Abstract
Purpose
To investigate the clinical importance of the inflammatory prognostic index (IPI) in patients with metastatic colorectal cancer treated with regorafenib.
Methods
A retrospective analysis of 65 metastatic CRC patients treated with regorafenib between 2015 and 2020 was performed. The association between NLR, PNLR, IPI, and overall survival (OS) and progression-free survival (PFS) was evaluated.
Results
According to the cut-off points, patients were divided into two groups. The patients in the high IPI group showed poorer OS compared to patients in the low IPI groups. The PFS was better in patients with low neutrophil-lymphocyte ratio (NLR) and platelet-neutrophil to lymphocyte ratio (PNLR), and the OS was better in patients with low IPI.
Conclusion
Among the immune inflammation scores analyzed in mCRC patients receiving regorafenib, NLR and PNLR were the best predictor of recurrence, whereas IPI was the best predictor of long-term survival. After being confirmed by better designed controlled trials, IPI can be used to identify the group of patients who will benefit more from regorafenib treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer is the third most common cancer in men and the second most common cancer in women worldwide. [1] By the introduction of targeted therapies, substantial progress in the management metastatic colorectal cancer (mCRC) has been obtained. Regorafenib is an oral multikinase inhibitor that inhibits the activity of several protein kinases, including kinases associated with tumour angiogenesis, oncogenesis, and the tumour microenvironment. [2] It is approved as monotherapy for the treatment of mCRC in patients who have failed fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy, anti-VEGF therapy and, if applicable, anti-EGFR therapy. Anti-angiogenic drugs are known to have effects not only on endothelial and tumour cells, but also on immune cells. [3] As the relationship between inflammation and tumour growth is well known, the potentially predictive role of inflammatory biomarkers during the use of these drugs is under investigation. [4]
Tumour-related inflammation could be determined by the levels of serum leukocytes, neutrophils, lymphocytes, platelets and acute phase proteins such as C-reactive protein (CRP) and albumin. [5] Recently, the combinations of these systemic inflammation parameters were reported as prognostic factors in some malignant solid tumours. [6] Indexes including hematological parameters neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) have been the subject of extensive research in the literature. Besides, Mercier et al. found significant different prognoses associated with elevated platelet-neutrophil to lymphocyte ratio (PNLR) in metastatic colorectal patients. [7] On the other hand, a new inflammatory prognostic index (IPI) consisting of serum CRP, NLR and serum albumin has been defined for lung cancer. High IPI values were associated with poor survival in patients with lung cancer. [8] Based on this data, we planned to evaluate IPI in mCRC patients using regorafenib. In addition, we aimed to analyze the difference in predictive power of IPI and other inflammatory markers, NLR and PNLR, in the same patient group.
Considering the cost and side effects of targeted therapies, it is important to identify the group of patients who will benefit more from these treatments. There are currently no validated biomarkers used to predict response to regorafenib in daily practice. We performed a retrospective analysis to identify the predictive role of a blood-based inflammation tool in mCRC patients treated with regorafenib.
Materials and Methods
A multicentric retrospective analysis of 65 metastatic CRC patients treated with regorafenib between 2015 and 2020 was performed. Patient demographics, Eastern Cooperative Oncology Group (ECOG) performance score, tumour location, metastatic sites, RAS mutation status, neutrophil, lymphocyte, albumin and CRP levels at the time of metastasis were recorded. NLR was calculated by dividing absolute neutrophil count by absolute lymphocyte count. IPI was calculated by the following formula: CRP × NLR / serum albumin. PNLR is calculated by platelet × neutrophil / lymphocyte × 1/1000.
The association between inflammatory markers and overall survival (OS) and progression-free survival (PFS) was evaluated. OS was calculated from the diagnosis of the patient to the date of death from any cause. PFS was calculated using the time interval between the onset of treatment and the date of the first visit in which progression was detected. Categorical variables were compared using chi-square or Fisher’s exact test with odds ratio (OR) and corresponding 95% confidence interval (CI). Receiver operating characteristic (ROC) curve analysis was used to determine the cut-off value for NLR, PNLR and IPI. According to cut-off points, patients were divided into two groups. These groups were compared in terms of OS and PFS rates. Statistical analyses were performed using SPSS 22.0 software (SPSS Inc. Chicago, IL). All statistical assessments were two-sided and a p value of 0.05 was considered statistically significant.
Results
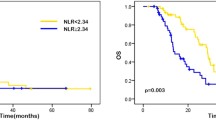
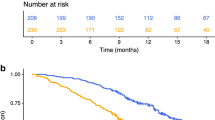
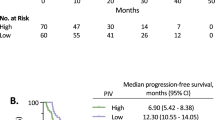
Table 1 presents the characteristics of 65 patients with mCRC. Median OS was 25 months. (%95 CI 21.7–36.3). Median PFS of the patients was 12 months. Using the ROC curve analysis, the optimal cut-off points of NLR, PNLR and IPI for the stratification of OS was found to be 5.87, 1277 and 5.97, respectively. According to the cut-off points, patients were divided into two groups. These groups were compared in terms of OS and PFS rates. Patients with NLR ≤ 5.87 had a PFS of 16 months, while those with a NLR > 5.87 had a PFS of 4 months (p: 0.004) (Fig. 1). Patients with PNLR < 1277 had a PFS of 16 months, while those with a PNLR ≥ 1277 had a PFS of 3 months (p:0.0007) (Fig. 2).
When the patients with high PNLR and high NLR scores were evaluated in the same group, the difference in PFS was found to be statistically significant compared to the patients with low scores (p:0.012) (Fig. 3).
As for the relationship of CRP with PFS, the cut-off value was taken as 5 because it is the upper limit of the CRP reference range in the laboratory. There were 29 (37.9%) patients with a CRP value below 5, and 36 (55.6%) patients with a CRP value of 5 and above. There was no statistically significant relationship between CRP and PFS (p: 0.471).
In univariate analysis, age, sex, ECOG performance status and the presence of liver metastasis were not associated with PFS. However, patients with lung and brain metastases had a worse PFS duration than those with metastases in other areas (p: 0.019 and p: 0.02, respectively). Compared to patients with metastases in a single site, patients with metastases in 2 or more sites had a shorter PFS duration (p: 0.024). Tumour location and RAS mutation status did not differ significantly between the high and low NLR, PNLR and IPI groups.
The patients in the high IPI group showed poorer OS compared to patients in the low IPI groups (Fig. 4).
The PFS were better in patients with low NLR, PNLR and the OS were better in patients with low IPI.
Discussion
Due to the different toxicity profile of a new compound such as regorafenib and lack of defined markers for therapeutic efficacy, it requires a good clinical evaluation before treatment. Undeniable number of patients may not provide any benefit from this treatment, and on top of that, they may be exposed to undeadly toxicity. Based on this rationale, we conducted a detailed evaluation of peripheral blood-based inflammatory markers in the absence of well-defined biomarkers predicting the clinical benefit of regorafenib therapy in mCRC.
The interactions between systemic inflammation and tumour progression alter the disease course for solid tumours. [9] Survival of tumour cells depends on signals generated from the tumour microenvironment, which is mainly composed of neutrophils and lymphocytes. [10] NLR is an indicator of inflammation and is a marker with prognostic value in cancer patients. [11] Lymphocytopenia is the component that raises the NLR and may indicate the extent of lymphocytic response to the tumour. [12] The low level of lymphocytes in the blood may reflect a silent or incomplete lymphocyte response to the tumour. [13] Lymphocytopenia is shown as an adverse prognostic factor in mCRC. [14] Furthermore according to the study of Del Prete et al., pre-treatment high NLR, high platelet and neutrophil count and low lymphocyte count are significantly associated with worse clinical outcome in mCRC patients treated with regorafenib. [15] Similarly, in our study, patients with NLR ≤ 5.87 had a PFS of 16 months, while those with a NLR > 5.87 had a PFS of 4 months (p: 0.004).
The prognostic role of NLR has also been prospectively studied in patients with mCRC receiving bevacizumab, another antiangiogenic agent such as regorafenib. The patients in this study were those who received bevacizumab as first- and second-line therapy for the metastatic disease. Results from this study confirmed the trend of worse PFS and OS with higher NLR. [16] By interpreting all these data together, NLR is considered as a potentially prognostic and predictive factor in mCRC patients treated with regorafenib.
Platelets play a role in tumour proliferation and invasion. [17] Considering that thrombocytosis, neutrophilia and lymphocytopenia may be associated with a worse prognosis, it has been hypothesized that PNLR could provide more reliable prognostic information than currently available prognostic markers. In the study of Mercier et al., PNLR with a cut-off value of 2000 was found to be a strong prognostic marker in patients with metastatic colorectal cancer. Patients with a PNLR above 2000 had a shorter median PFS (6.5 vs. 13.3 months, p: 0.001) than in patients with a PNLR below the threshold. In our study, patients with PNLR < 1277 had a PFS of 16 months, while those with a PNLR ≥ 1277 had a PFS of 3 months (p: 0.007). Accordingly, PNLR seems to be a marker compatible with the literature in terms of PFS in patients receiving regorafenib.
It is known that systemic inflammatory response is an important marker that predicts cancer prognosis. Low serum albumin is an important indicator of systemic inflammation as well as nutritional status. Hypoalbuminemia has been shown to be associated with poor prognosis in various cancers. [18] High level of IPI due to high serum CRP and neutrophil levels accompanied by low lymphocyte and serum albumin indicates a stronger inflammatory state. According to our findings, the patients in the high IPI group showed poorer OS compared to patients in the low IPI groups. These data suggest that patients under regorafenib therapy have worse survival if their inflammation burden is high. Since regorafenib is used in later lines of treatment, the inflammatory response may be increased due to the poor performance score and higher tumour burden of the patients.
Among the immune inflammation scores analyzed in our study, NLR and PNLR were the best predictor of recurrence, whereas IPI was the best predictor of long-term survival.
In the exploratory analysis of phase 3 CORRECT study evaluating the efficacy of regorafenib, it was stated that circulating DNA levels and plasma levels of IL-8 could predict the clinical results of regorafenib. [19] Also, in CONCUR, another phase 3 study of regorafenib, many other proteins, such as angiopoietin-2, VEGF-A and IL-8, were found to be associated with poor PFS. [20] However, these proteins are not easily available and cost effective for use in daily clinical practice. At this stage, the IPI score we evaluated in our study comes to the fore as a tool that can be easily calculated and evaluated quickly with the values obtained from routine blood tests. Among the other advantages of IPI include the low cost, high prognostic sensitivity, which could make this new marker a practical, inexpensive and reliable tool.
In our study, we evaluated the effect of IPI score for the first time in patients who received regorafenib in addition to previously studied NLR and PNLR in mCRC patients. The present study has some limitations due to the small number of patients and retrospective nature of the data. These results may not be generalizable, as they are derived from only three academic tertiary-care centres. Regardless of these limitations, our findings are clinically meaningful. These clinical tools, once confirmed by better designed controlled trials, may play a role in determining treatment and providing prognostic opinion for patients taking regorafenib.
Conclusion
We think that inflammatory scores can be used in order not to expose patients for unnecessary toxicity, not to initiate treatments that do not provide survival benefit, and not to create financial toxicity. This study suggests that IPI may be useful for evaluating survival in patients with mCRC and for determining the choice of later line treatments. Molecular mechanism of excessive systemic inflammatory response remains unclear, future studies with a prospective design are needed to validate our results.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Wilhelm SM, Dumas J, Adnane L et al. Regorafenib (BAY 73–4506): a new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int J Cancer. 2011;129(1):245–255.
Diakos CI, Charles KA, McMillan DC. SJ Clarke Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15:e493–503.
Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454 (7203): 436e444.
Lamb GW, McArdle PA, Ramsey S, McNichol AM, Edwards J, Aitchison M, McMillan DC. The relationship between the local and systemic inflammatory responses and survival in patients undergoing resection for localized renal cancer. BJU Int. 2008;102(6):756–61.
Chen JH, Zhai ET, Yuan YJ, Wu KM, Xu JB, Peng JJ, Chen CQ, He YL, Cai SR. Systemic immune-inflammation index for predicting prognosis of colorectal cancer. World J Gastroenterol. 2017;23(34):6261–72.
Mercier J, Voutsadakis IA. The platelets-neutrophils to lymphocytes ratio: a new prognostic marker in metastatic colorectal cancer. J Gastrointest Oncol. 2018;9(3):478–86.
Dirican N, Dirican A, Anar C, et al. A new inflammatory prognostic index, based on C-reactive protein, the neutrophil to lymphocyte ratio and serum albumin is useful for predicting prognosis in non-small cell lung cancer cases. Asian Pac J Cancer Prev. 2016;17:5101–6.
Ohki S, Shibata M, Gonda K, et al. Circulating myeloid-derived suppressor cells are increased and correlate to immune suppression, inflammation and hypoproteinemia in patients with cancer. Oncol Rep. 2012;28:453–8.
Carreau NA, Diefenbach CS. Immune targeting of the microenvironment in classical Hodgkin’s lymphoma: insights for the hematologist. Ther Adv Hematol. 2019;10:2040620719846451.
Pine JK, Morris E, Hutchins GG, West NP, Jayne DG, Quirke P, Prasad KR. Systemic neutrophil-to-lymphocyte ratio in colorectal cancer: the relationship to patient survival, tumour biology and local lymphocytic response to tumour. Br J Cancer. 2015;113(2):204–11.
Quigley DA, Kristensen V. Predicting prognosis and therapeutic response from interactions between lymphocytes and tumor cells. Mol Oncol. 2015;9:2054–62.
Zippelius A, Batard P, Rubio-Godoy V, Bioley G, Lienard D, Lejeune F, Rimoldi D, Guillaume P, Meidenbauer N, Mackensen A, Rufer N, Lubenow N, Speiser D, Cerottini JC, Romero P, Pittet MJ. Effector function of human tumor-specific CD8 T cells in melanoma lesions: a state of local functional tolerance. Cancer Res. 2004;64:2865–73.
Yoneyama Y, Ito M, Sugitou M, Kobayashi A, Nishizawa Y, Saito N. Postoperative lymphocyte percentage influences the long-term disease-free survival following a resection for colorectal carcinoma. Jpn J Clin Oncol. 2011;41(3):343e347.
Del Prete M, Giampieri R, Loupakis F, Prochilo T, Salvatore L, Faloppi L, Bianconi M, Bittoni A, Aprile G, Zaniboni A, Falcone A, Scartozzi M, Cascinu S. Prognostic clinical factors in pretreated colorectal cancer patients receiving regorafenib: implications for clinical management. Oncotarget. 2015;6(32):33982–92.
Clarke SJ, Burge M, Feeney K, et al. The prognostic role of inflammatory markers in patients with metastatic colorectal cancer treated with bevacizumab: a translational study [ASCENT]. PLoS One. 2020;15(3):e0229900.
Jain S, Harris J, Ware J. Platelets: linking hemostasis and cancer. Arterioscler Thromb Vasc Biol. 2010;30(12):2362–7.
Sun H, Hu P, Shen H, et al. Albumin and neutrophil combined prognostic grade as a new prognostic factor in non-small cell lung cancer: results from a large consecutive cohort. PLoS One. 2015;10:e0144663.
Tabernero J, Lenz HJ, Siena S, et al. Analysis of circulating DNA and protein biomarkers to predict the clinical activity of regorafenib and assess prognosis in patients with metastatic colorectal cancer: a retrospective, exploratory analysis of the CORRECT trial. Lancet Oncol. 2015;16(8):937–48.
Teufel M, Kalmus J, Rutstein MD, et al. Analysis of plasma protein biomarkers from the phase 3 CONCUR study of regorafenib in Asian patients with metastatic colorectal cancer (mCRC) [abstract no. 672]. J Clin Oncol. 2016;34(4 Suppl).
Acknowledgements
The authors would like to thank Prof. Dr. Gamze Göksel for providing guidance and feedback throughout this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Approval was obtained from the Medical Research Ethics Committee of the Manisa Celal Bayar University Faculty of Medicine with the number 80 dated 15.06.2020.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Erdoğan, A.P., Ekinci, F., Karabaş, A. et al. Could the Inflammatory Prognostic Index Predict the Efficacy of Regorafenib in Patients with Metastatic Colorectal Cancer?. J Gastrointest Canc 53, 45–51 (2022). https://doi.org/10.1007/s12029-021-00642-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-021-00642-w