Abstract
Purpose
Bevacizumab is a standard first-line (L1) treatment for metastatic colorectal cancer (mCRC) patients regardless of RAS status. This retrospective study examined treatment patterns and outcomes in a community oncology sample of KRAS mutant mCRC patients treated with chemotherapy (C) or C plus bevacizumab (CB) in L1.
Methods
This study used medical records from the Vector Oncology Data Warehouse. Eligible patients were confirmed KRAS mutant mCRC and received L1 C or CB. Kaplan-Meier analysis assessed L1 progression-free survival (PFS) and overall survival (OS). Cox regression models examined the interaction of tumor location (R/L) with treatment.
Results
CB (n = 264) compared to C (n = 109) patients were younger, less likely performance status (PS) impaired, and more likely with liver metastases. Median unadjusted PFS was 10.41 months (95% CI 9.0–11.3) in CB and 7.66 months (95% CI 6.5–9.1) in C patients (p = 0.174). Median unadjusted OS was 26.91 months (95% CI 24.3–29.3) in CB and 23.33 months (95% CI 19.7–29.2) in C patients (p = 0.571). For patients with right- vs. left-sided tumors, C (but not CB)-treated patients had higher adjusted risk for progression (HR = 1.715, 95% CI 1.108, 2.653; p = 0.015).
Conclusions
CB- vs. C-treated KRAS mutant mCRC patients may have a meaningful PFS benefit. Patients with right-sided tumors treated with C were at higher risk for disease progression than patients with left-sided tumors. Tumor location had no significant effect on outcomes in the CB cohort.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer death in the USA. In 2017, it is projected that 135,430 people will be diagnosed with CRC and 50,270 people will die from the disease [1]. About 20% of newly diagnosed patients present with metastatic disease, and among those, about 80–90% of patients’ tumors are unresectable [2].
The current approach to treating metastatic CRC (mCRC) includes doublet combinations of fluorouracil, leucovorin, and irinotecan (FOLFIRI); infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX); and capecitabine plus oxaliplatin (XELOX), as well as the triplet combination fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) [3,4,5]. The introduction of targeted biologic agents (cetuximab, panitumumab [epidermal growth factor receptor (EGFR) inhibitors], bevacizumab, ziv-aflibercept, ramucirumab [vascular endothelial growth factor (VEGF) inhibitors], regorafenib [multiple kinase inhibitor]) into clinical practice has improved response rates and overall survival (OS) [6,7,8,9,10,11].
Bevacizumab, a VEGF inhibitor, has demonstrated OS benefit in first-line and second-line mCRC [12], and retrospective analyses have shown that the clinical benefit of bevacizumab in mCRC is independent of KRAS mutation status [13]. In contrast, benefits of anti EGFR are limited to KRAS wild-type (WT) patients, and patients with KRAS mutations should not receive treatment with EGFR inhibitors [14,15,16].
This study was a retrospective, descriptive study of treatment patterns and outcomes among KRAS mutant mCRC patients who received chemotherapy alone (C) or chemotherapy plus bevacizumab (CB) as part of first-line treatment for metastatic disease in the community oncology setting. We also examined tumor location (right side vs. left side) as a predictor of outcomes. Previous investigation indicated that among KRAS wild-type patients, those treated with bevacizumab-containing regimens did not show significantly different outcomes by tumor side, and we sought to examine tumor sidedness comparably in a sample of KRAS mutant patients [17].
Patients and Methods
Study Design
This was a retrospective, observational study using data from the Vector Oncology Data Warehouse, a repository of electronic medical record (EMR) data, billing data, and health care provider notes for cancer patients. The provider notes supported the collection of key information not otherwise available in structured data fields (e.g., performance status, confirmation of and dates for disease progressions, tumor location) through review by experienced Clinical Research Nurses. The protocol for this study received institutional review board approval from IntegReview (Austin, TX).
Patients
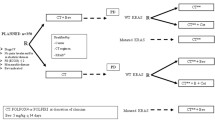
Patients were included if the following criteria were met: metastatic/stage IV CRC first diagnosed prior to 03/01/2014, documentation of KRAS testing with affirmation of mutant-type status at any point, received C or CB in the first-line metastatic setting, and at least 18 years old at metastatic diagnosis. Altogether, records from 3825 patients were examined for potential eligibility, and a final sample of 374 was accrued for this study. Of the potential sample, 2973 (77.7%) were screened out for not meeting the KRAS mutant confirmed criterion.
Study Endpoints and Assessments
The primary endpoints were progression-free survival (PFS) and overall survival (OS). PFS was defined as the interval from the start of a line of therapy until occurrence of disease progression, death, or end of the medical record, whichever occurred first. Dates of all disease progressions after diagnosis of mCRC were directly determined from radiological scan notes and oncologist progress notes. OS was defined as the interval from the start of first-line therapy until death. Dates of death were determined from the medical record and Social Security Death Index.
Treatment patterns included classification of patients to a first-line therapy group (C or CB) and included description of oxaliplatin- and irinotecan-based chemotherapy backbones. Lines of treatment were defined by occurrence of a documented disease progressions rather than mere changes in treatment regimen as some patients may have changed therapy before a disease progression. Eligibility for the study and classification to group (C or CB) was based on first-line treatment regimen.
Other study variables collected as part of this investigation included patient demographic (age, race, sex) and clinical (performance status, number of metastatic sites, side of primary tumor location, prior adjuvant therapy status, primary tumor resection status, and lines of therapy after first line) characteristics.
Statistical Analysis
Fisher’s exact test or Chi-square, and t test or Wilcoxon rank sum test were used to compare patient characteristics by treatment groups on categorical or continuous variables, respectively. Unadjusted PFS and OS were calculated using Kaplan-Meier estimator and compared across groups with a log rank test. PFS and OS were assessed from the start of line 1, and the primary comparison groups were C and CB regimen groups. We also conducted subset analyses of PFS and OS within each regimen group where patients were divided into those with left- vs. right-sided tumors. Multivariate Cox regression models with covariates (age, gender, race [minority vs. white]), body mass index (BMI), performance status [impaired vs. unimpaired], liver metastasis [present vs. absent], lung metastasis [present vs. absent], stage at diagnosis [I/II and III vs. IV], chemotherapy backbone [irinotecan, capecitabine/5FU monotherapy, and other vs. oxaliplatin] were also used to examine PFS and OS.
Results
Patient Characteristics
Table 1 shows the demographic and clinical characteristics of patients at the time of metastatic diagnosis and start of first-line treatment by first-line therapy group (C, n = 109 and CB, n = 264) and overall. On average, patients in the C group were older (62.7 years) than patients in the CB group (59.1 years), p = 0.0108. Otherwise, the two patient groups were not significantly different in basic demographic characteristics. A higher percentage of C patients (11.0%) were performance status impaired on a composite measure of performance status as compared to CB patients (4.5%), p = 0.0207. A higher percentage of CB patients (76.1%) had liver metastases as compared to C patients (65.1%), p = 0.0297. A higher percentage of CB patients had no history of liver resection (65.2%) compared to that of C patients (51.4%), p = 0.0418. There were no other significant differences in clinical characteristics between the two patient groups.
Treatment Patterns
Among the 109 C patients, the most common single regimen was FOLFOX, which comprised 43.1% (47/109) of that group, and the second most common was capecitabine monotherapy, which comprised 18.3% (20/109) of that group. The most common CB regimen was FOLFOX + Bev which comprised 61.7% (n = 163/264) of this group, and the second most common regimen was FOLFIRI + Bev which comprised 16.7% (n = 44/264) of this group.
PFS and OS in Line 1
Figure 1 shows the Kaplan-Meier plot of PFS from the start of line 1 by first-line therapy group. Median unadjusted PFS was 7.66 months (95% CI 6.5, 9.1) for C-treated patients and 10.41 months (95% CI 9.0, 11.3) for CB-treated patients. In multivariate survival analysis, the improvement in PFS among patients receiving CB compared to patients receiving C (HR = 0.817, 95% CI 0.627, 1.063) was not statistically significant. In this Cox model of PFS, the presence (vs. absence) of liver metastases (HR = 1.393, 95% CI 1.062, 1.828) and lung metastases (HR = 1.289, 95% CI 1.020, 1.628) were identified as significant predictors of risk for disease progression, all ps < 0.033.
Figure 2 shows Kaplan-Meier plot of OS from the start of line 1, by first-line treatment group. Median unadjusted OS was 23.33 months (95% CI 19.7, 29.2) for C-treated patients and 26.91 months (95% CI 24.3, 29.3) for CB-treated patients. In multivariate survival analysis, the improvement in OS among patients receiving CB compared to patients receiving C (HR = 0.879, 95% CI 0.658, 1.174) was not statistically significant. As in the analyses for PFS, the presence of liver metastases (HR = 1.391, 95% CI 1.032, 1.876) and impaired performance status (HR = 1.807, 95% CI 1.103, 2.962) were the only significant predictors of mortality, all ps < 0.030.
PFS and OS by Tumor Location
We conducted subset analyses for each treatment group to examine effects of tumor location (right vs. left, and unknown vs. left). For the CB group, neither Kaplan-Meier nor Cox regression analyses showed a significant effect for tumor location in predicting first-line PFS. For the C group, there was a significant effect in the multivariate model (though not in Kaplan-Meier) for tumor location such that right- vs. left-sided tumor location was associated with significant and meaningful increased risk (72% increase) for disease progression (HR = 1.715, 95% CI 1.108, 2.653), p = 0.015. The same subset analyses were conducted for OS from the start of first-line treatment, and there were no significant effects for tumor location.
Discussion
One aim of this study was to provide a retrospective description of patients and treatment patterns in a sample of community oncology mCRC patients who had a confirmed status of being KRAS mutant and who received first-line treatment with either CB or C. In terms of patient characteristics, the C patients were on average about 3 years older than the CB patients, and proportionately, more of the C patients were performance status impaired (11 vs. 5%). On the other hand, more of the CB patients had liver metastases (76 vs. 65%), but otherwise, the C- and CB-treated patients were generally comparable. As might be expected in a US sample, for both groups of patients, FOLFOX-based chemotherapy regimens were more common than other chemotherapy backbones [4, 18].
The second aim was to assess treatment outcomes. The comparison of outcomes for CB- vs. C-treated patients suggests that there might be clinically meaningful benefit in median PFS for the CB-treated patients (2.75 months), but that difference was not statistically significant in this study. The small sample size likely contributed to the lack of statistical significance. This interpretation of the present study is supported by evidence from two other investigations. First, in a phase III placebo-controlled trial of FOLFOX or XELOX plus bevacizumab, the median PFS difference between CB and C was 1.4 months, which was statistically significant with a large sample of 1401 patients [19]. Second, the point estimate for median PFS in the CB group from the present study (10.41 months [95% CI 9.0, 11.3]) was similar to that reported in a similar group of KRAS mutant mCRC patients (n = 658) treated with CB regimens from a European registry study (11.4 months) [20].
We found that CB-treated patients did not show a difference in risk for disease progression as a function of tumor sidedness. However, for C-treated patients, those with right-sided tumors had a significantly higher risk for disease progression as indicated by multivariate survival analyses. Other investigations of tumor sidedness have also suggested that patients with right-sided tumors may be at greater risk for disease progression and have worse outcomes compared to patients with left-sided tumors, but those previous observations were confined to patients with KRAS wild-type tumors and did not include patients with KRAS mutant tumors [17, 21]. Results showing less favorable outcomes for right-sided tumors in the KRAS wild-type population were most pronounced in patients treated with chemotherapy plus cetuximab. Our findings in a sample of KRAS mutant mCRC patients treated with chemotherapy without a biologic agent suggest that tumor sidedness may be a general predictor of worse outcomes. The fact that CB-treated patients did not show poorer outcomes associated with right- vs. left-sided tumor may be hypothesis generating. The addition of B to C may confer some survival benefit to patients with right-sided tumors even among those patients who are not KRAS wild type.
This study has some limitations. Sample size for the C group was small because many more patients were available who had treatment with CB. Bevacizumab approval for mCRC was first issued in February 2004 and has become widely used alongside standard chemotherapy backbones. It is noteworthy that our sample of C patients was more likely to be older and have some performance status impairment, factors that may well have led to the decision to treat without adding bevacizumab. It is also possible that these “selection” differences accounted for the apparent efficacy advantages of adding B to C.
Conclusion
This study provides additional real-world evidence that the addition of bevacizumab to standard chemotherapy backbones in KRAS mutant mCRC patients may add clinical benefit compared to standard chemotherapy without bevacizumab. Further research with larger sample sizes is required to confirm the findings of this exploratory study.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017; https://doi.org/10.3322/caac.21387.
Hu CY, Bailey CE, You YN, Skibber JM, Rodriguez-Bigas MA, Feig BW, et al. Time trend analysis of primary tumor resection for stage IV colorectal cancer: less surgery, improved survival. JAMA Surg. 2015;150(3):245–51. https://doi.org/10.1001/jamasurg.2014.2253.
de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18(16):2938–47.
Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, et al. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol. 2004;22(1):23–30. https://doi.org/10.1200/JCO.2004.09.046.
Falcone A, Ricci S, Brunetti I, Pfanner E, Allegrini G, Barbara C, et al. Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol. 2007;25(13):1670–6. https://doi.org/10.1200/JCO.2006.09.0928.
Starling N, Tilden D, White J, Cunningham D. Cost-effectiveness analysis of cetuximab/irinotecan vs active/best supportive care for the treatment of metastatic colorectal cancer patients who have failed previous chemotherapy treatment. Br J Cancer. 2007;96(2):206–12.
Capdevila J, Ramos FJ, Macarulla T, Elez E, Tabernero J. The role of salvage treatment in advanced colorectal cancer. Crit Rev Oncol Hematol. 2009;71(1):53–61.
Kelly H, Goldberg RM. Systemic therapy for metastatic colorectal cancer: current options, current evidence. J Clin Oncol. 2005;23(20):4553–60. https://doi.org/10.1200/JCO.2005.17.749.
Tabernero J, van Cutsem E, Lakomy R, Prausova J, Ruff P, van Hazel G, et al. Results from VELOUR, a phase III study of aflibercept versus placebo in combination with FOLFIRI for the treatment of patients with previously treated metastatic colorectal cancer Abstract 6LBA. Paper presented at the ECCO-ESMO 2011, Stockholm Sweeden,
Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381(9863):303–12. https://doi.org/10.1016/S0140-6736(12)61900-X.
Zaniboni A. New active drugs for the treatment of advanced colorectal cancer. World J Gastrointest Surg. 2015;7(12):356–9. https://doi.org/10.4240/wjgs.v7.i12.356.
Bennouna J, Sastre J, Arnold D, Osterlund P, Greil R, Van Cutsem E, et al. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol. 2013;14(1):29–37. https://doi.org/10.1016/S1470-2045(12)70477-1.
Kubicka S, Greil R, Andre T, Bennouna J, Sastre J, Van Cutsem E, et al. Bevacizumab plus chemotherapy continued beyond first progression in patients with metastatic colorectal cancer previously treated with bevacizumab plus chemotherapy: ML18147 study KRAS subgroup findings. Ann Oncol. 2013;24(9):2342–9. https://doi.org/10.1093/annonc/mdt231.
The National Comprehensive Cancer Network (NCCN) (2016) Clinical Practice Guidelines in Oncology: Colon Cancer, Version 2.2016. The National Comprehensive Cancer Network. http://www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed 22 Feb 2016.
Amgen (2015) Drug Label for Panitumumab. Amgen, Inc. http://pi.amgen.com/united_states/vectibix/vectibix_pi.pdf. Accessed 11 May 2015.
Bristol-Myers Squibb and Lilly (2015) Cetuximab Drug Label. Bristol-Myers Squibb and Lilly. http://packageinserts.bms.com/pi/pi_erbitux.pdf. Accessed 11 May 2015.
Venook AP, Niedzwiecki D, Innocenti F, Fruth B, Greene C, O’Neil BH, et al. (2016) Impact of primary (1º) tumor location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): analysis of CALGB/SWOG 80405 (Alliance). ASCO Meeting Abstracts 34 (15_suppl):3504.
Hallek M, Fischer K, Fingerle-Rowson G, Fink AM, Busch R, Mayer J, et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet. 2010;376(9747):1164–74. https://doi.org/10.1016/S0140-6736(10)61381-5.
Saltz LB, Clarke S, Diaz-Rubio E, Scheithauer W, Figer A, Wong R, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008;26(12):2013–9. https://doi.org/10.1200/JCO.2007.14.9930.
Bencsikova B, Bortlicek Z, Halamkova J, Ostrizkova L, Kiss I, Melichar B, et al. Efficacy of bevacizumab and chemotherapy in the first-line treatment of metastatic colorectal cancer: broadening KRAS-focused clinical view. BMC Gastroenterol. 2015;15:37. https://doi.org/10.1186/s12876-015-0266-6.
von Einem JC, Heinemann V, von Weikersthal LF, Vehling-Kaiser U, Stauch M, Hass HG, et al. Left-sided primary tumors are associated with favorable prognosis in patients with KRAS codon 12/13 wild-type metastatic colorectal cancer treated with cetuximab plus chemotherapy: an analysis of the AIO KRK-0104 trial. J Cancer Res Clin Oncol. 2014;140(9):1607–14. https://doi.org/10.1007/s00432-014-1678-3.
Funding
This work was funded by Genentech, Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
SO and NS are employed by and own stock in Genentech, Inc. SSH and MW are consultants for Genentech. JH is on an advisory board for Genentech, with her institution receiving her honorarium. In addition, JH’s institution has received research support from Genentech. YZ has received travel support from Genentech. AH reported no conflicts of interest. Genentech sponsored this study and provided financial support for the conduct of the research and for preparation of the article. Genentech collaborated on the design of the study, interpretation of the analyses, and in the decision to submit the article for publication, but did not have a direct role in data collection, data analysis, or writing of the report.
Research Involving Human Participants
This research was reviewed and approved by IntegReview Institutional Review Board, in Austin, Texas. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
For this type of study, formal consent is not required.
Availability of Data and Material
Vector Oncology does not make datasets publicly available because study data are used under license from source practices. Vector Oncology will consider requests to access study datasets on a case-by-case basis.
Rights and permissions
About this article
Cite this article
Houts, A.C., Ogale, S., Zafar, Y. et al. Progression-Free Survival in Patients Receiving Chemotherapy Alone (C) or Chemotherapy with Bevacizumab (CB) for First-Line Treatment of KRAS Mutant Metastatic Colorectal Cancer in Community Oncology Settings. J Gastrointest Canc 50, 16–22 (2019). https://doi.org/10.1007/s12029-017-0017-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-017-0017-8