Abstract
Purpose
Serum intact parathyroid hormone (iPTH) level is an early marker of post-thyroidectomy hypocalcemia. However, lack of methodological standardization to define timing and cut-off points of iPTH measurement limit its clinical applicability. Here, we evaluated the relationship between two distinct postoperative time sampling and iPTH accuracy on predicting hypocalcemia.
Methods
iPTH was measured within 4 h after surgery (iPTH 4 h) and on the morning of the first postoperative day (iPTH 1st PO). Hypocalcemia was defined by levels of total calcium corrected by serum albumin ≤ 8.0 mg/dL and/or by the presence of symptoms. The most accurate iPTH cut-off point for hypocalcemia prediction was established from a ROC curve comparing both time-points.
Results
The study included 101 patients. The mean age was 52.4 ± 12.9 years, 93 were women (92.1%) and 69 patients underwent total thyroidectomy (68.3%). Hypocalcemia occurred in 25 patients (24.8%), of whom 12 were symptomatic. Total thyroidectomy, longer duration of surgery, surgical complications related to parathyroid glands and lower levels of iPTH 4 h and iPTH 1st PO were associated with postoperative hypocalcaemia (all P < 0.05). Using the ROC curve, the optimal cut-off points were 19.55 pg/mL and 14.35 pg/mL for iPTH 4 h and iPTH 1st PO, respectively. The comparison of the AUC showed no significant difference between these two points of evaluation (0.935 vs. 0.940; P = 0.415).
Conclusions
Serum iPTH levels measured within 4 h or on the first morning after surgery are predictors of postoperative hypocalcemia. Notably, both time-points have the same accuracy to predict postoperative hypocalcemia (with different cutoff points).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Thyroidectomy is one of the most common head and neck surgeries currently performed and although considered a safe procedure with low morbidity, it may present some complications [1, 2]. Postoperative hypocalcemia is the most common complication (particularly following total thyroidectomy) and may increase the length of hospital stay [3,4,5]. The incidence of this event varies widely (1.6–68%), reflecting the considerable heterogeneity among the studies [6,7,8,9,10,11,12,13,14].
The occurrence of postoperative hypocalcemia may be influenced by various factors including surgeon/surgery features (surgeon’s experience, technical difficulties, surgical extent and duration, surgical complications related to parathyroid glands), patients’ characteristics (age, gender), underlying diseases/surgery indications (thyroid autoimmunity, large goiters, thyroid carcinoma, thyrotoxicosis) and biochemical factors (metabolism of calcium, vitamin D, phosphorus, magnesium) [8,9,10,11,12,13,14,15,16,17,18,19,20]. Regardless, the main reason for this complication is acute parathyroid dysfunction after surgery, which may be due to mechanical or thermal trauma, as well as devascularization or accidental removal of the parathyroid glands [9, 10].
Postoperative hypocalcemia may be an isolated laboratory finding or present with symptoms, usually about 24–48 h after the surgery although, less frequently, they may occur in up to 4 days [21,22,23]. In most cases the hypocalcemia is mild and presents with spontaneous resolution, however it may present in a severe form in some cases, leading to concerns in the clinical practice due to the difficulty to recognize it rapidly in those patients and to the possible delay until its manifestation [10, 24]. Serum calcium levels are controlled by the parathyroid hormone (PTH), which has a half-life of approximately 2–5 min [25, 26]. Because of this feature, the levels of intact PTH (iPTH) and/or the percent decline of its serum concentrations during the first hours or days after surgery has been studied and correlated with the occurrence of postoperative hypocalcemia [4,5,6,7,8,9, 20,21,22,23,24, 27,28,29,30,31]. However, the clinical application of postoperative iPTH is currently hindered by the absence of a methodological standardization to define cut-off points. The studies differ regarding hypocalcemia definition criteria, selection of patients, protocols for management and follow-up, as well as methodological flaws such as retrospective design in many of them.
The aim of this study was to evaluate the role of iPTH as an early marker of postoperative hypocalcemia. Using a prospective design, we were able to compare and determine the most accurate cut-off point of iPTH, as well the best time-point after surgery to measure it.
Materials and methods
Patients and study design
We prospectively evaluated a cohort of patients who underwent thyroidectomy (total or partial) between the months of January and November of 2015 at Hospital de Clínicas de Porto Alegre (HCPA), a tertiary care university teaching hospital in southern Brazil. Patients who had previous diagnosis of hypoparathyroidism, renal insufficiency with estimated glomerular filtration (eGFR) rate ≤ 30 mL/min/1.73 m² (according to chronic kidney disease epidemiology collaboration calculator, CKD-EPI) and/or who underwent parathyroid surgery for treatment of parathyroid diseases were excluded from the study. The study protocol was approved by the Institution Ethics Committee of the HCPA and all patients provided written informed consent.
Thyroidectomy
A surgical team composed of general surgeons dedicated to endocrine-related surgery (including thyroidectomies) and training residents, performed all the surgeries. Decisions regarding cervical lymph-node dissection were made at the surgeons’ discretion and based on the current guidelines [32]. During the surgery was not used any form or nerve monitoring neither advance hemostasis system. The indications for thyroidectomy were suspicion of thyroid cancer (current surgery for complementation due to diagnosis of carcinoma in a previous procedure, fine needle aspiration of thyroid nodule with cytological analysis suspect for carcinoma—Bethesda V or VI—as well as for follicular neoplasia—Bethesda IV—if the cell block analysis increased the suspicion of cancer in this last group), hyperthyroidism (Graves’ or Plummer’s disease), and/or nodular goiter (uninodular or multinodular and surgery indicated due to compressive symptoms or toxic goiter).
Surgical complications related to parathyroid glands were defined as accidentally surgical removals of these glands and/or alterations of glandular blood supply (evidenced as alterations in the color of parathyroid glands during the procedure).
Follow-up and treatment
Within the first 4 h after the end of surgery, peripheral venous blood sample was collected and levels of intact parathyroid hormone determined (iPTH 4 h). The majority of the patients (n = 62, 61.4%) collected samples for measurement of iPTH 4 h in the fourth hour after surgery, 12 patients (11.8%) collected the samples between 2 and 3 h after the surgery and the remaining patients collected in the first 2 h after surgery (median 4 h, range 1–4 h). A second laboratory analysis was performed on the first morning following the surgical procedure and iPTH levels determined again (iPTH 1st PO), together with serum phosphorus, albumin, total calcium and magnesium. The gap until this second blood sample collection varied according to the time of the end of surgery, being performed in general about 16–22 h after that. During hospitalization, physical examination was carried out daily focusing on hypocalcemia compatible signs and/or symptoms (paresthesia, Trousseau/Chvostek signs).
Patients with postoperative hypocalcemia during hospitalization (the criteria used for defining outcomes are described below) received the oral administration of calcium carbonate 1 g three times a day and calcitriol 0.5 mcg once daily with dose adjustments according to the periodic medical reassessments. Intravenous calcium gluconate was used in cases of severe hypocalcemia at the attending physician’s discretion. The clinicians were not blinded for the results of PTH measurements.
The time of hospital discharge was defined by the medical team based on postoperative course and laboratory results. The following data were recorded at this time: patient demographics, medical history and complications associated with surgery. All patients were educated on signs and symptoms of hypocalcemia and instructed about calling the research group and/or emergency department in such cases.
The follow-up protocol called for an initial outpatient assessment (4–6 weeks after hospital discharge) including anamnesis, physical examination and laboratory tests collected 2 weeks after surgery (phosphorus, albumin, total calcium, magnesium and iPTH). In a subsequent evaluation 6 months after surgery, those defined as having protracted hypoparathyroidism after the first post-discharge evaluation were reassessed clinically and with laboratory tests. At that time, it was defined whether there was recovery of the parathyroid function or the condition persisted.
Outcomes
Hypocalcemia was defined as total calcium values corrected for albumin ≤ 8.0 mg/dL and/or by presence of characteristic clinical signs in the postoperative period during hospitalization.
Protracted hypoparathyroidism was defined as hypocalcemia in patients with iPTH levels ≤15 pg/mL at the first ambulatory evaluation and/or who continued to need treatment for the condition after that. Permanent hypoparathyroidism was defined if the same situation persisted 6 months after the surgical procedure at the last visit included in the study protocol.
Transient hypoparathyroidism was defined if there was a recovery of parathyroid function at any time in the study follow-up, based on the same criteria described above.
Laboratory analysis
Samples were collected and immediately sent to the Central Laboratory of our Institution to be processed and analyzed, following the standard operating procedures. Serum iPTH and 25-OH-vitamin D (25-OH-D) were evaluated by chemiluminescent microparticle immunoassay method through the ARCHITECT ci 4100 equipment (Abbott Diagnostics, Abbott Park, IL, USA). For serum iPTH reference values were (RV), 15.0–68.3 pg/mL; intra-assay and inter-assay coefficients of variation were 6.1 and 6.4%, respectively. Total serum calcium was evaluated by the NM-BAPTA method and corrected by albumin levels (corrected calcium = 0.8 × [4.0−serum albumin] + serum calcium) (30, 31) with RV 8.6 to 10.0 mg/dL (8.8 to 10.2 mg/dL when aged over 60 years). Serum phosphorus and magnesium were evaluated by the Molybdate UV and colorimetric (xilidil blue) methods, with RV 2.5–4.5 mg/dL and 1.6 to 2.6 mg/dL, respectively. The electrolyte tests were made using Cobas 8000 c702 equipment (Roche Diagnostics, Indianapolis, IN, USA). Serum 25-OH-vitamin D was evaluated by all these tests were conducted at the central laboratory of our Institution.
Statistical analysis
Clinical and laboratory data are reported as the mean ± standard deviation or median and percentiles 25 and 75 (P25-75) for continuous variables and absolute numbers and percentages for categorical variables. Comparative analyses were performed using an unpaired Student’s t, Mann–Whitney U, Fisher test or chi-square test as appropriate.
Postoperative iPTH was evaluated using the area under the Receiver Operating Characteristic (ROC) curve with iPTH (both iPTH 4 h and iPTH 1st PO) as a continuous variable and hypocalcemia as the outcome variable. The two AUC were compared using the DeLong’s test with the R program (R Foundation for Statistical Computing), version 3.1.2.
All tests were two-tailed, and every other analysis was performed using the Statistical Package for Social Science Professional software version 20.0 (SPSS, Chicago, IL, USA). A two-tailed P < 0.05 was considered statistically significant.
Results
Clinical and surgical characteristics
Based on the inclusion and exclusion criteria, 101 patients were included in the study (Fig. 1). The mean age was 52.4 ± 12.9 years, and 93 were women (92.1%). The serum levels of calcium, phosphorus and magnesium on the first postoperative day were 8.42 ± 0.5 mg/dL, 3.76 ± 0.78 mg/dL and 1.8 (1.7–2.0) mg/dL, respectively. The clinical and surgical characteristics of the patients are described in Table 1.
Regarding the surgical approach, 69 (68.3%) underwent total thyroidectomy and 32 (31.7%) partial thyroidectomy. Eleven patients underwent reoperations due to recurrence of goiter or for completion of gland excision following a prior partial procedure. The reasons for thyroidectomy were nodular goiter in 78 (77.2%), suspected thyroid cancer in 20 (19.8%) and hyperthyroidism in 3 patients (3%—2 with Graves’ and 1 with Plummer’s disease). Eighteen (17.8%) patients underwent cervical lymph-node dissection at the discretion of the surgical team. The mean duration of surgery was 133.5 ± 42.6 min.
The postoperative histopathologic analysis results showed benign disease in 76 patients (75.2%) and malignancy in 25 (24.8%). Of the 20 patients who underwent thyroidectomy for suspected thyroid cancer, 16 have the final diagnosis of thyroid cancer and for the remaining 4 patients the malignancy was not confirmed. The nine additional cases were incidental thyroid cancer (primary indication of surgery was nodular or multinodular goiter).
Surgical complications related to parathyroid glands occurred in 18 (17.8%) patients. Other surgical complications included recurrent laryngeal nerve injury and transient vocal cord paresis in two individuals, vascular damage in two patients and postoperative cervical haematoma in one patient. None of these patients needed to re do surgery for hemostasis. There were no deaths related to the surgical procedure or later during the follow-up period.
The length of stay in hospital after surgery was up to 24 h for 8 patients (7.9%), from 24 to 48 h for 37 (36.6%), from 48 to 72 h for 25 (24.8%), and greater than 72 h for 31 patients (30.7%).
Postoperative hypocalcemia
Of the 101 patients, 25 (24.8%) developed postoperative hypocalcemia, 12 of them with signs and/or symptoms of the condition. The time between the end of surgery and onset of hypocalcemia was less than 24 h in 19 (76%) patients, between 24–48 h in 4 (16%) and ≥48 h in 2 (8%).
To investigate the factors associated with hypocalcemia, the patients were grouped according to its presence or absence during the postoperative period (Table 2). Univariate analysis indicated that the group with hypocalcemia had more patients who underwent total thyroidectomy (96 vs. 59.2%, P = 0.001), longer duration of surgery (160.6 ± 41.5 vs. 124.7 ± 39.3 min, P < 0.001) and higher proportion of surgical complications related to parathyroid glands (40 vs. 10.5%, P = 0.002). As expected, this group showed a greater proportion of patients requiring hospitalization time over to 48 h (100 vs. 40.8%, P < 0.001). Regarding lymph node dissection, those patients who underwent lymph node dissection had longer duration of surgery (190.8 ± 38.1 vs. 121.1 ± 32.2 min, P < 0.001), but the proportion of lymph node dissection was not higher in the group of patients with hypocalcemia (28 vs. 14.5%, P = 0.14). There was no significant difference between the groups in age, sex, previous use of possibly interfering medications, indication for surgery or final diagnostic of malignancy.
Regarding comparison of the electrolyte test results on the first postoperative day, the group of patients with hypocalcemia showed lower levels of calcium (7.82 ± 0.42 vs. 8.62 ± 0.35 mg/dL, P < 0.001) and magnesium (1.8 [1.7–1.8] vs. 1.9 [1.7–2.0] mg/dL, P < 0.001). There was no significant difference between the groups in serum levels of phosphorus (4.08 ± 0.89 vs. 3.66 ± 0.71 mg/dl, P = 0.19).
In an additional analysis, the hypocalcemic patients were grouped according to presence or absence of signs and/or symptoms. Symptomatic patients were younger (46.6 ± 12.3 vs. 58.4 ± 11.0 years, P = 0.02) and interestingly, there were no significant differences in the levels of iPTH between symptomatic or asymptomatic patients (6.0 vs. 5.5 and 4.4 vs. 4.4 for iPTH 4 h and iPTH 1st PO, respectively). There was also no significant difference between the groups in other variables (sex, indication, extension and duration of the surgery, biochemical electrolyte test results, lymph-node dissection or final diagnostic of malignancy).
Postoperative parathyroid hormone levels
The mean levels of iPTH within the first 4 h after surgery (iPTH 4 h) and on the first morning following the surgical procedure (iPTH 1st PO) were 46.7 [19.1–69.9] and 39.9 [18.5–63.2] pg/mL, respectively. There was no correlation between the eGFR rate and PTH values (P = 0.45).
Lower levels of both iPTH 4 h (5.5 [3.6–17.5] vs. 58.0 [34.9–81.6], P < 0.001) and iPTH 1st PO (4.4 [3.2–11.8] vs. 48.0 [33.8–68.3], P < 0.001) were observed in the group of patients with postoperative hypocalcemia as compared with the normocalcemic ones. Moreover, the proportion of patients with PTH < 15 pg/mL was higher in those with hypocalcemia at both time-points (73.9 vs. 4.3% and 88 vs. 1.3%, respectively, both P < 0.001). Interestingly, there was no significant difference in serum levels of 25-OH-D between the groups with or without hypocalcemia (Table 2).
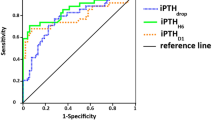
The ROC curves for iPTH 4 h and iPTH 1st PO are represented in Fig. 2. The AUC for iPTH 4 h was 0.93 and an iPTH level of 19.55 pg/mL was the optimal cut-off point to predict postoperative hypocalcemia with a sensibility of 86.9%, specificity of 94.2%, positive predictive value (PPV) of 83.3% and negative predictive value (NPV) of 95.6%. We found similar results for iPTH 1st PO: AUC was 0.94, sensitivity 88%, specificity of 98.6%, PPV of 95.6% and NPV of 96.1% to a cut-off point of 14.35 pg/ml. Indeed, the comparison of the two curves showed no significant difference (Z −0.81, P = 0.41). We also analyzed the presence of post-thyroidectomy hypocalcemia according to the cut-off value found as most accurate for each time-point (Table 3). Hypocalcemia still occurred in 3 patients with iPTH levels higher than these points at both time-points (the same patients, representing 13 and 12% of hypocalcemic ones for iPTH 4 h and 1st PO, respectively). Noteworthy, all these individuals presented hypocalcemia in a mildly and asymptomatic way and showed evolution resembling transient hypoparathyroidism, having treatment withdrawal within the first month after surgery.
Receiver Operating Characteristic (ROC) curve of both time-points of iPTH measurement and post-operative hypocalcemia. iPTH 4 h: intact parathyroid hormone levels measured within the first 4 h after the end of surgery. iPTH 1st PO: intact parathyroid hormone levels measured on the first morning following the surgical procedure
Of the 25 patients with postoperative hypocalcemia, 10 needed to maintain the use of calcium supplements and/or calcitriol until the revaluation of the sixth month (protracted hypoparathyroidism). At the last visit, four of these had a definition of permanent hypoparathyroidism (3.9% of all samples and 16% of patients with hypocalcemia). Their characteristics are shown in Table 4. Of note, the proportion of patients who underwent lymph node dissection was similar between patients with transient/permanent hypoparathyroidism and those who not presented these complications (28 vs. 14.5%; P = 0.14). Similarly, the lymph node dissection was not associated with permanent hypoparathyroidism occurrence (25 vs. 17.5%, P = 0.55).
Discussion
Postoperative hypocalcemia is a common and important complication of thyroid surgery. The identification of those patients at higher risk for this event could optimize their clinical management reducing expenses, hospital length of stay and unnecessary exams, as well as avoiding discomfort to the patient due to overtreatment [9, 30]. Our study indicates that postoperative iPTH measurements, either within 4 h after surgery or on the morning of the first postoperative day, are strong predictors of postoperative hypocalcemia. Interestingly, the performance of the two time-points was comparable, but the best cut-off points were different (19.55 and 14.35, respectively), an aspect that is critical in the clinical application of these data. Our data could have important clinical implications. First, our results indicate that measuring iPTH 4 h or on the first morning after the surgery has the same accuracy to predict postoperative hypocalcemia. Therefore, in the postoperative follow-up, one of these time-points can be chosen to evaluate parathyroid function taking into account the logistics of the hospital and the patient himself. Second, besides the prospective design, we also did not use the iPTH level as an indication for treatment and, and therefore we were able to determine the best cut-off point and its real value as a predictor of postoperative hypocalcemia without the interference of treatment. Serum PTH level is regarded as one of the most sensitive in vivo biomarkers of vitamin D activity [33]. However, of interest, we found no significant differences in serum levels of 25-OH-D between the groups with or without hypocalcemia. The role of iPTH as a predictor for postoperative hypocalcemia has been addressed by several studies [6, 30, 31, 34]. The studies differ regarding selection of patients, duration of follow-up, definitions of outcomes as well as methodological flaws like retrospective design, use of surrogate endpoints and lack of standardized criteria to interpret the iPTH (not all the studies used a receiver operator characteristics to define the cut-off of iPTH). Another important limitation of these studies is that most of them use the iPTH levels as a determinant criterion for treatment and, doing that, the natural course of the disease is not observed. All these aspects preclude the widespread applicability of the postoperative iPTH in clinical practice.
In a prospective study, including 200 patients, Barczyński et al. showed that a cutoff of 10 pg/mL for iPTH 4 h after surgery is most accurate to predict calcium levels under 8.0 mg/dL [24]. On the other hand, in another prospective study that included 1504 patients, Raffaelli et al. observed that even patients with iPTH above 10 pg/mL 4 h after surgery could present postoperative hypocalcemia (18% of this group of patients) [9]. In our study, we showed that the measurements of iPTH at two different time-points (4 h and on the first morning following the surgery) are excellent predictors for postoperative hypocalcemia (ROC curves with AUC of 0.93 and 0.94, respectively). Yet, similarly to the results shown in the studies cited above, 12–13% of the patients in our study presented hypocalcemia with iPTH levels higher than the cut-off found at both measurement times, two of them being asymptomatic and the other mildly symptomatic. It is, however, of interest that all of them recovered the parathyroid function in the first weeks after surgery.
Our study has some limitations and strengths that must be noted. Some concern can arise because we measured iPTH and electrolytes at few time-points and our follow-up time may be short for evaluation of permanent hypoparathyroidism. However, as demonstrated by Ritter et al. in a retrospective analysis of 1054 consecutive thyroidectomies, the majority of 189 patients with low postoperative iPTH (<10 pg/ml) recovered the parathyroid function within the initial months after surgery [35]. Other potential limitations are the relatively small sample size and the fact that the clinicians were not blinded for the results of PTH measurements. However, we believe that the prospective study design and a protocol that did not include the iPTH levels as a criterion for treatment constitute methodological strengths that could overcome these two aspects. The fact that all patients included in the present study were followed at a single institution ensures a similar therapeutic approach and follow-up strategy, thereby enhancing the validity of our data. The prospective design and the protocol that did not include the iPTH levels as a criterion for treatment are also strengths that must be acknowledged.
In conclusion, this prospective cohort study demonstrates that iPTH 4 h or on the first morning after surgery are good predictors of postoperative hypocalcemia. Notably, both time-points have the same accuracy to predict postoperative hypocalcemia (with different cut-off points). These findings might have important implications in the follow-up of those who underwent thyroidectomy enabling earlier and safer discharge for patients. Hence, patients with iPTH above 19.55 pg/ml 4 h after surgery or 14.35 pg/ml on the first morning after surgery can be safely discharged from hospital without supplemental calcium and/or vitamin D.
References
A.A. Padur, N. Kumar, A. Guru, S.N. Badagabettu, S.R. Shanthakumar, M.B. Virupakshamurthy, J. Patil, Safety and effectiveness of total thyroidectomy and its comparison with subtotal thyroidectomy and other thyroid surgeries: a systematic review. J. Thyroid Res. 2016, 7594615 (2016)
G.F. Fezer, R.R. Gama, R.A. Delfes, Nível de paratormônio pós-tireoidectomia total como preditor de hipocalcemia sintomática - estudo prospectivo. Rev. Bras. Cir. Cabeça Pescoço 41, 58–64 (2012)
L. Rosato, N. Avenia, P. Bernante, M. De Palma, G. Gulino, P.G. Nasi, M.R. Pelizzo, L. Pezzullo, Complications of thyroid surgery: analysis of a multicentric study on 14,934 patients operated on in Italy over 5 years. World J. Surg. 28, 271–276 (2004)
S.F. Al-Dhahri, M. Mubasher, F. Al-Muhawas, M. Alessa, R.S. Terkawi, A.S. Terkawi, Early prediction of oral calcium and vitamin D requirements in post-thyroidectomy hypocalcaemia. Otolaryngol. Head. Neck Surg. 151, 407–414 (2014)
C.P. Lombardi, M. Raffaelli, P. Princi, S. Santini, M. Boscherini, C. De Crea, E. Traini, A.M. D’Amore, C. Carrozza, C. Zuppi, R. Bellantone, Early prediction of postthyroidectomy hypocalcemia by one single iPTH measurement. Surgery 136, 1236–1241 (2004)
C.P. Lombardi, M. Raffaelli, P. Princi, C. Dobrinja, C. Carrozza, E. Di Stasio, A. D’Amore, C. Zuppi, R. Bellantone, Parathyroid hormone levels 4 h after surgery do not accurately predict post-thyroidectomy hypocalcemia. Surgery 140, 1016–1023 (2006)
C. Aggeli, G.N. Zografos, A. Nixon, I. Tsipras, Postoperative hypoparathyroidism after thyroid surgery. Hell. J. Surg. 87, 106–110 (2015)
P. Cmilansky, L. Mrozova, Hypocalcemia—the most common complication after total thyroidectomy. Bratisl. Lek. Listy 115, 175–178 (2014)
M. Raffaelli, C. De Crea, G. D’Amato, U. Moscato, C. Bellantone, C. Carrozza, C.P. Lombardi, Post-thyroidectomy hypocalcemia is related to parathyroid dysfunction even in patients with normal parathyroid hormone concentrations early after surgery. Surgery 159, 78–84 (2016)
F. Pattou, F. Combemale, S. Fabre, B. Carnaille, M. Decoulx, J.L. Wemeau, A. Racadot, C. Proye, Hypocalcemia following thyroid surgery: incidence and prediction of outcome. World J. Surg. 22, 718–724 (1998)
W.W. Kim, S.H. Chung, E.J. Ban, C.R. Lee, S.W. Kang, J.J. Jeong, K.H. Nam, W.Y. Chung, C.S. Park, Is preoperative vitamin d deficiency a risk factor for postoperative symptomatic hypocalcemia in thyroid cancer patients undergoing total thyroidectomy plus central compartment neck dissection? Thyroid 25, 911–918 (2015)
S.P. Balasubramanian, Latrogenic/post-surgical hypoparathyroidism: where do we go from here? Endocrine 47, 357–359 (2014)
M.R. Pelizzo, M. Variolo, C. Bernardi, M. Izuzquiza, A. Piotto, G. Grassetto, P.M. Colletti, I. Merante Boschin, D. Rubello, Complications in thyroid resurgery: a single institutional experience on 233 patients from a whole series of 4752 homogeneously treated patients. Endocrine 47, 100–106 (2014)
A. Puzziello, L. Rosato, N. Innaro, G. Orlando, N. Avenia, G. Perigli, P.G. Calò, M. De Palma, Hypocalcemia following thyroid surgery: incidence and risk factors. A longitudinal multicenter study comprising 2,631 patients. Endocrine 47, 537–542 (2014)
O. Edafe, R. Antakia, N. Laskar, L. Uttley, S.P. Balasubramanian, Systematic review and meta-analysis of predictors of post-thyroidectomy hypocalcaemia. Br. J. Surg. 101, 307–320 (2014)
J. Bollerslev, L. Rejnmark, C. Marcocci, D.M. Shoback, A. Sitges-Serra, W. van Biesen, O.M. Dekkers, Endocrinology European Society of Endocrinology Clinical Guideline: treatment of chronic hypoparathyroidism in adults. Eur. J. Endocrinol. 173, G1–G20 (2015)
D.M. Shoback, J.P. Bilezikian, A.G. Costa, D. Dempster, H. Dralle, A.A. Khan, M. Peacock, M. Raffaelli, B.C. Silva, R.V. Thakker, T. Vokes, R. Bouillon, Presentation of hypoparathyroidism: etiologies and clinical features. J. Clin. Endocrinol. Metab. 101, 2300–2312 (2016)
J.C. Nellis, R.P. Tufano, C.G. Gourin, Association between magnesium disorders and hypocalcemia following thyroidectomy. Otolaryngol. Head. Neck Surg. 155, 402–410 (2016)
D.S. Ross, H.B. Burch, D.S. Cooper, M.C. Greenlee, P. Laurberg, A.L. Maia, S.A. Rivkees, M. Samuels, J.A. Sosa, M.N. Stan, M.A. Walter, 2016 American Thyroid Association Guidelines for diagnosis and management of hyperthyroidism and other causes of thyrotoxicosis. Thyroid 26, 1343–1421 (2016)
J.N. Cho, W.S. Park, S.Y. Min, Predictors and risk factors of hypoparathyroidism after total thyroidectomy. Int. J. Surg. 34, 47–52 (2016)
A. Puzziello, R. Gervasi, G. Orlando, N. Innaro, M. Vitale, R. Sacco, Hypocalcaemia after total thyroidectomy: could intact parathyroid hormone be a predictive factor for transient postoperative hypocalcemia? Surgery 157, 344–348 (2015)
T.N. Le , P.D.Kerr , D.E.Sutherland , P.Lambert , Validation of 1-h post-thyroidectomy parathyroid hormone level in predicting hypocalcemia. J. Otolaryngol. Head Neck Surg. 43, 1–5 (2014).
F.A. Vanderlei, J.G. Vieira, F.C. Hojaij, O. Cervantes, I.S. Kunii, M.N. Ohe, R.O. Santos, M. Abrahao, Parathyroid hormone: an early predictor of symptomatic hypocalcemia after total thyroidectomy. Arq. Bras. Endocrinol. Metabol. 56, 168–172 (2012)
M. Barczyński, S. Cichoń, A. Konturek, Which criterion of intraoperative iPTH assay is the most accurate in prediction of true serum calcium levels after thyroid surgery? Lange. Arch. Surg. 392, 693–698 (2007)
M. Hosseini, H.A. Otaghvar, A. Tizmaghz, G. Shabestanipour, P.A. Vahid, Evaluating the time interval for presenting the signs of hypocalcaemia after thyroidectomy. J. Clin. Diagn. Res. 10, PC19–PC22 (2016)
K.M. Rosa, L.L. Matos, C.R. Cernea, L.G. Brandão, V.J. Araújo Filho, Postoperative calcium levels as a diagnostic measure for hypoparathyroidism after total thyroidectomy. Arch. Endocrinol. Metab. 59, 428–433 (2015)
A.A. Carr, T.W. Yen, G.G. Fareau, A.K. Cayo, S.M. Misustin, D.B. Evans, T.S. Wang, A single parathyroid hormone level obtained 4 h after total thyroidectomy predicts the need for postoperative calcium supplementation. J. Am. Coll. Surg. 219, 757–764 (2014)
A. Selberherr, C. Scheuba, P. Riss, B. Niederle, Postoperative hypoparathyroidism after thyroidectomy: efficient and cost-effective diagnosis and treatment. Surgery 157, 349–353 (2015)
P. Lecerf, D. Orry, E. Perrodeau, C. Lhommet, C. Charretier, C. Mor, C. Valat, P. Bourlier, L. de Calan, Parathyroid hormone decline 4 h after total thyroidectomy accurately predicts hypocalcemia. Surgery 152, 863–868 (2012)
M.T. Julian, J.M. Balibrea, M.L. Granada, P. Moreno, A. Alastrue, M. Puig-Domingo, A. Lucas, Intact parathyroid hormone measurement at 24 h after thyroid surgery as predictor of parathyroid function at long term. Am. J. Surg. 206, 783–789 (2013)
R. Promberger, J. Ott, C. Bures, M. Freissmuth, R. Seemann, M. Hermann, Perioperative kinetics of parathyroid hormone in unilateral, primary thyroid surgery. Endocrine 48, 293–298 (2015)
B.R. Haugen, E.K. Alexander, K.C. Bible, G.M. Doherty, S.J. Mandel, Y.E. Nikiforov, F. Pacini, G.W. Randolph, A.M. Sawka, M. Schlumberger, K.G. Schuff, S.I. Sherman, J.A. Sosa, D.L. Steward, R.M. Tuttle, L. Wartofsky, 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26, 1–133 (2016)
A. Valcour, F. Blocki, D.M. Hawkins, S.D. Rao, Effects of age and serum 25-OH-vitamin D on serum parathyroid hormone levels. J. Clin. Endocrinol. Metab. 97, 3989–3995 (2012)
J. Wu, B. Harrison, Hypocalcemia after thyroidectomy: the need for improved definitions. World J. End. Surg. 2, 17–20 (2010)
K. Ritter, D. Elfenbein, D.F. Schneider, H. Chen, R.S. Sippel, Hypoparathyroidism after total thyroidectomy: incidence and resolution. J. Surg. Res. 197, 348–353 (2015)
Acknowledgements
This work has been made possible by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundo de Incentivo a Pesquisa (FIPE) and Programa de Apoio a Núcleos de Excelência (PRONEX) / Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS). We wish to thank the surgeons of our Hospital, Dr. Alceu Migliavacca, Dr. José Ricardo Guimarães, and Dr. Diego Mossmann, for surgical management of our patients.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Filho, E.B.Y., Machry, R.V., Mesquita, R. et al. The timing of parathyroid hormone measurement defines the cut-off values to accurately predict postoperative hypocalcemia: a prospective study. Endocrine 61, 224–231 (2018). https://doi.org/10.1007/s12020-018-1601-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1601-9