Abstract
Multi-HIV, a multiepitopic protein derived from both gp120 and gp41 envelope proteins of the human immunodeficiency virus (HIV), has been proposed as a vaccine prototype capable of inducing broad immune responses, as it carries various B and T cell epitopes from several HIV strains. In this study, the immunogenic properties of a Multi-HIV expressed in tobacco chloroplasts are evaluated in test mice. BALB/c mice orally immunized with tobacco-derived Multi-HIV have elicited antibody responses, including both the V3 loop of gp120 and the ELDKWA epitope of gp41. Based on splenocyte proliferation assays, stimulation with epitopes of the C4, V3 domain of gp120, and the ELDKWA domain of gp41 elicits positive cellular responses. Furthermore, specific interferon gamma production is observed in both CD4+ and CD8+ T cells stimulated with HIV peptides. These results demonstrate that plant-derived Multi-HIV induces T helper-specific responses. Altogether, these findings illustrate the immunogenic potential of plant-derived Multi-HIV in an oral immunization scheme. The potential of this low-cost immunization approach and its implications on HIV/AIDS vaccine development are discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The worldwide need for developing effective HIV vaccines has prompted research efforts to develop a myriad of vaccine candidates against HIV/AIDS [1]. Thus far, several chimeric protein-based approaches have been reported, most consisting of a carrier protein conferring immunogenicity to a single HIV antigen. For example, a chimeric protein, comprising the Gag (p24) and the Fc region of IgG (Gag-Fc), has been found to bind efficiently to airway mucosa, as it is transported across epithelial surfaces. Upon intranasal co-administration with CpG as an adjuvant, this chimeric protein induces local and systemic immune responses in mice, including long-lasting B and T cell memories [2]. However, this approach tends to limit the number of targeted epitopes. In contrast, design of chimeric multiepitopic immunogens would offer more promising immune responses, as the inclusion of several immunoprotective epitopes within a single polypeptide may mediate the induction of broad immune responses that are likely to confer sterilizing immunity against the hypervariable pathogen [3]. In addition to efficacy, cost is an important factor for vaccination programs, particularly in developing countries wherein HIV/AIDS is a serious health problem [4]. Plant-based vaccines serve as a desirable platform for vaccine production that is both viable and low cost, and well suited for convenient oral administration [5].
Previously, we have reported on designing carrier-free multiepitopic HIV proteins targeting C4 and V3 sequences of gp120. The C4V3 and C4(V3)6 proteins have been shown to be capable of evoking humoral responses against the V3 loop of different HIV isolates, and without the use of co-administered adjuvants. These candidate chimeric proteins have been efficiently expressed in Escherichia coli, and more importantly in both tobacco and lettuce, as low-cost production systems, and found to induce systemic humoral responses following their oral administration, without adjuvants [6–9].
As HIV is a hypervariable virus, the design of chimeric proteins carrying additional immunoprotective epitopes of the enveloping protein, Env component, such as gp41, is an important consideration toward achieving broad immune responses. In particular, the ELDKWA epitope, located in the membrane-proximal external region (MPER) of HIV-1 gp41 and near the C-terminus of the gp41 ectodomain, is conserved among different HIV-1 strains [10]. This epitope is a target of the broad neutralizing human monoclonal IgG antibody, 2F5, and has been used in several vaccine models [11–13]. The ability of anti-ELDKWA antibodies to block viral transmission in vivo by passive immunization in the macaque model [14] further reinforces the importance of inducing antibodies against this region. However, elicitation of an antibody response against the linear ELDKWA epitope has proven to be difficult [15], and most similar multi-epitope antigens have been administered along with a complete Freund’s adjuvant, which is not suitable for human applications [16].
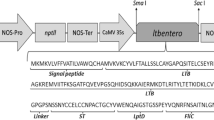
Recently, efforts have been made to improve the design of a candidate vaccine by extending the HIV epitope coverage. In this sense in a previous work we designed and produced a 32-kDa Multi-HIV protein in tobacco chloroplasts, targeting gp120 epitopes, consisting of C4, V1, V2, and V3 domains, as well as that of the ELDKWA epitope of gp41 [17]. Epitopes were chosen based on their reported capabilities to either induce a broad spectrum of neutralizing antibodies or serve as targets of broadly neutralizing antibodies. Configuration of the enhanced multiepitopic HIV (Multi-HIV) vaccine comprises the following sequences described herein. (i) A peptide sequence, KQIINMWQEVGKAMYA, beginning from the fourth conserved domain of gp120 (C4), which serves as a T helper cell, and CTL epitopes, and this has been associated with adjuvant properties [18]. (ii) A V3 consensus neutralizing antigenic determinant, RPNNNTRKSIHIGPGRAFYATE, capable of inducing neutralizing antibodies against 30 % of HIV primary isolates assessed by Haynes et al. [19]. (iii) Several gp41-derived epitopes, such as the conserved epitope VD20 along with the GIKQLQARVLAVERY sequence as this induces Env-specific long-lasting Th responses, and the promiscuous HLA-binding peptide [20, 21]; moreover, six ELDKWA epitope variants [10, 22] along with the peptide NWFDITNWLWKKKK [22, 23], recognized by the broadly neutralizing antibodies 2F5 and 4E10, respectively, are also included. (iv) The MWQEVGKAMYAPPIEG sequence of gp120, located within the b20–b21 hairpin of the bridging sheet region, involved in CD4 binding, and a target for HIV-neutralizing antibodies [24, 25]. (v) Eight variants of the conserved sequence STSIRGKV from gp120 V1/V2 loops, included as antibodies to this region, exhibiting potent neutralization activities [26–28]. (vi) The RSVNFTDNAKTII sequence from V2, as it is recognized by b12 broadly neutralizing antibodies [27, 29]. (vii) The EQELLELDKWAS peptide, a highly conserved epitope in 72 % of 79 HIV isolates assessed by Muster et al. [10], and capable of inducing neutralization of several HIV strains [30, 31].
It is noteworthy to point out that the above-described chimeric Multi-HIV protein is efficiently produced and appears to preserve its antigenicity as it is recognized by sera of HIV-positive patients. Furthermore, such a Multi-HIV protein is also immunogenic, as it is observed to be capable of evoking humoral responses in mice, when orally administered without adjuvants [17].
Therefore, in this study, immunogenic properties of the Multi-HIV protein were analyzed on a broad scale in order to assess its efficacy and potential as a candidate HIV vaccine. Mice were orally immunized with tobacco-derived Multi-HIV, and both humoral and cellular immune responses against specific HIV epitopes derived from gp41 and gp120 sequences were evaluated.
Materials and Methods
Plant Material
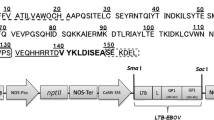
Previously, transplastomic tobacco lines expressing a novel multiepitopic protein, designated as Multi-HIV, have been developed as a collective platform for an oral candidate HIV vaccine [17]. This chimeric protein carries the following epitopes: a region from the fourth conserved domain of gp120 (C4) along with a set of epitopes derived from gp41 and gp120 sequences (Fig. 1), associated with the elicitation of T cells and neutralizing antibodies against HIV [32]. Among transplastomic lines recovered, line THIV1 was confirmed to express the highest levels of Multi-HIV protein and therefore was selected for further studies.
The Multi-HIV sequence used in this study. The 32-kDa Multi-HIV is composed of Env epitopes (gp120 and gp41). The sequence KQIINMWQEVGKAMYA or C4 from gp120 is a B-cell, CTL, and Th-cell epitope; RPNNNTRKSIHIGPGRAFYATE is a V3 consensus neutralizing antigenic determinant; VD20 epitope of gp41 (aa 62–76) induces Env-specific long-lasting Th responses (underline); ELDKWA epitope of gp41 is an important highly conserved region recognized by broadly neutralizing antibodies 2F5; the V3 loop from MN HIV isolated is also incorporated. The 4E10 epitope is recognized by broadly neutralizing antibodies and located between V3 loop and the epitope corresponding to the CD4-binding site (RSVNFTDNAKTII sequence from V2), identified as a target of neutralizing antibodies. The V1/V2 loops epitope STSIRGKV was incorporated as tandem repeats to enhance the immunogenicity. Finally, the last epitope included, RSVNFTDNAKTII, is recognized by the b12 broadly neutralizing antibody
Seeds of both transplastomic line THIV1 and wild-type tobacco were germinated in sunshine mix and maintained in a growth chamber under 16-h light/8-h photoperiod at 25 °C, at normal light [100 µmol/(m2 s)] and humidity (30 %) conditions. When plants were about 30 cm in length, leaves were collected, frozen at −80 °C, and then subsequently freeze-dried overnight in a Labconco chamber at 0.002 mbar and a collector temperature of −4 °C.
Leaves were ground into a fine powder using an analytical mill, maintained at 25 °C, and used for enzyme-linked immunosorbent assay (ELISA) analysis to estimate the amount of recombinant antigen (per gram of dried material) using an anti-Histidine tag antibody targeting the recombinant protein. Dried leaf tissues of tobacco line THIV1 expressing the Multi-HIV (TT) at 16 µg/g fresh leaf biomass or of wild-type (WT) plants were used for all subsequent immunological analyses.
Mice Immunization
Immunogenicity of tobacco-derived Multi-HIV protein was evaluated in female BALB/c mice (11 weeks of age), which were handled according to the guidelines of the Federal Regulations for Animal Experimentation and Care (NOM-062-ZOO-1999, Ministry of Agriculture, Mexico). The immunization scheme consisted of three groups of mice (n = 5), each receiving, by gavage, one of the following treatments: 500 µl Phosphate Buffered Saline (PBS group), 50 mg of freeze-dried tobacco leaves of THIV1 plants containing the Multi-HIV protein (~100 µg of Multi-HIV) (TT group), or 50 mg of freeze-dried WT tobacco leaves (WT group). Plant doses consisted of plant powder resuspended in 500 µl of PBS. These treatments were applied on days 0, 7, 14, and 28. Test animals were sacrificed at day 35, and both sera and spleen were collected to conduct antibody, proliferation, and interferon production assays.
Antibody Assays
Antibody responses against some of the epitopes included in Multi-HIV were determined by ELISA, as previously described [33]. Ninety-six-well polystyrene plates were individually coated overnight at 4 °C with one of the following proteins: 1 μg/well of E. coli-derived C4(V3)6 (purified as described by Govea-Alonso et al. [7] ), carrying a T helper epitope from the fourth conserved domain (C4) of gp120 along with six tandemly arranged V3 loops corresponding to five different HIV isolates, including MN, IIIB, RF, CC, and RU; 0.5 μg/well of a synthetic peptide carrying the V3 MN epitope (GPGRAFTYA; GenScript, NJ, USA); 1 μg/well of a synthetic peptide designated as gp41ELDKWAp, carrying tandem repeats of the conserved linear epitope within the envelope glycoprotein gp41 and having a core recognition sequence, ELDKWA, along variants (ELDKWAKLDQWAALDSWNALDLKWD; GenScript, NJ, USA); 0.025 μg/well of the gp120 native protein (Protein Sciences Corporation, CT, USA); and 0.25 μg/well of E. coli-derived Multi-HIV protein (purified as described in [17]). After a blocking step, serum samples were added in a 1:20 dilution, and plates were incubated overnight. Subsequently, a goat anti-mouse IgG-HRP (1:1000, Pierce, Rockford, IL) was added as a secondary antibody, and plates were incubated for 1 h at 37 °C. After washing, plates were incubated for 15 min with 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid (ABTS), used as a substrate (Sigma, St. Louis, MO, USA). Specific antibody levels in serum samples were expressed as corresponding optical density values, measured at OD415nm, using a Multiskan Ascent microplate reader (Thermo Electron Corporation, Waltham, MA, USA).
Cell Proliferation Assays
A carboxyfluorescein (CFSE) proliferation assay was conducted as previously described [6, 9]. Splenocytes were isolated from different groups of test mice using the following protocol. Spleens were disaggregated under sterile conditions, and red blood cells were lysed using a hypotonic ammonium chloride solution. Following two washes with PBS, cells were resuspended in a culture medium. The culture medium consisted of RPMI 1640 (Invitrogen, Carlsbad, CA, USA) supplemented with 1 mM sodium pyruvate, 0.1 mM non-essential amino acids, 2 mM glutamine, 25 mM HEPES, 100 U/ml penicillin, 100 µg/ml streptomycin, and 10 % (v/v) fetal bovine serum (FBS) (Invitrogen, Carlsbad, CA, USA). Freshly prepared splenocytes (10 x106 cells) were then stained for 10 min with 1 mL CFSE (5,6-carboxyfluorescein diacetate succinimidyl ester; Sigma-Aldrich, St. Louis, MO, USA) to a final concentration of 5 µM. After a subsequent washing step, a total of 2 × 106 cells were added per well in 1 ml volume to 6-well flat-bottom plates and incubated in a complete RPMI-1640 medium (GIBCO BRL, California, USA) for 65 h at 37 °C and in the presence of 5 % CO2. Cells were stimulated by adding 1 µg of either a pure recombinant E. coli-derived C4(V3)6 (rC4(V3)6), or a synthetic gp41ELDKWAp epitope, or a synthetic C4 epitope purchased from GenScript (NJ, USA). Unstimulated cell cultures were included as controls.
Subsequently, cells were harvested, washed with PBS-1 %, heat-inactivated fetal bovine serum (FBS)-0.1 % NaN3, and stained for 30 min (4 °C) with an appropriate combination of fluorochrome-labeled mAbs to conduct flow cytometry analysis. Antibodies labeled with phycoerythrin (PE), allophycocyanin (APC), cyanine 5 (Cy5), Cy5-PE, or fluorescein isothiocyanate (FITC) were used. CD4-PE/Cy5 and CD8-APC monoclonal antibodies were purchased from Caltag (Little Balmer Buckingham, UK). After labeling, cells were washed with PBS-1 %, FBS-0.1 % NaN3, resuspended in PBS, and analyzed by flow cytometry in a 4-color FACSCalibur (Becton–Dickinson, Franklin Lakes, NJ, USA) instrument using CellQuest Pro software.
Lymphocytes were gated on the basis of forward- and side-scattering, and a total of 30,000 events from these gated regions were obtained. Proliferating cells were identified as those populations with decreased mean FL1 fluorescence (CFSE), and CD3+ cells were gated to identify either CD4+ T helper cells or CD8+ T cytotoxic cells. To generate dot plots, data analysis was performed using summit software.
IFN-γ Production Assays
To estimate IFN-γ production, splenocytes were cultured on RPMI 1640 medium supplemented with 10 % FBS, 100 U/ml of penicillin G, and 100 µg/ml of streptomycin. Cell cultures were grown in either presence or absence of a C4(V3)6 pure protein (1 µg/ml) at 37 °C, high humidity, along with 5 % CO2 for 1 h. These were subsequently incubated with 10 µg/ml of brefeldin A (Sigma, Missouri, USA) for 4 h under the same conditions. Cells were washed in ice-cold PBS containing 3 % FBS and 0.05 % NaN3. Approximately 1 × 106 cells were labeled in a total volume of 100 µl of staining buffer for 30 min with either anti-CD8-APC or anti-CD4-PE/Cy5 monoclonal antibodies (Caltag, Little Balmer Buckingham, UK), along with isotype-matched controls (BD, California, USA). Cells were fixed and permeabilized with cytofix/cytoperm solution for 15 min, incubated with 0.1 µg of fluorescein isothiocyanate (FITC)-labeled anti-IFN-γ (BD, California, USA), and washed with a cold staining buffer containing 0.05 % NaN3.
Flow cytometry analysis was performed using a FACSCalibur™ flow cytometer (BD, California, USA), as described above. A total number of 30,000 events were generated and analyzed. The frequency of positive events was calculated against relevant stained controls.
Statistical Analysis
ELISA, proliferation assays, and IFN-γ production data were subjected to one-way ANOVA, followed by a post hoc Tukey test using Graph Pad Prism 5 software at P < 0.05.
Results
Tobacco-Derived Multi-HIV Elicits Broad Humoral Responses
To investigate the immunogenic potential of the tobacco-derived Multi-HIV protein, antibody levels against specific antigens were measured in test mice subjected to an oral immunization scheme. Sera from immunized mice were initially tested for reactivity against the Multi-HIV protein. Mice immunized with tobacco-derived Multi-HIV (TT group) showed significantly higher OD readings (p < 0.05) than mice treated with WT tissues (control) (Fig. 2a). Reactivity against C4(V3)6, carrying sequences of the V3 loop from gp120, yielded higher OD measurements for the TT group (P < 0.05). To assess humoral responses, i.e., levels of antibodies against the gp41 component, an ELISA assay targeting the ELDWKA sequence (ELDKWAKLDQWAALDSWNALDLKWD) was performed. Significantly higher ELISA readings were observed for the TT group than those for the control group of mice (Fig. 2b). Similarly, an ELISA assay targeting the Multi-HIV protein produced in E. coli revealed significantly higher ELISA readings for the TT group than those for the control group (Fig. 2c). These assays demonstrated that mice immunized with tobacco-derived Multi-HIV generated specific antibodies against the targeted epitopes.
Humoral immune responses against C4(V3)6 peptide, gp41-derived epitope gp41ELDKWAp (ELDKWAKLDQWAALDSWNALDLKWD), and Multi-HIV recombinant protein induced by tobacco-derived Multi-HIV. ELISA analysis was performed by coating plates with a C4(V3)6 protein (a), gp41ELDKWAp peptide (b), or E. coli-derived Multi-HIV (c). Sera from mice immunized with tobacco-derived Multi-HIV (TT), wild-type tobacco (WT), and PBS, both used as controls, were analyzed in a 1:20 dilution. Mean OD 415 nm values ± SD from each experimental group (n = 5, analyzed in triplicate) are shown. *p < 0.05 vs. the WT group
The reactivity against the V3 synthetic peptide (GPGRAFTYA) was significantly higher in the TT group than with either WT or PBS control group (Fig. 3a). Although ELISA assays targeting gp120 revealed higher signals in the TT group than those in either WT or PBS control group, these were not significantly different (Fig. 3b).
Humoral immune responses against the native V3MN induced by tobacco-derived Multi-HIV. ELISA analysis was performed by coating plates with a V3MN native synthetic peptide (a) and a native gp120 protein (b). Sera from mice immunized with tobacco-derived Multi-HIV (TT), wild-type tobacco (WT), and PBS, both used as controls, were analyzed in a 1:20 dilution. Mean OD 415 nm values ± SD from each experimental group (n = 5, analyzed in triplicate) are shown. *p < 0.05 vs. the WT group
Tobacco-Derived Multi-HIV Induces T Helper and CTL Responses
To assess whether significant T helper cell responses are induced by oral immunization with tobacco-derived Multi-HIV, isolated splenocytes were stimulated with each of recombinant C4(V3)6, synthetic gp41ELDKWAp, or C4 peptides. The proliferation assay performed the following stimulation with a recombinant C4(V3)6 indicated that in mice immunized with Multi-HIV, there was an increase from 2.69 % (without stimulus) to 5.07 % (with stimulus) in the proportion of proliferating CD4+ T cells (p < 0.05); whereas proliferating CD4+ T cell responses of mice treated with WT tobacco or the vehicle alone did not change (p > 0.05), and these were lower than those observed in TT-immunized mice (Fig. 4a). In the presence of a stimulus, there was a slight increase in the proportion of proliferating CD8+ T cells in the TT group (p < 0.05), while the proliferative responses observed in WT and PBS groups did not change significantly (p > 0.05) upon stimulation (Fig. 4b). Furthermore, the TT group showed a significantly higher proportion (p < 0.05) of CD4+ T and CD8+ T cells producing IFN-γ (an increase of 1.5 and 2.5 %, respectively) in the presence of the C4(V3)6 stimulus in comparison with the observed proportion elicited in splenocyte cultures of control PBS and WT groups (Fig. 5).
Cellular immune responses against C4(V3)6 induced by tobacco-derived Multi-HIV. Splenocytes from mice immunized with multi-HIV-producing tobacco (TT), wild-type tobacco (WT), or PBS were stained with CFSE, incubated for 72 h in either the presence or absence of a C4(V3)6 stimulus (peptide with six different V3 from different isolates), and labeled with anti-CD4-PE/Cy5 (a) and anti CD8-APC (b) monoclonal antibodies. Estimates of percentages of proliferating CD4+ and CD8+ cells in 30,000 events of lymphocyte-gated regions along with dot plots of representative results are presented. Mean percentage of proliferating cells is presented in the right graph. Augment in CD4 T cell proliferation was observed in TT-immunized mice (p < 0.05) but not in control groups (PBS- or WT-derived stimulated splenocytes). *p < 0.05 vs. unstimulated TT; $ p < 0.05 vs. the stimulated WT group
IFN-γ production by CD4+ and CD8+ T cells from mice immunized with tobacco-derived Multi-HIV. Splenocytes from mice immunized with either tobacco-derived multi-HIV (TT), wild-type tobacco (WT), or PBS were incubated in the presence of pure C4(V3)6 as a stimulus. IFN-γ-producing CD4+ T and CD8+ cells were detected by staining with labeled antibodies followed by flow cytometry analysis. Mean percentage of IFN-γ-positive cells is presented in the right graph. *p < 0.05 vs. stimulated WT group
In the presence of the gp41ELDKWAp peptide stimulus, CD4+ proliferation in the TT group increased from 2.7 to 5.5 % (p < 0.05), while no significant differences were observed upon stimulation in both control PBS and WT groups Fig. 6a). More importantly, it was observed that upon stimulation in the TT group, CD8+ proliferation showed a twofold increase compared to the WT group, which was significantly higher than that observed in unstimulated TT cells (Fig. 6b). Furthermore, the proportion of CD8+ proliferating cells in the PBS and WT groups did not change significantly upon simulation with gp41ELDKWAp.
Cellular immune responses against gp41GELDKWAp epitope induced by tobacco-derived Multi-HIV. Splenocytes were stained with CFSE, cultivated in the presence or absence of gp41EELDKWAp peptide containing ELDKWAKLDQWAALDSWNALDLKWD epitope as a stimulus, and labeled with anti-CD4-PE/Cy5 (a) or anti-CD8-APC (b) monoclonal antibodies. Estimates of percentages of proliferating CD4+ or CD8+ cells in 30,000 events of lymphocyte-gated region have been determined. Representative results obtained from mice immunized with multi-HIV-producing tobacco (TT), wild-type tobacco (WT), or PBS are presented. Mean percentage of proliferating cells is presented in the right graph. *p < 0.05 vs. unstimulated TT; $ p < 0.05 vs. the stimulated WT group
When splenocytes from the three immunized groups were stimulated with the C4 peptide, described as a CTL, and the Th-cell epitope, significantly higher specific proliferation responses of T lymphocytes were observed in the TT group compared to those of both WT and PBS control groups, as well as with unstimulated TT cells (p < 0.05). The observed responses in the TT group showed a three fold increase in CD4+ (Fig. 7a) and a two fold increase in CD8+ (Fig. 7b), which is consistent with the CTL and Th epitopes present in this sequence.
Cellular immune responses against C4 epitope induced by tobacco-derived Multi-HIV. Splenocytes were stained with CFSE, incubated in either the presence or absence of a C4 peptide (KQIINMWQEVGKAMYA) stimulus, and labeled with anti -CD4-PE/Cy5 (a) or anti-CD8-APC (b) monoclonal antibodies. Estimates of percentages of proliferating CD4+ and CD8+ cells in 30,000 events of lymphocyte-gated region have been determined. Representative results obtained from mice immunized with multi-HIV-producing tobacco (TT), wild-type tobacco (WT), or PBS are presented. Mean percentage of proliferating cells is presented in the right graph. *p < 0.05 vs. unstimulated TT; $ p < 0.05 vs. the stimulated WT group
Discussion
In this study, the immunogenic properties of a tobacco-derived Multi-HIV protein have been detected and characterized in orally immunized BALB/c mice without adjuvant co-administration in an effort to establish a ‘proof of concept’ of a plant-made multiepitopic HIV oral immunogenic protein. As specific antibodies against both gp120 (comprising epitopes of the C4 and V3 domains) and gp41 (comprising epitope ELDKWA) components of the chimeric Multi-HIV protein are induced, this Multi-HIV protein is deemed immunogenic. Although the sequence of the V3 loop is highly variable, antibodies directed against the consensus region included in the multiepitopic HIV protein are found to be capable of neutralizing approximately 30 % of the primary isolates analyzed by Haynes et al. [19]. It is proposed that the observed virus neutralization by this chimeric protein is achieved via interference of HIV-1 binding to CD4 and CCR-5 receptors, as previously reported by Trkola et al. [34].
Since the completion of the first clinical trial for an HIV vaccine that demonstrated a limited level of protection against HIV infection, characterization of immune profiles associated with observed protective effects has been strongly associated with the humoral responses against the Env component of the HIV pathogen [35, 36]. In this study, a Multi-HIV protein, carrying several epitopes such as those recognized by the broadly neutralizing antibodies 2F5, 4E10, and b12 and those of V1/V2 regions, has been designed in an effort to induce antibody responses that confer protection. Several human monoclonal antibodies against V2 epitopes are capable of recognizing multiple subtypes of the V2 loop [37–39]. Moreover, antibody-dependent cellular cytotoxicity (ADCC), Tier 1 neutralization, and capture of virions can be induced by the RV144 vaccine as this vaccine elicits monoclonal antibodies against V2 [38, 40] that are inversely correlated with the risk of HIV-1 infection [41]. As the Multi-HIV chimeric protein is capable of inducing broad humoral responses, it may induce long-lasting immune responses and confer immunoprotective properties.
In this study, a Multi-HIV comprising a KQIINMWQEVGKAMYA (C4) sequence derived from the fourth conserved domain of gp120, serving as a T helper, and CTL epitopes, is used, and its configuration has been associated with adjuvant properties [18]. It is found that this Multi-HIV protein is capable of eliciting cellular responses against different stimuli (Figs. 4, 5, 6 7). More specifically, proliferation assays following C4 and C4(V3)6 stimulation have provided evidence of the elicitation of CD8+ responses by the tobacco-derived Multi-HIV (Fig. 4). In the presence of the C4(V3)6 stimulus, IFN-γ production by CD8+ cells is induced (Fig. 5), and this cytokine serves as a critical antiviral mediator [42, 43]. This finding is of particular importance as CTL responses are typically poorly evoked in the majority of orally administered subunit vaccines due to the mode of antigen uptake, antigen design, and peculiarity of the mucosal immune system [44]. As distinct components of the Multi-HIV have demonstrated a capacity for inducing Th responses, which are critical for activating B and CTL responses, the Multi-HIV is a promising multiepitopic vaccine candidate. Although ELDKWA is typically recognized as a B-cell epitope, a notable CTL proliferation is observed following stimulation with gp41ELDKWAp. Therefore, further studies should be conducted to determine whether or not ELDKWA is also a CTL epitope. We presume that the Multi-HIV protein forms aggregates as suggested by the previous characterization by Western blots, where bands of higher molecular weight were detected [17]. Therefore, aggregation or precipitation of the protein within the chloroplasts or within the mouse biological compartments might account for the immunogenicity of the Multi-HIV protein, and also to its ability to elicit cellular immune responses. Although the precise way in which T cell responses against tobacco-derived Multi-HIV protein are elicited are to be determined, the antigen administered via gastric lavage must be uptaken by DCs, which exhibit the ability to internalize soluble exogenous Ags and process them for cross-presentation by class I MHC (MHC-I) to CD8+ T cells [45].
Production of multiepitopic proteins has been previously explored using conventional production platforms and with varied outcomes. For example, earlier clinical trials using HIV Env-based subunit vaccines have elicited antibodies that reacted with gp120, but have not resulted in neutralizing activities [46–49]. Similar findings were reported in studies wherein HIV Env mutants carrying the LLELDKWASL sequence, replacing V1, V2, V3, or V4 loops, have been used. Nevertheless, in such studies both mice and guinea pigs were immunized with plasmid DNA encoding the 2F5 epitope, which optimized within the V2 loop and produced the strongest antibody responses [50]. On the other hand, Coëffier et al. [15] incorporated ELDKWAS and LLELDKWASL sequences into the maltose-binding protein (MBP) of E. coli and reported that the immunized mice elicited LLELDKWASLK peptide-binding activity.
Given the wide genetic variability of HIV, designing multiepitopic proteins capable of evoking broad immune responses, preferably activating cellular and humoral arms of the immune system, is worthy of further exploration [51]. In this study, a multiepitopic protein capable of inducing both responses is produced in a low-cost platform, plant chloroplasts. Previously, we have reported on the production of two HIV antigens in plants. These antigens included C4V3, carrying a single sequence of the gp120 V3 loop from the MN isolate, and C4(V3)6, targeting six V3 loops that correspond to five different HIV isolates, including MN, IIIB, RF, CC, and RU. Both candidates are found to be immunogenic following oral administration and without the use of adjuvants, thus suggesting a notable immunogenic potential, likely due to the presence of the Th epitope from the C4 gp120 domain [7, 9, 10]. These earlier findings have prompted the development of a new candidate vaccine, designated as Multi-HIV, targeting additional epitopes of gp120 and the epitope ELDKWA from gp41, expected to serve as an elicitor of broader immune responses. In this study, the evaluation of this Multi-HIV protein, expressed in plants, following oral administration in test animals has demonstrated the efficacy of this protein in eliciting appropriate and broad humoral responses, Th responses. Thus, this chimeric protein can serve as a broad immunogen, and its production in edible crops has potential for use in HIV vaccine development, rendering it as a low-cost production vaccine system.
One of the advantages of plant-made vaccines is that these can bypass protein purification steps, as well as used for oral immunization in low-cost formulations. However, few studies have reported on the immunogenicity of HIV plant-derived antigens administered via mucosal routes [51]. Therefore in this study, the immunogenicity of a Multi-HIV complex has been explored as a model vaccination approach, consisting of a plant biomass administered via an oral route along with the absence of co-administered adjuvants. Under these conditions, the observed responses, although modest in potency, have demonstrated a promising potential of this candidate immunogen, as most antigen candidates tend to show no immunogenic responses when administered orally. Thus, it is proposed that the Multi-HIV antigen could be included in immunization schemes comprising priming of the parenteral route with antigens produced in conventional platforms, and subsequently boosted by the administration of a plant-made Multi-HIV antigen via an oral route [52]. This approach may lead to new schemes that confer appropriate immune profiles, in terms of potency and antibody diversity, against HIV.
References
Schiffner, T., Sattentau, Q. J., & Dorrell, L. (2013). Development of prophylactic vaccines against HIV-1. Retrovirology, 10, 72.
Lu, L., Palaniyandi, S., Zeng, R., Bai, Y., Liu, X., & Wang, Y. (2011). A neonatal Fc receptor-targeted mucosal vaccine strategy effectively induces HIV-1 antigen-specific immunity to genital infection. Journal of Virology, 85, 10542–10553.
Domingo, E., Grande-Pérez, A., & Martín, V. (2008). Future prospects for the treatment of rapidly evolving viral pathogens: insights from evolutionary biology. Expert Opinion on Biological Therapy, 8, 1455–1460.
Ebrahim, O., & Mazanderani, A. H. (2013). Recent developments in HIV treatment and their dissemination in poor countries. Infectious Disease Reports, 5(Suppl 1), e2.
Melnik, S., & Stoger, E. (2013). Green factories for biopharmaceuticals. Current Medicinal Chemistry, 20, 1038–1046.
Varona-Santos, J. T., Vazquez-Padrón, R. I., & Moreno-Fierros, L. (2006). Production of a short recombinant C4V3 HIV-1 immunogen that induces strong anti-HIV responses by systemic and mucosal routes without the need of adjuvants. Viral Immunology, 19, 237–249.
Govea-Alonso, D. O., Gómez-Cardona, E. E., Rubio-Infante, N., García-Hernández, A. L., Varona-Santos, J. T., Salgado-Bustamante, M., et al. (2013). Production of an antigenic C4(V3)6 multiepitopic HIV protein in bacterial and plant systems. Plant Cell Tissue Organ Culture, 113, 73–79.
Govea-Alonso, D. O., Rubio-Infante, N., García-Hernández, A. L., Varona-Santos, J. T., Korban, S. S., Moreno-Fierros, L., et al. (2013). Immunogenic properties of a lettuce-derived C4(V3)6 multiepitopic HIV protein. Planta, 238, 785–792.
Rubio-Infante, N., Govea-Alonso, D. O., Alpuche-Solís, Á. G., García-Hernández, A. L., Soria-Guerra, R. E., Paz-Maldonado, L. M., et al. (2012). A chloroplast-derived C4V3 polypeptide from the human immunodeficiency virus (HIV) is orally immunogenic in mice. Plant Molecular Biology, 78, 337–349.
Muster, T., Steindl, F., Purtscher, M., Trkola, A., Klima, A., Himmler, G., et al. (1993). A conserved neutralizing epitope on gp41 of human immunodeficiency virus type 1. Journal of Virology, 67, 6642–6647.
Arnold, G. F., Velasco, P. K., Holmes, A. K., Wrin, T., Geisler, S. C., Phung, P., et al. (2009). Broad neutralization of human immunodeficiency virus type 1 (HIV-1) elicited from human rhinoviruses that display the HIV-1 gp41 ELDKWA epitope. Journal of Virology, 83, 5087–5100.
Matoba, N., Magérus, A., Geyer, B. C., Zhang, Y., Muralidharan, M., Alfsen, A., et al. (2004). A mucosally targeted subunit vaccine candidate eliciting HIV-1 transcytosis-blocking Abs. Proceedings of the National Academy of Sciences of the United States of America, 101, 13584–13589.
Marusic, C., Rizza, P., Lattanzi, L., Mancini, C., Spada, M., Belardelli, F., et al. (2001). Chimeric plant virus particles as immunogens for inducing murine and human immune responses against human immunodeficiency virus type 1. Journal of Virology, 75(18), 8434–8439.
Mascola, J. R., Stiegler, G., VanCott, T. C., Katinger, H., Carpenter, C. B., Hanson, C. E., et al. (2000). Protection of macaques against vaginal transmission of a pathogenic HIV-1/SIV chimeric virus by passive infusion of neutralizing antibodies. Nature Medicine, 6, 207–210.
Coëffier, E., Clément, J. M., Cussac, V., Khodaei-Boorane, N., Jehanno, M., Rojas, M., et al. (2000). Antigenicity and immunogenicity of the HIV-1 gp41 epitope ELDKWA inserted into permissive sites of the MalE protein. Vaccine, 19, 684–693.
Li, H., Liu, Z. Q., Ding, J., & Chen, Y. H. (2002). Recombinant multi-epitope vaccine induce predefined epitope-specific antibodies against HIV-1. Immunology Letters, 84, 153–157.
Rosales-Mendoza, S., Rubio-Infante, N., Monreal-Escalante, E., Govea-Alonso, D. O., García-Hernández, A. L., Salazar-González, J. A., et al. (2014). Chloroplast expression of an HIV envelop-derived multiepitope protein: towards a multivalent plant-based vaccine. Plant Cell, Tissue and Organ Culture, 116, 111–123.
Patterson, L. J., Robey, F., Muck, A., Van Remoortere, K., Aldrich, K., Richardson, E., et al. (2001). A conformational C4 peptide polymer vaccine coupled with live recombinant vector priming is immunogenic but does not protect against rectal SIV challenge. AIDS Research and Human Retroviruses, 17, 837–849.
Haynes, B. F., Ma, B., Montefiori, D. C., Wrin, T., Petropoulos, C. J., Sutherland, L. L., et al. (2006). Analysis of HIV-1 subtype B third variable region peptide motifs for induction of neutralizing antibodies against HIV-1 primary isolates. Virology, 345, 44–55.
Kent, S. J., Greenberg, P. D., Hoffman, M. C., Akridge, R. E., & McElrath, M. J. (1997). Antagonism of vaccine-induced HIV-1-specific CD4+ T cells by primary HIV-1 infection: potential mechanism of vaccine failure. Journal of Immunology, 158, 807–815.
Malhotra, U., Holte, S., Zhu, T., Delpit, E., Huntsberry, C., Sette, A., et al. (2003). Early induction and maintenance of Env-specific T-helper cells following human immunodeficiency virus type 1 infection. Journal of Virology, 77, 2663–2674.
Zwick, M. B., Labrijn, A. F., Wang, M., Spenlehauer, C., Saphire, E. O., Binley, J. M., et al. (2001). Broadly neutralizing antibodies targeted to the membrane-proximal external region of human immunodeficiency virus type 1 glycoprotein gp41. Journal of Virology, 75, 10892–10905.
Mehandru, S., Wrin, T., Galovich, J., Stiegler, G., Vcelar, B., Hurley, A., et al. (2004). Neutralization profiles of newly transmitted human immunodeficiency virus type 1 by monoclonal antibodies 2G12, 2F5, and 4E10. Journal of Virology, 78, 14039–14042.
Kelker, H. C., Itri, V. R., & Valentine, F. T. (2010). A strategy for eliciting antibodies against cryptic, conserved, conformationally dependent epitopes of HIV envelope glycoprotein. PLoS ONE, 5, e8555. doi:10.1371/journal.pone.0008555.
Kwong, P. D., Wyatt, R., Robinson, J., Sweet, R. W., Sodroski, J., & Hendrickson, W. A. (1998). Structure of an HIV gp120 envelope glycoprotein in complex with the CD4 receptor and a neutralizing human antibody. Nature, 393, 648–659.
Gaudebout, P., Zeliszewski, D., Golvano, J. J., Pignal, C., Le Gac, S., Borras-Cuesta, F., et al. (1997). Binding analysis of 95 HIV gp120 peptides to HLA-DR1101 and -DR0401 evidenced many HLA-class II binding regions on gp120 and suggested several promiscuous regions. Journal of Acquire Immune Deficiency Syndromes and Human Retrovirology, 14, 91–101.
Geretti, A. M., Van Baalen, C. A., Borleffs, J. C., Van Els, C. A., & Osterhaus, A. D. (1994). Kinetics and specificities of the T helper-cell response to gp120 in the asymptomatic stage of HIV-1 infection. Scandinavian Journal of Immunology, 39, 355–362.
Warrier, S. V., Pinter, A., Honnen, W. J., Girard, M., Muchmore, E., & Tilley, S. A. (1994). A novel, glycan-dependent epitope in the V2 domain of human immunodeficiency virus type 1 gp120 is recognized by a highly potent, neutralizing chimpanzee monoclonal antibody. Journal of Virology, 68, 4636–4642.
Wahren, B., Rosen, J., Sandström, E., Mathiesen, T., Modrow, S., & Wigzell, H. (1989). HIV-1 peptides induce a proliferative response in lymphocytes from infected persons. Journal of Acquire Immune Deficiency Syndromes, 2, 448–456.
Broliden, P. A., von Gegerfelt, A., Clapham, P., Rosen, J., Fenyö, E. M., Wahren, B., & Broliden, K. (1992). Identification of human neutralization-inducing regions of the human immunodeficiency virus type 1 envelope glycoproteins. Proceedings of the National Academy of Sciences of the United States of America, 89, 461–465.
Vanini, S., Longhi, R., Lazzarin, A., Vigo, E., Siccardi, A. G., & Viale, G. (1993). Discrete regions of HIV-1 gp41 defined by syncytia-inhibiting affinity-purified human antibodies. AIDS, 7, 167–174.
Xiao, Y., Zhao, Y., Lu, Y., & Chen, Y. H. (2000). Epitope-vaccine induces high levels of ELDKWA-epitope-specific neutralizing antibody. Immunological Investigations, 1, 41–50.
Rosales-Mendoza, S., Alpuche-Solis, A., Soria-Guerra, R. E., Moreno-Fierros, L., Herrera-Díaz, A., & Korban, S. S. (2009). Expression of an Escherichia coli antigenic fusion protein comprising the heat labile toxin B subunit and the heat stable toxin, and its assembly as a functional oligomer in transplastomic tobacco plants. Plant Journal, 57, 45–54.
Trkola, A., Dragic, T., Arthos, J., Binley, J. M., Olson, W. C., Allaway, G. P., et al. (1996). CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature, 384, 184–187.
Rao, M., Peachman, K. K., Kim, J., Gao, G., Alving, C. R., Michael, N. L., et al. (2013). HIV-1 variable loop 2 and its importance in HIV-1 infection and vaccine development. Current HIV Research, 11, 427–438.
Gottardo, R., Bailer, R. T., Korber, B. T., Gnanakaran, S., Phillips, J., Shen, X., et al. (2013). Plasma IgG to linear epitopes in the V2 and V3 regions of HIV-1 gp120 correlate with a reduced risk of infection in the RV144 vaccine efficacy trial. PLoS ONE, 8, e75665. doi:10.1371/journal.pone.0075665.
Gorny, M. K., Pan, R., Williams, C., Wang, X. H., Volsky, B., O’Neal, T., et al. (2012). Functional and immunochemical cross-reactivity of V2-specific monoclonal antibodies from human immunodeficiency virus type 1-infected individuals. Virology, 427, 198–207.
Liao, H. X., Bonsignori, M., Alam, S. M., McLellan, J. S., Tomaras, G. D., Moody, M. A., et al. (2013). Vaccine induction of antibodies against a structurally heterogeneous site of immune pressure within HIV-1 envelope protein variable regions 1 and 2. Immunity, 38, 1–11.
Walker, L. M., Phogat, S. K., Chan-Hui, P. Y., Wagner, D., Phung, P., Goss, J. L., et al. (2009). Broad and potent neutralizing antibodies from an african donor reveal a new HIV-1 vaccine target. Science, 326, 285–289.
Liu, P., Yates, N. L., Shen, X., Bonsignori, M., Moody, M. A., Liao, H. X., et al. (2013). Infectious virion capture by HIV-1 gp120 specific IgG from RV144 vaccinees. Journal of Virology, 87, 7828–7836.
Zolla-Pazner, S., Decamp, A., Gilbert, P. B., Williams, C., Yates, N. L., Williams, W. T., et al. (2014). Vaccine-induced IgG antibodies to V1V2 regions of multiple HIV-1 subtypes correlate with decreased risk of HIV-1 infection. PLoS ONE, 9, e87572.
Sáez-Cirión, A., Lacabaratz, C., Lambotte, O., Versmisse, P., Urrutia, A., Boufassa, F., et al. (2007). HIV controllers exhibit potent CD8 T cell capacity to suppress HIV infection ex vivo and peculiar cytotoxic T lymphocyte activation phenotype. Proceedings of the National Academy of Sciences of the United States of America, 104, 6776–6781.
Chesler, D. A., & Reiss, C. S. (2002). The role of IFN-gamma in immune responses to viral infections of the central nervous system. Cytokine & Growth Factor Reviews, 13(441–54), 11.
Neutra, M. R., & Kozlowski, P. A. (2006). Mucosal vaccines: the promise and the challenge. Nature Reviews Immunology, 6, 148–158.
Brossart, P., & Bevan, M. J. (1997). Presentation of exogenous protein antigens on major histocompatibility complex class I molecules by dendritic cells: pathway of presentation and regulation by cytokines. Blood, 90, 1594–1599.
Belshe, R. B., Mendelman, P. M., Treanor, J., King, J., Gruber, W. C., & Piedra, P. (1998). The efficacy of live attenuated, cold-adapted, trivalent, intranasal influenzavirus vaccine in children. The New England Journal of Medicine, 338, 1405–1412.
Connor, R. I., Korber, B. T. M., Graham, B. S., Hahn, B. H., Ho, D. D., Walker, B. D., et al. (1998). Immunological and virological analyses of persons infected by human immunodeficiency virus type I while participating in trials of recombinant gp120 subunit vaccines. Journal of Virology, 72, 1552–1576.
VanCott, T. C., Mascola, J. R., Loomis-Price, L. D., Sinangil, F., Zitomersky, N., McNeil, J. G., et al. (1999). Cross-subtype neutralizing antibodies induced in baboons by a subtype E GP120 immunogen based on an R5 primary HIV-1 envelope. Journal of Virology, 73, 4640–4650.
Wrin, T., Crawford, L., Sawyer, L., Weber, P., Sheppard, H. W., & Hanson, C. V. (1994). Neutralizing antibody responses to autologous and heterologous isolates of human immunodeficiency virus. Journal of Acquire Immune Deficiency Syndromes and Human Retrovirology, 7, 211–219.
Liang, X., Munshi, S., Shendure, J., Mark, G., Davies, M. E., Freed, D. C., et al. (1999). Epitope insertion into variable loops of HIV-1 gp120 as a potential means to improve immunogenicity of viral envelope protein. Vaccine, 17, 2862–2872.
Rosales-Mendoza, S., Rubio-Infante, N., Govea-Alonso, D. O., & Moreno-Fierros, L. (2012). Current status and perspectives of plant-based candidate vaccines against the human immunodeficiency virus (HIV). Plant Cell Reports, 31, 495–511.
Youm, J. W., Jeon, J. H., Kim, H., et al. (2008). Transgenic tomatoes expressing human beta-amyloid for use as a vaccine against Alzheimer’s disease. Biotechnology Letters, 30, 1839–1845.
Acknowledgments
This research was funded by Grants 102109 from CONACYT and 173858 from CIBIOGEM to SRM.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rubio-Infante, N., Govea-Alonso, D.O., Romero-Maldonado, A. et al. A Plant-Derived Multi-HIV Antigen Induces Broad Immune Responses in Orally Immunized Mice. Mol Biotechnol 57, 662–674 (2015). https://doi.org/10.1007/s12033-015-9856-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-015-9856-3