Abstract
Little is known about the pathological characteristics of metastatic and nonmetastatic lymph nodes (LNs) in papillary thyroid carcinoma (PTC). The aim of this study was to elucidate the pathological characteristics of neck level VI nodes and correlations with clinicopathological parameters. We investigated the clinicopathological features of 124 classical PTCs and characteristics of 850 dissected neck level VI LNs. Immunohistochemistry for nuclear factor-κB (NF-κB) and Ki-67 was performed on primary tumors, and correlations with nodal characteristics were investigated. Nodal metastasis at neck level VI was identified in 68 of 124 PTCs (54.8 %) and was significantly correlated with tumor size, tumor multifocality, extrathyroidal extension, and tumor stage. LN metastasis was significantly correlated with larger mean LN size (P < 0.001), larger size of the largest LN (P < 0.001), and more LNs (P < 0.001). The mean size of metastatic LNs (n = 233) was significantly larger than nonmetastatic LNs (n = 617) (P < 0.001). Primary tumor diameter significantly correlated with the largest LN size (P = 0.014, R 2 = 0.049), but not mean size or number of LNs by linear regression analysis. NF-κB and Ki-67 proliferation indexes were not significantly correlated with larger nodal size. These results suggested that characteristics of nodes and primary tumor would be useful criteria for making decisions about preoperative surveillance of nodal metastasis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Papillary thyroid carcinomas (PTCs) are malignant tumors with lymph node (LN) metastasis occurring in 30–80 % of patients [1, 2]. Although most PTCs have favorable behavior, some tumors show aggressiveness including local invasion, LN metastasis, locoregional recurrence, and distant metastasis. In PTCs, neck LN dissection is recommended when LN involvement or advanced stage is suspected based on the American Thyroid Association management guidelines [3]. However, prophylactic neck level VI (central-compartment) LN dissection is controversial. Thus, the detection or suspicion of LN metastasis is important for decisions about surgical extent. Preoperative ultrasonography (US) is reported to be the most sensitive method for the detection of LN metastasis. However, preoperative US cannot detect all metastatic nodes because some LNs are too small [4]. Therefore, we need improved criteria for the evaluation of nodal metastasis.
Although nodal characteristics are useful for predicting LN metastasis, the pathological characteristics of metastatic nodes are not fully understood, and the criteria for predicting nodal metastasis vary [5]. In addition, little is known about whether proliferation of primary tumors is associated with LN metastasis, although primary tumor diameter is reported to be correlated with LN metastasis [6]. We previously reported that nuclear factor-κB (NF-κB) activation contributes to tumor growth and aggressiveness in human PTC [7]. Ki-67 is a cellular proliferation-associated antigen [8], and a higher Ki-67 proliferation index is associated with tumor growth and recurrence [9, 10]. However, the correlation between these factors and nodal characteristics is not fully understood in PTC.
Therefore, we investigated the pathological features of neck level VI LN to explore the correlation between nodal characteristics and the clinicopathological features of the primary tumor.
Patients and Methods
Patients
We investigated 124 surgically resected conventional PTCs at the Department of Pathology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea from January 1 to December 31, 2010. Patients without US examination were excluded. Patients’ data used in the present study was partly cited from our previous study [7]. Clinicopathological parameters, such as ultrasonographic findings of neck level VI LN, patient age and gender, tumor size, multifocality, pTNM stage, extrathyroidal extension, LN metastasis, and nodal characteristics, were evaluated by reviewing medical charts, pathological records, and glass slides following the AJCC seventh edition TNM classification system [11]. All patients underwent either total thyroidectomy (n = 97) or lobectomy (n = 27). All patients in the present study underwent therapeutic or prophylactic neck level VI LN dissection after clinical evaluations [12]. Modified radical neck dissection was selectively performed in patients with confirmed lateral neck nodal metastasis via preoperative fine-needle aspiration biopsy or frozen section. Mean patient age was 47.4 ± 12.1 years, mean size of the largest dominant tumor was 1.22 ± 0.67 cm, and the ratio of men to women was 1:3.96. Based on pTNM classification [11], 74 patients had stage I cancer, none had stage II, 45 had stage III, and 5 had stage IV. This protocol was reviewed and approved by the Institutional Review Board of Kangbuk Samsung Hospital (Approval No. KBSMC 2014-08-019).
Evaluation of Pathological Features
The pathological features investigated were primary tumor diameter, tumor multifocality, extrathyroidal extension, LN metastasis, and nodal characteristics. All dissected LNs were embedded for microscopic examination, and nodal characteristics were evaluated. Nodal characteristics were the number of dissected LNs, number of metastatic LNs, largest LN size, largest metastatic LN size, and mean LN size. Psammoma bodies may form through the necrosis and calcification of intravascular or intralymphatic tumor thrombi or by intracellular calcifications in the viable cells of the nidus [13]. In the present study, of 124 patients, psammoma bodies without tumor cells were considered metastatic PTCs. In the cases of multifocal PTCs, the largest single tumor was defined as the primary tumor.
Tissue Array Methods
Three array blocks containing 124 PTC tissue cores were prepared for immunohistochemistry. A single core was obtained from each primary tumor. Core tissue biopsies (2 mm in diameter) were taken from individual paraffin-embedded PTCs (donor blocks) and arranged in recipient paraffin tissue array blocks using a trephine apparatus. The sections of 4 μm were cut from each block and stained with hematoxylin and eosin for histological assessment. Each block contained nonneoplastic thyroid tissue as an internal control.
Immunohistochemical Staining
Sections were deparaffinized and hydrated by routine xylene-alcohol procedures before incubation with 0.01 M citrate buffer (pH 6.0) for 5 min in a microwave oven for antigen retrieval and treatment with 3 % H2O2 to quench endogenous peroxidase. Sections were treated with normal serum from the animal used to generate secondary antibodies to block nonspecific binding, then incubated with anti-RelA (1:100, Santa Cruz Biotechnology, Santa Cruz, CA, USA) or anti-Ki-67 (1:50, Santa Cruz Biotechnology). Immunohistochemical staining used a compact polymer method from Bond Intense detection kits (Leica Biosystems, Newcastle, UK). Visualization was with 3,3′-diaminobenzidine (Vector Laboratories, Burlingame, CA, USA). To confirm reaction specificity, a negative control without primary antibody was included. All immunostained sections were lightly counterstained with Mayer’s hematoxylin.
Evaluation of Immunohistochemistry
NF-κB RelA staining, regardless of cytoplasmic staining, was considered as showing NF-κB activation, and NF-κB positivity was defined as a positive NF-κB stain in ≥5 % of tumor cell nuclei [14–16]. Because NF-κB is constitutively expressed in the cytoplasm and we used an antibody against the nonphosphorylated form of RelA, immunoreactivity was localized in the cytoplasm and nucleus [7, 15].
For Ki-67 staining, we evaluated 300 cells and counted cells with nuclear staining for each specimen. Proliferation index (%) was 100 × Ki-67-positive cells/300 cells.
Statistical Analysis
All statistical analyses used SPSS version 18.0 software (SPSS, Chicago, IL, USA). The significance of correlation between LN metastasis and clinicopathological parameters was determined by either χ 2 test or Fisher exact test (two sided). Two-tailed Student’s t test and linear regression tests were used to evaluate correlation between nodal characteristics and clinicopathological parameters. Results were considered statistically significant at P < 0.05.
Results
Correlation Between Lymph Node Metastasis and Clinicopathological Parameters
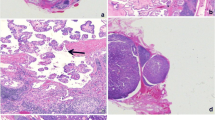
Correlation between lymph node metastasis and clinicopathological parameters of 124 PTCs were investigated (Fig. 1a, b and Table 1). LN metastasis was identified in 68 out 124 PTCs (54.8 %) and was significantly associated with tumor size (P < 0.001), tumor multifocality (P = 0.005), extrathyroidal extension (P = 0.001), and tumor stage (P = 0.029), but not age or gender, by univariate analysis. In multivariate analysis, larger tumor size (>1 cm), tumor multifocality, and extrathyroidal extension, but not older age or gender, were risk factors for LN metastasis. Among 68 PTCs with LN metastasis, 30 (44.1 %) showed suspicious findings on preoperative ultrasonography.
Comparison of Nodal Characteristics by Clinicopathological Features
We evaluated the nodal characteristics of neck level VI by clinicopathological parameters. PTCs with nodal metastasis showed larger mean LN size (0.29 ± 0.12 vs. 0.22 ± 0.18 cm, P < 0.001), larger size of the largest LN (0.54 ± 0.32 vs. 0.32 ± 0.16 cm, P < 0.001), and more LNs (8.97 ± 7.44 vs. 4.29 ± 2.82, P < 0.001) than PTCs without nodal metastasis (Table 2). NF-κB activation was significantly correlated with a higher number of LNs (P = 0.030), but not with mean LN size (P = 0.757) or largest LN size (P = 0.940, Fig. 1c). No significantly different nodal characteristics were observed between PTCs with higher (≥2 %) or lower (<2 %) Ki-67 proliferation index (Fig. 1d). Of 850 dissected neck level VI LNs, 233 (26.2 %) show identified metastasis. Mean sizes were 0.39 ± 0.25 cm for metastatic LNs and 0.25 ± 0.14 cm for nonmetastatic LNs. Metastatic LNs were significantly larger than nonmetastatic LNs (P < 0.001). We divided all dissected LNs with a cutoff of 0.3 cm; 68.2 % of metastatic LNs and 38.9 % of nonmetastatic LNs were larger than 0.3 cm (Table 3). Larger LNs (≥0.3 cm) exhibited a higher rate of metastasis than smaller LNs (<0.3 cm) (P < 0.001). In addition, all LNs larger than 1 cm were metastatic.
Correlation Between Node Size and Tumor Size
We investigated the correlation between LN size and tumor size by linear regression. The largest LN size, but not the mean LN size, increased with increasing primary tumor diameter (P = 0.014, R 2 = 0.049 for largest LN; P = 0.161, R 2 = 0.016 for mean LN size) (Fig. 2). The number of LNs did not correlate with primary tumor diameter in linear regression tests (P = 0.079, R 2 = 0.025).
Discussion
Although PTCs have favorable prognosis, cervical LN metastasis occurs in up to 80 % of patients, and locoregional recurrence and distant metastasis are common [1, 2]. However, the contributing factors of LN that predict nodal metastasis are poorly understood, and treatment guidelines including prophylactic neck level VI (central-compartment) LN dissection are controversial.
Although preoperative US findings have low sensitivity, US on preoperative examination may be the most useful method for evaluating LN metastasis [5]. In our study, only 30 patients (44.1 %) were predicted to have LN metastasis by preoperative US, out of 68 patients with PTCs with cervical LN metastasis. On preoperative US, nodal features suspicious of metastasis included node size, hyperechogenicity, echogenicity of hilum, calcification, and intranodal cystic necrosis. However, the criterion of node size on preoperative US is not completely understood. None of the features we studied completely contributed to LN metastasis, and all have limitations for use in interpretation such as small LN size and complex anatomy of the neck. Therefore, more simple, but easily adjustable, criteria for LN metastasis are needed. Our data showed that nodal size is a simple marker with significant correlation with largest LN size, mean LN size, and the presence of metastasis. However, the mean size difference between metastatic and nonmetastatic LNs was only 0.15 cm. This difference might not be useful as a criterion for preoperative US examinations. Therefore, we investigated the size distributions of LNs, which were 0.05–1.6 cm for metastatic LNs and 0.05–1.0 cm for nonmetastatic LNs. In our study, all LNs larger than 1 cm were metastatic. This criterion could be useful for suspicious preoperative US; however, our results were limited because we had few patients who had LNs larger than 1 cm (10.6 %).
Based on a node size of 0.3 cm, larger LNs (≥0.3 cm) showed a higher rate of metastasis than smaller LNs (<0.3 cm) (P < 0.001). Among 451 LNs smaller than 0.3 cm, metastatic LNs were 16.4 % and nonmetastatic LNs were 83.6 % suggesting that almost LNs smaller than 0.3 cm were nonmetastatic. Moreover, the largest LN size was larger than 0.3 cm in 56 of 68 patients with LN metastasis (82.4 %). Our data indicated that LN sizes larger than 0.3 cm on preoperative US might be a useful predictive factor of LN metastasis. However, this is not an absolute criterion because LNs detected on preoperative US might not match postoperative pathological examinations. An increased number of enlarged LNs would reinforce the findings. However, we found no other studies with results similar to ours, so further large-scale studies may be needed.
PTC prognosis uses systems such as AJCC TNM classification [17–19], AGES (age, grade, extent, size), AMES (age, distant metastasis, extent, size), MACIS (distant metastasis, age, completeness of resection, local invasion, size), and GAMES (grade, age, distant metastasis, extent, size). Primary tumor diameter is an important prognostic factors based on reports that primary tumor diameter correlates with LN metastasis [6], consistent with our results. In addition, node size proportionally increased with tumor size, consistent with a previous report [6]. Although several studies reported correlation between primary tumor diameter and nodal characteristics [6], no studies reported on whether the largest LN coincided with metastatic nodes. In this study, the largest LNs were metastatic LNs in 56 of 68 patients (82.4 %).
We previously reported that NF-κB activation is associated with larger tumor size, nodal metastasis, and higher Ki-67 proliferation index [7]. However, whether tumor proliferation-associated factors affect nodal characteristics is unknown. In the current study, NF-κB activation did not correlate with mean LN size or largest LN size. The Ki-67 proliferation index of the primary tumor did not correlate with largest LN size (P = 0.567, R 2 = 0.003) or mean LN size by linear regression (P = 0.390, R 2 = 0.006) (data not shown). Other factors might influence tumor size or presence of LN metastasis. Thus, nodal characteristics might be directly affected by primary tumor diameter rather than primary tumor proliferation-associated factors such as NF-κB and Ki-67. However, we found no pathogenesis that explained correlation between number of LNs and higher NF-κB activation, which requires a further study.
In conclusion, our data showed that LN metastasis strongly correlated with nodal characteristics, such as mean and largest LN size, and number. Thus, the nodal characteristics of size and number, combined with the nature of the primary tumor, might be useful for predicting LN metastasis on preoperative US and could be a useful criterion for decisions about surgical extent or modalities.
References
Mazzaferri EL (1993) Management of a solitary thyroid nodule. N Engl J Med 328:553-559.
Sugitani I, Fujimoto Y, Yamada K et al (2008) Prospective outcomes of selective lymph node dissection for papillary thyroid carcinoma based on preoperative ultrasonography. World J Surg 32:2494-2502.
American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer, Cooper DS, Doherty GM, Haugen BR et al (2009) Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19:1167-214.
Roh JL, Kim JM, Park CI (2008) Central cervical nodal metastasis from papillary thyroid microcarcinoma: pattern and factors predictive of nodal metastasis. Ann Surg Oncol 15:2482-2486.
Rosário PW, de Faria S, Bicalho L et al (2005) Ultrasonographic differentiation between metastatic and benign lymph nodes in patients with papillary thyroid carcinoma. J Ultrasound Med 24:1385-1389.
Lee YS, Lim YS, Lee JC et al (2013) Nodal status of central lymph nodes as a negative prognostic factor for papillary thyroid carcinoma. J Surg Oncol 107:777-782.
Pyo JS, Kang G, Kim DH et al (2013) Activation of nuclear factor-κB contributes to growth and aggressiveness of papillary thyroid carcinoma. Pathol Res Pract 209:228-232.
Gerdes J, Li L, Schlueter C (1991) Immunobiochemical and molecular biologic characterization of the cell proliferation-associated nuclear antigen that is defined by monoclonal antibody Ki-67. Am J Pathol 138:867-873.
Ranjbari N, Rahim F (2013) The Ki-67/MIB-1 index level and recurrence of papillary thyroid carcinoma. Med Hypotheses 80:311-314.
Miyauchi A, Kudo T, Hirokawa M (2013) Ki-67 labeling index is a predictor of postoperative persistent disease and cancer growth and a prognostic indicator in papillary thyroid carcinoma. Eur Thyroid J 2:57-64.
Edge SB, Byrd DR, Compton CC (2009) Thyroid The AJCC cancer staging manual (7th ed.) Springer-Verlag, New York
Cooper DS, Doherty GM, Haugen BR et al (2009) Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19:1167–1214.
Moon WJ, Jung SL, Lee JH et al (2008) Benign and malignant thyroid nodules: US differentiation—multicenter retrospective study. Radiology 247:762–770
Pacifico F, Leonardi A (2006) NF-kappaB in solid tumors. Biochem Pharmacol 72:1142-52.
Pyo JS, Ko YS, Kim WH (2010) Impairment of nuclear factor-kappaB activation increased glutamate excitotoxicity in a motoneuron-neuroblastoma hybrid cell line expressing mutant (G93A) Cu/Zn-superoxide dismutase. J Neurosci Res 88:2494-2503.
Kim B, Byun SJ, Kim YA (2010) Cell cycle regulators, APC/beta-catenin, NF-kappaB and Epstein-Barr virus in gastric carcinomas. Pathology 42:58–65.
Rodriguez-Cuevas S, Labastida-Almendaro S, Cortes-Arroyo H et al (2002) Multifactorial analysis of survival and recurrences in differentiated thyroid cancer. Comparative evaluation of usefulness of AGES, MACIS, and risk group scores in Mexican population. J Exp Clin Cancer Res 21:79-86.
Voutilainen PE, Siironen P, Franssila KO (2003) AMES, MACIS and TNM prognostic classifications in papillary thyroid carcinoma. Anticancer Res 23:4283-4288.
Mazzaferri EL (2008) Managing small thyroid cancers. JAMA 295:2179-2182.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pyo, JS., Sohn, J.H., Kang, G. et al. Characteristics of Neck Level VI Lymph Nodes in Papillary Thyroid Carcinoma: Correlation Between Nodal Characteristics and Primary Tumor. Endocr Pathol 26, 15–20 (2015). https://doi.org/10.1007/s12022-014-9353-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12022-014-9353-4