Abstract
Introduction
Acute neurological injury and several medications commonly administered in the Neuro ICU pose a risk of fatal cardiac dysrhythmias. The objective of this study is to identify the predictors of ventricular dysrhythmias in the Neuro ICU patients with prolonged QTc, thereby helping the clinicians make important treatment decisions.
Methods
We performed a retrospective review of all consecutive adults admitted to the Neuro ICU from January 2015 to September 2015 with a QTc interval ≥ 450 ms on electrocardiogram.
Results
A total of 170 patients with a mean age of 66 years (SD ± 16) were included in the final analysis. Eighty-seven patients (51%) were women. Median duration of hospitalization was 9 days (IQR 4–16). Most common primary diagnosis was ischemic stroke (38%) followed by cerebral hemorrhage (19%) and subarachnoid hemorrhage (8%). Mean QTc was 487 ms (SD ± 35, range 450–659 ms). There were 27 episodes (16%) of monomorphic non-sustained ventricular tachycardia and one episode of Torsades (1%). Three cardiac arrests (2%) were recorded, none resulting from ventricular dysrhythmias. In multivariate analysis, prolonged QTc ≥ 492 ms (p = 0.0008), supratentorial acute ischemic stroke (p = 0.005), prolonged hospitalization (p = 0.03), and premature ventricular complexes on ECG (p = 0.047) were all independently associated with increased risk of ventricular dysrhythmias.
Conclusions
In this group of patients with prolonged QTc in the Neuro ICU, we observed several episodes of non-sustained ventricular tachycardia and identified important risk factors associated with their occurrence. This knowledge is essential to inform clinical decisions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The QT interval on the electrocardiogram (ECG) represents ventricular depolarization and repolarization. QT interval is inversely proportional to the heart rate (RR interval) and is therefore often corrected for heart rate (QTc). Two commonly used methods are Bazett’s formula (using square root of the RR interval) or Fridericia formula (using cube root of the RR interval) [1, 2]. Delayed ventricular repolarization leads to QTc prolongation and is associated with ventricular dysrhythmias and sudden cardiac death [3, 4]. A form of polymorphic ventricular tachycardia known as Torsades de Pointes (TdP) is specifically associated with prolongation of QTc interval [1]. Prolonged QTc can be either congenital due to channelopathies (such as long QT syndrome [LQTS]) or acquired due to factors such as ischemic heart disease, cardiomyopathies, and electrolyte imbalances [5]. Several drugs commonly administered in a critical care setting such as psychotropic drugs, macrolides, and fluoroquinolones can cause QT prolongation by interfering with potassium efflux from cardiomyocytes [6,7,8,9]. In addition, acute neurological injury is known to cause a variety of cardiac rhythm abnormalities [10,11,12]. The risk of fatal cardiac dysrhythmias has never been studied in the Neurology Intensive Care Unit (Neuro ICU), the knowledge of which is important in routine clinical practice. The purpose of this study is to evaluate the factors increasing the risk of ventricular dysrhythmias (monomorphic and polymorphic ventricular tachycardia and ventricular fibrillation) in Neuro ICU patients with prolonged QTc interval.
Methods
This is a retrospective review of all consecutive adults (18 years of age or older) admitted to Wake Forest Baptist Medical Center Neuro ICU through the Emergency Department from January 2015 to September 2015. Patients who (1) had prolonged QTc on ECG defined as ≥ 450 ms, (2) were admitted from the emergency room, and (3) had QRS duration ≤ 120 ms were included in the study. We chose to include only patients admitted from the emergency room as they are more likely to be critically ill (and therefore at higher risk of ventricular dysrhythmias) than the already screened and selected neurosurgical patients admitted to the ICU following elective surgeries. At our center, all craniotomies and some spine surgeries (most of which are non-emergent scheduled cases) are routinely admitted to the Neuro ICU for close monitoring following surgery and are usually ready to be discharged the next day to the regular floor.
Bazett’s formula was used for QTc calculation. The definition of abnormal QTc is variable throughout the literature with some references suggesting a cutoff value > 450 ms for men and > 470 ms for women as prolonged and a value > 450 ms for women as borderline prolonged [13, 14]. We chose to have a single QTc cutoff for both genders as having different QTc thresholds of inclusion for men and women would have introduced a substantial gender bias for which we would then have to adjust the model and potentially weaken the study. Prolonged QRS duration can affect QTc interval and does not truly represent repolarization, abnormality especially when QRS duration exceeds 120 ms [15]. Consequently, patients with QRS duration > 120 ms were excluded. This study was approved by the Wake Forest University Health Sciences Institutional Review Board (IRB). Waiver of written informed consent was requested and approved by the IRB.
Information gathered included demographics, admission diagnosis, comorbidities, electrolyte abnormalities, renal failure, myocardial infarction, critical care interventions (mechanical ventilation, sepsis, vasopressor use), QTc-prolonging medications, and length of stay. At our center, we do not have a step-down unit and consequently have a low threshold to admit patients to the Neuro ICU. Patients with single-organ failure not needing mechanical ventilation (otherwise sometimes referred to as level 2 ICU) as well as multiple-organ dysfunction or single-organ dysfunction needing mechanical ventilation (otherwise sometimes referred to as level 3 ICU) are admitted to our unit. A laboratory result closest to the ECG used for QTc measurement or closest to the occurrence of the ventricular dysrhythmia (for patients who had one) was used for collecting electrolyte data. A list of QTc-prolonging drugs was generated using the database maintained by the Arizona Center for Education and Research on Therapeutics (www.azcert.org). Only drugs with a “known risk” and “possible risk” were included.
ECG data included heart rate, QRS interval, and presence of premature ventricular complexes (PVC). For patients who had multiple ECGs during the hospitalization, the ECG showing the highest QTc was used for patients without ventricular dysrhythmia event, and in patients who did have such an event, ECG showing the highest QTc obtained prior to the event was used when available and if not then ECG done after the event was reviewed. At our institution, all ECGs are signed off by board-certified cardiologists in the medical records of every patient. All Neuro ICU patients are continuously monitored with bedside telemetry, and any dysrhythmia noted by the monitor sounds an alarm. The bedside nurse/provider then interprets whether that is a false alarm or a true event. A strip if not automatically printed by then, is then printed and is subsequently scanned in the patient’s medical record. In our study, all episodes of ventricular dysrhythmias were identified based upon manual review of all the scanned bedside continuous monitor strips for every patient. Episodes of cardiac arrest were recorded in the electronic medical record. Poor outcome was defined as discharge to nursing home, hospice, or death. Ventricular tachycardia was defined as ≥ 3 consecutive beats arising below the atrioventricular node at a heart rate of > 100 beats/min. Ventricular tachycardia was considered to be non-sustained with duration < 30 s.
Statistical Analysis
All statistical analysis was done using SAS software JMP Pro version 13.0. Participant characteristics were described using descriptive statistics. Means and medians as appropriate with corresponding standard deviations and interquartile ranges, respectively, were used as measurements of central tendencies. Comparisons between groups were done using Chi-square tests for proportions and Wilcoxon rank sum test for nonparametric data. An univariate logistic regression model was used to estimate the receiver operating characteristics curves (ROC) and to identify a threshold QTc with an optimum combination of sensitivity and specificity for predicting the risk of cardiac dysrhythmias. All significant variables (p < 0.05) in univariate analysis were simultaneously included in an automated multivariate logistic regression model using JMP Pro 13.0, and only those retaining significance (p < 0.05) are reported with their respective odds ratios.
Results
One hundred and seventy consecutive patients with a mean age of 66 years (SD ± 16) were included in the final analysis (Fig. 1). Eighty-seven (51%) were women. Demographic and clinical characteristics of those with and without ventricular dysrhythmias are outlined in Table 1. The most common primary diagnosis was ischemic stroke (38%), followed by cerebral hemorrhage (19%), and subarachnoid hemorrhage (8%). Median duration of hospitalization was 9 days (IQR 4–16). Seventy-seven patients (45%) received two or more QTc-prolonging drugs simultaneously. Forty-three (25%) patients had heart failure, and 60 (35%) had coronary artery disease. Renal failure was present in 61 patients (36%), of which ten (6%) were on dialysis. Fifty-nine patients (35%) had sepsis, and seven (4%) were in septic shock. Sixty-three patients (37%) received mechanical ventilation during the hospitalization. Eighty-two patients (48%) had poor outcome. Electrolyte abnormalities involving low calcium, potassium, and magnesium were common (Table 1). Right-sided supratentorial strokes were associated with a greater likelihood of ventricular dysrhythmias.
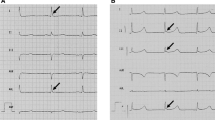
Mean QTc was 487 ms (SD ± 35, range 450–659 ms). Twenty-seven patients (16%) had non-sustained monomorphic ventricular tachycardia. There was one episode of non-sustained TdP. These ventricular dysrhythmias occurred after a median of 5 days (IQR 2 to 15) following admission. All but two ECGs showing prolonged QTc were obtained a median of 3 days (IQR 1 to 9) prior to the occurrence of the dysrhythmia, one was obtained 1 day after the event, and another was obtained 6 days after the event. Three cardiac arrests (2%) occurred, none resulting from ventricular dysrhythmia (two patients were in pulseless electrical activity and the third in asystole).
Using ROC table, we identified a threshold QTc of 492 with an optimum combination of sensitivity and specificity for predicting the risk of cardiac dysrhythmias. The patients were then dichotomized into two groups, one with QTc of < 492 ms and the other with QTc ≥ 492 ms. The following variables with p < 0.05 in univariate analysis (Table 1) were simultaneously included in an automated multivariate logistic regression model, and only those retaining significance (p < 0.05) are reported in Table 2 along with their respective odds ratios; supratentorial stroke, coronary artery disease, QTc ≥ 492 ms, low serum calcium, duration of hospitalization, PVCs on ECG, atrial fibrillation, heart failure, and acute myocardial infarction. Area under curve (AUC) for the model was 0.90 with a sensitivity of 93% and specificity of 75%. The model was adjusted by adding the following well-known confounders of QTc prolongation: age, sex, serum potassium level, and QT-prolonging medications. Full model has been included in the supplementary materials.
Next, we analyzed the data using poor outcome as the outcome variable and the following variables were significant in univariate analysis: primary diagnosis (p = 0.03), heart failure (p = 0.03), coronary artery disease (0.01), QTc ≥ 492 (p = 0.005), atrial fibrillation (p = 0.006), ventricular dysrhythmia (p = 0.007), renal failure (p = 0.006), mechanical ventilation (p = 0.002), sepsis (p = 0.0002), older age (median 75 vs 59 years, p < 0.0001), diabetes mellitus (p = 0.01), and longer duration of hospitalization (median 13 vs 7 days, p = 0.002). These variables were then simultaneously included in an automated multivariate logistic regression analysis, and only those retaining significance (p < 0.05) are reported in Table 3 along with their respective odds ratios. Full model has been included in the supplementary materials.
Discussion
In this group of selected patients at high risk of ventricular dysrhythmias in the Neuro ICU, several episodes of non-sustained monomorphic ventricular tachycardia were observed. The prevalence of TdP was found to be very low. There were three cardiac arrests although none were caused by ventricular dysrhythmias. In addition to QTc ≥ 492 ms, other risk factors for ventricular dysrhythmias were prolonged duration of hospitalization, supratentorial acute ischemic stroke, and PVCs on ECG. Older age, QTc ≥ 492 ms, sepsis, and mechanical ventilation were associated with poor outcome. We examined the effect of stroke laterality on the risk of ventricular dysrhythmias and found that right supratentorial lesions increased the risk significantly. Insular cortex lesions particularly on the right side have been previously reported to have a higher incidence of ECG abnormalities and prolonged QTc thought to be related to sympathetic over activation [16, 17].
Non-sustained ventricular tachycardia (NSVT) has been recorded in a wide range of conditions from apparently healthy individuals to patients with severe heart disease, both ischemic and non-ischemic [18]. In addition, certain inherited channelopathies such as LQTS and drug-associated QT prolongation can be present with ventricular tachycardia, which are usually polymorphic episodes [5]. It is generally accepted that QTc of > 500 ms is associated with high risk of dysrhythmias but these data are derived from long-term, follow-up studies of patients with LQTS in the outpatient setting [19, 20]. The physiology of critically ill patients in the ICU is different from clinic patients and therefore needs to be studied separately. In our study, we found a threshold QTc of 492 ms beyond which the risk of dysrhythmias increased significantly. To our best knowledge, this is the first study evaluating the predictors of cardiac dysrhythmias specifically in the Neuro ICU population with prolonged QTc. Acute neurological injury, underlying cardiovascular comorbidities, avoidance of sedation to preserve neurological examination, and instead the use of medications such as antipsychotics and dexmedetomidine, all place these patients at risk of cardiac dysrhythmias. Estimating that risk helps inform clinical decisions; however, the available data are limited and derived from medical, surgical, and cardiac ICUs. In a prospective observational single-center study by Reinelt et al., the authors examined the incidence of cardiac dysrhythmias in a group of 756 patients admitted to medical and cardiac ICU. There were five patients with TdP (< 1%) and 54 patients (7%) with monomorphic ventricular tachycardia [21]. Utility of continuous QT interval measurement in critically ill patients was examined by Pickham and colleagues in a single-center, prospective, observational study which shares several similarities with our study [22]. This study was conducted in six ICUs across the institution, and the authors concluded that QT prolongation is common in this patient population with a prevalence of 24% and is associated with increased length of hospitalization and mortality. There were 17 “code blue” events, 16 of which were cardiac. Only one episode of TdP (representing 6% of cardiac arrests) was evident. Whether prolonged QTc is simply a surrogate marker of the illness severity remains to be proven. Ischemic stroke particularly involving the right hemisphere increased the risk of ventricular dysrhythmias in our study, which aligns with findings previously reported in the literature. Ischemic stroke has been described to cause QTc prolongation and ventricular dysrhythmias, especially with lesions involving the right insular cortex [1, 4]. Patients with acute stroke often share several of the common comorbidities associated with heart disease such as hypertension, diabetes, hypercholesterolemia, coronary artery disease, and need for QTc-prolonging medications, among others, that increase the risk of malignant cardiac dysrhythmias. Catecholamine surge occurring as a result of acute cerebral injury can lead to cardiomyocyte necrosis and stress cardiomyopathy. Finally, genetic and epigenetic factors might modify the individual phenotype of stroke–heart syndrome [23].
In spite of having 26 episodes of ventricular tachycardia and one episode of TdP, there were no cardiac arrests due to ventricular dysrhythmias. One likely explanation for this observation is the sample size. It is likely that with a bigger sample size, some of these ventricular dysrhythmias may lead to cardiac arrest; however, based upon our results, the risk appears to be low. The strength of this study lies in the identification of a “threshold QTc” and factors increasing the chance of ventricular dysrhythmias in the Neuro ICU population, which may help the clinician identify patients at the highest risk and remain extra vigilant during their care.
Our study has some limitations. All of our patients were treated at a single tertiary care referral center. This and the retrospective design could potentially lead to a selection bias. The decision to obtain ECG, and therefore our ability to identify patients with prolonged QTc, was at the discretion of the treating physicians. However, we believe that patients perceived at “high risk” were more likely to get ECGs than others, a factor that may help mitigate this bias to some extent. We studied only those with borderline or prolonged QTc at baseline (≥ 450 ms) excluding the ones with normal QTc. Our goal is to study the predictors of ventricular dysrhythmias, of which QTc is an important one. Since the outcome of interest is rare, we chose to include only patients with prolonged QTc, thereby improving the probability of the outcome occurring in a reasonable sample size and thus allowing us to study the associated factors. Due to the retrospective nature of the study, we relied on proper medical documentation of events and monitor strips of dysrhythmias, thus potentially introducing information bias. However, all of our Neuro ICU patients undergo continuous ECG monitoring with automated alarms and printouts of abnormal rhythms, which are then manually reviewed for accuracy and entered into the medical record. Lastly, we studied the entire Neuro ICU population and not a single disease process, thus creating heterogeneity in the sample, which may affect the applicability of our results to other Neuro ICUs with a different patient population.
Conclusion
In spite of the limitations, our study does provide useful information for practicing neurointensivists. We found that the risk of ventricular dysrhythmias is increased in patients with prolonged QTc ≥ 492 ms, supratentorial acute ischemic stroke, PVCs on ECG, and prolonged hospitalization. Prolonged QTc is also associated with poor outcome. Ability to identify which patients are at a higher risk of dysrhythmias will help the clinicians make important treatment decisions, especially regarding the choice of medications and optimum treatment of electrolyte imbalances.
Future Direction
Future prospective studies are needed to validate our findings in a larger sample. Capture and analysis of continuous QTc data throughout the hospitalization can increase our understanding of the relationship between QTc and the risk of ventricular dysrhythmia. Detailed review of all QTc-prolonging drugs administered with regard to the class, pharmacokinetics, dosage, and interactions can help analyze the impact of these drugs, particularly in patients with normal baseline QTc on admission. Lastly, patients after neurosurgical procedures need to be studied as a separate group.
References
Drew BJ, Ackerman MJ, Funk M, et al. Prevention of torsade de pointes in hospital settings: a scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121:1047–60.
Bazett HC. An analysis of the time-relations of electrocardiograms. Heart. 1920;7:353–70.
Niemeijer MN, van den Berg ME, Deckers JW, et al. Consistency of heart rate-QTc prolongation consistency and sudden cardiac death: The Rotterdam Study. Heart Rhythm. 2015;12:2078–85.
Zipes DP, Wellens HJ. Sudden cardiac death. Circulation. 1998;98:2334–51.
Katritsis DG, Zareba W, Camm AJ. Nonsustained ventricular tachycardia. J Am Coll Cardiol. 2012;60:1993–2004.
Ng TM, Olsen KM, McCartan MA, et al. Drug-induced QTc-interval prolongation in the intensive care unit: incidence and predictors. J Pharm Pract. 2010;23:19–24.
Tisdale JE, Wroblewski HA, Overholser BR, Kingery JR, Trujillo TN, Kovacs RJ. Prevalence of QT interval prolongation in patients admitted to cardiac care units and frequency of subsequent administration of QT interval-prolonging drugs: a prospective, observational study in a large urban academic medical center in the US. Drug Saf. 2012;35:459–70.
Tseng PT, Lee Y, Lin YE, Lin PY. Low-dose escitalopram for 2 days associated with corrected QT interval prolongation in a middle-aged woman: a case report and literature review. Gen Hosp Psychiatry. 2012;34(210):e13–5.
Wenzel-Seifert K, Wittmann M, Haen E. QTc prolongation by psychotropic drugs and the risk of Torsade de Pointes. Deutsch Arzteblatt Int. 2011;108:687–93.
Samuels MA. The brain-heart connection. Circulation. 2007;116:77–84.
Gregory TSM. Cardiovascular complications of brain injury. Contin Educ Anaesth Crit Care Pain. 2012;12:67–71.
Frontera JA, Parra A, Shimbo D, et al. Cardiac arrhythmias after subarachnoid hemorrhage: risk factors and impact on outcome. Cerebrovasc Dis. 2008;26:71–8.
Al-Khatib SM, LaPointe NM, Kramer JM, Califf RM. What clinicians should know about the QT interval. JAMA. 2003;289:2120–7.
Moss AJ. Long QT syndrome. JAMA. 2003;289:2041–4.
Crow RS, Hannan PJ, Folsom AR. Prognostic significance of corrected QT and corrected JT interval for incident coronary heart disease in a general population sample stratified by presence or absence of wide QRS complex: the ARIC study with 13 years of follow-up. Circulation. 2003;108:1985–9.
Katsanos AH, Korantzopoulos P, Tsivgoulis G, Kyritsis AP, Kosmidou M, Giannopoulos S. Electrocardiographic abnormalities and cardiac arrhythmias in structural brain lesions. Int J Cardiol. 2013;167:328–34.
Marafioti V, Rossi A, Carbone V, Pasqualin A, Vassanelli C. Prolonged QTc interval is a powerful predictor of non-cardiac mortality in patients with aneurysmal subarachnoid hemorrhage independently of traditional risk factors. Int J Cardiol. 2013;170:e5–6.
Katritsis DG, Camm AJ. Nonsustained ventricular tachycardia: where do we stand? Eur Heart J. 2004;25:1093–9.
Moss AJ, Schwartz PJ, Crampton RS, et al. The long QT syndrome. Prospective longitudinal study of 328 families. Circulation. 1991;84:1136–44.
Sauer AJ, Moss AJ, McNitt S, et al. Long QT syndrome in adults. J Am Coll Cardiol. 2007;49:329–37.
Reinelt P, Karth GD, Geppert A, Heinz G. Incidence and type of cardiac arrhythmias in critically ill patients: a single center experience in a medical-cardiological ICU. Intensive Care Med. 2001;27:1466–73.
Pickham D, Helfenbein E, Shinn JA, et al. High prevalence of corrected QT interval prolongation in acutely ill patients is associated with mortality: results of the QT in Practice (QTIP) Study. Crit Care Med. 2012;40:394–9.
Scheitz JF, Nolte CH, Doehner W, Hachinski V, Endres M. Stroke-heart syndrome: clinical presentation and underlying mechanisms. Lancet Neurol. 2018;17:1109–20.
Funding
Funding was received from the Dubie Hulliman Heart and Cancer Fund through the Wake Forest School of Medicine Medical Student Research Program.
Author information
Authors and Affiliations
Contributions
Megan Garzon contributed towards study concept and design, data collection and writing the first draft of the manuscript. Kelly Fuhrmann contributed towards data collection from pharmacy standpoint. Christopher McLouth contibuted towards data analysis. Dr. Vachharajani contributed towards interpretation of data and revising the manuscript for content. Dr. Datar contributed towards study concept and design, data collection, analysis, data interpretation and revising the manuscript for content.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed Consent
We confirm adherence to ethical guidelines and IRB approval for this study. Waiver of written informed consent was requested and approved by the IRB.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garzon, M.E., Fuhrmann, K.M., McLouth, C.J. et al. Predictors of Ventricular Dysrhythmias in Neurology Intensive Care Unit Patients with Prolonged QTc. Neurocrit Care 33, 769–775 (2020). https://doi.org/10.1007/s12028-020-00954-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-020-00954-y