Abstract
Occupational skin disease is common. It affects workers more often than reported. Contact dermatitis, both irritant and allergic, accounts for the majority of occupational skin diagnoses. Occupational contact dermatitis (OCD) frequently affects the hands and may have a profound impact on an employee’s ability to perform a job. Severe OCD can affect a worker’s activities of daily living and can even lead to job loss. Numerous irritants have been described in the workplace, from the common (wet work) to the more obscure (warm, dry air). Several contact allergens may be work-related, and the majority of established occupational allergens are also known nonoccupational allergens. Emerging occupational allergens are continually described in the literature. Patch testing is the gold standard for the workup of allergic contact dermatitis. Patch testing in the setting of OCD may require extended or unique allergen trays, as well as a thorough occupational history and collection of workplace Material Safety Data Sheets (MSDS). These MSDS contain valuable information but may not be complete or accurate. Proof of occupational causation can be aided by employing the Mathias criteria. Certain industries and occupations are associated with higher rates of OCD, and as expected, the industries with direct contact with irritants and allergens are highly represented. The differential diagnosis for occupational dermatitis is broad and should be considered when evaluating an employee with suspected OCD. Some other diagnoses to consider include atopic dermatitis, psoriasis, and manifestations of internal disease, as well as an overlap syndrome of more than one diagnosis. OCD treatment should ideally follow the public health hazard controls’ stepwise approach. Prevention and early intervention are key to promoting occupational health and preventing OCD. Multidisciplinary teams have been successful in the treatment of OCD, and newly described topical treatments may provide additional modalities for use in the occupational setting.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Humans typically use their hands to explore the immediate environment. At home, at work, and at play, the hands are one of the main points of human interface. Because of this, we rely strongly on the sensory, tactile, strength, gripping, grasping, pinching, and gross and fine motor skills that our hands provide. Given the numerous interactions our hands have with our environment, it is not entirely surprising that the hand skin can become compromised. However, the skin on the palmar aspect of the hand is particularly adept at resisting physical injury. The epidermis on the palms is 30 times thicker than the epidermis on the eyelid [1]. The stratum corneum, the outermost layer of the epidermis, is a protective barrier that can also thicken in response to repeated trauma or microtrauma in the form of a callus. Alternately, the epidermis can be overcome and damaged by caustic agents or sharp objects. Hands may be the site of sensitization to allergens in our environment, or they may be the site of recurrent exposure to irritants or allergens. Numerous underlying conditions may increase the risk of hand dermatitis.
Ultimately, the cause of dermatitis may be occupational, nonoccupational, or both. While this distinction is not always clear, we will provide tools to help distinguish occupational from nonoccupational dermatitis. Additionally, dermatitis may involve the body, hands, or both. Hands are typically involved in 80–90% of all OCD [2, 3] (see Fig. 1). Several types of dermatitis will be reviewed with emphasis on specific patterns and clues to help the clinician distinguish them. Some dermatoses may be exacerbated by work, and others may be caused by work. Still, others may not be affected by work altogether. We will shed light on a complex and often burdensome subject, stressing both the art and the science of diagnostics and observation in occupational and hand dermatitis.
Epidemiology
Contact dermatitis (CD) accounts for up to 30% of all occupational disease in industrialized nations [4]. CD is the most common occupational skin disorder and represents about 95% of all cases of occupational skin diseases [5].
In the USA, occupational skin disease accounts for 15.2% of all private nonfatal occupational injury [6]. The incidence rate of occupational skin disease is 0.5–1.9 cases per 1000 full-time workers per year [4]. Furthermore, there is a 1-year occupational contact dermatitis prevalence estimate of 20% in the general working population and 21–22% in healthcare workers [7, 8].
However, true epidemiologic data regarding occupational contact dermatitis (OCD) are lacking. The statistics presented are likely underestimated due to gross underreporting. It is thought that mild cases are specifically underreported due to many factors. The Bureau of Labor Statistics has rigorous inclusion criteria and may therefore exclude cases not meeting all measures. Because of the delayed onset of some dermatitis, the eruption may not be obviously work-related. Other reasons for underreporting include cases that are self-treated or only requiring first aid. Luckhaupt et al. estimate that OCD cases are underreported by 85–88%, but even this may be too low an estimate [9].
Selected Global OCD Epidemiology Literature
A review of the literature by Keegel et al. from 1990 to 2007 found that the international incidence of OCD ranged from 1.3 to 8.1 per 10,000 full-time workers per year [10]. Despite OCD being a worldwide issue, OCD epidemiology data from the last decade is lacking. Selected international data is presented.
Australia
In Australia, the incidence of OCD in 2005 was reported to be 2.15 per 10,000 full-time workers per year [11].
China
The 1-year prevalence of OACD in all workers of textile factories in China was 8.5%. However, there was a difference between the prevalence of OACD in managers (3.2%) compared to factory workers (10.8%) [12].
Indonesia
At an Indonesian shoe manufacturing plant, a 29% point prevalence of occupational contact dermatitis was reported [13].
Netherlands
Dutch apprentice nurses were followed prospectively, and a 1-year period prevalence of hand eczema was 23% in the first year of employment, 25% in the second year of employment, and 31% in the third year of employment. The authors correlated increased risk of hand eczema with frequent hand washing at work as well as at home, and wet work outside of nursing [14]. In one study, Dutch construction workers self-reported hand contact dermatitis at a point prevalence rate 32.9%, but experts reported a higher point prevalence rate of 61.4% [15]. Additionally, construction workers had a 25.4% point prevalence of hand skin symptoms [16].
Turkey
A study of pediatric inpatient nurses found that 47.5% of nurses had hand dermatitis, with higher likelihood working in the intensive care unit or infectious disease unit [17].
USA
In poultry processing and manual laborers, 23% of workers noted skin symptoms in a 1-year prevalence study [18]. Latino farm workers were surveyed in North Carolina, and in the early season, 24% reported skin symptoms, while 37% reported skin symptoms in the late season [19]. In a US hospital, 55% of inpatient nurses had hand dermatitis, with higher prevalence of hand dermatitis among intensive care unit (ICU) nurses (65%) than non-ICU nurses (50%) [20].
Societal Impact
The impact of OCD is immense. Workers with OCD may suffer in both their personal and professional lives. In a survey of established cases of OCD, it was reported that over 1 year, 19.9% of cases reported prolonged sick leave and 23% reported job loss [21]. In a 12-year follow-up study, 48% of patients reported prolonged sick leave (a week or longer), 82% of patients changed jobs due to OCD, and, 15% reported job loss or exclusion [22]. There is likely a “healthy worker effect,” described by occupational epidemiologists as “the reduction of mortality or morbidity of occupational cohorts when compared with the general population” [23]. There is also a self-selection that occurs over time; this is the loss of workers who are ill or have bothersome symptoms at work. These employees may find alternate work that suits their health situation better. Because of these factors, we may not be adequately measuring the true attrition of workers over time due to difficulty performing their job because of OCD.
The financial impact of OCD has been estimated. The costs of OCD are numerous and may include the cost of healthcare, workers’ compensation, presenteeism, absenteeism, loss of productivity, and job retraining, among other metrics. In 1985, Mathias estimated annual costs of OCD to be between $222 million and $1 billion [24]. These estimates from over three decades ago are some of the only approximations available, and it is assumed that these costs have only grown with time and inflation. The National Institute of Occupational Safety and Health (NIOSH) estimated the impact of OCD at $1.2 billion in 2004 [25]. Over a decade later, it is likely that these costs have increased.
Occupational contact dermatitis is a term encompassing several diagnoses. The most common dermatoses falling under this term are irritant contact dermatitis (ICD) and allergic contact dermatitis (ACD). Irritant contact dermatitis accounts for 60–80% of all contact dermatitis, while ACD accounts for remaining 20–40%. However, in reality, overlap of these two entities is common (see Figs. 2 and 3).
Allergic Contact Dermatitis
ACD is a type IV delayed-type hypersensitivity reaction. This means that a person must first be sensitized to a hapten prior to eliciting an immune response on re-exposure. The reaction that occurs after re-exposure to an allergen (or cross-reactor) is delayed, which essentially means that it is not immediate in most cases. The delay is usually several hours to a few days. This delay in response may make it difficult to determine the cause of the allergic reaction. Therefore, work-relatedness of dermatitis may be challenging to prove without careful analysis and heightened suspicion.
Most Commonly Reported Allergens in the Workplace (See Table 1)
Rubber Accelerators
Carbamates and thiurams are common occupational allergens [5, 26, 27]. These chemicals are used in the rubber processing (vulcanization) to speed up the reaction. Rubber accelerators are found in elastic which is commonly used in undergarments, socks, waistbands, surgical bonnets, wrists of surgical gowns, hair ornaments, shoe covers, and shoes, among others. In the workplace, they can also be found commonly in both sterile and nonsterile gloves. Rubber accelerators are present in gloves that are both latex and latex-free. Rubber accelerator-free gloves exist in both a sterile and nonsterile form (Table 2). Office workers are often exposed to these allergens in the workplace as rubberized office equipment and computer accessories are common and can cause ACD.
Epoxy Resin
Epoxy resin is a sensitizer and is created by the polymerization process of epichlorhydrin with bisphenol A. Completely cured, this substance is inert, while incompletely cured the substance is allergenic. This allergen can be found in the maritime industry, the electronics industry, dentistry, flooring industry, and industries working with epoxy glues. When epoxy resin is positive on patch testing, it is likely to be clinically relevant [28]. Epoxy resin is a frequent occupational allergen [5, 27, 29].
Formaldehyde
Formaldehyde is a common occupational allergen, and its sources are numerous [27]. First, formaldehyde itself is an allergen and can be found in many occupations such as anatomic pathology, farming, furniture making, wood manufacturing, laboratory work, pest control, and construction [30]. Second, formaldehyde resins are used in clothing to prevent wrinkling; therefore, the textile industries and launderers may contact formaldehyde resins in this form [31]. Third, formaldehyde-releasers are preservatives that release molecules of formaldehyde over time to prevent contamination. These compounds can be found in numerous industries and products and are commonly found in cleansers, detergents, and protective creams [32]. Some formaldehyde-releasers are quaternium-15, imidazolidinyl urea (Germall 115), diazolidinyl urea (Germall II), DMDM hydantoin (Glydant), 2-bromo-2-nitropropane-1,3-diol (bronopol, though this molecule does not require formaldehyde release to act as preservative), and sodium hydroxymethyl glycinate (Table 3).
Nickel
A strong metal used in alloys, nickel is a ubiquitous allergen. It is also the most common contact allergen in North America [35]. Nickel is found in many workplaces, including those involving machines, office supplies, tools, electronics, uniforms, jewelry, keys, and coins, among others. Avoiding this allergen is challenging due to its widespread use. Nickel-free items are available but must be sought out. Where possible, personal protective equipment may be useful or substitution to an alternate item that is nickel-free.
The dimethylglyoxime test can be used to evaluate up to 10 ppm nickel release from items without altering the object [36]. It is a simple, inexpensive in-office test. When the test liquid is placed on a cotton swab and rubbed against an object, the cotton swab turns a shade of pink if nickel release within detection limits is present.
Methyldibroglutaronitrile
A recent study from Europe reported that methyldibromoglutaronitrile (MDGN) is a common occupational allergen [27]. MDGN is a preservative and can be found by itself or in combination with phenoxyethanol. Multiple industries may be exposed to this chemical as it can be found in cleansers, cutting and drilling oils, adhesives, and coolants.
Recently Described Occupational Allergens
Coconut Derivatives
Cocamide diethanolamine (cocamide DEA) is a surfactant that is found in industrial, household, and cosmetic products. In a study in Finland, 19 of 25 reactions were noted to be occupational, with most exposures noted to be in the metal industry in soaps and metalworking fluids [37]. Cocamide MEA, tall oil fatty acids monoethanolamide, is another recently described allergen found in metalworking fluid [38]. Also a coconut derivative, capryldiethanolamine was also found to be an occupational allergen in metalworking fluids, and in this case the ingredient was not listed on the Material Safety Data Sheet [39]. Sodium cocoamphopropionate is a surfactant derived from coconut fatty acids condensed with amino-ethyl ethanolamines. It is found in soaps, shampoos, and conditioners and found to be an allergen in Swedish fast-food workers [40].
Medications
Benzodiazepines were noted to be the cause of facial airborne contact from crushing pills in a cohort of patients [41]. Sevofluorane caused airborne allergic contact dermatitis in a surgeon [42].
Omeprazole is used in the equine industry to treat and prevent stomach ulcers in horses. Cases of occupational contact dermatitis to omeprazole have been reported recently in a horse caregiver and in a horse trainer [43, 44].
Fragrance
Citral caused hand and arm contact dermatitis in a group of beauticians in a high-end spa [45]. Another fragrance, d-limonene, was the causative allergen of OCD and was found in the workplace in machine cleaners, hand soaps, moisturizers, surface cleanser, and dishwashing soaps [46].
In a UK study analyzing OCD 1996–2015, beauticians, hairdressers, and beauty industry workers had a 47 times higher incidence rate ratio of allergy to fragrance when compared to the average rate of all other occupations combined [47].
Isothiazolinones
There have been increased reactions noted to this group of preservatives [27]. Industries and trades at higher risk due to exposure to the isothiazolinones include painting, welding (blacksmiths), machine operating, cosmetology and working in a water cooling tower [48, 49]. Occupational sources of isothiazolinones, some in high concentrations, include paints, varnishes, cleaners and cleansers, and polishing liquids [50]. One unique case reported OCD from an isothiazolinone in the ultrasound gel used by an ultrasonographer [51].
Irritant Contact Dermatitis
Irritant contact dermatitis (ICD) is the most common type of occupational dermatitis, accounting for about 70% of all OCD [5]. ICD is not immune-mediated and requires no prior sensitization, in contrast to allergic contact dermatitis. It results from direct contact with a substance that causes abruption or injury to the skin. It can be clinically difficult to distinguish ICD from ACD. There are numerous irritants in the workplace, and recent literature has helped describe some that are less obvious.
Irritants in the Workplace (See Table 4)
Some irritants in the workplace are well known. Alkalis such as soaps, detergents, and cleansers can cause ICD. Acids can be caustic, as can hydrocarbons such as petroleum and oils. Solvents work to defat the skin and as such can cause ICD. Frictional dermatitis is a subtype of ICD and can be caused by the repetitive handling of objects or materials. Some common examples of items causing frictional dermatitis in the workplace include fabrics, paper, metal objects, and even a steering wheel [52]. Organic materials such as food may also cause ICD [53].
Wet work is perhaps the most common cause of ICD. Wet work can be considered any task where the hands are in contact with moisture or liquids. Of the ICD noted in one study, 68% was due to wet work [5]. Washing hands and performing wet work contributes to hand dermatitis, particularly in healthcare workers [14].
While often thought of as a protective barrier, gloves may actually cause ICD [53]. Prolonged contact of gloves with skin affects the epidermal barrier, and the glove itself may be an irritant even in short contact with skin [54]. Once the hand skin is irritated, gloves may (ironically) make the epidermal barrier more susceptible to allergens or other irritants.
Likely underappreciated, air, particularly warm, dry air, can be an irritant as well [53].
Occupations at Higher Risk for OCD
Many occupations increased the risk for worker occupational hand dermatitis. Some of these occupations are shoemakers and repairers, machinery fitters, mechanics, plumbers, agricultural workers, factory workers, electronics workers, launderers, printers, builders, painters, childcare workers [55], cleaners/washers, food industry workers [5, 55], metal workers, toolmakers, hairdressers, and medical and dental workers [5, 27, 55].
Occupations at higher risk specifically for ICD include hairdressers, nurses, doctors, mechanics, cooks, cleaners, painters, and plumbers [53].
Differential Diagnosis of Hand Dermatitis
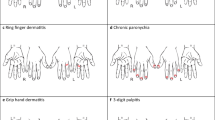
The differential diagnosis of hand dermatitis is broad, including common and less common diagnoses. Aside from contact dermatitis, including allergic and irritant subtypes, one common hand dermatosis to consider is atopic dermatitis (AD). Atopic dermatitis is an immunologic disease and may be found within the clinical triad of atopic dermatitis, allergic rhinitis, and asthma. Investigating a patient’s medical history is helpful when considering AD, and asking about childhood skin eruptions in the antecubital fossae or popliteal fossae (common locations for childhood AD) can aid in eliciting a positive history of atopy. Patients with AD report having “sensitive skin,” and they tend to avoid irritants out of necessity.
Atopic dermatitis may occur on the skin in many areas, including the hands (Fig. 4). Hyperlinear palms, which are palms with increased skin markings, may be found in the setting of AD (but not exclusively to AD) (Fig. 5). Atopic dermatitis on the hands can present in many patterns. Typically, AD appears as eczematous, scaly pruritic papules, and plaques on the dorsum and palmar aspect of the hands, including the fingertips and wrists. Painful fissuring can occur, and retracted cuticles may occur with chronic disease. Chronic AD may be hyperkeratotic and some patients tend to manipulate the thicker scale. A pruritic vesicular or pustular hand dermatitis may occur in the setting of AD, sometimes controversially referred to as “pompholyx” or “dyshidrosis” (Fig. 6). Diagnosis of AD is made clinically or sometimes with skin biopsy. Skin biopsies taken from the hand may not be very helpful in diagnosis. Even skilled dermatopathologists cannot often differentiate ICD, ACD, or AD; therefore, clinical context is of the utmost importance in diagnosis of hand dermatitis.
Staphylococcus is an opportunistic AD infection (or colonization) that can cause a honey-crusted or weeping appearance of the dermatitis. Stable AD with a recent flare should be investigated for secondary staphylococcus infection.
Recent research has elucidated some of the barrier issues that occur in AD. These patients lack essential building blocks in the skin that help protect the epidermis from the environment. Filaggrin mutations are associated with increased risk of skin barrier dysfunction and AD [56]. With an innately impaired epidermal barrier, patients with AD are susceptible to irritants and allergens, thus also have an increased risk of hand dermatitis. AD can co-occur with ICD and/or ACD.
Another inflammatory dermatosis, psoriasis can manifest as hand dermatitis. Common areas of the skin involved in psoriasis include the extensor elbows, extensor knees, nuchal scalp, postauricular scalp, and gluteal cleft. Lesions are classically salmon-colored, erythematous, papules, and plaques with a silvery, thickened scale. Pruritus is common and patients may manipulate the thick scale. Psoriatic fingernails and toenails may appear pitted and thickened. Distal onycholysis and “oil spots” under the nail are other psoriatic nail findings. Psoriasis may occur on the soles as well. In the setting of a history of psoriatic arthritis, psoriasis can be considered as a cause for hand dermatitis. Diagnosis is made clinically and sometimes with skin biopsy.
Though there are many types, keratoderma is a consideration in the differential of hand dermatitis. Keratoderma is a thickening of the palmar and plantar skin that can appear somewhat homogenous and yellowish or waxy, or simply desquamative. It may be inherited (ask family history) or may be acquired. Variations include partial involvement of the palms and soles, striated, and inflammatory, among others. Diagnosis is based on history and sometimes skin biopsy.
Tinea manuum is an uncommon superficial fungal skin infection. It tends to occur in the setting of tinea pedis and onychomycosis, and a classic “two foot, one hand” distribution, but can be bilateral. Tinea manuum affects the palm and appears as dry, somewhat powdery scale that is KOH positive on microscopic examination. Rarely, an advanced fungal skin infection of the dorsum of the hands called “Majocchi’s granuloma” may be present. In this case, erythematous pustules occur at areas where fungus has invaded the hair follicle. Diagnosis can be made clinically, with KOH examination if an adequate sample can be obtained, or by biopsy.
Squamous cell carcinoma (SCC) can masquerade as hand dermatitis. Typically on the (sun-exposed) dorsum of the hand, SCC may appear as persistent scaly papules and plaques. Actinic keratoses (AKs), pre-cancerous lesions caused by sun exposure, may present in a similar fashion, though perhaps with thinner scale. The diagnosis of AKs may be made clinically, and diagnosis of SCC is made by skin biopsy.
Very rarely, cutaneous T cell lymphoma (CTCL) can occur on the hands. The clinical appearance of CTCL can vary and diagnosis is made by skin biopsy.
Keratolysis exfoliativa is a condition found more often in younger patients and is characterized by recurrent peeling of the palms. It is often worsened with hand sweating and summer heat. Vesicles may precede the superficial desquamation.
Hand Dermatitis as a Skin Manifestation of Internal Disease or Immune Reaction
Bullae and scarring on the dorsum of the hands can be associated with select porphyrias and pseudoporphyria. Biopsy, direct immunofluorescence, medical history, and labwork help make this diagnosis.
Connective tissue diseases such as dermatomyositis, polymyositis, scleroderma, and lupus can affect the hands. Prominent capillaries surrounding the cuticle may be present in any of these diseases. Gottron’s sign in dermatomyositis is the appearance of violaceous, erythematous papules (Gottron’s papules), and plaques on the dorsum of the metacarpophalangeal joints and fingers. A rare manifestation of polymyositis or dermatomyositis is called “mechanic’s hands.” The hands are hyperkeratotic, rough, and fissured, mimicking those of a mechanic. Scleroderma affects the fingers with sclerotic tapering and even ulceration of the fingertips.
Granuloma annulare (GA) is an inflammatory dermatosis with a predilection for joints and the dorsum of the hands. GA appears as annular, granulomatous, somewhat pruritic papules and plaques.
Though rare, paraneoplastic states can be suggested by hand dermatitis. Mechanic’s hands, as described above, are one state suggestive of underlying malignancy. Tripe palms are usually associated with internal malignancy and presents with velvety, thickened palms suggestive of animal intestine, thus “tripe.” Bazex syndrome, acrokeratosis paraneoplastica, may occur in association with aerodigestive cancers. It is characterized by psoriasiform papules and plaques on the acral skin.
Patch Testing and the Occupational Patient
The gold standard in the workup of a patient with possible ACD is patch testing. Patch testing elicits a delayed-type hypersensitivity reaction, a type IV allergic response. Patch testing is at least a week-long test in which haptens are placed on the back under occlusion and skin reaction is observed. Typically, allergens are standardized and chosen for the patient based on exposure history and clinical suspicion. Allergens are placed in chambers under occlusion on the skin, usually the back, for 48 h. The patches are then removed 48 h later, and a preliminary reading can be performed, but a final reading is performed at 96 or more hours. Positive reactions are noted and correlated to the patient’s dermatitis and exposures, including those in the workplace. Avoidance is then pursued for at least 2 months and if the patient improves then the positive allergen(s) may be the cause of the eruption.
Extended patch testing is recommended for occupational contact dermatitis given the unique nature and broad implication of workplace allergens. First, an extensive history, including a work history is important. Obtaining a thorough work history will enable targeted patch testing for optimal results. Several questions should be included and are summarized in Table 5.
If substances are contacted at work and may be causative, obtaining Material Safety Data Sheets (MSDS) from the workplace may be helpful. While MSDS are often useful in the workup of a patient with suspected OCD, they also have many deficiencies. MSDS are mandated to include hazardous chemical name, chemical properties, physical hazards, route of entry, known exposure limits, carcinogenicity, cleanup practices, control measures, and company contact information. With regard to OCD, much information is lacking in the MSDS. Unfortunately, the following are not required: if the substance is proprietary, specific chemical names are often avoided and a general category may be used, irritants and sensitizers do not need to be listed if < 1% concentration, and the MSDS often do not address prevention of sensitization and irritancy [57]. Contacting the manufacturing company may be helpful in some cases. Otherwise, it is important to avoid placing any unknown allergen on the skin. Some chemicals can be compounded or diluted to an acceptable concentration as outlined by DeGroot, but otherwise caution is advised [58]. Sometimes a surrogate standardized allergen can be used.
Standardized trays are available for patch testing. The only FDA-approved patch test tray is the thin-layer rapid use epicutaneous (T.R.U.E.) test [59]. It consists of 35 allergens along with a negative control and may not detect all relevant allergens [35]. Off-label use of standardized allergens available from several manufacturers is recommended for suspected OCD. This extended patch testing is paramount to possibly detect all relevant allergens. It is a time-consuming and resource-heavy process. Numerous standard and specialty trays are available from several manufacturers. Some trays that have occupational impact include hairdressing series, oils and coolants, metals, plastics and glues, shoe series, baker series, and textile and dyes. These are usually tested in conjunction with a standard series of 60–80 allergens.
Determining dermatitis causation can be challenging, and the Mathias criteria (Table 6) can be of help. The Mathias criteria is a series of seven questions to help determine occupational causation [60]. An answer of “yes” to four or more of the seven criteria yields a greater than 50% probability of occupational cause [61]. This, in turn, provides a “reasonable degree of medical certainty” and can be used in occupational cases. These questions are as follows: Is the clinical appearance consistent with contact dermatitis? Are there workplace exposures to potential cutaneous irritants or allergens? Is the anatomic distribution of the dermatitis consistent with cutaneous exposure in relation to work tasks? Is there a temporal association between onset of dermatitis and exposure consistent with contact dermatitis? Are nonoccupational exposures excluded as probable causes? Does dermatitis improve away from work exposure to the suspected allergen or irritant? Do patch or provocation tests identify a probable causal agent [60]? Again, it is important to consider ICD, AD, as well as ACD and overlap dermatitis.
Approach to Occupational Exposure
The preferred approach to occupational exposure follows the stepwise approach to hazard controls in the workplace (Fig. 7) [62]. First, elimination is the removal of the hazard in the workplace; this is the most effective preventive health approach. Second, substitution is replacing the hazard itself. There have been some successes using this approach with latex in gloves and chromates in cement. Third, isolation and engineering controls is the removal of contact of the hazard with workers. Fourth, administrative controls are changing the way a job is performed, such as rotating workers through tasks. Finally, personal protection is often the first considered but the least effective. This is protecting the worker from the hazard with personal protective equipment. Matching the correct glove to the task can be helpful [63].
National Institute of Occupational Safety and Health (NIOSH) hierarchy of hazard controls [62]
Ultimately, the goal of worker recovery and skin health will be avoidance of any allergens and irritants in the workplace. This can be challenging with omnipresent allergens and cross-reactors. Additionally, modified work may need to be considered with clear limitations. Coordination with employee health or an occupational health provider in the workplace can be helpful. Ultimately, permanent work restrictions may be required if the stepwise approach to hazard control is not possible or the approach does not improve the employee.
Ideally, prevention is the best approach to occupational health. A thorough review of reported primary prevention strategies was written by Zack et al. and suggests that primary prevention of OCD through education can be successful [64]. For example, evidence-based education intervention was successful in a study. Full-time hospital cleaners were offered a 1-h voluntary hand protective behavior lecture/intervention at hire. At 3 months post-intervention, the cleaners who experienced the education had decreased hand cleansing, better knowledge of hand preservation, and improved hand dermatitis compared to cleaners who did not attend the education/intervention [65].
Recent literature has proven that early intervention can be helpful in the workplace. Multidisciplinary teams aid in successful return to work for injured workers with OCD. A German model included inpatient and outpatient care, and 87% remained in workforce [66]. A Netherlands model included a dermatologist, education nurse, occupational medicine physician and was helpful with return to work [67]. A similar Canadian model employing a return to work coordinator and physicians in a graduated return to work program was also reported [68].
Early educational intervention in employees with occupational hand dermatitis may be helpful. Training on hand dermatitis prevention was offered to workers with OCD as a secondary prevention strategy. Workers who adhered to training recommendations fared better both in symptoms and objective skin barrier findings [69].
What Is New in Treating Hand Dermatitis
Aside from the traditional approach to treating hand dermatitis, new treatments have been described. Barrier repair topical treatments aim to “repair” the impacted epidermal barrier. Ceramides are one of the described ingredients that can help restore a more intact barrier in hand dermatitis. In contrast, barrier protect topical treatments aim to shield the epidermis from additional irritants. Barrier protect creams with dimethicone may help prevent ICD at high dose of application (2–20 times the recommended application) [70]. Studies are variable in demonstrating any benefit from barrier creams compared to regular emollients. As more data helps determine whether these barrier protect or repair topical treatments have a role in CD treatment and prevention, a recommendation of frequent use of moisturizers is important.
Keeping in mind that gloves themselves can be irritating, one method has been helpful, at least in short-contact tasks. Wearing cotton liners under gloves can be helpful in hand dermatitis [71]. While this is not a long-term solution, it may be one that allows an employee tolerate a task if gloves and cotton liners can be changed frequently. In personal experience (HPL), this approach is variable in patient tolerance and improvement.
The traditional approach to hand dermatitis includes decreasing inflammation. Topical corticosteroids are usually the first treatment considered. Starting with a potent topical steroid application once to twice daily will help to decrease dermatitis, but should be decreased and ultimately discontinued as soon as possible. Side effects of chronic use of topical steroids can include epidermal atrophy, which in turn can actually make the skin more susceptible to irritants and even allergens. When possible, topical calcineurin inhibitors can be used as an alternative or adjunct to topical steroids. A topical phosphodiesterase-4 (PDE4) inhibitor, crisaborole, was approved in 2016 for mild to moderate AD treatment.
Phototherapy is an excellent option for hand dermatitis when available. Traditionally, ultraviolet A light devices have been used for hand dermatitis and can be used with or without a topical or oral psoralen. Ultraviolet B light can also be used for hands.
Systemic medications are an option for patients with recalcitrant hand dermatitis. Oral retinoids such as acitretin can be used for psoriasiform or hyperkeratotic hand dermatitis. Systemic immunosuppressants, such as mycophenolate mofetil, cyclosporine, methotrexate, and azathioprine can be helpful in these patients. Each systemic medication has potential side effects and risks and should be used judiciously. Recently approved for treatment of psoriasis, apremilast is an oral PDE4 inhibitor. It can be considered for treatment of hand psoriasis. Finally, a new injectable biologic medication has been approved for AD. Dupilumab is an antagonist of the interleukin-4 receptor alpha and is used to treat moderate to severe AD.
Discussion
Hand dermatitis is a challenging diagnosis that can be multifactorial. Occupational hand dermatitis has even greater implication as an affected employee may not be able to complete work tasks successfully. Brisk identification of hand dermatitis in the worker and subsequent treatment of cause(s) is the ideal course in this patient population. Unfortunately, the cause of hand dermatitis is not often obvious and is often a “layered” diagnosis. That is, multiple diagnoses can co-exist making the diagnosis more difficult.
Approach to the patient with suspected OCD is somewhat distinct. While a thorough family and past medical history is important, an extensive work history is also helpful. The more a provider knows about a workplace and how a task is completed, the better the chance the provider will have in determining if there is a workplace contribution or cause to the dermatitis. Although workplace visits are rarely reimbursed, sometimes videos of a task can be of use. Encouraging patients to bring items from work that may be of concern, such as personal protective equipment, used gloves, uniforms, and MSDS helps to provide a full picture of the environment in which the patient works.
Once causation of an OCD is established, it is important to work closely with the patient’s employer. Work restrictions may be necessary, but working through the hazard control approach is preferred. Employers with robust occupational health resources may be more likely to implement changes in hazard controls and accommodate restrictions. Smaller companies may not be able to accommodate work restrictions, and there is the possibility for any worker to have job loss from OCD. Job retraining is not often available or feasible. For these reasons, patients with OCD should be managed with care and resolve.
Conclusions/Summary
Occupational skin disease is a largely under-captured entity. Contact dermatitis is the most common form of occupational skin disease. Allergic and irritant contact dermatitis can be difficult to distinguish and they can co-exist. Hand dermatitis is the most common presentation of OCD. Other diagnoses should be considered when evaluating worker hand dermatitis.
Numerous allergens and irritants exist in the workplace. Patch testing for ACD is the gold standard for diagnosis. While common occupational allergens are known, novel and emerging allergens should also be considered. A thorough workplace history is paramount for a successful patch test. Patch testing with the T.R.U.E.™ test can be considered, with referral for comprehensive panels when indicated.
Occupational dermatitis will continue if the exposure continues, and approach to workplace dermatitis is unique. Using hazard controls in the workplace is the preferred stepwise approach to worker protection. Multidisciplinary teams and educational interventions may play a role in prevention and rehabilitation of patients with OCD. Diagnosing and treating OCD in a worker can greatly improve the patient’s quality of life and hopefully maintain employment.
References
SEER Training: Layers of the skin (n.d.) Retrieved from https://training.seer.cancer.gov/melanoma/anatomy/layers.html
Bhatia R, Sharma VK, Ramam M, Sethuraman G, Yadav CP (2015) Clinical profile and quality of life of patients with occupational contact dermatitis from New Delhi, India. Contact Dermatitis 73(3):172–181. https://doi.org/10.1111/cod.12411
Koch P (2001) Occupational contact dermatitis. Am J Clin Dermatol 2(6):353–365. https://doi.org/10.2165/00128071-200102060-00002
Diepgen TL, Coenraads PJ (1999) The epidemiology of occupational contact dermatitis. Int Arch Occup Environ Health 72(8):496–506. https://doi.org/10.1007/s004200050407
Caroe TK, Ebbehoj N, Agner T (2013) A survey of exposures related to recognized occupational contact dermatitis in Denmark in 2010. Contact Dermatitis 70(1):56–62
US Bureau of Labor Statistics. 2014 Survey of occupational injuries & illnesses. Summary estimates charts package. Curr Popul Surv 2015:1Y16. Available at: http://www.bls.gov/iif/oshwc/osh/os/osch0054.pdf. Accessed November 18, 2017
Ibler KS, Jemec GBE, Flyvholm MA, Diepgen TL, Jensen A, Agner T (2012) Hand eczema: prevalence and risk factors of hand eczema in a population of 2274 healthcare workers. Contact Dermatitis 67(4):200–207
Luk N-MT, Lee H-CS, Luk C-KD, Cheung YYA, Chang MC, Chao VKD, Ng SC, Tang LSN (2011) Hand eczema among Hong Kong nurses: a self-report questionnaire survey conducted in a regional hospital. Contact Dermatitis 65(6):329–335
Luckhaupt SE, Dahlhamer JM, Ward BW et al (2010) Prevalence of dermatitis in the working population, United States, 2010 National Health Interview Survey. Am J Ind Med 56(6):625–634
Keegel T, Moyle M, Dharmage S, Frowen K, Nixon R (2009) The epidemiology of occupational contact dermatitis (1990–2007): a systematic review. Int J Dermatol 48(6):571–578
Keegel T, Cahill J, Noon A et al (2005) Incidence and prevalence rates for occupational contact dermatitis in an Australian suburban area. Contact Dermatitis 52(5):254–259
Chen YX, Gao BA, Cheng HY, Li LF (2017) Survey of occupational allergic contact dermatitis and patch test among clothing employees in Beijing. Biomed Res Int 2017:10 pages
Febriana SA, Hardyanto S, Coenraads PJ (2014) Occupational skin hazards and prevalence of occupational skin diseases in shoe manufacturing workers in Indonesia. Int Arch Occup Environ Health 87(2):185–194
Visser MJ, Verberk MM, van Dijk FJ, Bakker JG, Bos JD, Kezic S (2013) Wet work and hand eczema in apprentice nurses; part I of a prospective cohort study. Contact Dermatitis 70(1):44–55
Timmerman JG, Heederik D, Spee T, van Rooy FG, Krop EJM, Rustemeyer T, Smit LAM (2017) Contact dermatitis is an unrecognized problem in the construction industry: comparison of four different assessment methods. Am J Ind Med 60(10):879–888
Timmerman JG, Heederik D, Spee T, Smit LAM (2014) Skin symptoms in the construction industry: occurrence and determinants. Am J Ind Med 57(6):660–668
Özyazıcıoğlu N, Sürenler S, Tanrıverdi G (2010) Hand dermatitis among paediatric nurses. J Clin Nurs 19((11–12))
Quandt SA, Newman JC, Pichardo-Geisinger R et al (2013) Self-reported skin symptoms and skin-related quality of life among Latino immigrant poultry processing and other manual workers. Am J Ind Med 57(5):605–614
Arcury TA, Quandt SA, Mellen BG (2003) An exploratory analysis of occupational skin disease among Latino migrant and seasonal farmworkers in North Carolina. J Agric Saf Health 9(3):221–232
Lampel HP, Patel N, Boyse K, O’Brien SH, Zirwas MJ (2007) Prevalence of hand dermatitis in inpatient nurses at a United States hospital. Dermatitis 18(3):140–142
Cvetkovski R, Rothman K, Olsen J et al (2005) Relation between diagnoses on severity, sick leave and loss of job among patients with occupational hand eczema. Br J Dermatol 152(1):93–98. https://doi.org/10.1111/j.1365-2133.2005.06415.x
Meding B, Lantto R, Lindahl G, Wrangsjö K, Bengtsson B (2005) Occupational skin disease in Sweden—a 12-year follow-up. Contact Dermatitis 53(6):308–313. https://doi.org/10.1111/j.0105-1873.2005.00731.x
Shah D (2009) Health worker effect phenomenon. Indian J Occup Environ Med 13(2):77–79
Mathias CG (1985) The cost of occupational skin disease. Arch Dermatol 121(3):332–334. https://doi.org/10.1001/archderm.121.3.332
Blanciforti LA (2011) Eonomic burden of dermatitis in US workers. J Occup Environ Med 53(1):99. https://doi.org/10.1097/jom.0b013e318209a6b9
Rietschel RL, Mathias CG, Fowler JF Jr et al (2002) North American Contact Dermatitis Group. Relationship of occupation to contact dermatitis (OCD): evaluation in patients tested from 1998 to 2000. Am J Contact Dermat 13(4):170–176
Pesonen M, Jolanki R, Filon FL et al (2015) Patch test results of European baseline series among patients with occupational contact dermatitis across Europe—analyses of European Surveillance System on Contact Allergy network, 2002-2010. Contact Dermatitis 72(3):154–163
Bangsgaard N, Thysse JP, Menne T et al (2012) Contact allergy to epoxy resin: risk occupations and consequences. Contact Dermatitis 67(2):73–77. https://doi.org/10.1111/j.1600-0536.2012.02072.x
Arrandale VH, Liss GM, Tario SM et al (2012) Occupational contact allergens: are they also associated with occupational asthma? Am J Ind Med 55(4):353–360
Goyer N, Bégin D, Beaudry C et al (2006) Prevention guide: formaldehyde in the workplace. Available at http://biology.mcgill.ca/safety/Formaldehyde_guide.pdf. Retrieved on November 19, 2017
Reich HC, Warshaw EM (2010) Allergic contact dermatitis from formaldehyde textile resins. Dermatitis 21(2):65–76. https://doi.org/10.2310/6620.2010.09077
Aalto-Korte K, Kuuliala O, Suuronen K, Alanko K (2008) Occupational contact allergy to formaldehyde and formaldehyde releasers. Contact Dermatitis 59(5):280–289. https://doi.org/10.1111/j.1600-0536.2008.01422.x
Cameo Chemicals “1-(3-CHLOROALLYL)-3,5,7-TRIAZA-1-AZONIAADAMANTANE CHLORIDE” Website. Available at: https://cameochemicals.noaa.gov/chemical/18075. Accessed November 18, 2017
Chempoint “Suttocide A Hydroxymethylglycinate” Website. Available at: https://www.chempoint.com/products/catalog/ashland/ashland-personal-care-preservatives/suttocide-a-sodium-hydroxymethylglycinate. Accessed November 18, 2017
Dekoven JG, Warshaw EM, Belsito DV et al (2017) North American Contact Dermatitis Group Patch Test Results 2013–2014. Dermatitis 28(1):33–46. https://doi.org/10.1097/DER.0000000000000225
Chemo Nickel Test. Chemotechnique Diagnostics. Available at: https://www.chemotechnique.se/ckfinder/userfiles/files/Nickel%20Test%20Package%20Insert,%20version%201%20-%20Digital.pdf. Accessed November 18, 2017
Aalto-Korte K, Pesonen M, Kuuliala O, Suuronen K (2014) Occupational allergic contact dermatitis caused by coconut fatty acids diethanolamide. Contact Dermatitis 70(3):169–174
Aalto-Korte K, Pesonon M, Kuuliala O et al (2013) Contact allergy from metalworking fluid traced to tall oil fatty acids monoethanolamide. Contact Dermatitis 69(5):316–317
Suuronen K, Aalto-Korte K, Suomela S (2014) Contact allergy to capryldiethanolamine in metalworking fluids. Contact Dermatitis 72(2):120–121
Hagvall L, Bråred-Christensson J, Inerot A (2014) Occupational contact dermatitis caused by sodium cocoamphopropionate in a liquid soap used in fast-food restaurants. Contact Dermatitis 71(2):122–124
Swinnen I, Ghys K, Kerre S, Constandt L, Goossens A (2014) Occupational airborne contact dermatitis from benzodiazepines and other drugs. Contact Dermatitis 70(4):227–232
Burches E, Revert A, Martin J et al (2015) Occupational systemic allergic dermatitis caused by sevoflurane. Contact Dermatitis 71(1):62–63
Alwan W, Banerjee P, White IR (2014) Occupational contact dermatitis caused by omeprazole in a veterinary medicament. Contact Dermatitis 71(6):376–376
Al-Falah K, Schachter J, Sasseville D (2014) Occupational allergic contact dermatitis caused by omeprazole in a horse breeder. Contact Dermatitis 71(6):377–378
De Mozzi P, Johnston GA (2014) An outbreak of allergic contact dermatitis caused by citral in beauticians working in a health spa. Contact Dermatitis 70(6):377–379
Pesonen M, Suomela S, Kuuliala O, Henriks-Eckerman ML, Aalto-Korte K (2014) Occupational contact dermatitis caused by D-limonene. Contact Dermatitis 71(5):273–279
Montgomery RL, Agius R, Wilkinson SM, Carder M (2017) UK trends of allergic occupational skin disease attributed to fragrances 1996-2015. Contact Dermatitis 78:33–40. https://doi.org/10.1111/cod.12902
Schwensen JF, Menné T, Andersen KE, Sommerlund M, Johnasen JD (2014) Occupations at risk of developing contact allergy to isothiazolinones in Danish contact dermatitis patients: results from a Danish multicenter study (2009-2012). Contact Dermatitis 71(5):295–302
Maor D, Nixon R (2015) Allergic contact dermatitis to methylchloroisothiazolinone/methylisothiazolinone in cooling tower technicians. Dermatitis 26(1):62–64
Friis UF, Menné T, Flyvholm MA (2014) Isothiazolinones in commercial products at Danish workplaces. Contact Dermatitis 71(2):65–74. https://doi.org/10.1111/cod.12235
Madsen JT, Broesby-Olsen S, Andersen KE (2014) Undisclosed methylisothiazolinone in an ultrasound gel causing occupational allergic contact dermatitis. Contact Dermatitis 71(5):312–313
McMullen E, Gawkrodger DJ (2006) Physical friction is under-recognized as an irritant that can cause or contribute to contact dermatitis. Br J Dermatol 154(1):154–156. https://doi.org/10.1111/j.1365-2133.2005.06957.x
Friis UF, Menné T, Schwensen JF, Flyvholm MA, Bonde JPE, Johansen JD (2014) Occupational irritant contact dermatitis diagnosed by analysis of contact irritants and allergens in the work environment. Contact Dermatitis 71(6):364–370
Ramsing DW, Agner T (1996) Effect of glove occlusion on human skin (II). Contact Dermatitis 34(4):258–262
Meding B, Jarvholm B (2002) Hand eczema in Swedish adults—changes in prevalence between 1983 and 1996. J Investig Dermatol 118(4):719–723. https://doi.org/10.1046/j.1523-1747.2002.01718.x
Weidinger S, Illig T, Baurecht H, Irvine AD, Rodriguez E, Diaz-Lacava A, Klopp N, Wagenpfeil S, Zhao Y, Liao H (2006) Loss-of-function variations within the filaggrin gene predispose for atopic dermatitis with allergic sensitizations. J Allergy Clin Immunol 118(1):214–219. https://doi.org/10.1016/j.jaci.2006.05.004
Friis UF, Menné T, Flyvholm MA, Bonde JPE, Johansen JD (2015) Difficulties in using the material safety data sheets to analyse occupational exposures to contact allergens. Contact Dermatitis 72(3):147–153. https://doi.org/10.1111/cod.12314
deGroot AC (2008) Patch testing: test concentrations and vehicles for 4350 chemicals, 3rd edn. acdegroot publishing, Wapserveen
Thin-layer rapid-use epicutaneous test. Thin-layer rapid-use epicutaneous test (TRUE-test). Available at: https://www.smartpractice.com/dermatologyallergy/pdfs/TRUE%20TEST%20Prescribing%20Information.pdf. Accessed November 19, 2017
Mathias CG (1989) Contact dermatitis and workers’ compensation: criteria for establishing occupational causation and aggravation. J Am Acad Dermatol 20(5):842–848
Ingber A, Merims S (2004) The validity of the Mathias criteria for establishing occupational causation and aggravation of contact dermatitis. Contact Dermatitis 51(1):9–12
Centers for Disease Control and Prevention. National Institute for Occupational Safety and Health (NIOSH) “Hierarchy of Controls” Website. Available at: https://www.cdc.gov/niosh/topics/hierarchy/default.html. Accessed November 5, 2017
Forsberg K, Mansdorf SZ (2001) Quick selection guide to chemical protective clothing. Wiley, Hoboken
Zack B, Arrandale VH, Holness DL (2017) Preventing occupational skin disease: a review of training programs. Dermatitis 28(3):169–182
Clemmensen KK, Randdboll I, Ryborg MF, Ebbehoj NE, Agner T (2015) Evidence-based training as primary prevention of hand eczema in a population of hospital cleaning workers. Contact Dermatitis 72(1):47–54
Weisshaar E, Skudlik C, Scheidt R, Matterne U, Wulfhorst B, Schönfeld M, Elsner P, Diepgen TL, John SM, for the ROQ Study Group (2013) Multicentre study rehabilitation of occupational skin diseases—optimization and quality assurance of inpatient management (ROQ)—results from 12-month follow-up. Contact Dermatitis 68(3):169–174
van Gils RF, Groenewoud K, Boot CR et al (2012) Process evaluation of an integrated, multidisciplinary intervention programme for hand eczema. Contact Dermatitis 66(5):254–263
Chen J, Gomez P, Kudla I, DeKoven J, Holness DL, Skotnicki S (2016) Return to work for nurses with hand dermatitis. Dermatitis 27(5):308–312. https://doi.org/10.1097/DER.0000000000000215
Mauro M, De Giusti V, Bovenzi M et al (2017) Effectiveness of a secondary prevention protocol for occupational contact dermatitis. J Eur Acad Dermatol Venereol 31(4):656–663. https://doi.org/10.1111/jdv.13947
Schliemann S, Petri M, Elsner P (2014) Preventing irritant contact dermatitis with protective creams: influence of the application dose. Contact Dermatitis 70(1):19–26
Adisesh A, Robinson E, Nicholson PJ, Sen D, Wilkinson M, Standards of Care Working Group (2013) UK standards of care for occupational contact dermatitis and occupational contact urticaria. Br J Dermatol 168(6):1167–1175
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Lampel, H.P., Powell, H.B. Occupational and Hand Dermatitis: a Practical Approach. Clinic Rev Allerg Immunol 56, 60–71 (2019). https://doi.org/10.1007/s12016-018-8706-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-018-8706-z