Abstract
Recently, more and more clinical trials have been performed to evaluate the effects of anti-interleukin (IL)-5 antibodies in eosinophilic asthma. However, a confirm conclusion has not been well established. We therefore sought to conduct a meta-analysis to assess the overall efficacy and safety of anti-interleukin 5 treatments in eosinophilic asthma. RCTs of anti-interleukin 5 treatments in eosinophilic asthma published up to June 2016 in PubMed, Embase, Cochrane library databases, and CBM, which reported pulmonary functions, quality-of-life scores, asthmatic exacerbations, and adverse events were included. Fixed-effect models were used to calculate mean difference, relative risks (RR), and 95 % CIs. Twelve studies involving 3340 patients were identified. Pooled analysis revealed significant improvements in FEV1 (nine trials, 1935 subjects; MD = 0.12; 95 % CI, 0.08–0.16), and Asthma Quality-of-Life Questionnaire scores (five trials, 1334 subjects; MD = 0.23; 95 % CI, 0.13–0.34). Anti-interleukin 5 treatment was also associated with significantly decreased exacerbation risk than placebo (six trials, 875 subjects; RR = 0.52; 95 % CI, 0.46 to 0.59) and a lower incidence of adverse events (eight trials, 1754 subjects; RR = 0.93; 95 % CI, 0.89 to 0.97). Anti-interleukin 5 treatment is well tolerated and could significantly improve FEV1, quality of life, and reduced exacerbations risk in patients with eosinophilic asthma. Further trials are necessary to assess the baseline blood eosinophil count to identify the optimal patients of eosinophilic asthma that could benefit from anti-interleukin 5 therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Asthma is a heterogeneous disease that can be divided into different clinical phenotypes [1]. Eosinophilic asthma, which is the most predominant phenotype, accounts for approximately 50 to 60 % of the total asthma population [2]. The presence of eosinophils and helper T cell 2 (Th2) cytokines have been confirmed to participate in the eosinophilic asthmatic airways [3, 4]. Interleukin 5 (IL-5), the Th2 cell cytokine, plays an important role in eosinophil maturation, differentiation, recruitment, and survival [5]. Thus, the clinical application of antibodies that target IL-5 and its receptor has been considered for asthma treatment in recent years. The humanized anti-interleukin 5 monoclonal antibodies, mepolizumab (formerly termed SCH55700), reslizumab (formerly termed Res-5-0010), and benralizumab (formerly termed MEDI-563) have been developed for clinical application.
Earlier clinical trials showed mepolizumab was effective in mild-moderate asthmatics at reducing sputum and blood eosinophils but had no effect on clinical signs and symptoms [6]. Subsequent studies in a selected group of patients with severe asthma and persistent sputum or blood eosinophilia, mepolizumab decreased exacerbations, reduced use of oral corticosteroids, and improved symptoms and lung function compared with placebo [7–10]. Moreover, several trials evaluated the role of other alternative IL-5 blockers such as reslizumab and benralizumab. However, results from single studies have been less consistent and some analyses may have been underpowered to detect statistically significant differences. Therefore, we conducted a meta-analysis of randomized controlled trials (RCTs) to assess the overall efficacy and safety of anti-interleukin 5 treatments on eosinophilic asthma.
Methods
Data Sources and Searches
We searched PubMed, Embase, the Cochrane Library, and the Chinese Biological Medicine (CBM) database for articles published up to June 2016 to identify all trials assessing anti-interleukin 5 therapy for patients with eosinophilic asthma, using the following search strategies: (mepolizumab or benralizumab or reslizumab or monoclonal antibody or anti-interleukin 5 or anti-IL-5) and (“pulmonary eosinophilia” [Mesh] or eosinophilia, pulmonary or simple pulmonary eosinophilia or pneumonia, eosinophilic or eosinophilic pneumonias or pneumonias, eosinophilic or eosinophilic pneumonia). Publication species of humans was limited. No language restrictions were applied. In addition, the relevant review articles and their references were checked as well.
Study Selection
Specific inclusion criteria were as follows: (1) adults/adolescents (12 years or older) with a diagnosis of eosinophilic asthma, eosinophilic inflammation was shown by one or more criteria at study entry or in the previous year: a sputum eosinophil count ≥2.5 % or the eosinophil/lymphocyte and eosinophil/neutrophil (ELEN; a surrogate blood-based marker of sputum eosinophilia) index was positive, an exhaled nitric oxide concentration (FENO) ≥50 ppb, and an asthma-related peripheral blood eosinophil count ≥300 μL [11, 12]; (2) participants with anti-interleukin 5 therapy at any dose; (3) randomized (parallel group) placebo-controlled trials; and (4) RCTs reporting the following outcomes: lung function, asthma exacerbations, asthma control and quality-of-life scores, and adverse events. An exacerbation was defined as a worsening of asthma symptoms requiring treatment with systemic corticosteroids or increased doses of rescue medication, and/or the need for asthma-related hospitalization or an emergency room visit, or an unscheduled physician visit. Two authors (FPW) and (XFX) independently screened all references according to the selection criteria. Any disagreements were resolved through discussion or adjudicated by a third author (HM) when necessary.
Data Extraction and Quality Assessment
The Preferred Reporting Items for Systemic Reviews and Meta-Analyses (PRISMA) statement was followed [13]. Using a standardized data extraction form, FPW and XFX independently extracted data in blinded fashion from eligible studies based on authors, publication year, study design, patient demographic characteristics (age, gender, etc.), type of anti-interleukin 5, dose, and therapy duration and outcome definitions. Disagreements were resolved through consultation with a third author (HM). In addition, we assessed risk of bias using the Cochrane Collaboration’s domains which included adequate sequence generation, allocation concealment, blinding of participants and personnel, incomplete outcome data, selective reporting, and other bias [14].
Statistical Analyses
Intervention effects were expressed with risk ratios (RR) and 95 % confidence intervals (CIs) for dichotomous data and mean differences (MD) and 95 % CIs for continuous data. If a study presents more than two interventions, we combined two or three intervention groups into a single-intervention group in accordance with the Cochrane handbook [14]. Dichotomous variables (asthma exacerbations, adverse events) were reported as frequency and proportion, while continuous (FEV1, Asthma Quality-of-Life Questionnaire (AQLQ) were shown as mean change from baseline and standard derivation (SD). Heterogeneity was quantified by I 2 statistic and the chi-squared test. I 2 value of 50 % indicates significant heterogeneity [15]. Fixed-effect models were used except where we identified statistical heterogeneity when we used a random effect model. Publication bias was tested using funnel plot with the Begg’s and Egger’s tests [16]. All analyses were performed with according to the intention-to-treat principle. All statistical analysis was performed using Review Manager (version 5.3, The Cochrane Collaboration) and Stata (version 12.0, Stata Corporation, USA), and a P value <0.05 was considered statistically significant.
Meta-analyses may result in type I errors due to sparse data and repetitive testing of accumulating data [17]. To assess the risk of type I errors, we applied trial sequential analysis, a method which can determine whether the evidence in a meta-analysis is reliable and conclusive. If the cumulative z curve crosses the trial the boundaries and the required information size, evidence to reach a conclusion is sufficient and no further trials are needed. We estimated the required information size for FEV1 using α = 0.05 (two-sided), β = 0.20 (power of 80 %). TSA version 0.9 beta (http://www.ctu.dk/tsa) were used for the analyses [18].
Results
Search Results
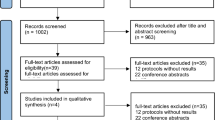
A total of 457 potentially relevant articles were identified. Among all the potential studies, 34 duplicate records were removed, leaving 423 articles for screening. After reviewing the titles and abstracts, we identified and retrieved 45 database references in full text for review. Of these articles, 33 articles were excluded owing to wrong population (n = 16), no placebo control (n = 5), and data unavailable (n = 12). Ultimately, 12 studies were included for our systematic review and meta-analysis (Fig. 1).
Characteristics of Studies
We included 12 studies [7–12, 19–24] with 3340 participants (Table 1). The sample sizes ranged from 20 to 621 subjects. Of these, five studies used mepolizumab [7–11], four reslizumab [19–22], and three benralizumab [12, 23, 24]. Treatment duration ranged from 1 day to 52 weeks and follow-up ranged from 12 to 52 weeks. The mean age of patients was 46.8 years old. The anti-interleukin 5 was administered exclusively through intravenous infusion (IV) in seven studies [7, 8, 11, 19–22], and five studies [9, 10, 12, 23, 24] had a subcutaneous (SC) arm. Outcome reporting was varied among studies. FEV1 was reported in liters in nine trials [7, 8, 10–12, 19–22], as percent of predicted in another two [9, 24], and as both in three trials [8, 10, 19]. Airway hyper-reactivity was reported as the dose of histamine required to produce a 20 % drop in FEV1 (PC20) in one trial [7]. Five studies included severe eosinophilic asthmatics [8–11, 19], three studies included refractory or uncontrolled eosinophilic asthmatics [7, 20, 22], and the remaining studies did not specify asthma severity [12, 21, 23, 24].
Lung Function
FEV1
Nine trials reported the data on FEV1. Mepolizumab was used in four studies showed significant effect on FEV1 (MD = 0.09; 95 % CI, 0.03 to 0.14; P = 0.002). Reslizumab was reported in four studies, also could significantly improve FEV1 (MD = 0.15, 95 % CI, 0.09 to 0.22; P < 0.001). Benralizumab was used in only one study (MD = 0.14, 95 % CI, 0.02 to 0.26; P = 0.02). Overall, anti-interleukin 5 treatment were associated with significant improvements in FEV1 (MD = 0.12; 95 % CI, 0.08 to 0.16; P < 0.001) (Fig. 2), with minimal heterogeneity (I 2 = 15 %, P = 0.3), with no evidence of publication bias (Egger’s P = 0.61; Begg’s P = 0.86) (Fig. 3a). Trial sequential analysis found that the optimal sample size needed to reliably detect a plausible effect of anti-interleukin 5 treatment on FEV1 of eosinophilic asthma was 1789 patients, and the patients including in our analysis is far more than it. TSA showed that the cumulative z curve crossed both the conventional boundary and the sequential monitoring boundary, which suggested that the cumulative evidence is reliable and conclusive. Thus, further trials were not required and were unlikely to alter the conclusions (Fig. 4).
Trial sequential analysis of 12 trials comparing anti-interleukin 5 with placebo for FEV1. Trial sequential analysis of ten groups (one trial contains two groups) illustrating that the cumulative z curve crossed both the conventional boundary and the trial sequential monitoring boundary, establishing sufficient and conclusive evidence and suggesting that further trials are not required. Using α = 0.05 (two-sided) and β = 0.20 (power of 80 %) calculate that the optimal sample size was 1789 patients
Peak Expiratory Flow and Airway Hyper-reactivity
Only one trial depicted the change of peak expiratory flow (PEF) [24] and histamine PC20 [7] after treatment with anti-interleukin 5. However, both demonstrated that there was no difference between anti-interleukin 5 treatment and placebo in terms of PEF and histamine PC20 values.
Asthma Quality-of-Life Questionnaire
Five trials provided data about AQLQ scores. The pooled analysis showed anti-interleukin 5 treatment was associated with a significant increase in AQLQ scores (MD = 0.23; 95 % CI, 0.13–0.34; P < 0.001), with no significant heterogeneity (I 2 = 0 %; P = 0.81) (Fig. 5). When it comes to subgroups, AQLQ scores improved both in mepolizumab treatment (MD = 0.18; 95 % CI, 0.01–0.36; P = 0.04) and reslizumab (MD = 0.27; 95 % CI, 0.13–0.42; P < 0.001). Benralizumab only used in one study (MD = 0.21; 95 % CI, −0.12–0.54; P = 0.22). Egger and Begg test showed no evidence of publication bias (P = 0.34; P = 0.45) (Fig. 3b).
Asthma Exacerbations
Six studies were included. Overall, compared with placebo, asthma exacerbations risk was significantly decreased with anti-interleukin 5 treatment (RR = 0.52; 95 % CI, 0.46 to 0.59; P < 0.001) (Fig. 6), and there was no heterogeneity among studies (I 2 = 0 %, P = 0.5). When looking at subgroups, mepolizumab (RR = 0.55; 95 % CI, 0.47 to 0.64; P < 0.001) and reslizumab (RR = 0.46; 95 % CI, 0.37 to 0.58; P < 0.001) were also linked to markedly lower asthma exacerbations. There was no evidence of publication bias (Egger’s P = 0.41; Begg’s P = 0.37) (Fig. 3c).
Adverse Events
Eight studies mentioned adverse events. Anti-interleukin 5 treatment was associated with a trend of lower adverse events incidence (RR = 0.93; 95 % CI, 0.89 to 0.97; P = 0.001) (Fig. 7), with no heterogeneity (I 2 = 0 %, P = 0.55). In subgroup analysis, however, we found no significant differences in both mepolizumab (RR = 0.96; 95 % CI, 0.9–1.03; P = 0.3) and benralizumab treatment groups (RR = 0.91; 95 % CI, 0.81–1.02; P = 0.09). Only treatment with reslizumab was associated with a trend of lower adverse events incidence (RR = 0.92; 95 % CI, 0.87–0.97; P = 0.003). Publication bias was not found (Egger’s P = 0.16; Begg’s P = 0.21) (Fig. 3d). The incidence of serious adverse events was low in the anti-interleukin 5 treatment group (1–16 %). Common adverse events were nasopharyngitis, headache, asthma worsening, injection-site reactions and upper respiratory tract infection (Table 2).
Discussion
Our meta-analysis indicated that anti-interleukin 5 treatment was well tolerated and could significantly improve FEV1, and quality of life, and reduced the incidence of asthma exacerbations in patients with eosinophilic asthma.
Asthma is a heterogeneous condition that affects more than 300 million people worldwide [25]. Eosinophilic asthma phenotype is characterized by persistent eosinophilic airway inflammation. IL-5 is central to this asthma phenotype because it is responsible for eosinophil production, survival, maturation, recruitment, and activation at sites of allergic inflammation [26]. Given the relationship of IL-5 to eosinophilia and asthma severity, humanized monoclonal antibodies targeting IL-5 have shown great promise in eosinophilic asthma, especially for refractory/severe eosinophilic asthma. Several clinical trials have been performed evaluating the role of anti-interleukin 5 therapy in eosinophilic asthma. However, the evidence is inadequate for drawing robust conclusions, as the sample sizes of these studies are varied and their conclusions inconsistent. Therefore, it seems reasonable to explore this issue further.
Based on the pooled analyses, we found that anti-interleukin 5 could significantly improve FEV1. The clinical relevance of this finding to patients may be clinically important. However, previous two systematic reviews [27, 28] failed to show a significant effect in lung function, because they were based only on studies with mepolizumab, and Liu et al. [27] only selected one pair of interventions and excluded the others when studies with multiple intervention groups, which is not generally recommended by Cochrane Collaboration [14]. Additionally, both the two systematic reviews included an unselected population of patients with asthma. In contrast to previous systematic reviews [27, 28], we identified a selected population of patients with eosinophilic asthma and included all trials involving anti-interleukin 5 treatment (mepolizumab, reslizumab, benralizumab), not only just mepolizumab. In addition, we used TSA to calculate a required information size for meta-analysis, which can correct for the increased risk of random errors. Therefore, our results may be more believable. On the other hand, the different outcomes between our meta-analysis and the previous two systematic reviews suggested that anti-interleukin 5 therapy might be effective only in a targeted subgroup with an eosinophilic phenotype. For example, early trials of mepolizumab were disappointing because the recruitment of asthmatics did not include specifying the presence of eosinophilic airway inflammation [6, 29]. In subsequent studies, when anti-interleukin 5 therapies have been used in a patient population selected for eosinophils, results have been more promising [7–11]. In addition, an FDA advisory committee has recently approved the use of mepolizumab for patients with eosinophilic severe asthma. Therefore, the clinical effects of anti-interleukin 5 treatment are much more influenced by patient selection.
AQLQ is a disease-specific health-related quality-of-life instrument which contains 32 items, and it has proved responsive in before-after studies and in clinical trials [30]. Asthma Control Questionnaire (ACQ) applied in studies are used variously, some used ACQ-6, while others used ACQ-5 or ACQ-7, which makes this parameter was unsuitable for analysis. Therefore, our meta-analysis used AQLQ score to evaluate the life quality of patients. In our study, there was a significant improvement in AQLQ. This result was consistent with previous systematic reviews [27, 28]. However, the mean change in AQLQ is less the clinical minimally important difference of 0.5 units [31]. So, the clinical relevance of this finding to patients may not be clinically important.
Asthma exacerbations are associated with substantial morbidity and mortality [32]. Decreasing the asthma exacerbations rate is a key goal of asthma management. Previous studies revealed that increased blood and sputum eosinophil counts have emerged as independent risk factors for future asthma exacerbations [33, 34]. Studies using specific inhibitors of have also shown a link between eosinophils and the pathogenesis of asthma exacerbations [35, 36]. These foundings strongly suggested that the presence of airway eosinophilia is a clinically relevant finding, and anti-interleukin 5 treatments might be a promising strategy to treat asthma. Further support of this idea, our meta-analysis showed a significant reduction in exacerbation rates. The improvements in asthma-related quality of life with anti-IL-5 treatment may probably be attributed to preventing asthma exacerbations.
Not all studies reported adverse events. The safety profile of mepolizumab has been up to par with the placebo in the RCTs we included, and the most frequently reported adverse events were nasopharyngitis and headache. Consistent with mepolizumab, the overall safety profile of reslizumab was similar to that of placebo based on studies we included, and the most widely reported adverse events were asthma worsening, nasopharyngitis, upper respiratory tract infection, and sinusitis. As for benralizumab, nasopharyngitis and injection-site reactions were most common adverse events. One RCT which assessed the efficacy and safety of benralizumab both in patients with eosinophilic asthma and non-eosinophilic asthma revealed the overall incidence of adverse events irrespective of causality was slightly higher in the benralizumab treatment groups than in the placebo groups [12]. However, adverse events were summarized for eosinophilic and non-eosinophilic participants combined in this study, which makes us unable to get detailed data, and we only included two studies on benralizumab to analysis. Pooled analysis found treatment with reslizumab was associated with a trend of lower adverse event incidence, but the numbers of patients reporting adverse events was similar in both mepolizumab and benralizumab treatment groups when compared with the placebo. Therefore, benralizumab needs more data to support its safety profile.
There are several potential limitations that need to be considered. Firstly, the severity and baseline therapy of eosinophilic asthma varied among studies, so it is not possible to investigate the impact of those on the results. Secondly, several studies were of a small scale, which may affect the power to explore the real outcomes. Moreover, given the variety of the anti-interleukin 5 therapy, we were unable to assess factors that may impact the effects of anti-interleukin 5 therapy, such as dose, and treatment duration. Lastly, as commended by the Cochrane handbook, we combined two or three intervention groups into a single intervention group regardless of different intervention dosage and administration routine, which made it difficult to determine the optimal dose. Another important aspect that we should pay attention is that we used a blood eosinophil count more than 300 cells/μL at baseline to select patients with the eosinophilic phenotype. However, a recent study re-examined baseline blood eosinophil counts from previous two studies [10, 11] on mepolizumab found that the use of the baseline at a threshold of at least 150 cells/μL will select patients with the phenotype that is likely to achieve important reductions in the rate of exacerbations and resultant improvements in the quality of life and asthma control with mepolizumab [37]. However, given the lack of individual patient data among all studies, we failed to further analysis the relationship between blood eosinophil counts ≥150 cells/μL at baseline and outcomes of treatment.
Conclusions
In summary, the current meta-analysis indicated that anti-interleukin 5 treatment was well tolerated and could significantly improve FEV1, quality of life, and reduced asthma exacerbation risk in patients with eosinophilic asthma. Therefore, the humanized anti-interleukin 5 monoclonal antibodies may be effective and safe for eosinophilic asthma. The results highlight the importance of selection asthma phenotypes could derive clinical benefit from anti-interleukin 5 therapy. Nasopharyngitis was the most frequently reported adverse event in either study involving anti-interleukin 5 treatments, and benralizumab needs more data to support its safety profile. Further trials are necessary to assess the baseline blood eosinophil count to identify the optimal patients of eosinophilic asthma that could benefit from anti-interleukin 5 treatment.
References
Gibson PG (2009) Inflammatory phenotypes in adult asthma: clinical applications. Clin Respir J 3(4):198–206. doi:10.1111/j.1752-699X.2009.00162.x
Aleman F, Lim HF, Nair P (2016) Eosinophilic endotype of asthma. Immunol Allergy Clin North Am 36(3):559–568. doi:10.1016/j.iac.2016.03.006
Garcia G, Taille C, Laveneziana P, Bourdin A, Chanez P, Humbert M (2013) Anti-interleukin-5 therapy in severe asthma. Eur Respir Rev 22(129):251–257. doi:10.1183/09059180.00004013
Durham AL, Caramori G, Chung KF, Adcock IM (2016) Targeted anti-inflammatory therapeutics in asthma and chronic obstructive lung disease. Transl Res 167(1):192–203. doi:10.1016/j.trsl.2015.08.004
Takatsu K, Nakajima H (2008) IL-5 and eosinophilia. Curr Opin Immunol 20(3):288–294. doi:10.1016/j.coi.2008.04.001
Flood-Page P, Swenson C, Faiferman I, Matthews J, Williams M, Brannick L, Robinson D, Wenzel S, Busse W, Hansel TT, Barnes NC (2007) A study to evaluate safety and efficacy of mepolizumab in patients with moderate persistent asthma. Am J Respir Crit Care Med 176(11):1062–1071
Haldar P, Brightling CE, Hargadon B, Gupta S, Monteiro W, Sousa A, Marshall RP, Bradding P, Green RH, WardIaw AJ, Pavord ID (2009) Mepolizumab and exacerbations of refractory eosinophilic asthma. N Engl J Med 360(10):973–984
Nair P, Pizzichini MMM, Kjarsgaard M, Inman MD, Efthimiadis A, Pizzichini E, Hargreave FE, O’Byrne PM (2009) Mepolizumab for prednisone-dependent asthma with sputum eosinophilia. N Engl J Med 360(10):985–993
Bel EH, Wenzel SE, Thompson PJ, Prazma CM, Keene ON, Yancey SW, Ortega HG, Pavord ID (2014) Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N Engl J Med 371(13):1189–1197. doi:10.1056/NEJMoa1403291
Ortega HG, Liu MC, Pavord ID, Brusselle GG, FitzGerald JM, Chetta A, Humbert M, Katz LE, Keene ON, Yancey SW, Chanez P, Investigators M (2014) Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J Med 371(13):1198–1207
Pavord ID, Korn S, Howarth P, Bleecker ER, Buhl R, Keene ON, Ortega H, Chanez P (2012) Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet (London, England) 380(9842):651–659. doi:10.1016/s0140-6736(12)60988-x
Castro M, Wenzel SE, Bleecker ER, Pizzichini E, Kuna P, Busse WW, Gossage DL, Ward CK, Wu Y, Wang B, Khatry DB, Merwe R, Kolbeck R, Molfino NA, Raible DG (2014) Benralizumab, an anti-interleukin 5 receptor alpha monoclonal antibody, versus placebo for uncontrolled eosinophilic asthma: a phase 2b randomised dose-ranging study. Lancet Respir Med 2(11):878–890. doi:10.1016/S2213-2600%2814%2970201-2
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62(10):1006–1012. doi:10.1016/j.jclinepi.2009.06.005
Higgins J, Green S (2013) Cochrane handbook for systematic reviews of interventions version 5.1. 0, The Cochrane Collaboration, 2011
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558. doi:10.1002/sim.1186
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ (Clinical research ed) 315(7109):629–634
Brok J, Thorlund K, Wetterslev J, Gluud C (2009) Apparently conclusive meta-analyses may be inconclusive—trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta-analyses. Int J Epidemiol 38(1):287–298
Thorlund K, Engstrøm J, Wetterslev J, Brok J, Imberger G, Gluud C (2011) User manual for trial sequential analysis (TSA). Copenhagen Trial Unit, Centre for Clinical Intervention Research, Copenhagen
Castro M, Mathur S, Hargreave F, Boulet LP, Xie F, Young J, Wilkins HJ, Henkel T, Nair P (2011) Reslizumab for poorly controlled, eosinophilic asthma: a randomized, placebo-controlled study. Am J Respir Crit Care Med 184(10):1125–1132. doi:10.1164/rccm.201103-0396OC
Castro M, Zangrilli J, Wechsler ME, Bateman ED, Brusselle GG, Bardin P (2015) Reslizumab for inadequately controlled asthma with elevated blood eosinophil counts: results from two multicentre, parallel, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet Respir Med 3(5):355–366. doi:10.1016/S2213-2600(15)00042-9
Corren J, Weinstein S, Janka L, Zangrilli J, Garin M (2016) Phase 3 study of reslizumab in patients with poorly controlled asthma: effects across a broad range of eosinophil counts. Chest. doi:10.1016/j.chest.2016.03.018
Bjermer L, Lemiere C, Maspero J, Weiss S, Zangrilli J, Germinaro M (2016) Reslizumab for inadequately controlled asthma with elevated blood eosinophil levels: a randomized phase 3 study. Chest. doi:10.1016/j.chest.2016.03.032
Laviolette M, Gossage DL, Gauvreau G, Leigh R, Olivenstein R, Katial R, Busse WW, Wenzel S, Wu Y, Datta V, Kolbeck R, Molfino NA (2013) Effects of benralizumab on airway eosinophils in asthmatic patients with sputum eosinophilia. J Allergy Clin Immunol 132(5):1086–1096.e1085. doi:10.1016/j.jaci.2013.05.020
Park HS, Kim MK, Imai N, Nakanishi T, Adachi M, Ohta K (2016) A phase 2a study of benralizumab for patients with eosinophilic asthma in South Korea and Japan. Int Arch Allergy Immunol 169(3):135–145. doi:10.1159/000444799
The Global Asthma Report (2014) Auckland. Global Asthma Network, New Zealand, Available at: http://www.globalasthmareport.org/resources/Global_Asthma_Report_2014.pdf
Cardet JC, Israel E (2015) Update on reslizumab for eosinophilic asthma. Expert Opin Biol Ther 15(10):1531–1539. doi:10.1517/14712598.2015.1090972
Liu Y, Zhang S, Li DW, Jiang SJ (2013) Efficacy of anti-interleukin-5 therapy with mepolizumab in patients with asthma: a meta-analysis of randomized placebo-controlled trials. PLoS One 8:3. doi:10.1371/journal.pone.0059872
Powell C, Milan SJ, Dwan K, Bax L, Walters N (2015) Mepolizumab versus placebo for asthma. Cochrane Database Syst Rev 7:CD010834
Leckie MJ, Ten Brinke A, Khan J, Diamant Z, O’Connor BJ, Walls CM, Mathur AK, Cowley HC, Chung KF, Djukanovic R, Hansel TT, Holgate ST, Sterk PJ, Barnes PJ (2000) Effects of an interleukin-5 blocking monoclonal antibody on eosinophils, airway hyper-responsiveness, and the late asthmatic response. Lancet (London, England) 356(SUPPL):2144–2148
Juniper EF, Guyatt G, Epstein R, Ferrie P, Jaeschke R, Hiller TK (1992) Evaluation of impairment of health related quality of life in asthma: development of a questionnaire for use in clinical trials. Thorax 47(2):76–83
Juniper EF, Guyatt GH, Willan A, Griffith LE (1994) Determining a minimal important change in a disease-specific Quality of Life Questionnaire. J Clin Epidemiol 47(1):81–87
Masoli M, Fabian D, Holt S, Beasley R, Global Initiative for Asthma P (2004) The global burden of asthma: executive summary of the GINA Dissemination Committee report. Allergy 59(5):469–478. doi:10.1111/j.1398-9995.2004.00526.x
Schatz M, Li Q, Chen W, Khatry D, Tran T, Zeiger R (2014) Elevated blood eosinophil level is a risk factor for exacerbations in adult persistent asthma. In: C33. Cytokines and asthma mediators. Am Thoracic Soc A4235–A4235
Malinovschi A, Fonseca JA, Jacinto T, Alving K, Janson C (2013) Exhaled nitric oxide levels and blood eosinophil counts independently associate with wheeze and asthma events in National Health and Nutrition Examination Survey subjects. The Journal of Allergy and Clinical Immunology 132 (4):821–827 e821–825. doi:10.1016/j.jaci.2013.06.007
Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, Wardlaw AJ, Pavord ID (2002) Asthma exacerbations and sputum eosinophil counts: a randomised controlled trial. Lancet (London, England) 360(9347):1715–1721. doi:10.1016/S0140-6736(02)11679-5
Nadif R, Siroux V, Oryszczyn MP, Ravault C, Pison C, Pin I, Epidemiological Study on the Genetics and Environment of Asthma (EGEA) (2009) Heterogeneity of asthma according to blood inflammatory patterns. Thorax 64(5):374–380. doi:10.1136/thx.2008.103069
Ortega HG, Yancey SW, Mayer B, Gunsoy NB, Keene ON, Bleecker ER, Brightling CE, Pavord ID (2016) Severe eosinophilic asthma treated with mepolizumab stratified by baseline eosinophil thresholds: a secondary analysis of the DREAM and MENSA studies. Lancet Respir Med 4(7):549–556. doi:10.1016/S2213-2600(16)30031-5
Acknowledgments
We are grateful to the authors of the primary studies included in this meta-analysis. We thank Jiao-Qing Tang for his help and editorial advice during the preparation of the review.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study did not receive any funding.
Conflict of Interest
The authors declare no conflict of interest.
Author Contributions
Fa-Ping Wang and Xiao-Feng Xiong performed the literature searches, selected the studies, analyzed the data, and wrote the manuscript draft. Ting Liu and Su-Yun Li aided in the data analysis. Hui Mao and De-Yun Cheng designed the study and revised manuscript.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Fa-Ping Wang and Xiao-Feng Xiong contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, FP., Xiong, XF., Liu, T. et al. Anti-interleukin 5 Therapy for Eosinophilic Asthma: a Meta-analysis of Randomized Clinical Trials. Clinic Rev Allerg Immunol 54, 318–330 (2018). https://doi.org/10.1007/s12016-016-8588-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-016-8588-x