cancer [25] and pancreatic cancer [22] and miR-34a is also down-regulated in GC [18]. Here, the down-regulation of miR-34a-5p was also observed in GC tissues and cells, which suggested that miR-34a-5p was closely linked to GC progression. More importantly, copious papers have demonstrated the importance of miR-34a/miR-34a-5p in cancers by serving as a tumor suppressor [25, 40]. For instance, increased miR-34a-5p suppresses migration, proliferation, and invasion of liver cancer cells [25]. Overexpressed miR-34a-5p attenuates proliferative, invasive, and migratory capacities of prostate cancer cells [40]. In particular, increased miR-34a attenuates proliferative and invasive abilities of GC cells [19]. In the current study, we discovered that up-regulation of miR-34a-5p made inhibitory effects on cell viability, migration and invasion of GC cells. These findings strongly suggested that miR-34a-5p may be a tumor suppressor in GC and miR-34a-5p inhibitors may be promising agents for GC therapy. Concurrently, we discovered that miR-34a-5p was inversely modulated by lncRNA LINC01106 in GC cells and was inversely correlated with lncRNA LINC01106 in GC tissues. The inhibitory impacts of LINC01106 knockdown on migration, invasion, and viability of GC cells were abolished by miR-34a-5p down-regulation. Taken together, we speculated that LINC01106 knockdown exerted an anti-tumor role through interacting with miR-34a-5p in GC cells. Additionally, LINC01106 has been reported to interact with many signaling pathways in human cancers, such as LINC01106-STAT3 pathway [33] or -Hedgehog pathway [35] in CRC and LINC01106-ELK pathway [36] in BC. We speculated that LINC01106 may be involved in the progression of GC by regulating these signaling pathways. Further researches on the regulatory relations of LINC01106 with these signaling pathways are urgently needed.
It is generally accepted that miRNAs possess multiple functions by targeting diverse genes [41]. In previous study, proto-oncogene MYCN is demonstrated to be targeted by several miRNAs in cancers, such as miR-493-5p in hepatic cancer [29] and miR-34a in lung cancer [42]. In the current study, MYCN was targeted by miR-34a-5p. Increased expression of MYCN has been unveiled in NSCLC and HCC [28, 29]. Consistent with the above researches, we also observed the higher expression of MYCN in GC tissues and cells than that of their controls, indicating the interconnection between GC and MYCN. Likewise, the pivotal role of MYCN has been revealed in several cancers [28, 29]. Liu et al. have reported that MYCN contributes to NSCLC development via promoting cell proliferation [28]. Yasukawa et al. have indicated that down-regulation of MYCN displays the anti-cancer potential by repressing invasion and growth of HCC cells [29]. According to these results, we speculated that MYCN acted as an oncogene in GC and targeting MYCN down-regulation may provide beneficial effects for the clinical therapy of GC. Simultaneously, we discovered that MYCN was negatively regulated by miR-34a-5p in GC cells and inversely correlated with miR-34a-5p in GC tissues. We deduced that miR-34a-5p restrained malignant behaviors of GC cells by targeting MYCN. Considering the existence of the lncRNAs-miRNAs-mRNAs network [43, 44], we made an assumption that lncRNA LINC01106 participated in the regulation of malignant behaviors in GC cells by binding to miR-34a-5p to regulate MYCN. Specifically, we found a positive regulation and a positive correlation between MYCN and lncRNA LINC01106 in GC cells and in GC tissues. Meanwhile, we discovered that the suppressive effects of LINC01106 knockdown on cell viability, migration, and invasion were reversed by MYCN overexpression in GC cells. Collectively, we concluded that silencing of lncRNA LINC01106 directly bound to miR-34a-5p to regulate MYCN, thereby retarding GC progression in vitro. Our conclusion indicated that targeting the LINC01106/miR-34a-5p/MYCN axis may be a promising approach for GC treatment.
There are also some other limitations of this study. First, exploring the effects of miR-34a-5p overexpression or MYCN silencing on tumor size in vivo may be more rigorous for this study. Second, the roles of some important downstream genes of miR-34a-5p such as c-Myc have been confirmed in several human cancers and the interaction between miR-34a-5p and c-Myc in GC should be further explored.
In a word, the present study demonstrated that relative expression of lncRNA LINC01106 was boosted in GC tissues and cells compared to their controls. Silencing of lncRNA LINC01106 repressed the viability, migratory, and invasive abilities of GC cells as well as the tumor growth of xenograft mice. On the mechanism, knockdown of lncRNA LINC01106 was identified as a sponge of miR-34a-5p to target MYCN in GC cells, leading to the repression of GC progression. Our findings strongly supported that lncRNA LINC01106 was a valuable target for prognosis and treatment of GC.
References
Crew, K. D., & Neugut, A. I. (2006). Epidemiology of gastric cancer. World Journal of Gastroenterology, 12(3), 354–362. https://doi.org/10.3748/wjg.v12.i3.354
Siegel, R. L., Miller, K. D., & Jemal, A. (2019). Cancer statistics. CA: A Cancer Journal for Clinicians, 69(1), 7–34. https://doi.org/10.3322/caac.21551
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A., & Jemal, A. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 68(6), 394–424. https://doi.org/10.3322/caac.21492
Sitarz, R., Skierucha, M., Mielko, J., Offerhaus, G. J. A., Maciejewski, R., & Polkowski, W. P. (2018). Gastric cancer: Epidemiology, prevention, classification, and treatment. Cancer Manag Res, 10, 239–248. https://doi.org/10.2147/CMAR.S149619
Cervantes, A., Roda, D., Tarazona, N., Rosello, S., & Perez-Fidalgo, J. A. (2013). Current questions for the treatment of advanced gastric cancer. Cancer Treatment Reviews, 39(1), 60–67. https://doi.org/10.1016/j.ctrv.2012.09.007
Wang, X., Arai, S., Song, X., Reichart, D., Du, K., Pascual, G., Tempst, P., Rosenfeld, M. G., Glass, C. K., & Kurokawa, R. (2008). Induced ncRNAs allosterically modify RNA-binding proteins in cis to inhibit transcription. Nature, 454(7200), 126–130. https://doi.org/10.1038/nature06992
Fatica, A., & Bozzoni, I. (2014). Long non-coding RNAs: New players in cell differentiation and development. Nature Reviews Genetics, 15(1), 7–21. https://doi.org/10.1038/nrg3606
Huang, Y., Zhang, J., Hou, L., Wang, G., Liu, H., Zhang, R., Chen, X., & Zhu, J. (2017). LncRNA AK023391 promotes tumorigenesis and invasion of gastric cancer through activation of the PI3K/Akt signaling pathway. Journal of Experimental & Clinical Cancer Research, 36(1), 194. https://doi.org/10.1186/s13046-017-0666-2
Luo, M., & Liang, C. (2020). LncRNA LINC00483 promotes gastric cancer development through regulating MAPK1 expression by sponging miR-490-3p. Biological Research, 53(1), 14. https://doi.org/10.1186/s40659-020-00283-6
Pan, H., Ding, Y., Jiang, Y., Wang, X., Rao, J., Zhang, X., Yu, H., Hou, Q., & Li, T. (2021). LncRNA LIFR-AS1 promotes proliferation and invasion of gastric cancer cell via miR-29a-3p/COL1A2 axis. Cancer Cell International, 21(1), 7. https://doi.org/10.1186/s12935-020-01644-7
Han, W., Zhang, Z., He, B., Xu, Y., Zhang, J., & Cao, W. (2017). Integrated analysis of long non-coding RNAs in human gastric cancer: An in silico study. PLoS ONE, 12(8), e0183517. https://doi.org/10.1371/journal.pone.0183517
Mao, R., Wang, Z., Zhang, Y., Chen, Y., Liu, Q., Zhang, T., & Liu, Y. (2020). Development and validation of a novel prognostic signature in gastric adenocarcinoma. Aging (Albany, NY), 12(21), 22233–22252. https://doi.org/10.18632/aging.104161
Guo, L. L., Song, C. H., Wang, P., Dai, L. P., Zhang, J. Y., & Wang, K. J. (2015). Competing endogenous RNA networks and gastric cancer. World Journal of Gastroenterology, 21(41), 11680–11687. https://doi.org/10.3748/wjg.v21.i41.11680
Bhaskaran, M., & Mohan, M. (2014). MicroRNAs: History, biogenesis, and their evolving role in animal development and disease. Veterinary Pathology, 51(4), 759–774. https://doi.org/10.1177/0300985813502820
Meng, H., Li, Y. Y., Han, D., & Zhang, C. Y. (2019). MiRNA-93-5p promotes the biological progression of gastric cancer cells via Hippo signaling pathway. European Review for Medical and Pharmacological Sciences, 23(11), 4763–4769.
Wang, C., Huang, Y., Zhang, J., & Fang, Y. (2020). MiRNA-339-5p suppresses the malignant development of gastric cancer via targeting ALKBH1. Experimental and Molecular Pathology. https://doi.org/10.1016/j.yexmp.2020.104449
Hu, S., Zheng, Q., Wu, H., Wang, C., Liu, T., & Zhou, W. (2017). miR-532 promoted gastric cancer migration and invasion by targeting NKD1. Life Sciences, 177, 15–19. https://doi.org/10.1016/j.lfs.2017.03.019
Hu, Y., Pu, Q., Cui, B., & Lin, J. (2015). MicroRNA-34a inhibits tumor invasion and metastasis in gastric cancer by targeting Tgif2. International Journal of Clinical and Experimental Pathology, 8(8), 8921–8928.
Wei, B., Huang, Q. Y., Huang, S. R., Mai, W., & Zhong, X. G. (2015). MicroRNA34a attenuates the proliferation, invasion and metastasis of gastric cancer cells via downregulation of MET. Molecular Medicine Reports, 12(4), 5255–5261. https://doi.org/10.3892/mmr.2015.4110
Zheng, F., Li, J., Ma, C., Tang, X., Tang, Q., Wu, J., Chai, X., Xie, J., Yang, X. B., & Hann, S. S. (2020). Novel regulation of miR-34a-5p and HOTAIR by the combination of berberine and gefitinib leading to inhibition of EMT in human lung cancer. Journal of Cellular and Molecular Medicine, 24(10), 5578–5592. https://doi.org/10.1111/jcmm.15214
Ma, Y., Fan, B., Ren, Z., Liu, B., & Wang, Y. (2019). Long noncoding RNA DANCR contributes to docetaxel resistance in prostate cancer through targeting the miR-34a-5p/JAG1 pathway. Oncotargets and Therapy, 12, 5485–5497. https://doi.org/10.2147/OTT.S197009
Sun, Z., Zhang, B., & Cui, T. (2018). Long non-coding RNA XIST exerts oncogenic functions in pancreatic cancer via miR-34a-5p. Oncology Reports, 39(4), 1591–1600. https://doi.org/10.3892/or.2018.6245
Bartel, D. P. (2009). MicroRNAs: Target recognition and regulatory functions. Cell, 136(2), 215–233. https://doi.org/10.1016/j.cell.2009.01.002
Zuo, Y., Zheng, W., Liu, J., Tang, Q., Wang, S. S., & Yang, X. S. (2020). MiR-34a-5p/PD-L1 axis regulates cisplatin chemoresistance of ovarian cancer cells. Neoplasma, 67(1), 93–101. https://doi.org/10.4149/neo_2019_190202N106
Xu, X. P., Peng, X. Q., Yin, X. M., Liu, Y., & Shi, Z. Y. (2020). miR-34a-5p suppresses the invasion and metastasis of liver cancer by targeting the transcription factor YY1 to mediate MYCT1 upregulation. Acta Histochemica, 122(6), 151576. https://doi.org/10.1016/j.acthis.2020.151576
He, J., Zhao, H., Liu, X., Wang, D., Wang, Y., Ai, Y., & Yang, J. (2020). Sevoflurane suppresses cell viability and invasion and promotes cell apoptosis in colon cancer by modulating exosomemediated circHMGCS1 via the miR34a5p/SGPP1 axis. Oncology Reports, 44(6), 2429–2442. https://doi.org/10.3892/or.2020.7783
Meyer, N., & Penn, L. Z. (2008). Reflecting on 25 years with MYC. Nature Reviews Cancer, 8(12), 976–990. https://doi.org/10.1038/nrc2231
Liu, K., Wang, S., Liu, Y., Gu, J., Gu, S., Xu, Z., Zhang, R., Wang, Z., Ma, H., Chen, Y., & Ji, L. (2016). Overexpression of MYCN promotes proliferation of non-small cell lung cancer. Tumour Biology, 37(9), 12855–12866. https://doi.org/10.1007/s13277-016-5236-2
Yasukawa, K., Liew, L. C., Hagiwara, K., Hironaka-Mitsuhashi, A., Qin, X. Y., Furutani, Y., Tanaka, Y., Nakagama, H., Kojima, S., Kato, T., Ochiya, T., & Gailhouste, L. (2020). MicroRNA-493-5p-mediated repression of the MYCN oncogene inhibits hepatic cancer cell growth and invasion. Cancer Science, 111(3), 869–880. https://doi.org/10.1111/cas.14292
Liu, T., Liu, Y., Wei, C., Yang, Z., Chang, W., & Zhang, X. (2020). LncRNA HULC promotes the progression of gastric cancer by regulating miR-9-5p/MYH9 axis. Biomedicine & Pharmacotherapy, 121, 109607. https://doi.org/10.1016/j.biopha.2019.109607
Wu, F., Gao, H., Liu, K., Gao, B., Ren, H., Li, Z., & Liu, F. (2019). The lncRNA ZEB2-AS1 is upregulated in gastric cancer and affects cell proliferation and invasion via miR-143-5p/HIF-1alpha axis. Oncotargets and Therapy, 12, 657–667. https://doi.org/10.2147/OTT.S175521
Smyth, E. C., Nilsson, M., Grabsch, H. I., van Grieken, N. C., & Lordick, F. (2020). Gastric cancer. Lancet, 396(10251), 635–648. https://doi.org/10.1016/S0140-6736(20)31288-5
Gu, Y., Huang, Y., Sun, Y., Liang, X., Kong, L., Liu, Z., & Wang, L. (2020). Long non-coding RNA LINC01106 regulates colorectal cancer cell proliferation and apoptosis through the STAT3 pathway. Nan Fang Yi Ke Da Xue Xue Bao, 40(9), 1259–1264. https://doi.org/10.12122/j.issn.1673-4254.2020.09.06
Gao, X., Yu, L., Zhang, J., & Xue, P. (2020). Silencing of long non-coding RNA LINC01106 suppresses the proliferation, migration and invasion of endometrial cancer cells through regulating the miR-449a/MET axis. Oncotargets and Therapy, 13, 9643–9655. https://doi.org/10.2147/OTT.S264642
Guo, K., Gong, W., Wang, Q., Gu, G., Zheng, T., Li, Y., Li, W., Fang, M., Xie, H., Yue, C., Yang, J., & Zhu, Z. (2020). LINC01106 drives colorectal cancer growth and stemness through a positive feedback loop to regulate the Gli family factors. Cell Death & Disease, 11(10), 869. https://doi.org/10.1038/s41419-020-03026-3
Meng, L., Xing, Z., Guo, Z., & Liu, Z. (2020). LINC01106 post-transcriptionally regulates ELK3 and HOXD8 to promote bladder cancer progression. Cell Death & Disease, 11(12), 1063. https://doi.org/10.1038/s41419-020-03236-9
Paraskevopoulou, M. D., & Hatzigeorgiou, A. G. (2016). Analyzing MiRNA-LncRNA interactions. Methods in Molecular Biology, 1402, 271–286. https://doi.org/10.1007/978-1-4939-3378-5_21
Jiang, X., Guo, S., Zhang, Y., Zhao, Y., Li, X., Jia, Y., Xu, Y., & Ma, B. (2020). LncRNA NEAT1 promotes docetaxel resistance in prostate cancer by regulating ACSL4 via sponging miR-34a-5p and miR-204-5p. Cellular Signalling. https://doi.org/10.1016/j.cellsig.2019.109422
Li, S., Zhu, K., Liu, L., Gu, J., Niu, H., & Guo, J. (2020). lncARSR sponges miR-34a-5p to promote colorectal cancer invasion and metastasis via hexokinase-1-mediated glycolysis. Cancer Science, 111(10), 3938–3952. https://doi.org/10.1111/cas.14617
Ma, E., Wang, Q., Li, J., Zhang, X., Guo, Z., & Yang, X. (2020). LINC01006 facilitates cell proliferation, migration and invasion in prostate cancer through targeting miR-34a-5p to up-regulate DAAM1. Cancer Cell International, 20, 515. https://doi.org/10.1186/s12935-020-01577-1
Bracken, C. P., Scott, H. S., & Goodall, G. J. (2016). A network-biology perspective of microRNA function and dysfunction in cancer. Nature Reviews Genetics, 17(12), 719–732. https://doi.org/10.1038/nrg.2016.134
Song, C., Lu, P., Sun, G., Yang, L., & Wang, Z. (2017). miR-34a sensitizes lung cancer cells to cisplatin via p53/miR-34a/MYCN axis. Biochemical and Biophysical Research Communications, 482(1), 22–27. https://doi.org/10.1016/j.bbrc.2016.11.037
Muller, S., Raulefs, S., Bruns, P., Afonso-Grunz, F., Plotner, A., Thermann, R., Jager, C., Schlitter, A. M., Kong, B., Regel, I., Roth, W. K., Rotter, B., Hoffmeier, K., Kahl, G., Koch, I., Theis, F. J., Kleeff, J., Winter, P., & Michalski, C. W. (2015). Next-generation sequencing reveals novel differentially regulated mRNAs, lncRNAs, miRNAs, sdRNAs and a piRNA in pancreatic cancer. Molecular Cancer, 14, 94. https://doi.org/10.1186/s12943-015-0358-5
Li, S., Chen, X., Liu, X., Yu, Y., Pan, H., Haak, R., Schmidt, J., Ziebolz, D., & Schmalz, G. (2017). Complex integrated analysis of lncRNAs-miRNAs-mRNAs in oral squamous cell carcinoma. Oral Oncology, 73, 1–9. https://doi.org/10.1016/j.oraloncology.2017.07.026
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hong, S., Li, Q., Yang, Y. et al. Silencing of Long Non-coding RNA LINC01106 Represses Malignant Behaviors of Gastric Cancer Cells by Targeting miR-34a-5p/MYCN Axis. Mol Biotechnol 64, 144–155 (2022). https://doi.org/10.1007/s12033-021-00402-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-021-00402-y