Abstract
Celiac disease is a complex immune-mediated gluten-sensitive enteropathy with protean clinical manifestations. It is manifest in genetically predisposed individuals who ingest gluten in varying amounts. In broad terms, it is thought to affect 1% of the population in the USA. More specifically, the prevalence increases drastically from 1:133 in patients not-at-risk, to 1:56 in symptomatic patients, to 1:39 in patients with a second-degree relative with the diagnosis, and to 1:22 in patients with a first-degree relative with the diagnosis. It may be associated with several immune-mediated phenomena, autoimmune diseases, and complicated by vitamin and other trace element deficiencies, bone disease, and malignancy. Our understanding of celiac disease has evolved rapidly over the past two decades. This has led to several lines of enquiry on the condition and potential treatment options. More recently, several entities including gluten intolerance, non-celiac gluten sensitivity, and seronegative celiac disease have been described. These conditions are distinct from allergies or intolerance to wheat or wheat products. There are challenges in defining some of these entities since a large number of patients self-report these conditions. The absence of confirmatory diagnostic tests poses an added dilemma in distinguishing these entities. The differences in spectrum of symptoms and highlights of the variability between the pediatric and adult populations have been studied in some detail. The role of screening for celiac disease is examined in both the general population and “at risk” populations. Diagnostic strategies including the best available serologic testing, utility of HLA haplotypes DQ2 and DQ8 which are seen in over 90% of patients with celiac disease as compared with approximately 40% of the general population, and endoscopic evaluation are also reviewed. Comprehensive nutritional management after diagnosis is key to sustained health in patients with celiac disease. Simple algorithms for care based on a comprehensive multidisciplinary approach are proposed. Refractory and non-responsive celiac diseases in the setting of a gluten-free diet are examined as are novel non-dietary therapies. Finally, the association of other disease states including psychiatric illness, infertility, lymphoproliferative malignancy, and mortality is explored with special attention paid to autoimmune and atopic disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Celiac disease (CD) and non-celiac gluten sensitivity have been seemingly novel topics of great discussion over the last few years. Yet from a historical perspective, descriptions of celiac disease are by no means novel. The first recorded account of the condition is attributed to the ancient Greek physician Aretaeus, who described in adult patients “a chronic disturbance of digestion and malabsorption, leading to a malnourished state and whose symptoms are relieved with fasting” [1]. Samuel Jones Gee wrote of what he called the “Coeliac Affection” in 1888, as a chronic indigestion occurring in persons of all ages but principally in children and characterized by diarrhea and signs of malabsorption [1, 2]. The disease was then known as “Gee-Herter” after Gee in England and Christian Herter who described the disease in the USA until the mid-twentieth century.
Perhaps the most significant and fascinating historical description of celiac disease came from Dutch pediatrician Willem Dicke during the 1930s and 1940s. During this period, the accepted treatment of children with celiac disease centered around a “banana diet” or “Fanconi diet” consisting of fruits and vegetables [2], but the potential toxicities of wheat ingestion had not yet been elucidated. Dicke proposed a wheat-free diet in 1941 after discoveries he made when counseling families to experiment with various diets, including a wheat-free diet, and report back to him. During the so-called winter of starvation in World War II era in the Netherlands, the unavailability of breads led to the clinical improvement of many of Dicke’s patients with celiac disease and reintroduction of breads saw drastic relapse of disease. Henceforth, the toxic effects related to wheat became more generally accepted [2].
Definitions—Celiac Disease
There is a lack of consensus on the use of terms related to celiac disease and disorders associated with ingestion of gluten. Other historical terms which have been used as descriptors of celiac disease include sprue, celiac sprue, gluten intolerance, and gluten-sensitive enteropathy. Approximately four decades ago in 1970, the first consensus definition of celiac disease was published in Acta Paediatrica. At that time, celiac disease was defined as a permanent condition of gluten intolerance with mucosal flattening that (a) reversed on a gluten-free diet (GFD) and (b) then relapsed on reintroduction of gluten [3]. With increasing recognition of the broader spectrum of disorders associated with gluten ingestion, a collaborative effort on extensive literature review on definitions was published in 2013 as the Oslo definitions [4]:
Celiac Disease (CD) is defined as a chronic small intestinal immune-mediated enteropathy precipitated by exposure to dietary gluten in genetically predisposed individuals.
Classical CD is defined as CD presenting with signs and symptoms of malabsorption. Diarrhea, steatorrhea, weight loss, or growth failure is required.
Non-classical CD refers to patients with CD but no associated features of malabsorption. Such patients may present with constipation, abdominal discomfort, and other symptoms not typically associated with malabsorption.
Symptomatic CD is characterized by clinically evident gastrointestinal and/or extraintestinal symptoms attributable to gluten intake.
Gluten-related disorders is an umbrella term that encompasses all diseases triggered by gluten. These include gluten ataxia, dermatitis herpetiformis (DH), non-celiac gluten sensitivity, and CD.
In addition, it recommended that terms gluten intolerance, overt CD (characterized by diarrhea, dyspepsia, and bloating associated with gluten ingestion), and silent CD which is equivalent to asymptomatic CD should not be used. Other frequently used terms in the celiac literature include:
Latent CD is defined in varied ways. Perhaps the encompassing definition is in patients with positive CD serology with normal mucosa, absence of villous atrophy (VA), and lack of symptoms. Latent CD has been used interchangeably with potential CD. The term Genetically at risk for CD refers to family members of patients with CD that test positive for human leucocyte antigen (HLA) DQ2 and/or DQ8. Genetic susceptibility to CD is contributed to by HLA and non-HLA genes which account for approximately 65% of the predisposition [5].
Non-celiac Gluten Sensitivity
Non-celiac gluten sensitivity (NCGS) is characterized by celiac-like symptom improvement after gluten withdrawal in absence of serologic or histologic evidence of CD. It has also been defined as a variety of immunological, morphological, or symptomatic manifestations that are precipitated by the ingestion of gluten in individuals in whom CD has been excluded. For completeness of diagnosis, there must be an absence of wheat allergy [6, 7]. The estimated prevalence of NCGS in the USA is 0.548%, almost half that of patients with CD [8]. More recent literature suggests that symptoms in patients with self-reported NCGS may be precipitated by fructan ingestion [9]. A recent double-blind, randomized, placebo-controlled trial evaluating patients with NCGS found that reduction in dietary FODMAPs leads to a significant improvement in symptoms and upon re-challenge of these patients with gluten showed no specific or dose-dependent effects [10].
There are no definitive tests to confirm a diagnosis of NCGS; however, this is an important distinction to make for two reasons. First, NCGS patients do not appear to be at risk for the nutritional deficiencies and associated-disease states that their CD counterparts may inherit. Second, the possibility for reintroduction of a gluten-containing diet may exist for the NCGS group as well.
Seronegative Celiac Disease
One of the diagnostic challenges to emerge over the last few years has been that of a group of patients with so-called seronegative celiac disease (SNCD), i.e., patients with duodenal mucosa damage Marsh I, II, and III stages, HLA DQ2/DQ8 haplotype, and clinical features suggestive of CD but negative for CD serology. A recent series of 200 patients over a 16-year period shed some light on this group. Of patients referred with seronegative villous atrophy (SNVA), up to 69% were felt to have a cause other than CD; 27% were blamed on infections, 17.5% on an immune or inflammatory process, and 6.5% on medications (particularly the angiotensin-2-receptor blocker class of medications). SNCD was seen in 31% of patients. The HLA-DQ2 and/or DQ8 genotype was present in 61%, with a 51% positive predictive value for SNCD. For 18% of these patients, no identifiable cause was determined; however, 72% of this group saw spontaneous resolution of their villous blunting while consuming a diet rich in gluten. To date, suffice it to say that the diagnosis of “seronegative” CD should be used with extreme caution, as most causes of seronegative villous atrophy have an alternative and identifiable cause, and the histologic findings are often transient [11].
Pathophysiology
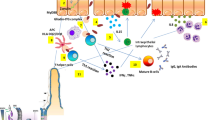
The term “gluten” refers to the alcohol-soluble protein fraction of wheat and is a prolamin or storage protein. Rye and barley have components that are extremely similar to wheat gluten in amino acid sequence and while not technically glutens, are often described as such in the literature [12]. Glutens can be subdivided into the monomeric gliadins and polymeric glutenins, and due to the abundance of proline and glutamine amino acids in these proteins, gastrointestinal enzymatic digestion is limited and yields rather large peptides, up to 33-mer in length [12, 13]. Under normal physiologic conditions, these large peptides translocate from the lumen to the lamina propria via a transcellular or paracellular route [14, 15]. Some studies suggest that gliadins can lead to a temporary increase in gut permeability, even in normal individuals, the result of which seems to have no clinical significance [12, 16, 17]. In addition to the genetic factors and proposed environmental factors, the pathogenesis of CD involves both the innate and adaptive immune systems. A great deal has been learned about the pathogenesis of CD, the adaptive and innate responses, and their interplay over the last few decades. In the adaptive immune response, gluten is deamidated by tissue transglutaminase when it reaches the lamina propria, increasing its immunogenicity. The gliadin fragments are bound by HLA-DQ2 or HLA-DQ8 on antigen-presenting cells, and in turn activate CD4+ T cells. A Th1 response mediated by IFN-gamma [18] and to a lesser extent a Th2 response ensues, leading to a pro-inflammatory, tissue destructive, and remodeling cascade through matrix metalloproteinases. The Th2 response may lead to further production of antibodies to tissue transglutaminase and gluten; the implication of which is unclear but may play a role in the development of extraintestinal CD [19, 20]. In the innate immune response within the epithelial layer, gliadin stimulates secretion of IL-15 which leads to the upregulation of natural killer receptors on intraepithelial lymphocytes, enhancing cell death and permeability of the gut wall [21].
Genetic Factors
It is well-established that CD occurs almost exclusively in the presence of the HLA-DQ2 or HLA-DQ8 haplotypes [22, 23]. The HLA-DQ2 haplotype (DQA1*0501/DQB1*0201) is expressed in approximately 90% of patients with CD. In contrast, it is expressed in approximately 30 to 40% of the general population. HLA-DQ8 haplotype (DQA1*0301/DQB1*0302) is expressed in another 5% of patients, and almost all the remaining 5% of patients have at least one of the two genes encoding DQ2 (DQB1*0201 or DQA1*0501). The DQ2 and DQ8 haplotypes are necessary but not sufficient for the development of CD [24]. So far, at least 39 non-HLA genes that confer a predisposition to the disease have been identified, most of which are involved in inflammatory and immune responses [25]. Other noteworthy genetic predisposing factors may include family history and gender. Despite sex being discussed as a potential genetic risk factor, the available literature shows mixed results. Three screening studies from the USA show equal prevalence among men and women [26,27,28]. However, a Swedish study and the study by Liu et al. showed a higher prevalence of CD among women [22, 29].
Overall, HLA DQ2 haplotype confers the highest genetic risk to the development of CD. In addition, it is generally accepted that female sex and a family history are also genetic risk factors.
Environmental Factors
Aside from the obvious consideration which is the degree of gluten exposure, a variety of other environmental triggers have been explored in CD. This is particularly relevant given that the concordance of disease in monozygotic twins is high but not universal, and concordance is seemingly only modestly related to the HLA genotype [30, 31].
None of the environmental triggers theorized to lead to increased risk of CD have garnered any overwhelming evidence-based support to date, and thus this remains an area of significant interest and attention. A great deal of effort has focused on neonatal and early-life exposure and the risk of CD. A recent nested case-control based in a Norwegian pregnancy cohort evaluated maternal and neonatal 25-hydroxy vitamin D status and found no evidence to support an increased risk for childhood CD [32]. Similarly, despite earlier concerns, two recent multicenter randomized trials have shown no enhanced risk of CD with respect to the timing of gluten exposure nor protective benefit from breast feeding [33, 34].
Attention has also been given to H. pylori status, infectious triggers, and the microbiome of celiac patients as possible risk factors, with mixed results. In 2013, a cross-sectional study looked at over 100,000 patients with gastric and duodenal biopsies and compared H. pylori colonization status in patients with and without CD and found that H. pylori prevalence was significantly lower in CD patients when compared to those without [35], perhaps supporting a “hygiene hypothesis” that decreased exposure to bacteria may contribute to autoimmunity. Unfortunately, subsequent data have been conflicting, and more research is needed in this area. Socioeconomic status has also been explored with mixed results. On the one hand, a cross-sectional study looked at children in Wales and indicated that CD was more common in children of higher socioeconomic status [36]. While the authors felt that some of this could be explained by more health-seeking behavior in the more affluent group, they also queried whether or not lack of exposure or the hygiene hypothesis could explain the enhanced risk [36]. Conversely, survey data from a large tertiary care center suggest that adult patients with lower incomes may have worse celiac-related health and higher symptom burden [37]. Additionally, multiple studies have suggested a link between early infections and risk of celiac autoimmunity, including both respiratory [38] and gastrointestinal infections [38, 39], particularly Campylobacter [40] and frequent rotavirus infections [41]. Recent animal studies have suggested that infection with even the seemingly innocuous reovirus may disrupt intestinal immune homeostasis by suppressing peripheral regulatory T cells and T helper 1 immunity to dietary antigens [42].
Lastly, differences in the CD patients’ microbiome have been explored. There is ongoing debate on the role of infant gut colonization and risk of developing CD. Some recent studies provide supporting data on method of delivery, timing and duration of exposure to breastfeeding and introduction of other foods. A key postulated environmental predisposing factor includes gut colonization. Initial gut colonization in infants is highly influenced by the diet and method of delivery. Normal post-partum gut colonization occurs when full-term neonates are born by vaginal delivery and are exclusively breast-fed during the first 6 months of life. On the other hand, infants born by cesarean section or who are bottle-fed have altered or inadequate initial colonization and mucosal immune dysfunction. It is thought that this leads to an increased risk of allergic and autoimmune diseases [43]. In addition, there is supportive evidence that disruption of the normal colonization process with aberrant probiotic flora can lead to alterations in the symbiotic relationship that is necessary for immune homeostasis and may be involved in the development of autoimmunity (e.g., in CD and type 1 diabetes) [44, 45].
Epidemiology
CD has a prevalence estimated to be about 1% of the general population [46, 47]. It is worth noting that this figure is considered a moving target, as both the prevalence of CD and the rate of diagnosis seem to be increasing though not necessarily proportionally due to the persistent variables of under diagnosis and potential for unfounded diagnosis [48]. With this said, prevalence of CD certainly seems widely variable depending on a number of factors. In the USA, the prevalence of CD increases drastically from 1:133 in patients not-at-risk, to 1:56 in symptomatic patients, to 1:39 in patients with a second-degree relative with the diagnosis, to 1:22 in patients with a first-degree relative with the diagnosis [28].
Similarly, a number of autoimmune diseases increase the celiac risk profile. Type 1 diabetes mellitus seems to be the most commonly recognized autoimmune disease state with this association. A recent prospective birth cohort study followed clinically significant type 1 diabetes and CD as well as persistently positive islet autoantibodies and tissue transglutaminase autoantibodies at quarterly intervals from 3 to 48 months and semiannually thereafter [49]. Of the 5891 children included in the analysis with a median follow-up of 66 months, 367 children developed islet autoantibodies, 808 children developed tissue transglutaminase autoantibodies, and 90 children developed both [49], which greatly exceeded the expected co-occurrence and could not be accounted for location or multiple genetic factors. The development of type 1 diabetes generally preceded that of CD and was felt to significantly increase risk of tissue transglutaminase antibodies later in childhood. Additional autoimmune diseases and developmental diseases have been felt to increase the prevalence of CD including autoimmune liver disease (13.5% prevalence), autoimmune thyroid disease (3% prevalence), Down syndrome (5.5% prevalence), Turner syndrome (6.5% prevalence), and Williams syndrome (9.5% prevalence) to name a few [46].
The celiac risk profile also appears to be altered by country of origin or geographic region, ethnicity, and perhaps latitude as well. Differences in celiac risk dependent on nationality and ethnicity have been well-described. For instance, in the USA, the prevalence of CD is significantly higher in Caucasians than that in Blacks and Hispanics [50, 51]. In Europe, Finland has a much higher prevalence of CD among adults than their counterparts in Germany and Italy (2.4 vs 0.3 and 0.7%) [52]. It is also noteworthy that the 2014 study by Lui et al. also indicated residence in Sweden as risk factor for development of CD [22]. Whether these differences can be attributed to specific environmental or genetic risk factors is not entirely clear in all cases. Interestingly, a recent study from India showed that in the northern part of the country, celiac autoantibodies are much more prevalent than those in the northeastern and southern parts of the country (1.23 vs 0.87 and 0.10%) [53]. This difference, at least in part, seems to be accounted for by dietary differences as while HLA haplotypes were virtually identical between the three populations, mean daily wheat intake was significantly higher in northern India when compared to that in northeastern and southern India (455 vs 37 and 25 g) [53]. A recent population-based study analyzed data on gluten-related conditions of 22,277 patients from the NHANES study group from 2009 through 2014. This showed that in the USA, a higher proportion of persons at latitudes north of 35° have CD or avoid gluten than persons living south of this latitude, and that this difference could not be accounted for by race or ethnicity, socioeconomic status, or body mass index (BMI) [54]. This latitude-dependent increased risk has been demonstrated in other autoimmune diseases and has been proposed to be related to vitamin D deficiency and mediated by vitamin D receptors; however, these data were not sufficiently available in the aforementioned study group.
Clinical Presentation (Adults, Children)
The classical paradigm of CD, i.e., the children described by Willem Dicke and predecessors, with a malabsorptive syndrome characterized by diarrhea and failure to thrive has largely been replaced. Today, CD can be diagnosed at any age and with a wide variety of clinical presentations.
Up to 20% of adults diagnosed with CD may have a subclinical presentation [55]. However, even among those presenting with symptomatic CD, two thirds present with so-called non-classical symptoms. Only 27% of patients in a recent retrospective review presented with diarrhea. Other intestinal manifestations frequently described were bloating (20%), aphthous stomatitis (18%), alternating bowel habits (15%), constipation (13%), and GERD (12%). Extraintestinal manifestations included osteopenia/osteoporosis (52%), anemia (34%), cryptogenic transaminitis (29%), and recurrent miscarriages (12%) [55]. This shift from the classical presentation with age seems to even be augmented when comparing geriatric patients to younger adults [56].
Children still seem more likely than adults to demonstrate “classical” malabsorptive symptoms of CD [57]. While up to two thirds of children exhibit “classical” presentations, atypical symptoms can be the chief complaint in the remainder. Abdominal pain and poor growth seem to be among the more common atypical presentations in children [57]. However, in children, the paradigm appears to be shifting as well. Whereas the classic description of an underweight child with failure to thrive, a recent cohort study suggested that in North America, up to 20% of children may be obese or overweight at the time of diagnosis of CD [58]. Interestingly, when adherent to a gluten-free diet (GFD), overweight or obese children may actually improve or normalize their BMI, whereas children of normal weight at the time of diagnosis are seemingly at risk for becoming overweight when starting a GFD [58].
Diagnosis—Adults and Children
Testing for CD should be initiated in any child or adult with signs or symptoms of intestinal or extraintestinal manifestations or laboratory evidence suggestive of the disease [59]. Testing generally involves serologic screening followed by small bowel biopsy. Perhaps one caveat and distinction between the diagnosis of children and adults is that the European Society for Pediatric Gastroenterology and Hepatology (ESPGHAN) has proposed that children meeting certain criteria need not undergo endoscopic exam to confirm the diagnosis of CD [46]. If clinical suspicion is very high, small bowel biopsies should still be pursued even in the setting of negative serologic testing [59].
Serologic testing should be performed on a gluten-containing diet, as serologies can become negative within weeks to months of initiation of a gluten-free diet [60]. The primary means by which to screen patients older than 2 years of age is the immunoglobulin A anti-tissue transglutaminase (tTG) antibody, which has both a sensitivity and specificity of at least 95% [59]. Due to the possibility of immunoglobulin A (IgA) deficiency leading to a false negative test, total IgA should be measured concurrently. If there is any concern for IgA deficiency affecting test results, CD screening should be performed employing IgG-based antibody testing [59]. Additional serologies for the primary detection of celiac disease include anti-endomysial antibodies (EMA) as well as antibodies to deamidated gliadin peptides (DGP). Analysis of the former may be expensive, subjective, and of limited availability. Antibodies to DGP are similar in sensitivity and specificity to anti-tTG antibodies and both are superior to the previously employed native gliadin (AGA) antibodies, which are no longer recommended [59, 61]. A summary of common serologic tests is provided (Table 1).
Confirmatory testing in most cases is accomplished with upper endoscopy, specifically, 1–2 biopsies in the duodenal bulb and at least four biopsies in the distal duodenum [59]. Endoscopic findings in CD include scalloping, flattening, and fissuring of the mucosal folds (Figs. 1 and 2), though they are nonspecific and their absence does not preclude the diagnosis as disease may be patchy in nature [62]. Hence, the histologic Marsh modified (Oberhuber) and Corazza criteria which characterize the degree of increased intraepithelial lymphocytes, crypt hyperplasia, and villous atrophy are useful in confirming the diagnosis [59] (Table 2).
Endoscopic/histologic findings in celiac disease. Courtesy: Presutti RJ, Cangemi JR, Cassidy HD, et al. Celiac disease. Am Fam Physician. 2007 Dec 15;76(12):1795–802. Endoscopic and biopsy findings in patients with and without celiac disease. a High-definition endoscopic photo of normal small intestine. The villi are clearly visible with no evidence of atrophy or scalloping of the folds. b Biopsy specimen of normal small intestine (hematoxylin-eosin; original magnification, × 100). c PillCam image of small intestine in a patient with celiac disease, showing scalloping of the mucosal folds (arrows) characteristic of a malabsorption pattern. There is also evidence of villous atrophy compared with normal. d Biopsy specimen of small intestine in a patient with celiac disease (hematoxylin-eosin; original magnification, × 100). Note the loss of villous architecture
Role of Primary Screening for Celiac/Screening of High-Risk Individuals
To date, there is insufficient evidence to favor primary screening of asymptomatic individuals for CD [63]. However, screening of high-risk individuals including first-degree relatives of a celiac patient and type 1 diabetics with signs, symptoms, or laboratory findings suggestive of the disease is appropriate [59]. Similarly, patients with transaminitis of unknown etiology should be screened [59]. Screening of asymptomatic first-degree relatives and second-degree relatives remains controversial [59]. A diagnostic algorithm based on risk profile has been suggested, Fig. 3.
Role of HLA-DQ2/DQ8 Testing
As the HLA-DQ2 or DQ8 haplotypes are present in virtually all patients with celiac disease, HLA-DQ typing yields a high negative predictive value for the disease. However, addition of HLA-DQ typing to standard diagnostic measures does not seem to augment test performance [64]. Thus, its routine use is not recommended [59]. HLA-DQ2/DQ8 genotyping may be useful in effectively ruling out the disease in difficult clinical situations, such as in a patient with equivocal histological findings and negative serologies or evaluation of a patient on a GFD if no testing was performed prior to initiation of the GFD [59].
Screening Special Groups
Studies on screening using serological testing indicate that only about 1:5 cases of CD are identified [52]. As indicated earlier in this review, the prevalence is higher among women compared to that among men. Prevalence is also increased in certain specific groups: persons who have an affected first-degree relative (10 to 15%), type 1 diabetes (3 to 16%), Hashimoto’s thyroiditis (5%) or other autoimmune diseases (including autoimmune liver diseases, Sjögren’s syndrome, and IgA nephropathy), Down’s syndrome (5%), Turner’s syndrome (3%), and IgA deficiency (9%) [65,66,67,68,69,70].
In addition to type 1 diabetics and first-degree relatives of celiac patients as described above, it is prudent to have a low threshold for screening individuals with any of these other conditions as well.
Nutritional Considerations
The role of a trained dietician or nutritionist in the care of patients with CD is pivotal. Perhaps, they play the most important role in the patients’ follow-up care and may be the key person in any celiac center. Their role includes general education on celiac disease, teaching label reading, providing tips on eating out of the home, and avoiding cross contamination. In addition, a trained dietician/nutritionist serves as an added resource in patients with limited response or non-response to a gluten-free diet. With careful added history from a dietician/nutritionist, patients usually labeled as non-responsive celiac disease may have had inadvertent exposure to gluten-free products.
There is a risk of malabsorption and other associated nutritional deficiencies in patients with CD. Pediatric patients with celiac disease are at risk of deficiencies of several micronutrients. Deora et al. evaluated the prevalence of micronutrient deficiencies in children with CD at diagnosis, at 6 months, and at 18 months after the start of a GFD. In addition, they sought to examine any correlation between micronutrient deficiencies, serum tissue transglutaminase immunoglobulin A antibody titers, and the degree of mucosal damage at diagnosis. A total of 140 CD patients (including 87 girls [621%]) under 17 years were included. The serum vitamins, minerals, and anti-tTG IgA antibodies were measured at diagnosis and at 6 and 18 months after starting a GFD. Histopathological changes of duodenal biopsies at diagnosis were documented using modified Marsh classification. The mean age at diagnosis was 7.8 ± 4.01 years. At diagnosis, serum vitamin D was the most commonly deficient vitamin in 70% of children. Serum ferritin was subnormal in 34.5% with zinc in 18.6% children but only 12 (10.9%) children had iron deficiency anemia. There was no correlation between micronutrient deficiencies at diagnosis and serum tTG IgA antibody titers or the degree of villous atrophy. Reassuringly, majority of serum levels of measured micronutrients had normalized after 6 months of starting GFD except for vitamin D, which improved but remained subnormal [71]. The risk of vitamin A deficiency in celiac disease has been thought to be due to the presumed reduced absorption of fat-soluble vitamins. However, the available data is limited to letters to the editor and there are no large studies assessing the extent of the presumed association [72].
The prevalence of CD in patients with iron deficiency (ID) is estimated at 0–6% in European countries. The prevalence of celiac disease in patients with iron deficiency in the USA is not well described. Abdalla et al. used the NHANES database, a nationally representative health survey conducted from 2009 to 2010, to explore this further. Their study included 2105 females aged 6 years or older. Iron deficiency was defined as serum ferritin level < 20 ng/mL; patients were diagnosed with CD if they tested positive for both IgA tissue transglutaminase antibody and IgA endomysial antibody. Subjects were divided into two groups, ID and non-ID respectively. Five hundred sixty-nine patients out of 2105 subjects had ID and 1536 did not have ID. Five people were identified as having CD among the ID group, as were two people in the non-ID group. After adjusting for selected covariates, the prevalence of CD was higher in female subjects with ID with OR of 12.5 (95% CI 1.74–90). From their study, the overall prevalence of CD in the US female population is low; however, the prevalence is higher in subjects with ID. The study results may not be generalizable since it only included females [73].
The recent prospective observational study conducted in an Indian patient cohort by Beniwal et al. sought to evaluate the prevalence and clinical features of CD among children with severe acute malnutrition (SAM). All consecutively admitted children with SAM were recruited. Subjects were screened for CD using IgA-tTG antibodies. Patients who tested positive underwent upper gastrointestinal endoscopy with small bowel biopsy for the confirmation. Clinical features of patients with and without CD were compared. The seroprevalence (IgA-tTG positivity) of CD was found to be 15.38% while prevalence of biopsy-confirmed CD was 14.42% among SAM children. Abdominal distension, diarrhea, anorexia, constipation, pain in the abdomen, vitamin deficiencies, edema, clubbing, and mouth ulcers were more common in patients of CD compared to patients without CD. However, there was a statistically significant-observed difference only for abdominal distension and pain [74].
Dermatitis Herpetiformis
Dermatitis herpetiformis (DH) is an immune-mediated itchy blistering skin disease which may be a manifestation of CD (Fig. 4). It is typically seen on the elbows, knees, and buttocks, but occasionally in the scalp and upper back. The diagnosis is confirmed by skin biopsy with special stains which show granular immunoglobulin A deposits in perilesional skin. Five percent of first-degree relatives may be affected by CD or DH. Whereas tissue transglutaminase (TG2) is the autoantigen in CD, epidermal transglutaminase (TG3) is the autoantigen in DH.
A few other distinguishing features are worth mentioning. First, up to a quarter of DH, patients do not have small bowel mucosal villous atrophy, but virtually all have celiac-type inflammatory changes. The skin symptoms respond slowly to a GFD. While the incidence of CD is increasing, for unclear reasons, the opposite is true for DH. Lastly, DH may be seen more commonly in males as opposed to CD which is more prevalent in females. As discussed later in this review, there is a small risk of small intestinal T cell lymphoma associated with CD. The associated lymphoma in DH is a B cell type and not limited to the small intestine. Adult CD may carry a risk of increased mortality [27]. On the other hand, DH is associated with a significantly decreased relative mortality rate. Both CD and DH show tissue transglutaminase (TG2)-specific autoantibodies in serum and small bowel mucosa. In addition, TG3-targeted IgA antibodies are found in the skin of DH patients.
Treatment is with a GFD which results in healing of the skin manifestations. Small bowel biopsy is not required to initiate a gluten-free diet in patients with DH. Dapsone and rituximab have also been used, typically for refractory cases [75].
Psychological/Psychiatric Associations
Schizophrenia
There has been a historical interest in the association of CD and schizophrenia. While older studies described an association, other studies questioned causality [76, 77]. The 2005 study by West et al. perhaps provides some clarity on this issue. In that study, patients with CD, Crohn’s disease, and ulcerative colitis were matched individually with five age-, sex-, and general practice-matched controls. The prevalence of schizophrenia was calculated and compared between disease groups and their respective controls. The adjusted odds ratio for CD showed no association with schizophrenia (CD vs. controls 0.76, 95% CI 0.41–1.4) [78].
Anxiety, Depression, and Panic Disorder
The data on association of anxiety and depressive spectrum disorders with CD is conflicting. The variations noted in study results may be the result of methodology, small numbers, or study design. A few are reviewed in this section.
A questionnaire-based study by the German Celiac Society compared anxiety and depression levels in adult patients with CD on a GFD with controls. The levels of anxiety, of depression, and of a probable anxiety or depressive disorder were assessed using the Hospital Anxiety and Depression Scale. Cases consisted of 441 adult patients with CD recruited by the German Celiac Society and 441 adult controls from the German general population (GP) sample. Potential demographic (age, sex, social class, family status) and disease-related (latency to diagnosis, duration of GFD, compliance with GFD, thyroid disease) predictors of anxiety and depression in CD were tested for by regression analyses. The level of anxiety in CD patients was predicted (R2 = 0.07) by female gender (P = 0.01). In addition, female sex was associated with a probable anxiety disorder (OR = 3.6, 95% CI 1.3–9.4, P = 0.01). Living alone (OR = 0.5, 95% CI 0.2–0.9, P = 0.05) was associated with a reduced risk of an anxiety disorder. The level of depression and a probable depressive disorder were not predicted by any of the demographic and medical variables tested for. The levels of depression in persons with CD (4.2 ± 3.4) and of the GP (4.2 ± 3.8) did not differ (P = 0.3). The prevalence of a probable anxiety disorder in persons with CD (16.8%) was higher than that of the GP (5.7%) (P < 0.001). The prevalence of a probable depressive disorder did not differ significantly between the three groups (P = 0.1). The study concluded that anxiety in adult German female patients with CD on a GFD is higher than that observed in the general population; the same was not seen in depression or probably depressive disorder. There is a potential sampling bias in that all patients were members of the German Celiac Society and several patients with CD who were non-members were excluded. The authors recommended screening for anxiety in female patients with CD [79].
Carta et al. evaluated the association between CD and specific anxiety and depressive disorders and sought to identify potential common pathogenetic links, with particular regard to thyroid function and autoimmunity. The study included only 36 adult CD patients, (25 females and 11 males), aged 18–64 years. The 144 controls consisted of healthy subjects matched by sex and age with no clinical evidence or family history of CD. Diagnosis of celiac disease was made by clinical history and serology. Psychiatric diagnoses were formulated using the International Composite Diagnostic Interview, according to DSM-IV criteria. Thyroid status was evaluated by palpation, imaging (ultrasonography), and measurement of serum-free thyroid hormones (FT4, FT3), thyroid-stimulating hormone (TSH), and antithyroid autoantibodies (anti-TPO). Compared to controls, a significantly higher number of CD patients met criteria for lifetime [15 (41.7%) versus 30 (29.8%), P < .01] and 6-month [7 (19.4%) versus 9 (6.2%), OR = 3.2, chi (2) = 5.2, P < .05] major depressive disorder (MDD). A similar trend was noted for panic disorder (PD)—lifetime [5 (13.9%) versus 3 (2.1%), P < .001] and 6 months [3 (8.1%) versus 2 (1.4%), P < 0.05]. Anti-TPO prevalence was significantly higher in CD patients than that in the control group (11/36 = 30.5% versus 14/144 = 9.7%, P < 0.001). Comparing PD and MDD in celiac patients with positive anti-TPO to patients with negative TPO, there was a higher frequency found in those with positive anti-TPO (4/11 = 36.4% PD in TPO+ versus 1/25 = 4% PD in TPO−, P < 0.01; 9/11 = 81.8% MDD in TPO+ versus 6/25 = 9.5% MDD in TPO−, P < 0.01). Celiac disease patients tend to show a high prevalence of PD and MDD. This observation is associated with subclinical thyroid disease [80].
The 2017 study by Zylberberg et al. evaluated a large representative sample in the USA. Data from 22,274 participants from the 2009–2014 National Health and Nutrition Examination Survey were evaluated to compare the prevalence of depression, insomnia, quality of life variables, and psychotropic medication use in CD and people who avoid gluten (PWAGs) to controls. Depression was present in 8.2% of controls compared with 3.9% of participants with CD (P = 0.18) and 2.9% of PWAGs (P = 0.002). After adjustment for age, sex, race, income, and access to healthcare, PWAGs maintained lower odds of depression compared with controls (odds ratio = 0.25; 95% confidence interval 0.12–0.51; P = 0.0001) [81].
The preponderance of available data does not support a definitive association of anxiety and depression with CD.
Treatment
Although NCGS may be a transient entity allowing for eventual gluten tolerance [82], in patients with CD, indefinite adherence to a strict GFD is currently the only treatment option. This equates to not only wheat avoidance, but also avoidance of barley and rye given the similarity to gluten of their prolamin proteins. Oat prolamins are more distantly related to glutens, and thus oats may be reintroduced with caution. Most patient with CD will tolerate oats without harm, so long as they are not contaminated with other grains [83, 84]. While seemingly simple enough, a strict GFD fraught with numerous challenges. Among the most challenging obstacles to a GFD is that it is substantially more expensive [85, 86], such that those with a lower socioeconomic standing may be a high risk of noncompliance. Further, a strict GFD may be socially isolating and unknown source of gluten in medications and when dining out is a source of great anxiety for patients. Because of these many challenges, the self-rated burden of disease in patients with CD is actually as high as that of patients with diabetes mellitus or end-stage renal disease [87].
Evolving Non-dietary Therapies
Due to the great burden of adhering to a strict GFD among patients with both CD and NCGS and avoiding inadvertent gluten exposures, non-dietary treatments have been proposed and are evolving. Two pharmacologic therapies and potential adjuncts to a gluten-free diet have received the most attention thus far. Latiglutenase, previously described as ALV003, is an orally administered mixture of two gluten-targeting recombinant proteases theorized to decrease the immunogenicity of ingested gluten in the small intestine by degrading it in the stomach. While a recent phase II clinical trial of patients with symptomatic CD already following a GFD for at least 12 months did not show any benefit of latiglutenase in reducing villous atrophy or improving symptoms, a subset analysis suggests that seropositive CD patients may gain significant symptom improvement from this therapy [88, 89]. Another novel oral agent, larazotide acetate, works by regulating intestinal tight junctions and thereby preventing gluten from reaching the small intestinal submucosa and triggering an immune response. In a large randomized controlled trial, larazotide acetate plus a GFD was shown to reduce the signs and symptoms of CD relative to a GFD alone [90]. Additional non-dietary therapies including desensitization, genetically modified gluten, probiotics, parasites, transglutaminase inhibition, polymer connectors, and TNF-alpha inhibitors and other anti-inflammatory strategies are under investigation [91].
Non-responsive/Refractory CD
Non-responsive celiac disease (NRCD) and refractory CD (RCD) are two important clinical entities to be aware of and distinguish. NRCD refers to persistent signs, symptoms, or laboratory abnormalities consistent with the disease despite 6 to 12 months of reported strict compliance with a GFD and may be present in up to 20% of those diagnosis with CD [92]. The most common etiology of non-responsive CD is felt to be inadvertent gluten exposure in up to 36% of patients [92], but other causes include lactose intolerance, SIBO, pancreatic insufficiency, IBS, microscopic colitis, and refractory RCD [59].
After the aforementioned disease entities have been evaluated and treated, in particular nutritional evaluation for exposures or lack of compliance with a GFD, and repeat small intestinal histology and serologies confirm the CD diagnosis, a diagnosis of truly “refractory” CD may be considered. RCD represents only 1–2% of all CD patients [93]. Type 1 RCD generally exhibits a similar population of intraepithelial lymphocytes seen in CD, whereas type 2 RCD generally exhibits a phenotypically abnormal population of T cells and is associated with a worse prognosis. In addition to standard care of the CD patient, systemic steroid therapy and immunosuppressive medications may be employed; however, there is a paucity of evidence-based data to deem these strategies effective [59].
Autoimmune and Atopic Disease Disease/Considerations for the Allergist
Distinction from Wheat Allergy/Wheat-Dependent Exercise-Induced Asthma
Wheat allergy is a rare but clinically distinct entity affecting only up to 0.5% of the general population; however, the omega-5 gliadin fraction is the most common allergen implicated in food-dependent exercise-induced asthma [94]. Clinical manifestations of wheat allergy are felt to be mostly IgE-mediated and may include vomiting, diarrhea, eczema, urticaria, or anaphylaxis [94]. Wheat-dependent exercise-induced asthma appears to be more common in adults, whereas IgE-mediated wheat allergy occurs in children. Most children will outgrow wheat allergy [94]; however, a small European study showed that children with severe food allergies may actually be at five times risk of CD [95]. Allergic skin testing or IgE-specific antigen serologies remain the most common used diagnostic tests and treatment is based on eliminating exposures [94].
Selective IgA Deficiency/Partial IgA Deficiency
A recent North American study confirmed European results suggesting that selective IgA deficiency does not appear to be more common in CD patients than that in the general population [96]. As previously mentioned, these patients seem to require special consideration as they often do not present with gastrointestinal symptoms and of course exhibit decrease IgA-tTG sensitivity [96]. Partial IgA deficiency is commonly seen in patients with gastrointestinal symptoms and is defined as detectable serum IgA level less than two standard deviations below age standards in the presence of normal serum IgM and IgG levels [96]. Partial IgA deficiency is much more commonly encountered and does not appear to alter the presentation of CD nor preclude the use of standard IgA-tTG serologies for diagnosis [96].
CVID and CD
Common variable immunodeficiency is characterized by defective B-cells with reduced antibody production, however, T cell and macrophage dysfunction and systemic inflammation may also exist. It is well-established that patients with common variable immunodeficiency (CVID) experience a wide range of gastrointestinal symptoms, pain, bloating, and diarrhea, due not only to infectious etiologies in the setting of immunodeficiency, but also inflammatory processes throughout the gut, and a possible connection between CVID and CD had been previously described [97]. Of course, standard antibody diagnosis of CD proves challenging in this patient group. Further, although villous blunting is usually not seen, increased intraepithelial lymphocytes are found routinely in a CVID patient’s duodenal biopsy. With that said, based on recent microarray analyses, a “celiac-like” disease seen in CVID patients seems to be phenotypically distinct from true CD at this time [98]. However, more investigation is warranted.
Atopic Disease and CD
The associations with CD and a variety of other autoimmune conditions including type 1 diabetes mellitus, autoimmune thyroid disease, autoimmune hepatitis, and Sjogren’s syndrome are well reported in the literature [99]. The evidence supporting a link between CD and atopic disease seems to show a link between cutaneous atopy and CD. Multiple case reports and small studies through the literature have described atopic dermatitis or chronic urticaria presenting in patients with CD [100,101,102,103]. A large series of celiac patients, their relatives, and their spouses were surveyed at the time of CD diagnosis for allergy. Those who reported allergic symptoms underwent further testing with total and antigen-specific IgE as well as PRICK testing in some. The authors compared presence of allergy, asthma, rhinitis, conjunctivitis, food allergy, occasional urticaria, and atopic dermatitis. The only significantly increased risk was found for atopic dermatitis, reported in 3.8% of CD patients, 2.3% of their relatives, and 1.3% of their spouses [104]. More recently, a large population study suggested an increased risk of asthma in CD patients (OR 1.5), although less so than type 1 diabetes mellitus (OR 5.5) and thyroid disease (OR 1.8) in keeping with previous reports [105]. A second population-based cohort study claimed CD confers a 1.6 times increased risk for asthma [106]. Clearly, some atopic disease appears to be associated with CD; however, these associations need to be further elucidated.
Eosinophilic Esophagitis and CD
As each has separately received a great deal of increased attention in the literature, so too has attention been paid to a possible association between eosinophilic esophagitis (EoE) and CD. Unfortunately, it is hard to make any definitive conclusions from the existing literature, and more studies are needed. A recent retrospective cohort study involving over 10,000 children undergoing at least one endoscopy found that 595 had EoE, 546 had CD, and the risk of having EoE was not increased relative to the diagnosis of CD (OR 0.29, 95% CI 0.154 to 0.545) [107]. The same authors performed a meta-analysis and identified 45 cases of concomitant EoE and CD, with similar results (OR 0.525, 95% CI 0.364 to 0.797) [107]. Prior to this, multiple cross-sectional studies and cohort studies involving both adults and children have appeared to show an association between EoE and CD [108,109,110]. However, it is important to consider that the diagnosis of EoE is one that generally requires multiple endoscopies over a period of time. Perhaps, these studies prove an association between esophageal eosinophilia, as opposed to EoE, as EoE is only one cause of many leading to esophageal eosinophilia. Further, the resolution of eosinophilia in at least two studies in patients of CD patients with the implementation of a GFD [108, 111] that examined multiple endoscopies over time suggests that perhaps patients are being mislabeled as EoE when in fact, esophageal eosinophilia is related to their CD as opposed to concomitant EoE.
Pregnancy
Women with CD appear to have similar overall fertility rates when compared to the general population [112,113,114]. However, there is some evidence to suggest that they have their children at a later age [114] and patients with CD may have decreased fertility up to 2 years before their diagnosis, suggesting that very high disease activity may portend a poorer prognosis with respect to fertility [113].
Malignancy
There is a well-established increased risk for lymphoproliferative malignancies, particularly enteropathy-associated T cell lymphoma and other non-Hodgkin lymphomas reported in the literature in patients with celiac disease [115,116,117,118,119]. However, this risk now seems to be significantly lower than previously reported [120]. Further, recent evidence suggests that patients with persistently positive celiac serologies and patients with persistent villous atrophy may be those that bear the burden of this association, whereas patients with truly latent CD and mucosal healing may be spared a strong association [121, 122].
Mortality
The mortality of patients with celiac disease as compared to the general population has been investigated in many population-based studies over the last few decades, with highly variable results. While some studies have conferred an excess mortality on patients CD, or one that is elevated but improves from the time of diagnosis [119, 123], more recent studies including a large cohort study from the UK have shown no increased mortality at all [124]. There are similarly varied results from studies on stored serum studies on people with undiagnosed CD [125]. Though there is concern about increased mortality associated with CD, the current literature contains conflicting data.
Follow-up
The cornerstone of celiac management in addition to the strict adherence to a GFD as discussed includes at least annual clinical follow-up with a physician and a registered dietician /nutritionist [59]. In addition to clinical response to a GFD and growth in children, seroconversion following a GFD should be monitored and approaches 95% in patients with systematic 5-year follow-up [126, 59]. Endoscopic follow-up is recommended starting at about 2 years after initiation of a GFD, as mucosal healing may take 2 to 3 years [59, 127, 128].
Perhaps the most important additional considerations in these patients include screening for and following low bone mineral density and nutritional deficiencies [59]. Both can be seen with untreated CD. The former seems to improve after initiation of a GFD, though the change is gradual and bone mineral density may not entirely normalize [129, 130]. Micronutrient deficiencies may include iron, folic acid, vitamins B12 and B6, vitamin D, copper, zinc, and carnitine [59] as described above. Some of the vitamin deficiencies proposed to be associated with a GFD may be mitigated by the addition of oats for those with CD tolerant to oats [131]. A follow-up algorithm has been suggested (Fig. 5).
Proposed celiac patient follow-up algorithm. A suggested plan of follow-up management of patients with celiac disease. Letter a indicates celiac serology, FBC, electrolytes, LFTs, thyroid function, iron studies, calcium, phosphate, vitamin D, folate, B12, fasting glucose, ± zinc, and Mg. Letter b indicates at 1–2 years, then as indicated on clinical grounds. Letter c indicates 3–5 yearly in high-risk groups (noncompliance, refractory celiac disease, female ≥ 50 years, fractures, men ≥ 55 years); yearly if osteoporosis on treatment. Courtesy: Haines ML; Anderson RP; Gibson PR., Systematic review: the evidence base for long-term management of celiac disease. Alimentary Pharmacology and Therapeutics, 2008
Current Misconceptions on Celiac Disease and Celiac Sensitivity
Improvement of gastrointestinal symptoms with avoidance of gluten is not diagnostic of celiac disease. In addition, nonspecific symptoms including fatigue, “brain fog,” which may mean different things to different people are not indicative of celiac disease or gluten intolerance.
More recently, there are several patients who avoid gluten despite a complete evaluation by a medical provider which has ruled out celiac disease. Such individuals report a significant sense of improved health including resolution of nonspecific gastrointestinal symptoms.
Gluten-free diets are low on FODMAPs. It is not surprising that symptoms that are attributable to irritable bowel syndrome may show some improvement.
A true diagnosis of gluten sensitivity or intolerance should involve a careful and comprehensive dietary history, blinded exposure of patients to identical diets with one containing gluten and the other gluten free. Reliance on self-reported diagnosis of gluten sensitivity or intolerance leads to challenges in accurately characterizing this patient subgroup and good study design to improve our understanding.
Patients who do not have celiac disease should not have to go on a gluten-free diet. There are no known advantages but there are potential consequences. These include the relative increased costs of gluten-free foods and a sense of social isolation when eating out of their own homes.
In a recent paper by Kamycheva et al., CD seropositivity was associated with lower levels of blood lead and mercury in children (β = − 0.14, P = 0.03 for lead and β = − 0.22, P = 0.008 for mercury), but not in adults [132]. Although the clinical implication of this finding is unclear, it signals unintended biochemical changes associated with a gluten-free diet. Further long-term studies are needed to get a better sense of the effect of a gluten-free diet in patients who do not have celiac disease.
The purported benefits of a gluten-free diet by celebrities, athletes, and in the lay press may also insinuate potential health and social benefits not backed by scientific enquiry.
Conclusions
CD is a complex immune-mediated condition triggered by ingestion of gluten. Our understanding of the disease continues to evolve, and there is a better recognition of unique characteristics of subgroups of patients, association with other immune-related conditions, methods of testing, and screening of appropriate populations. There are still areas of uncertainty and debate especially about the entity of NCGS/GS. This is largely a self-reported condition where patients report varying reactions to gluten ingestion with a clinical response to a GFD in the absence of serologic or histologic evidence of CD. However, the frequency, pathophysiology, and natural history of gluten sensitivity and its relationship to CD, if any, remain to be clarified.
The growing media attention on potential harms of gluten and the perceived benefits of a GFD has resulted in a significant number of people switching to GFD without prior professional consultation. The reported symptom reduction in non-CD patients on a gluten-free diet is not diagnostic of any known clinical entity. There is the placebo effect as well as improvement of symptoms that can be ascribed to the low FODMAP content of GFD. Patients with a wheat allergy may also benefit from a GFD. For now, we recommend that patients with concerns about undiagnosed CD, gluten intolerance, or any of the associated conditions including DH should consult a healthcare professional for appropriate evaluation. Treatment of CD should also include evaluation for associated conditions and potential nutritional deficiencies.
References
Dowd B, Walker-Smith J (1974) Samuel Gee, Aretaeus, and the coeliac affection. Br Med J 2(5909):45–47
Yan D, Holt PR (2009) Willem Dicke. Brilliant clinical observer and translational investigator. Discoverer of the toxic cause of celiac disease. Clin Transl Sci 2(6):446–448
Meeuwisse GW (1970) Round table discussion. Diagnostic criteria in coeliac disease. Acta Paediatr 59:461–463
Ludvigsson JF, Leffler DA, Bai JC, Biagi F, Fasano A, Green PHR, Hadjivassiliou M, Kaukinen K, Kelly CP, Leonard JN, Lundin KEA, Murray JA, Sanders DS, Walker MM, Zingone F, Ciacci C (2013) The Oslo definitions for coeliac disease and related terms. Gut 62(1):43–52
van Heel DA et al (2007) A genome-wide association study for celiac disease identifies risk variants in the region harboring IL2 and IL21. Nat Genet 39(7):827–829
Sapone A, Lammers KM, Casolaro V, Cammarota M, Giuliano MT, de Rosa M, Stefanile R, Mazzarella G, Tolone C, Russo MI, Esposito P, Ferraraccio F, Cartenì M, Riegler G, de Magistris L, Fasano A (2011) Divergence of gut permeability and mucosal immune gene expression in two gluten-associated conditions: celiac disease and gluten sensitivity. BMC Med 9:23
Potter M, Walker MM, Talley NJ (2017) Non-coeliac gluten or wheat sensitivity: emerging disease or misdiagnosis? Med J Aust 207(5):211–215
DiGiacomo DV, Tennyson CA, Green PH, Demmer RT (2013) Prevalence of gluten-free diet adherence among individuals without celiac disease in the USA: results from the continuous National Health and Nutrition Examination Survey 2009-2010. Scand J Gastroenterol 48(8):921–925
Skodje GI, et al. (2017) Fructan, rather than gluten, induces symptoms in patients with self-reported non-celiac gluten sensitivity. Gastroenterology
Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR (2013) No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 145(2):320–8 e1–3
Aziz I, Peerally MF, Barnes JH, Kandasamy V, Whiteley JC, Partridge D, Vergani P, Cross SS, Green PH, Sanders DS (2017) The clinical and phenotypical assessment of seronegative villous atrophy; a prospective UK centre experience evaluating 200 adult cases over a 15-year period (2000-2015). Gut 66(9):1563–1572
Silano M, Vincentini O, De Vincenzi M (2009) Toxic, immunostimulatory and antagonist gluten peptides in celiac disease. Curr Med Chem 16(12):1489–1498
Shan L, Molberg Ø, Parrot I, Hausch F, Filiz F, Gray GM, Sollid LM, Khosla C (2002) Structural basis for gluten intolerance in celiac sprue. Science 297(5590):2275–2279
Matysiak-Budnik T, Moura IC, Arcos-Fajardo M, Lebreton C, Ménard S, Candalh C, Ben-Khalifa K, Dugave C, Tamouza H, van Niel G, Bouhnik Y, Lamarque D, Chaussade S, Malamut G, Cellier C, Cerf-Bensussan N, Monteiro RC, Heyman M (2008) Secretory IgA mediates retrotranscytosis of intact gliadin peptides via the transferrin receptor in celiac disease. J Exp Med 205(1):143–154
Visser J, Rozing J, Sapone A, Lammers K, Fasano A (2009) Tight junctions, intestinal permeability, and autoimmunity: celiac disease and type 1 diabetes paradigms. Ann N Y Acad Sci 1165:195–205
Leonard MM, Sapone A, Catassi C, Fasano A (2017) Celiac disease and nonceliac gluten sensitivity: a review. JAMA 318(7):647–656
Picarelli A et al (1999) 31-43 amino acid sequence of the alpha-gliadin induces anti-endomysial antibody production during in vitro challenge. Scand J Gastroenterol 34(11):1099–1102
Nilsen EM, Jahnsen FL, Lundin KEA, Johansen F–E, Fausa O, Sollid LM, Jahnsen J, Scott H, Brandtzaeg P (1998) Gluten induces an intestinal cytokine response strongly dominated by interferon gamma in patients with celiac disease. Gastroenterology 115(3):551–563
Hadjivassiliou M, Aeschlimann P, Sanders DS, Maki M, Kaukinen K, Grunewald RA, Bandmann O, Woodroofe N, Haddock G, Aeschlimann DP (2013) Transglutaminase 6 antibodies in the diagnosis of gluten ataxia. Neurology 80(19):1740–1745
Taylor TB, Schmidt LA, Meyer LJ, Zone JJ (2015) Transglutaminase 3 present in the IgA aggregates in dermatitis herpetiformis skin is enzymatically active and binds soluble fibrinogen. J Investig Dermatol 135(2):623–625
Abadie V, Jabri B (2014) IL-15: a central regulator of celiac disease immunopathology. Immunol Rev 260(1):221–234
Liu E, Lee HS, Aronsson CA, Hagopian WA, Koletzko S, Rewers MJ, Eisenbarth GS, Bingley PJ, Bonifacio E, Simell V, Agardh D, TEDDY Study Group (2014) Risk of pediatric celiac disease according to HLA haplotype and country. N Engl J Med 371(1):42–49
Lundin KE, Scott H, Hansen T, Paulsen G, Halstensen TS, Fausa O, Thorsby E, Sollid LM (1993) Gliadin-specific, HLA-DQ(alpha 1*0501,beta 1*0201) restricted T cells isolated from the small intestinal mucosa of celiac disease patients. J Exp Med 178(1):187–196
Karell K, Louka AS, Moodie SJ, Ascher H, Clot F, Greco L, Ciclitira PJ, Sollid LM, Partanen J, European Genetics Cluster on Celiac Disease (2003) HLA types in celiac disease patients not carrying the DQA1*05-DQB1*02 (DQ2) heterodimer: results from the European Genetics Cluster on Celiac Disease. Hum Immunol 64(4):469–477
Trynka G et al (2011) Dense genotyping identifies and localizes multiple common and rare variant association signals in celiac disease. Nat Genet 43(12):1193–1201
Katz KD, Rashtak S, Lahr BD, Melton LJ, Krause PK, Maggi K, Talley NJ, Murray JA (2011) Screening for celiac disease in a North American population: sequential serology and gastrointestinal symptoms. Am J Gastroenterol 106(7):1333–1339
Rubio-Tapia A et al (2009) Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology 137(1):88–93
Fasano A, Berti I, Gerarduzzi T, Not T, Colletti RB, Drago S, Elitsur Y, Green PHR, Guandalini S, Hill ID, Pietzak M, Ventura A, Thorpe M, Kryszak D, Fornaroli F, Wasserman SS, Murray JA, Horvath K (2003) Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med 163(3):286–292
Ivarsson A, Myleus A, Norstrom F, van der Pals M, Rosen A, Hogberg L, Danielsson L, Halvarsson B, Hammarroth S, Hernell O, Karlsson E, Stenhammar L, Webb C, Sandstrom O, Carlsson A (2013) Prevalence of childhood celiac disease and changes in infant feeding. Pediatrics 131(3):e687–e694
Kuja-Halkola R, Lebwohl B, Halfvarson J, Wijmenga C, Magnusson PKE, Ludvigsson JF (2016) Heritability of non-HLA genetics in coeliac disease: a population-based study in 107 000 twins. Gut 65(11):1793–1798
Nistico L et al (2006) Concordance, disease progression, and heritability of coeliac disease in Italian twins. Gut 55(6):803–808
Marild K et al (2017) Maternal and neonatal vitamin D status, genotype and childhood celiac disease. PLoS One 12(7):e0179080
Vriezinga SL, Auricchio R, Bravi E, Castillejo G, Chmielewska A, Crespo Escobar P, Kolaček S, Koletzko S, Korponay-Szabo IR, Mummert E, Polanco I, Putter H, Ribes-Koninckx C, Shamir R, Szajewska H, Werkstetter K, Greco L, Gyimesi J, Hartman C, Hogen Esch C, Hopman E, Ivarsson A, Koltai T, Koning F, Martinez-Ojinaga E, te Marvelde C, Pavic A, Romanos J, Stoopman E, Villanacci V, Wijmenga C, Troncone R, Mearin ML (2014) Randomized feeding intervention in infants at high risk for celiac disease. N Engl J Med 371(14):1304–1315
Lionetti E, Castellaneta S, Francavilla R, Pulvirenti A, Tonutti E, Amarri S, Barbato M, Barbera C, Barera G, Bellantoni A, Castellano E, Guariso G, Limongelli MG, Pellegrino S, Polloni C, Ughi C, Zuin G, Fasano A, Catassi C, SIGENP (Italian Society of Pediatric Gastroenterology, Hepatology, and Nutrition) Working Group on Weaning and CD Risk (2014) Introduction of gluten, HLA status, and the risk of celiac disease in children. N Engl J Med 371(14):1295–1303
Lebwohl B, Blaser MJ, Ludvigsson JF, Green PHR, Rundle A, Sonnenberg A, Genta RM (2013) Decreased risk of celiac disease in patients with Helicobacter pylori colonization. Am J Epidemiol 178(12):1721–1730
Whyte LA, Kotecha S, Watkins WJ, Jenkins HR (2014) Coeliac disease is more common in children with high socio-economic status. Acta Paediatr 103(3):289–294
Oza SS, Akbari M, Kelly CP, Hansen J, Theethira T, Tariq S, Dennis M, Leffler DA (2016) Socioeconomic risk factors for celiac disease burden and symptoms. J Clin Gastroenterol 50(4):307–312
Marild K et al (2015) Infections and risk of celiac disease in childhood: a prospective nationwide cohort study. Am J Gastroenterol 110(10):1475–1484
Kemppainen KM, Lynch KF, Liu E, Lönnrot M, Simell V, Briese T, Koletzko S, Hagopian W, Rewers M, She JX, Simell O, Toppari J, Ziegler AG, Akolkar B, Krischer JP, Lernmark Å, Hyöty H, Triplett EW, Agardh D, TEDDY Study Group (2017) Factors that increase risk of celiac disease autoimmunity after a gastrointestinal infection in early life. Clin Gastroenterol Hepatol 15(5):694–702 e5
Riddle MS, Murray JA, Cash BD, Pimentel M, Porter CK (2013) Pathogen-specific risk of celiac disease following bacterial causes of foodborne illness: a retrospective cohort study. Dig Dis Sci 58(11):3242–3245
Stene LC, Honeyman MC, Hoffenberg EJ, Haas JE, Sokol RJ, Emery L, Taki I, Norris JM, Erlich HA, Eisenbarth GS, Rewers M (2006) Rotavirus infection frequency and risk of celiac disease autoimmunity in early childhood: a longitudinal study. Am J Gastroenterol 101(10):2333–2340
Bouziat R, Hinterleitner R, Brown JJ, Stencel-Baerenwald JE, Ikizler M, Mayassi T, Meisel M, Kim SM, Discepolo V, Pruijssers AJ, Ernest JD, Iskarpatyoti JA, Costes LMM, Lawrence I, Palanski BA, Varma M, Zurenski MA, Khomandiak S, McAllister N, Aravamudhan P, Boehme KW, Hu F, Samsom JN, Reinecker HC, Kupfer SS, Guandalini S, Semrad CE, Abadie V, Khosla C, Barreiro LB, Xavier RJ, Ng A, Dermody TS, Jabri B (2017) Reovirus infection triggers inflammatory responses to dietary antigens and development of celiac disease. Science 356(6333):44–50
Walker WA (2013) Initial intestinal colonization in the human infant and immune homeostasis. Ann Nutr Metab 63(Suppl 2):8–15
Sanchez E et al (2011) Influence of environmental and genetic factors linked to celiac disease risk on infant gut colonization by Bacteroides species. Appl Environ Microbiol 77(15):5316–5323
Peng J, Narasimhan S, Marchesi JR, Benson A, Wong FS, Wen L (2014) Long term effect of gut microbiota transfer on diabetes development. J Autoimmun 53:85–94
Husby S, Koletzko S, Korponay-Szabó IR, Mearin ML, Phillips A, Shamir R, Troncone R, Giersiepen K, Branski D, Catassi C, Lelgeman M, Mäki M, Ribes-Koninckx C, Ventura A, Zimmer KP, ESPGHAN Working Group on Coeliac Disease Diagnosis, ESPGHAN Gastroenterology Committee, European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (2012) European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr 54(1):136–160
Rubio-Tapia A, Ludvigsson JF, Brantner TL, Murray JA, Everhart JE (2012) The prevalence of celiac disease in the United States. Am J Gastroenterol 107(10):1538–1544 quiz 1537, 1545
Gasbarrini G, Miele L, Malandrino N, Grieco A, Addolorato G, Gasbarrini A, Cammarota G, Bonvicini F (2009) Celiac disease in the 21st century: issues of under- and over-diagnosis. Int J Immunopathol Pharmacol 22(1):1–7
Hagopian W, Lee HS, Liu E, Rewers M, She JX, Ziegler AG, Lernmark Å, Toppari J, Rich SS, Krischer JP, Erlich H, Akolkar B, Agardh D, the TEDDY Study Group (2017) Co-occurrence of type 1 diabetes and celiac disease autoimmunity. Pediatrics 140(5):e20171305
Choung RS, Ditah IC, Nadeau AM, Rubio-Tapia A, Marietta EV, Brantner TL, Camilleri MJ, Rajkumar SV, Landgren O, Everhart JE, Murray JA (2015) Trends and racial/ethnic disparities in gluten-sensitive problems in the United States: findings from the National Health and Nutrition Examination Surveys from 1988 to 2012. Am J Gastroenterol 110(3):455–461
Mardini HE, Westgate P, Grigorian AY (2015) Racial differences in the prevalence of celiac disease in the US population: National Health and Nutrition Examination Survey (NHANES) 2009-2012. Dig Dis Sci 60(6):1738–1742
Mustalahti K, Catassi C, Reunanen A, Fabiani E, Heier M, McMillan S, Murray L, Metzger MH, Gasparin M, Bravi E, Mäki M, the members of the Coeliac EU Cluster, Epidemiology (2010) The prevalence of celiac disease in Europe: results of a centralized, international mass screening project. Ann Med 42(8):587–595
Ramakrishna BS, Makharia GK, Chetri K, Dutta S, Mathur P, Ahuja V, Amarchand R, Balamurugan R, Chowdhury SD, Daniel D, Das A, George G, Gupta SD, Krishnan A, Prasad JH, Kaur G, Pugazhendhi S, Pulimood A, Ramakrishna K, Verma AK (2016) Prevalence of adult celiac disease in India: regional variations and associations. Am J Gastroenterol 111(1):115–123
Unalp-Arida A, Ruhl CE, Choung RS, Brantner TL, Murray JA (2017) Lower prevalence of celiac disease and gluten-related disorders in persons living in southern vs northern latitudes of the United States. Gastroenterology 152(8):1922–1932 e2
Volta U, Caio G, Stanghellini V, de Giorgio R (2014) The changing clinical profile of celiac disease: a 15-year experience (1998-2012) in an Italian referral center. BMC Gastroenterol 14:194
Kalkan, C., F. Karakaya, and I. Soykan (2017) Similarities and differences between older and young adult patients with celiac disease. Geriatr Gerontol Int
Vivas S, Ruiz de Morales JM, Fernandez M, Hernando M, Herrero B, Casqueiro J, Gutierrez S (2008) Age-related clinical, serological, and histopathological features of celiac disease. Am J Gastroenterol 103(9):2360–2365 quiz 2366
Reilly NR, Aguilar K, Hassid BG, Cheng J, Defelice AR, Kazlow P, Bhagat G, Green PH (2011) Celiac disease in normal-weight and overweight children: clinical features and growth outcomes following a gluten-free diet. J Pediatr Gastroenterol Nutr 53(5):528–531
Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA, American College of Gastroenterology (2013) ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol 108(5):656–676 quiz 677
Casella S, Zanini B, Lanzarotto F, Villanacci V, Ricci C, Lanzini A (2012) Celiac disease in elderly adults: clinical, serological, and histological characteristics and the effect of a gluten-free diet. J Am Geriatr Soc 60(6):1064–1069
Rashtak S, Ettore MW, Homburger HA, Murray JA (2008) Comparative usefulness of deamidated gliadin antibodies in the diagnosis of celiac disease. Clin Gastroenterol Hepatol 6(4):426–432 quiz 370
Lee SK, Green PH (2005) Endoscopy in celiac disease. Curr Opin Gastroenterol 21(5):589–594
Kivela L, Kurppa K (2017) USPSTF celiac disease screening recommendations. J Pediatr 188:308–311
Hadithi M, von Blomberg B, Crusius JB, Bloemena E, Kostense PJ, Meijer JW, Mulder CJ, Stehouwer CD, Peña AS (2007) Accuracy of serologic tests and HLA-DQ typing for diagnosing celiac disease. Ann Intern Med 147(5):294–302
Frost AR, Band MM, Conway GS (2009) Serological screening for coeliac disease in adults with Turner’s syndrome: prevalence and clinical significance of endomysium antibody positivity. Eur J Endocrinol 160(4):675–679
Lenhardt A, Plebani A, Marchetti F, Gerarduzzi T, Not T, Meini A, Villanacci V, Martelossi S, Ventura A (2004) Role of human-tissue transglutaminase IgG and anti-gliadin IgG antibodies in the diagnosis of coeliac disease in patients with selective immunoglobulin A deficiency. Dig Liver Dis 36(11):730–734
Rubio-Tapia A et al (2008) Predictors of family risk for celiac disease: a population-based study. Clin Gastroenterol Hepatol 6(9):983–987
Sattar N, Lazare F, Kacer M, Aguayo-Figueroa L, Desikan V, Garcia M, Lane A, Chawla A, Wilson T (2011) Celiac disease in children, adolescents, and young adults with autoimmune thyroid disease. J Pediatr 158(2):272–5 e1
Volta U, Tovoli F, Caio G (2011) Clinical and immunological features of celiac disease in patients with type 1 diabetes mellitus. Expert Rev Gastroenterol Hepatol 5(4):479–487
Wouters J, Weijerman ME, van Furth AM, Schreurs MWJ, Crusius JBA, von Blomberg BME, de Baaij LR, Broers CJM, Gemke RJBJ (2009) Prospective human leukocyte antigen, endomysium immunoglobulin A antibodies, and transglutaminase antibodies testing for celiac disease in children with Down syndrome. J Pediatr 154(2):239–242
Deora V, Aylward N, Sokoro AR, el-Matary W (2017) Serum vitamins and minerals at diagnosis and follow-up in children with celiac disease. J Pediatr Gastroenterol Nutr 65(2):185–189
Alwitry A (2000) Vitamin A deficiency in coeliac disease. Br J Ophthalmol 84(9):1079–1080
Abdalla A, Saifullah SM, Osman M, Baniya R, Sidahmed S, LaChance J, Bachuwa G (2017) Prevalence of occult celiac disease in females with iron deficiency in the United States: an NHANES analysis. J Community Hosp Intern Med Perspect 7(6):347–350
Beniwal N, Ameta G, Chahar CK (2017) Celiac disease in children with severe acute malnutrition (SAM): a hospital based study. Indian J Pediatr 84(5):339–343
Collin P, Salmi TT, Hervonen K, Kaukinen K, Reunala T (2017) Dermatitis herpetiformis: a cutaneous manifestation of coeliac disease. Ann Med 49(1):23–31
Dohan FC (1983) More on celiac disease as a model for schizophrenia. Biol Psychiatry 18(5):561–564
Ross-Smith P, Jenner FA (1980) Diet (gluten) and schizophrenia. J Hum Nutr 34(2):107–112
West J et al (2006) Risk of schizophrenia in people with coeliac disease, ulcerative colitis and Crohn’s disease: a general population-based study. Aliment Pharmacol Ther 23(1):71–74
Hauser W et al (2010) Anxiety and depression in adult patients with celiac disease on a gluten-free diet. World J Gastroenterol 16(22):2780–2787
Carta MG, Hardoy MC, Boi MF, Mariotti S, Carpiniello B, Usai P (2002) Association between panic disorder, major depressive disorder and celiac disease: a possible role of thyroid autoimmunity. J Psychosom Res 53(3):789–793
Zylberberg HM, Demmer RT, Murray JA, Green PHR, Lebwohl B (2017) Depression and insomnia among individuals with celiac disease or on a gluten-free diet in the USA: results from a national survey. Eur J Gastroenterol Hepatol 29(9):1091–1096
Fasano A, Sapone A, Zevallos V, Schuppan D (2015) Nonceliac gluten sensitivity. Gastroenterology 148(6):1195–1204
Janatuinen EK, Kemppainen TA, Julkunen RJ, Kosma VM, Mäki M, Heikkinen M, Uusitupa MI (2002) No harm from five year ingestion of oats in coeliac disease. Gut 50(3):332–335
Thompson T (2004) Gluten contamination of commercial oat products in the United States. N Engl J Med 351(19):2021–2022
Lee AR, Ng DL, Zivin J, Green PHR (2007) Economic burden of a gluten-free diet. J Hum Nutr Diet 20(5):423–430
Panagiotou S, Kontogianni M (2016) The weekly economic burden of a gluten-free diet. Clin Nutr ESPEN 13:e56
Shah S, Akbari M, Vanga R, Kelly CP, Hansen J, Theethira T, Tariq S, Dennis M, Leffler DA (2014) Patient perception of treatment burden is high in celiac disease compared with other common conditions. Am J Gastroenterol 109(9):1304–1311
Murray JA, Kelly CP, Green PHR, Marcantonio A, Wu TT, Mäki M, Adelman DC, Ansari S, Ayub K, Basile A, Behrend C, Bercik P, Bressler B, Byrnes V, Chandan VS, Cheekati V, Chipps B, Coates A, Collatrella A, Condemi J, Corder C, Corren J, Curtis C, DeMeo M, Desta T, Devereaux C, DiMarino A, DuPree M, Ennis C, Fedorak R, Fogel R, Freeman S, Freilich B, Friedenberg K, Geenen D, Gill K, Goldsobel A, Goldstein J, Goldstein M, Gordon G, Hardi R, Harris L, Holmes R, Jagarlamundi K, James G, Kaplan M, Kirstein J, Knoll A, Kotfila R, Krause R, Kravitz A, Kreines M, Lähdeaho ML, Lamet M, Laskin K, Lebwohl B, Leffler D, Lewis S, Liakos S, Lundin K, Marks K, Merkes K, Minton S, Moussa S, Narayen V, Nehra V, Newton E, Nyberg A, Oosthuizen J, Otrok T, Patel D, Pepin C, Phillips R, Pyle G, Reichelderfer M, Reid B, Ritter T, Saini S, Sanders D, Schulman M, Semrad C, Shah S, Stockwell D, Strout C, Suez D, Tatum H, Torbenson MS, Turner M, Varunok P, Vazquez-Roque M, Vento A, Welton T, Wohlman R, Wood M, Woods S, Yousef K (2017) No difference between latiglutenase and placebo in reducing villous atrophy or improving symptoms in patients with symptomatic celiac disease. Gastroenterology 152(4):787–798 e2
Syage JA, Murray JA, Green PHR, Khosla C (2017) Latiglutenase improves symptoms in seropositive celiac disease patients while on a gluten-free diet. Dig Dis Sci 62:2428–2432
Leffler DA, Kelly CP, Green PHR, Fedorak RN, DiMarino A, Perrow W, Rasmussen H, Wang C, Bercik P, Bachir NM, Murray JA (2015) Larazotide acetate for persistent symptoms of celiac disease despite a gluten-free diet: a randomized controlled trial. Gastroenterology 148(7):1311–9 e6
Szaflarska-Poplawska A (2015) Non-dietary methods in the treatment of celiac disease. Prz Gastroenterol 10(1):12–17
Leffler DA, Dennis M, Hyett B, Kelly E, Schuppan D, Kelly CP (2007) Etiologies and predictors of diagnosis in nonresponsive celiac disease. Clin Gastroenterol Hepatol 5(4):445–450
Roshan B, Leffler DA, Jamma S, Dennis M, Sheth S, Falchuk K, Najarian R, Goldsmith J, Tariq S, Schuppan D, Kelly CP (2011) The incidence and clinical spectrum of refractory celiac disease in a north american referral center. Am J Gastroenterol 106(5):923–928
Burkhardt JG, Chapa-Rodriguez A, Bahna SL (2017) Gluten sensitivities and the allergist: threshing the grain from the husks. Allergy
Pillon R, Ziberna F, Badina L, Ventura A, Longo G, Quaglia S, de Leo L, Vatta S, Martelossi S, Patano G, Not T, Berti I (2015) Prevalence of celiac disease in patients with severe food allergy. Allergy 70(10):1346–1349
Pallav K, Xu H, Leffler DA, Kabbani T, Kelly CP (2016) Immunoglobulin A deficiency in celiac disease in the United States. J Gastroenterol Hepatol 31(1):133–137
Venhoff N, Emmerich F, Neagu M, Salzer U, Koehn C, Driever S, Kreisel W, Rizzi M, Effelsberg NM, Kollert F, Goldacker S, Voll RE, Warnatz K, Thiel J (2013) The role of HLA DQ2 and DQ8 in dissecting celiac-like disease in common variable immunodeficiency. J Clin Immunol 33(5):909–916
Jorgensen SF et al (2016) A cross-sectional study of the prevalence of gastrointestinal symptoms and pathology in patients with common variable immunodeficiency. Am J Gastroenterol 111(10):1467–1475
Denham JM, Hill ID (2013) Celiac disease and autoimmunity: review and controversies. Curr Allergy Asthma Rep 13(4):347–353
Ress K, Annus T, Putnik U, Luts K, Uibo R, Uibo O (2014) Celiac disease in children with atopic dermatitis. Pediatr Dermatol 31(4):483–488
Enroth S, Dahlbom I, Hansson T, Johansson Å, Gyllensten U (2013) Prevalence and sensitization of atopic allergy and coeliac disease in the Northern Sweden Population Health Study. Int J Circumpolar Health. 72
Peroni DG et al (2010) Chronic urticaria and celiac disease: a case report. Pediatr Dermatol 27(1):108–109
Mery A (2004) Celiac disease presenting as urticaria. Allergy Asthma Proc 25(2):97–99
Ciacci C, Cavallaro R, Iovino P, Sabbatini F, Palumbo A, Amoruso D, Tortora R, Mazzacca G (2004) Allergy prevalence in adult celiac disease. J Allergy Clin Immunol 113(6):1199–1203
Assa A, Frenkel-Nir Y, Tzur D, Katz LH, Shamir R (2017) Large population study shows that adolescents with celiac disease have an increased risk of multiple autoimmune and nonautoimmune comorbidities. Acta Paediatr 106(6):967–972
Ludvigsson JF, Hemminki K, Wahlström J, Almqvist C (2011) Celiac disease confers a 1.6-fold increased risk of asthma: a nationwide population-based cohort study. J Allergy Clin Immunol 127(4):1071–1073
Hommeida S, Alsawas M, Murad MH, Katzka DA, Grothe RM, Absah I (2017) The association between celiac disease and eosinophilic esophagitis: Mayo experience and meta-analysis of the literature. J Pediatr Gastroenterol Nutr 65(1):58–63
Johnson JB, Boynton KK, Peterson KA (2016) Co-occurrence of eosinophilic esophagitis and potential/probable celiac disease in an adult cohort: a possible association with implications for clinical practice. Dis Esophagus 29(8):977–982
Dharmaraj R, Hagglund K, Lyons H (2015) Eosinophilic esophagitis associated with celiac disease in children. BMC Res Notes 8:263
Jensen ET, Eluri S, Lebwohl B, Genta RM, Dellon ES (2015) Increased risk of esophageal eosinophilia and eosinophilic esophagitis in patients with active celiac disease on biopsy. Clin Gastroenterol Hepatol 13(8):1426–1431
Ari A, Morgenstern S, Chodick G, Matar M, Silbermintz A, Assa A, Mozer-Glassberg Y, Rinawi F, Nachmias-Friedler V, Shamir R, Zevit N (2017) Oesophageal eosinophilia in children with coeliac disease. Arch Dis Child 102(9):825–829
Dhalwani NN et al (2014) Women with celiac disease present with fertility problems no more often than women in the general population. Gastroenterology 147(6):1267–74 e1 quiz e13–4
Zugna D, Richiardi L, Akre O, Stephansson O, Ludvigsson JF (2010) A nationwide population-based study to determine whether coeliac disease is associated with infertility. Gut 59(11):1471–1475
Tata LJ, Card TR, Logan RFA, Hubbard RB, Smith CJP, West J (2005) Fertility and pregnancy-related events in women with celiac disease: a population-based cohort study. Gastroenterology 128(4):849–855
Askling J, Linet M, Gridley G, Halstensen TS, Ekström K, Ekbom A (2002) Cancer incidence in a population-based cohort of individuals hospitalized with celiac disease or dermatitis herpetiformis. Gastroenterology 123(5):1428–1435
Catassi C, Fabiani E, Corrao G, Barbato M, de Renzo A, Carella AM, Gabrielli A, Leoni P, Carroccio A, Baldassarre M, Bertolani P, Caramaschi P, Sozzi M, Guariso G, Volta U, Corazza GR, Italian Working Group on Coeliac Disease and Non-Hodgkin’s-Lymphoma (2002) Risk of non-Hodgkin lymphoma in celiac disease. JAMA 287(11):1413–1419
Green PH et al (2003) Risk of malignancy in patients with celiac disease. Am J Med 115(3):191–195
Smedby KE, Akerman M, Hildebrand H, Glimelius B, Ekbom A, Askling J (2005) Malignant lymphomas in coeliac disease: evidence of increased risks for lymphoma types other than enteropathy-type T cell lymphoma. Gut 54(1):54–59
West J, Logan RFA, Smith CJ, Hubbard RB, Card TR (2004) Malignancy and mortality in people with coeliac disease: population based cohort study. BMJ 329(7468):716–719
Ilus T, Kaukinen K, Virta LJ, Pukkala E, Collin P (2014) Incidence of malignancies in diagnosed celiac patients: a population-based estimate. Am J Gastroenterol 109(9):1471–1477
Lebwohl B, Granath F, Ekbom A, Smedby KE, Murray JA, Neugut AI, Green PHR, Ludvigsson JF (2013) Mucosal healing and risk for lymphoproliferative malignancy in celiac disease: a population-based cohort study. Ann Intern Med 159(3):169–175
Elfstrom P, Granath F, Ekstrom Smedby K, Montgomery SM, Askling J, Ekbom A, Ludvigsson JF (2011) Risk of lymphoproliferative malignancy in relation to small intestinal histopathology among patients with celiac disease. J Natl Cancer Inst 103(5):436–444
Tio M, Cox MR, Eslick GD (2012) Meta-analysis: coeliac disease and the risk of all-cause mortality, any malignancy and lymphoid malignancy. Aliment Pharmacol Ther 35(5):540–551
Abdul Sultan A, Crooks CJ, Card T, Tata LJ, Fleming KM, West J (2015) Causes of death in people with coeliac disease in England compared with the general population: a competing risk analysis. Gut 64(8):1220–1226
Godfrey JD, Brantner TL, Brinjikji W, Christensen KN, Brogan DL, van Dyke CT, Lahr BD, Larson JJ, Rubio–Tapia A, Melton LJ III, Zinsmeister AR, Kyle RA, Murray JA (2010) Morbidity and mortality among older individuals with undiagnosed celiac disease. Gastroenterology 139(3):763–769
Zanini B, Lanzarotto F, Mora A, Bertolazzi S, Turini D, Cesana B, Donato F, Ricci C, Lonati F, Vassallo F, Scarcella C, Lanzini A (2010) Five year time course of celiac disease serology during gluten free diet: results of a community based “CD-Watch” program. Dig Liver Dis 42(12):865–870
Rubio-Tapia A, Rahim MW, See JA, Lahr BD, Wu TT, Murray JA (2010) Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am J Gastroenterol 105(6):1412–1420
Wahab PJ, Meijer JW, Mulder CJ (2002) Histologic follow-up of people with celiac disease on a gluten-free diet: slow and incomplete recovery. Am J Clin Pathol 118(3):459–463
Kalayci AG, Kansu A, Girgin N, Kucuk O, Aras G (2001) Bone mineral density and importance of a gluten-free diet in patients with celiac disease in childhood. Pediatrics 108(5):E89
McFarlane XA, Bhalla AK, Robertson DA (1996) Effect of a gluten free diet on osteopenia in adults with newly diagnosed coeliac disease. Gut 39(2):180–184
Kemppainen TA, Heikkinen MT, Ristikankare MK, Kosma VM, Julkunen RJ (2010) Nutrient intakes during diets including unkilned and large amounts of oats in celiac disease. Eur J Clin Nutr 64(1):62–67
Kamycheva E, Goto T, Camargo CA Jr (2017) Blood levels of lead and mercury and celiac disease seropositivity: the US National Health and Nutrition Examination Survey. Environ Sci Pollut Res Int 24(9):8385–8391
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest. However, for full disclosure, Dr. Clarke is on the speakers bureau for AbbVie, Takeda, and Janssen. In addition, he has served on an Ad Board for Pfizer.
Ethical Approval and Informed Consent
This is a review article, no patients were involved and informed consent was not required. Permission was obtained for all images used and appropriate attribution/acknowledgement stated.
Rights and permissions
About this article
Cite this article
McAllister, B.P., Williams, E. & Clarke, K. A Comprehensive Review of Celiac Disease/Gluten-Sensitive Enteropathies. Clinic Rev Allerg Immunol 57, 226–243 (2019). https://doi.org/10.1007/s12016-018-8691-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-018-8691-2