Abstract
Background
Early hematoma expansion in intracerebral hemorrhage (ICH) patients is associated with poor outcome. We aimed to investigate whether the minimal computed tomography (CT) attenuation value predicted hematoma expansion and poor outcome.
Methods
This study involved spontaneous ICH patients of two cohorts who underwent baseline CT scan within 6 h after ICH onset and follow-up CT scan within 24 h after initial CT scan. We determined the critical value of the minimal CT attenuation value via retrospective analysis of the data from a derivation cohort. Then, a prospective study on the validation cohort of three clinical centers was performed for determining the association between the minimal CT attenuation value and hematoma expansion as well as poor outcome (modified Rankin Scale scores > 3) at 90 days by using univariate and multivariate logistic regression analyses.
Results
One hundred and forty eight ICH patients were included in the derivation cohort. Minimal CT attenuation value ≤ 31 Hounsfield units (HU) was demonstrated as the critical value to predict hematoma expansion by using receiver operating characteristic analysis. A total of 311 ICH patients were enrolled in the validation cohort, 86 (27.7%) and 133 (42.8%) of which were found hematoma expansion and poor outcome. Minimal CT attenuation value ≤ 31 HU was positive in 73 patients (23.5%). The multivariate logistic regression analysis demonstrated minimal CT attenuation value and minimal CT attenuation value ≤ 31 HU independently predicted hematoma expansion (p < 0.001) and poor outcome (p < 0.001). The sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of minimal CT attenuation value ≤ 31 HU for hematoma expansion and poor outcome prediction were 64.0, 92.0, 75.3, 87.0, 84.2 and 45.1%, 92.7%, 82.2%, 69.3%, 72.3%, respectively.
Conclusions
The minimal CT attenuation value independently predicts early hematoma expansion and poor outcome in patients with ICH.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Intracerebral hemorrhage (ICH) accounts for approximately 7–15% of all cerebrovascular diseases, contributing the second most common cause of stroke. ICH has a high rate of mortality and morbidity with poor outcomes [1, 2]. However, although enormous progress has been made for management of ICH due to the unremitting efforts, ICH treatment is still limited in its ability to reduce the poor prognosis. It is well known that early intensive intervention is very crucial for ICH treatment [3]. Hematoma expansion, which often occurs within 6 h after ICH onset, is a major determinant of early deterioration and associated with poor outcomes [4]. Based on the aforementioned theory, it is hypothesized that early identification of the risk of hematoma expansion facilitates adequate intervention to prevent or reduce it, which may lead to clinical benefit for ICH treatment. However, prediction of hematoma expansion by existing predictors has not been translated to improved outcomes.
Radiological examination plays an important role in ICH diagnosis. Computed tomography angiography (CTA) spot sign, presenting as the active contrast extravasation, is a well-known predictor for hematoma expansion and disability [5]. The noncontrast CT (NCCT) scan is required for each ICH patient and can be achieved within a short time as it can be performed in almost every emergency room. As a result, NCCT scan has its particular advantages over CTA scan. It has been reported NCCT density heterogeneity may independently predict hematoma expansion [6]. Correspondingly, several NCCT signs are able to be used for predicting hematoma expansion, including swirl sign, blend sign, and black hole sign, which are also predictors for poor outcome [7,8,9,10]. In clinic, we found that patients with hematoma expansion usually had lower CT attenuation within the hematoma. The purpose of the current study was to investigate whether the minimal CT attenuation value was associated with hematoma expansion and poor outcome.
Methods
Study Population
This study contained two cohorts of spontaneous ICH patients aged 18 years or older.
Derivation Cohort
Consecutive patients with primary ICH from Huashan Hospital, Fudan University (Center 1), admitted between January 1, 2011, and December 31, 2014, were enrolled for retrospective analysis. The enrolled patients all underwent baseline NCCT scan within 6 h after ICH onset, and the follow-up NCCT scan was performed within 24 h after initial CT scan. The exclusion criteria included: (1) secondary ICH (cerebral aneurysm, Moyamoya syndrome, arteriovenous malformation, tumor, trauma, or hemorrhagic transformation from brain infarction); (2) anticoagulant-associated ICH; (3) undergoing surgical evacuation before follow-up NCCT scan; (4) primary intraventricular hemorrhage (IVH); (5) a baseline ICH volume of less than 1 ml; (6) historical modified Rankin Scale (mRS) score > 1. We retrospectively examined whether minimal CT attenuation value was appropriate for diagnosing hematoma expansion and determined the critical value via receiver operating characteristic (ROC) analysis.
Validation Cohort
Consecutive patients with primary ICH admitted to Department of Neurology from three clinical centers of Fudan University: Huashan Hospital (Center 1), Zhongshan Hospital (Center 2), and Jinshan Hospital (Center 3) between January 1, 2015, and December 31, 2017, were enrolled for prospective analysis. The inclusion and exclusion criteria were the same as in the derivation cohort. We prospectively investigated whether minimal CT attenuation value predicted hematoma expansion and poor outcome in ICH patients of this cohort. All patients or their next-of-kin gave their informed consent prior to inclusion in this study. This study was approved by and studied in accordance with the ethical standards of the Ethics Committee of Fudan University.
Imaging Analysis
NCCT examinations were performed using a multidetector CT scanner with contiguous axial 5-mm section thickness (Brilliance iCT; Philips Medical Systems, Cleveland, OH, USA). The hematoma was three dimensionally reconstructed, and the volume was measured by 3D Slicer software, which is a free open-source software platform for biomedical research (http://www.slicer.org). CT image data sets were acquired in the DICOM format, and hematomas were automatically identified pixel by pixel in each slice after setting the threshold range at 50–100 Hounsfield units (HU). A 3D model was constructed, and the hematoma volume was calculated by the accumulating volume of the pixels [11]. Hematoma expansion was defined as an absolute growth greater than 6 mL or a relative growth of more than 33% [2].
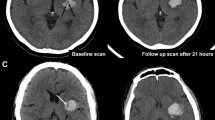
For the heterogeneous hematoma, we should first acquire one or more hypoattenuating area(s) within the hematoma. The hypoattenuating area connecting to the outer surface of the hematoma (normal brain tissue) should be excluded (Fig. 1a). Then, we obtained the minimal CT attenuation value of the hypoattenuating area by setting regions of interest (ROIs) on the CT images. The ROI should be carefully drawn to cover most part of the hypoattenuating area and avoid the surrounding hyperattenuating area. In case of a small hypoattenuating area, the CT image should be zoomed in to acquire more accurate ROI (Fig. 1b). Then, the software automatically calculated the minimal HU attenuation value in each ROI. If no obvious hypoattenuating area was observed in a homogeneous hematoma, the ROI should include most part of the hematoma in every CT section to obtain the minimal CT attenuation value. The minimal CT attenuation values were evaluated independently by two neuroradiologists with experience of 10 years or more who were blinded to the clinical data, follow-up CT scans, and outcomes.
a Hypoattenuating area (arrow) connecting to the outer surface of the hematoma should be excluded when calculating the minimal CT attenuation value. b To obtain the minimal CT attenuation value of the hypoattenuating area, a region of interest (ROI) was set to cover most part of the hypoattenuating area and avoid the surrounding hyperattenuating area. The minimal CT attenuation value was 28 Hounsfield units (HU). c The minimal CT attenuation value was 28 HU (arrow), and the follow-up CT displayed 40.7% of hematoma expansion. d No hematoma expansion was observed when the minimal CT attenuation value was 40 HU (arrow)
Clinical Data
The essential clinical data including sex, age, history of hypertension, diabetes mellitus, smoking, alcohol consumption, systolic and diastolic blood pressure, prothrombin time, activated partial thromboplastin time, international normalized ratio, platelet count on admission and the time of ICH onset, and Glasgow coma scale (GCS) scores were recorded after admission. We also recorded the time of initial CT, location of the hematoma, presence of IVH, hematoma volume, and the minimal CT attenuation values after the initial NCCT scan.
The functional outcome was assessed by using mRS at 90 days after ICH onset in the validation cohort. It was performed through in-person interviews by trained senior physicians or a phone call by trained study staffs. Poor outcome was defined as mRS > 3, and good outcome was defined as mRS ≤ 3 [12, 13].
Statistical Analysis
All statistical data were analyzed with SPSS22.0. Data for categorical variables are expressed as a percentage and compared using χ2 test or Fisher’s exact test (2-tailed). Data for continuous variables were presented as means (standard deviations [SDs]) or medians (interquartile ranges [IQRs]) when appropriate and analyzed using two-tailed Student t test, one-way analysis of variance, Mann–Whitney U test, or Kruskal–Wallis H test depending on the data distribution and the number of variables. Differences were regarded statistically significant at a value of P < 0.05. The ROC analysis was performed to determine the critical value of minimal CT attenuation value for predicting hematoma expansion. We used Cohen κ interagreement test as agreement statistics (interrater and intrarater reliability) for judgment of the minimal CT attenuation value. Univariate analysis was used for comparing the variables to discover the possible significant predictors for hematoma expansion and poor outcome. When significance appeared, multivariate logistic regression analysis was performed to investigate the independent predictors for hematoma expansion and poor outcome. Variables known to be associated with poor outcome based on multiple external data sets were also included in the multivariate model. Meanwhile, the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of minimal CT attenuation value for hematoma expansion and poor outcome prediction were also calculated.
Results
Baseline Characteristics of the Two Cohorts
After application of the eligibility criteria, 148 of 454 patients with primary ICH in the derivation cohort and 311 of 684 patients with primary ICH in the validation cohort were enrolled for analysis (Fig. 2). In comparison of the baseline characteristics between included and excluded patients, no significant differences were demonstrated except time to the baseline NCCT (135.0 [IQR 85.0–226.0] min vs. 375.0 [IQR 195.0–578.0] min).
The baseline characteristics of the patients are shown in Table 1. In the validation cohort, hypodensities (defined as 4 types [14]) were found in 127 patients (40.8%). We demonstrated excellent interrater agreement (κ = 0.90, 95% CI 0.72–0.97) and intrarater agreement (κ = 0.91, 95% CI 0.82–0.95) for the minimal CT attenuation values. Hematoma expansion was observed in 86 patients (27.7%). Patients with poor outcome (mRS > 3) were 133 (42.8%), and the median of mRS was 3 (IQR 2–4) (Table 1).
The Critical Value of Minimal CT Attenuation Value Determined by ROC Analysis
We listed the minimal CT attenuation values within the hematoma and the corresponding existence of hematoma expansion from the data of the derivation cohort. Then a ROC curve was established. The area under the curve (AUC) of ROC curve was 0.874 with 95% confidence interval (CI) 0.807–0.941 (p < 0.001), indicating that minimal CT attenuation value was appropriate for diagnosing hematoma expansion. The cutoff representing the optimal minimal CT attenuation value for predicting hematoma expansion was 31 HU with the sensitivity, specificity, and Youden index (sensitivity + specificity-1) of 71.0%, 91.5%, and 0.625%, respectively (Fig. 3a). As a result, we further validated whether minimal CT attenuation value ≤ 31 HU is independently associated with hematoma expansion and poor outcome.
Receiver operating characteristic (ROC) curves. a ROC analysis for determining the critical value of the minimal CT attenuation value. b ROC curve of minimal CT attenuation value ≤ 31 HU for predicting hematoma expansion. c ROC curve of minimal CT attenuation value ≤ 31 HU for predicting poor outcome
Association Between Minimal CT Attenuation Value and Hematoma Expansion
Of all the clinical and radiological variables, baseline GCS scores, time to the baseline NCCT, baseline hematoma volume, hematoma expansion, mRS scores, and poor outcome were significantly different among different minimal CT attenuation values (p < 0.05) (Table 2). Minimal CT attenuation value ≤ 31 HU was found in 73 patients (23.5%) and observed in 55 patients with hematoma expansion (64.0%) (Table 2). In this table, 31 HU, 43 HU, and 51 HU represent the critical value, second quartile, and third quartile, respectively. For hematoma expansion prediction, univariate analysis revealed significant differences in baseline GCS scores, time to the baseline NCCT, baseline hematoma volume, minimal CT attenuation value, and minimal CT attenuation value ≤ 31 HU (Table 3). Subsequently, the multivariate logistic regression analysis demonstrated minimal CT attenuation value (p < 0.001) and minimal CT attenuation value ≤ 31 HU (p < 0.001) were independently associated with hematoma expansion (Table 4). As shown in Fig. 1c, d, hematoma expansion occurred in a patient with the minimal CT attenuation value of 28 HU, while no hematoma expansion was observed when the value was 40 HU. The sensitivity, specificity, PPV, NPV, and accuracy of minimal CT attenuation value ≤ 31 HU for hematoma expansion prediction were 64.0%, 92.0%, 75.3%, 87.0%, and 84.2%, respectively. In ROC curve, the AUC was 0.796 with the 95% CI of 0.740–0.851 (p < 0.001) (Fig. 3b).
Association Between Minimal CT Attenuation Value and Poor Outcome
Minimal CT attenuation value ≤ 31 HU was found in 60 patients with poor outcome (45.1%). mRS scores in patients with or without minimal CT attenuation value ≤ 31 HU were also detailed (Fig. 4). Significant differences were demonstrated in baseline GCS scores, IVH, baseline hematoma volume, minimal CT attenuation value, and minimal CT attenuation value ≤ 31 HU by univariate analysis (Table 3). The multivariate logistic regression analysis demonstrated age, baseline GCS scores, baseline hematoma volume, minimal CT attenuation value, and minimal CT attenuation value ≤ 31 HU were independent predictors for poor outcome (Table 4). Moreover, the sensitivity, specificity, PPV, NPV, and accuracy of minimal CT attenuation value ≤ 31 HU for predicting poor outcome were 45.1%, 92.7%, 82.2%, 69.3%, and 72.3%, respectively. The AUC of ROC curve was 0.658 with the 95% CI of 0.600–0.716 (p < 0.001) (Fig. 3c).
Discussion
Since hematoma expansion mainly occurs within the first 6 h after ICH onset, early identification and prevention are critical for acute ICH treatment [15]. Several previous studies have focused on the prediction of early hematoma expansion by NCCT based on heterogeneous in density, including the blend sign, black hole sign, and hypodensities [6, 7, 14]. Here, we demonstrated both minimal CT attenuation value and minimal CT attenuation value ≤ 31 HU could independently predict hematoma expansion. Since hematoma expansion is associated with a poor prognosis [16], it should be more beneficial if the predictor for hematoma expansion is also associated with poor outcome. It has been reported that other NCCT signs such as blend sign, black hole sign, and hypodensities are also able to predict unfavorable outcome in ICH patients [17,18,19]. Correspondingly, our data also revealed minimal CT attenuation value and minimal CT attenuation value ≤ 31 HU were independently associated with poor outcome assessed by using mRS at 90 days after ICH. However, some common predictors such as IVH and infratentorial ICH were not displayed independent predictors. It is possible that patients with infratentorial ICH and (or) IVH who are inclined to unfavorable outcome are more likely received surgery. However, in order to evaluate hematoma expansion, we excluded the patients undergoing surgical evacuation before follow-up NCCT scan. Meanwhile, the enrolled ICH patients were from Department of Neurology other than Neurosurgery, which further excluded such patients. Thus, the two factors failed to be regarded as independent predictors in our studies.
To tailor intensive therapies timely and effectively for high-risk patients of hematoma expansion, the predictors should be easy to utilize as early as possible. Predicting hematoma expansion in virtue of neuroradiological findings is a hot topic. Numerous studies have indicated CTA spot sign is a strong predictor which represents contrast extravasation through ruptured vessels due to active bleeding or rebleeding [16]. Meanwhile, NCCT scan, which is a rapid and effective tool for ICH diagnosis, can be easily performed in almost every emergency room. Therefore, the early identifiable imaging characteristics on NCCT scan become good candidates for predicting hematoma expansion. The shape and density of hematoma are two key points, and the latter has been intensively investigated. In 2009, Barras and the colleagues created 5-point categorical scales for evaluating hematoma heterogeneous and demonstrated density heterogeneity independently predicted ICH growth [6]. Li et al. disclosed two signs: blend sign and black hole sign based on specific CT attenuation differences for definition, both of which have high specificities according to their work [8, 9]. However, the sensitivity to predict hematoma expansion (39.3% and 31.9%) [8, 9] and poor outcome (8% and 11%) [20] are relatively low (Table 5). Furthermore, the identification of minimal CT attenuation value only requires one ROI, while two ROIs are essential for blend sign and black hole sign, which makes our sign easier to distinguish. Moreover, hypodensities within the hematoma of different types have been reported to independently predict hematoma expansion and poor outcome (including 48 h and 90 days) [14, 18, 19]. Although hypodensities are easy to identify compared with other signs, the specificity is not satisfied (77% in hematoma expansion and 79.6% in poor outcome) [14, 18]. We listed the diagnostic efficacy of the above signs in Table 5 and demonstrated although the specificity of minimal CT attenuation value ≤ 31 HU was lower than blend sign and black hole sign, the accuracy was the highest both in predicting hematoma expansion and poor outcome (both nearly 10% higher).
It is well known that hyperdensity represents ICH in NCCT scan, while the density of hematoma dynamically varies in the early course. The attenuation of the hematoma depends on the content of hemoglobin, blood cells, and plasma, which are important components of the whole blood [21]. The early hematoma at the initial phase of ICH is comprised of a heterogeneous mass formed by various blood cells and platelet thrombi mixed with protein-rich plasma [22]. The hypodensity of the heterogeneous attenuation at this phase is credited to the plasma. Accompanying the clots retraction and cellular element sedimentation, the lower attenuating plasma is extruded, resulting in the formation of higher attenuating hematoma [23, 24]. As a result, hypoattenuating area may appear within the hyperattenuating hematoma in case of the existence of active bleeding or rebleeding. Therefore, the lower density the hypoattenuating area with hematoma is, the greater the possibly that incoagulable blood is inclined to exist, which indicates the high risk of hematoma expansion.
Since the minimal CT attenuation value is associated with hematoma expansion and poor outcome, the potential clinical implications should be further investigated. Single predictor for hematoma expansion has its own limitation. Therefore, the prediction scores comprising of several predictors have been created [25, 26]. Moreover, the predictors from laboratory tests, e.g., lactate dehydrogenase, may also be useful [27]. Correspondingly, due to the higher accuracy of minimal CT attenuation value ≤ 31 HU compared with other signs, it can be combined with other predictors to form a new prediction score with higher sensitivity and specificity. Moreover, our sign can also be represented numerically, different from yes or no in other signs. We hypothesize that it can be directly combined with other numerically represented predictors such as laboratory tests and clinical features to establish specific mathematical models, which may better predict hematoma expansion and poor outcome. In addition, the minimal CT attenuation value may also be applied in ICH treatment. It has been studied whether intensive blood pressure reduction improves outcome in ICH patients with NCCT and CTA markers [20, 28]. Meanwhile, CTA spot sign has been used as a key inclusion criterion of the trial to evaluate the effect of tranexamic acid [29]. Similarly, further studies may investigate whether ICH patients with minimal CT attenuation value ≤ 31 HU benefit from intensive blood pressure reduction or hemostasis therapy.
Several limitations of this study should be noted. The sample size is relatively small. Meanwhile, although the minimal CT attenuation value of 31 HU in our work better predicted hematoma expansion and poor outcome, the optimal HU threshold may be changed supposing the increase in sample size. Moreover, to simultaneously observe hematoma expansion, we excluded the patients undergoing surgical evacuation before follow-up NCCT scan and those without follow-up CT or follow-up CT > 24 h, which may reduce the sample size for predicting poor outcome.
Conclusions
The current study using derivation and validation cohorts suggests the minimal CT attenuation value is independently associated with hematoma expansion and poor outcome. Meanwhile, our findings indicate minimal CT attenuation value ≤ 31 HU can be regarded as a predictor for early hematoma expansion and poor outcome due to its feasibility for clinical practice.
References
van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol. 2010;9:167–76.
Demchuk AM, Dowlatshahi D, Rodriguez-Luna D, et al. Prediction of haematoma growth and outcome in patients with intracerebral haemorrhage using the CT-angiography spot sign (PREDICT): a prospective observational study. Lancet Neurol. 2012;11:307–14.
Hemphill JC 3rd, Greenberg SM, Anderson CS, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46:2032–60.
Dowlatshahi D, Demchuk AM, Flaherty ML, et al. Defining hematoma expansion in intracerebral hemorrhage: relationship with patient outcomes. Neurology. 2011;76:1238–44.
Delgado Almandoz JE, Yoo AJ, Stone MJ, et al. Systematic characterization of the computed tomography angiography spot sign in primary intracerebral hemorrhage identifies patients at highest risk for hematoma expansion: the spot sign score. Stroke. 2009;40:2994–3000.
Barras CD, Tress BM, Christensen S, et al. Density and shape as CT predictors of intracerebral hemorrhage growth. Stroke. 2009;40:1325–31.
Sporns PB, Schwake M, Kemmling A, et al. Comparison of spot sign, blend sign and black hole sign for outcome prediction in patients with intracerebral hemorrhage. J Stroke. 2017;19:333–9.
Li Q, Zhang G, Huang YJ, et al. Blend sign on computed tomography: novel and reliable predictor for early hematoma growth in patients with intracerebral hemorrhage. Stroke. 2015;46:2119–23.
Li Q, Zhang G, Xiong X, et al. Black hole sign: novel imaging marker that predicts hematoma growth in patients with intracerebral hemorrhage. Stroke. 2016;47:1777–81.
Selariu E, Zia E, Brizzi M, Abul-Kasim K. Swirl sign in intracerebral haemorrhage: definition, prevalence, reliability and prognostic value. BMC Neurol. 2012;12:109.
Xu X, Chen X, Zhang J, et al. Comparison of the Tada formula with software slicer: precise and low-cost method for volume assessment of intracerebral hematoma. Stroke. 2014;45:3433–5.
Qureshi AI, Palesch YY, Barsan WG, et al. Intensive blood-pressure lowering in patients with acute cerebral hemorrhage. N Engl J Med. 2016;375:1033–43.
Hanley DF, Lane K, McBee N, et al. Thrombolytic removal of intraventricular haemorrhage in treatment of severe stroke: results of the randomised, multicentre, multiregion, placebo-controlled CLEAR III trial. Lancet. 2017;389:603–11.
Boulouis G, Morotti A, Brouwers HB, et al. Association between hypodensities detected by computed tomography and hematoma expansion in patients with intracerebral hemorrhage. JAMA Neurol. 2016;73:961–8.
Kazui S, Naritomi H, Yamamoto H, Sawada T, Yamaguchi T. Enlargement of spontaneous intracerebral hemorrhage. Incidence and time course. Stroke. 1996;27:1783–7.
Goldstein JN, Fazen LE, Snider R, et al. Contrast extravasationon CT angiography predicts hematoma expansion inintracerebral hemorrhage. Neurology. 2007;68:889–94.
Sporns PB, Schwake M, Kemmling A, Minnerup J, Schwindt W, Niederstadt T, et al. Comparison of spot sign, blend sign and black hole sign for outcome prediction in patients with intracerebral hemorrhage. J Stroke. 2017;19:333–9.
Boulouis G, Morotti A, Brouwers HB, et al. Noncontrast computed tomography hypodensities predict poor outcome in intracerebral hemorrhage patients. Stroke. 2016;47:2511–6.
Dong J, Yang X, Xiang J, et al. Hypodensities detected at 1.5–3 h after intracerebral hemorrhage better predicts secondary neurological deterioration. J Neurol Sci. 2019;396:219–24.
Morotti A, Boulouis G, Romero JM, et al. Blood pressure reduction and noncontrast CT markers of intracerebral hemorrhage expansion. Neurology. 2017;89:548–54.
New PF, Aronow S. Attenuation measurements of whole blood and blood fractions in computed tomography. Radiology. 1976;121:635–40.
Schlunk F, Greenberg SM. The pathophysiology of intracerebral hemorrhage formation and expansion. Transl Stroke Res. 2015;6:257–63.
Wolverson MK, Crepps LF, Sundaram M, Heiberg E, Vas WG, Shields JB. Hyperdensity of recent hemorrhage at body computed tomography: incidence and morphologic variation. Radiology. 1983;148:779–84.
Boulouis G, Morotti A, Charidimou A, Dowlatshahi D, Goldstein JN. Noncontrast computed tomography markers of intracerebral hemorrhage expansion. Stroke. 2017;48:1120–5.
Brouwers HB, Chang Y, Falcone GJ, et al. Predicting hematoma expansion after primary intracerebral hemorrhage. JAMA Neurol. 2014;71:158–64.
Morotti A, Dowlatshahi D, Boulouis G, et al. Predicting intracerebral hemorrhage expansion with noncontrast computed tomography: the BAT score. Stroke. 2018;49:1163–9.
Chu H, Huang C, Dong J, et al. Lactate dehydrogenase predicts early hematoma expansion and poor outcomes in intracerebral hemorrhage patients. Transl Stroke Res. 2019;1:1. https://doi.org/10.1007/s12975-019-0686-7.
Morotti A, Brouwers HB, Romero JM, et al. Intensive blood pressure reduction and spot sign in intracerebral hemorrhage: a secondary analysis of a randomized clinical trial. JAMA Neurol. 2017;74:950–60.
Meretoja A, Churilov L, Campbell BC, et al. The spot sign and tranexamic acid on preventing ICH growth–AUStralasia Trial (STOP-AUST): protocol of a phase II randomized, placebo-controlled, double-blind, multicenter trial. Int J Stroke. 2014;9:519–24.
Author information
Authors and Affiliations
Contributions
HC and CH contributed to study design, data collection, and analysis and drafting of the manuscript. JD, XY, JX, and YM involved in data collection and analysis. QD and YT took part in conception and design, acquisition of clinical data, revision and approval of the manuscript. All authors gave the final approval of the version to be published. Authorship requirements have been met, and the final manuscript was approved by all authors. This manuscript has not been published elsewhere and is not under consideration by another journal.
Corresponding authors
Ethics declarations
Source of Support
This research was supported by grants from the National Natural Science Foundation of China (Nos. 81500998; 81701244; 81801290), Science and Technology Commission of Shanghai Municipality (No. 16140903200), and Shanghai Sixth People’s Hospital Medical Group (2017LY01).
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical Approval/Informed Consent
All patients or their next-of-kin gave their informed consent prior to inclusion in this study. This study was approved by and studied in accordance with the ethical standards of the Ethics Committee of Fudan University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chu, H., Huang, C., Dong, J. et al. Minimal Computed Tomography Attenuation Value Within the Hematoma is Associated with Hematoma Expansion and Poor Outcome in Intracerebral Hemorrhage Patients. Neurocrit Care 31, 455–465 (2019). https://doi.org/10.1007/s12028-019-00754-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-019-00754-z