Abstract
Coffee is one of the most widely consumed beverages in the world. However, coffee plants are often exposed to potentially toxic elements (PTEs) pollution. The main aims of current study were to detect the PTEs in instant coffee and health risk assessment of consumers in Bandar Abbas city. To achieve this, 40 samples of instant coffee were randomly collected from various points in the city in 2023 and PTEs concentrations were measured using flame atomic absorption spectrometry (FAAS). The non-carcinogenic and carcinogenic risks were calculated using Monte Carlo simulation (MCS) method. The concentrations of Fe and Cu were higher than other PTEs, equaling 404.41 mg/kg and 0.0046 mg/kg, respectively. The non-carcinogenic risk assessment revealed that THQ (Fe > Pb > As > Cd > Ni > Cu) and TTHQ levels were less than 1 based on the 95% percentile in adults and children, indicating there is no possibility of a non-carcinogenic risk associated with instant coffee. The carcinogenic risk due to inorganic As in instant coffee was acceptable (2.63E-5 and 1.27E-5 based on the 95% percentile for adults and children, respectively), therefore PTEs in instant coffee do not endanger the health of consumers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Potentially toxic elements (PTEs) refer to metals and metalloids such as copper (Cu), iron (Fe), manganese (Mn), arsenic (As), zinc (Zn), lead (Pb), chromium (Cr), cadmium (Cd), and nickel (Ni) that can have harmful effects on organisms in high concentrations [1,2,3]. Both natural processes, such as the weathering of rocks, and human activity, such as industrial emissions, mining, and inappropriate waste management, can release these metals into the environment [4, 5]. The impact of PTEs on human health varies depending on the particular metal, its concentration, the length of exposure, and each person's vulnerability [1, 6, 7]. The central nervous system, liver [8], and kidneys are just a few of the essential organs that might affected by various factors such as PTEs [9,10,11]. Additionally, it may result in a number of medical conditions, such as developmental and neurological difficulties, respiratory problems, cardiovascular diseases, and some malignancies [12,13,14]. PTEs can contaminate water, soil, and air as they accumulate in the environment [15,16,17]. The intake of tainted food or water, skin contact, inhalation, and other methods can all expose people to PTEs [18,19,20,21]. Once PTEs enter the body, they can bond to tissues and organs, which can have a variety of negative health impacts [22,23,24,25]. In recent decades, with the emergence of various chemical pollutants and their entry into the food chain, concerns about the increase in disease have increased [26,27,28,29,30]. The increase levels of various contaminations in food, such as mycotoxins in dairy products [31, 32], aflatoxins in corn [33, 34] and heavy metals in dairy products and meat [35], together with other variables has increased risk of cancers [36,37,38,39,40,41] and other non-communicable diseases [42, 43].
Coffee is one of the most extensively consumed beverages in the world and is renowned for its energizing properties as well as its flavorful aroma [44]. Caffeine, a natural stimulant that has an impact on the central nervous system, is present in this beverage since it is made from the roasted seeds of the Coffea plant. When drunk in moderation, coffee has been linked to various potential health advantages. Coffee's caffeine content can improve cognitive function overall as well as mental alertness and attention. Additionally, it might lessen weariness and speed up reflexes. Because it helps speed up the metabolism and boost fat oxidation, caffeine is a common component in pre-workout supplements [45]. Coffee is known to increase stomach acid production, which in some people can lead to heartburn, acid reflux, or gastrointestinal discomfort. Additionally, if ingested in high amounts, it may have a slight diuretic effect, increasing urine production and perhaps leading to dehydration. Coffee contains substances that can bind to specific minerals like calcium and iron, potentially preventing the body from absorbing such minerals. When coffee is drunk with food, this impact is more pronounced [46]. As with any dietary choice, it is advisable to consult with a healthcare professional or nutritionist to determine what level of coffee consumption is suitable for individual circumstances and overall health goals. Different nations around the world have different per capita coffee consumption rates. Finland (12 kg/year) is the country with the highest per capita coffee consumption, according to data from the International Coffee Organization for 2021, (6.1 kg /year), including Norway, Iceland, Denmark, Sweden, Switzerland, Netherlands, Belgium, and Luxembourg. It's crucial to remember that these figures reflect average per-person consumption for each country and may change annually. Additionally, cultural preferences, accessibility, and other elements can affect coffee consumption.
According to some research, coffee, especially instant coffee, contains PTEs. Pb and Cd were identified in substantial amounts in instant coffee samples [47]. Several studies showed have shown that coffee infusions contain the highest levels of leached Pb and Cr elements. Noel et al. (2012) previously reported that Cr levels in coffee infusion averaged 0.0023 mg/50ml. According to the World Health Organization (2000), adults are allowed a daily consumption of 50 to 200 µg (0.05 to 0.2 mg) of Cr [48]. Since the consumption of instant coffee, instant coffee drinks, and natural coffee is rather high in Poland [49, 50], the level of PTEs is typically examined in ground coffee. The weekly Pb consumption from cereal coffee is 1.49 g (about 0.2% BMDL01 and 0.5% BMDL10). In contrast, hazard index (HI) due to Cd and Pb ranged from 0.036 (chicory and coffee blend) to 0.109 (cappuccino) [51]. Despite the widespread consumption of instant coffee in Iran, there was no study on the concentration of PTEs in instant coffee and human health risk assessment. The main aims of current study were determining the concentration of PTEs in instant coffee and human health risk assessment.
Materials and Methods
Chemical and Reagents
Standard solutions of Fe, Pb, Cd, Cu, Ni, As and other analytical chemical were purchased from Merck (Darmstadt, Germany).
Sampling, Sample Preparation, and Analysis of PTEs in Rapid Coffee
In this descriptive cross-sectional study, in total, 40 samples of rapid coffee, were randomly collected from local markets, supermarkets, and grocery stores in Bandar Abbas Province, in South of Iran in 2023. At first, 0.5 g of each sample was heated at 600°C for 24 h to remove organic compounds. The samples were digested in a mixture of 25 mL (3:1 v/v) of HNO3 and HCl[52]. The solutions samples were heated in a microwave at 800 W for 30 min until to 210 °C. The concentration of PTEs was detected using Graphite furnace atomic absorption spectroscopy (GFAAS) (AA-7000, Shimadzu, Kyoto, Japan) (Table 1).
Method Validation
To ensure the precision of the analytical method, quality assurance and quality control (QA/QC) were ensured based on previous studies [53]. The validation protocol included limit of quantification (LOQ), limit of detection (LOD) and recovery were measured [54]. Triplicate analyses were conducted on samples, blanks, and standards. The calibration curves, regression coefficient (R2), linearity equations, LOQ, LOD, and recovery of PTEs are indicated in Table 2 [55].
Health Risk Assessment
Non-Carcinogenic Risk Assessment
Lifetime Average Daily Dose (LADD) was estimated by the below equation [56,57,58,59]:
where C, represents the concentration of PTEs (mg/kg); IR, ingestion rate of coffee consumption (g/n-d); CF, conversion factor (g/n-d); EF, exposure frequency (350 days/year); ED, exposure duration (adults = 30 years and children = 6 years); BW, body weight (adults = 70 kg and children = 29 kg) and AT average lifetime. AT for non-carcinogenic risk for adults and children is equal to 10,950 days and 2,190 days, respectively and for carcinogenic risk for both is 25,550 days.
The non-carcinogenic risk was estimated by the current equation [60,61,62]:
In this equation, THQ represents target hazard quotient; RfD, oral reference dose (mg/kg-d) and TDI, tolerable daily intake (mg/kg-d). TDI for Pb and RfD for Cd, Fe, Ni, Cu and As is 0.0036, 0.001, 0.7, 0.011, 0.04 and 0.0003 mg/kg-d, respectively [63].
Total non-carcinogenic risk was calculated current equation [60]:
where, TTHQ is total target hazard quotient.
When TTHQ is lower than 1, non-cancer risk is acceptable.
Carcinogenic Risk
The carcinogenic risk of As was estimated by current equation [56]:
In this equation, CSF is cancer slope factor (mg/kg-d)−1. CSF for inorganic As is 1.5 (mg/kg-d)−1 [64]. When CR in the range of 10–6 to 10–4 is acceptable [64].
Uncertainty Analysis
To increase the precision of risk assessment, the Monte Carlo Simulation (MCS) model was applied to identify an uncertain event's possible outcomes [65]. For this objective, the Oracle Crystal Ball software (version 11.1.2.4.600) was applied. Based on this technique, the parameters such as the concentration of PTEs, and ingestion rate were considered as lognormal distribution and body weight as normal distribution. The cut point of health risk was set at 10,000 repetitions was selected 95% of TTHQ.
Statistical Analysis
The results were indicated as Mean ± standard deviation. Concentration of PTEs were compared using one-way ANOVA. A p-value less than 0.05 was reported as significant difference in data. SPSS 11.5.1 software was utilized for statistical analysis.
Results and Discussion
Method Validation
The method validation parameters, including linearity, calibration curve, equation, R2, recovery, RSDs, LOD, and LOQ, are shown in Table 2. LOD and LOQ ranged 0.006 to 0.097μg/L and 0.025 to 0.321 μg/L in the rapid coffee products respectively. As given in Table 2, the recoveries were in the range of 91.13%–123.67%, and RSDs obtained for all selected PTEs in rapid coffee products were in the range of 1.11–3.31%.
Concentration of PTEs
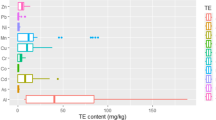
The rank order of PTEs in rapid coffee products was as follows: Fe (404.41 mg/kg) > Pb (0.0046 mg/kg) > As (< LOD) ~ Ni (< LOD) ~ Cd (< LOD) ~ Cu (< LOD) (Table 3). Heavy metals are chemical elements with relatively high density and low reactivity, and most of them are toxic in low concentrations. However, some metals are necessary in low concentrations for the body's metabolism and immune system [66].
Entry of high concentration of this group of metals into the body also causes poisoning. Fe and Cu are essential elements for the body [67]. Other heavy metals including Pb, Cd, Ni and As are dangerous toxins that are not metabolized in the body and gradually accumulate in human fat tissues, muscles, bones and joints. Therefore, long-term exposure to PTEs leads to damage to vital organs [68]. In general, neurological disorders (Parkinson's, Alzheimer's, schizophrenia), various types of cancer, nutrient deficiency, hormone imbalance, obesity, abortion, kidney and brain damage are some of the dangers of PTEs in the body [69].The International Agency for Research on Cancer (IARC) classified metals such as As, Cd, Cr and Ni as carcinogens due to their toxicity at low concentrations [70]. Accordingly, WHO has set permissible limits (PLs) for the concentration of PTEs in foodstuffs and beverages. Therefore, PTEs poisoning occurs when more than permissible amounts enter the body through food or drinking [70, 71].Since the entry of PTEs in high concentrations into the food chain causes damage to human health, it is necessary to determine their concentration in foodstuffs and drinking such as coffee.
Coffee is one of the most consumed and popular drinks in the world due to its energizing and therapeutic effects as well as its good taste. High consumption of this drink can be a source of PTEs entering the human body[69]. Typically, the concentration and types of PTEs in instant coffee powders brands. Usually, the concentration and types of PTEs in instant coffee powders vary among different brands. In general, coffee powder production and distribution industries prepare coffee beans from regions with diverse geographical, chemical and organoleptic conditions that may affect their chemical composition. Furthermore, factors such as regional soil characteristics, pesticide and fertilizer usage, coffee processing methods, and the inclusion of additives can influence the distribution of PTEs in instant coffee powders [72]. The elevated levels of Fe and Pb observed in the rapid coffee powder in this study could be attributed to soil pollution in the agricultural area, groundwater contamination, the utilization of micronutrients in chemical fertilizers and pesticides. Additionally, advancements in coffee bean grinding, roasting, and processing technologies, along with the inclusion of flavorings, may contribute to increased metal content in coffee [73, 74]. Toni et al., study have identified lead, nickel and cadmium in packaged coffee samples [75]. The most detected metal was related to nickel.They also attributed the high lead content in the coffee powder to the packaging coating. The results related to measuring the concentration of PTEs in Gogoasa et al. study showed that iron and cadmium had the highest and lowest concentrations in coffee, respectively [76]. Alkherraz et al., study determined the content of PTEs in coffee, tea and cocoa. The results showed that concentrations of Fe, Cu and Pb were above the limits whereas Cd concentration was below the permissible limits [77].
Human Health Risk Assessment
The rank order of PTEs based on percentile 95% of THQ in both adults and children was Fe > Pb > As > Cd > Ni > Cu (Figs. 1 and 2). Since the concentration of iron was higher than other PTEs in instant coffee, the value of THQ related to Fe was also higher. Similarly, low values of THQ (≤ 1) were obtained by Kowalska et al., in a study conducted to assess the risk of Cd, Pb, As, and Hg in coffee [78]. Due to the low value of THQ, there was no possibility of non-carcinogenic risk associated with coffee consumption. Studies carried out by Winiarska-Mieczan et al. showed that THQ and HI for PTEs consumed with coffee were lower than 1 [79]. According to the results of this research, drinking even 3 cups of coffee a day does not lead to disease.
TTHQ in adults and children due to PTEs in the rapid coffee products was 0.023 and 0.030, respectively (Fig. 3). Consumers are in the acceptable non-carcinogenic risk (TTHQ < 1).
The Total target hazard quotient (TTHQ)of PTEs in rapid coffee was higher in children than in adults. Therefore, considering that TTHQ was less than 0.1 in both age groups, there is no carcinogenic risk for consumers [80]. In the study of Winiarska et al., THQ value for coffee containing Pb and Cd was less than 1, which means that the risk of disease related to Pb and Cd due to long-term coffee consumption was very low[51]. In a study conducted by Khunlert et al., TTHQ value for cadmium, lead, iron, copper and nickel was found to be about 0.1–1[81]. The reason for high TTHQ can be mentioned to the daily consumption of more coffee. In a study conducted by Kowalska et al., THQ values related to coffee consumption for Cd, Pb, As and Hg were found to be less than 0.1, which was in conflict with the non-carcinogenic risk results of rapid coffee in the present study [78].
Percentile 95% of CR in the adults and children due to inorganic As in the rapid coffee products was 2.63E-5 and 1.27E-5, respectively (Fig. 4). Percentile 95% of CR for both adults and children was in the acceptable range (CR < 1E-6). In the present study, As concentration was lower than the detection limit, however, the carcinogenic risk caused by As in rapid coffee was higher than the carcinogenic limit, which could be due to per capita consumption of coffee and high toxicity of As. In the study of Kowalska et al., the carcinogenic risk for As in coffee samples could not be calculated considering that the concentration of As was lower than the detection limit[78]. In the study conducted by Taghizadeh et al., value of CR for As was obtained about 3.84E-6, which indicated an average level and no risk of cancer[82].
Conclusion
The concentration of Fe and Pb was higher than other PTEs in rapid coffee, Hence, source reduction plans for decrease concentration of Pb especially should be conducted. TTHQ values with PTEs in instant coffee were less than 0.1 (0.023 and 0.030 for adults and children, respectively). Therefore, consumers are not in considerable non-carcinogenic risk also carcinogenic risk caused by As was acceptable, hence, consuming instant coffee does not endanger the health of consumers.
Data Availability
No datasets were generated or analysed during the current study.
References
Gao L et al (2022) Concentrations and health risk assessment of 24 residual heavy metals in Chinese mitten crab (Eriocheir sinensis). Qual Assur Saf Crops Foods 14(1):82–91
Cai S, Zeng B, Li C (2023) Potential health risk assessment of metals in the muscle of seven wild fish species from the Wujiangdu reservoir, China. Qual Assur Saf Crops Foods 15(1):73–83
Luo C et al (2022) Comparison of the health risks associated with exposure to toxic metals and metalloids following consumption of freshwater catches in China. Qual Assur Saf Crops Foods 14(4):1–12
Rai PK et al (2019) Heavy metals in food crops: Health risks, fate, mechanisms, and management. Environ Int 125:365–385
Nie G et al (2023) Accumulation characteristics and evaluation of heavy metals in soils and vegetables of plastic-covered sheds in typical red soil areas of China. Qual Assur Saf Crops Foods 15(3):22–35
Talema A (2023) Causes, negative effects, and preventive methods of water pollution in Ethiopia. Qual Assur Saf Crops Foods 15(2):129–139
Li X et al (2023) Role of arbuscular mycorrhizal fungi in cadmium tolerance in rice (Oryza sativa L): a meta-analysis. Qual Assur Saf Crops Foods 15(2):59–70
Bahardoust M et al (2023) Effect of ABO blood group on postoperative overall survival and recurrence-free survival rate in patients with hepatocellular carcinoma after hepatectomy: a multi-center retrospective cohort study. BMC Surg 23(1):324
Onyeaka H et al (2022) A review of the top 100 most cited papers on food safety. Qual Assur Saf Crops Foods 3(1):1–15
Nematollahi A et al (2021) The concentration of potentially toxic elements (PTEs) in sausages: a systematic review and meta-analysis study. Environ Sci Pollut Res 28:55186–55201
Fakhri Y et al (2021) The concentration of potentially hazardous elements (PHEs) in the muscle of blue crabs (Callinectes sapidus) and associated health risk. Chemosphere 279:130431
Lee J-G et al (2019) Effects of food processing methods on migration of heavy metals to food. Appl Biol Chem 62(1):64
Pasalari N et al (2023) Potentially toxic elements (PTES) concentration in anchovy fish sauce from Hormozgan province, Iran: a probabilistic health risk study. Int J Environ Anal Chem 5(3):1–16
Mahmudiono T et al (2024) Potentially toxic elements (PTEs) in coastal sediments of Bandar Abbas city, North of Persian Gulf: an ecological risk assessment. Int J Environ Health Res 34(3):1255–1269
Mohammadpour A et al (2023) Novel modelling approach to assess elements contamination in drinking water from Southern Iran. Expos Health 3(1):1–18
Mohammadpour A et al (2023) Seasonal variations of potentially toxic elements (PTEs) in drinking water and health risk assessment via Monte Carlo simulation and Sobol sensitivity analysis in southern Iran’s largest city. Appl Water Sci 13(12):237
Liao S et al (2023) Establishment of dairy product risk rank model based on the perspective of time, space, and potential contaminants. Qual Assur Saf Crops Foods 15(4):143–155
Bai B et al (2020) Temperature-driven migration of heavy metal Pb2+ along with moisture movement in unsaturated soils. Int J Heat Mass Transf 153:119573
Bai B et al (2022) The remediation efficiency of heavy metal pollutants in water by industrial red mud particle waste. Environ Technol Innov 28:102944
Xiao-Mei H et al (2023) Impact of watershed habitat quality based on land use: a case study of taking Ciyao River Basin. Qual Assur Saf Crops Foods 15(1):18–31
Xiao-Mei H et al (2023) Analysis of hydrochemical characteristics and genesis of water-deficient rivers in China: a case study of the Ciyao River Basin in Shanxi Province. Qual Assur Saf Crops Foods 15(1):32–43
Kumar S et al (2019) Hazardous heavy metals contamination of vegetables and food chain: Role of sustainable remediation approaches - A review. Environ Res 179:108792
Bai B et al (2024) Corrosion effect of acid/alkali on cementitious red mud-fly ash materials containing heavy metal residues. Environ Technol Innov 33:103485
Gavahian M (2023) Valorized pineapple waste by conventional and energy-saving ohmic extraction: potentially toxic elements and mycotoxin contamination. Qual Assur Saf Crops Foods 15(4):11–20
Gao P et al (2023) Effects of vitamins A, C, and D and zinc on urinary tract infections: a systematic review and meta-analysis. Qual Assur Saf Crops Foods 15(3):88–95
Chen X et al (2023) Near-infrared spectroscopy of Chinese soy sauce for quality evaluation. Qual Assur Saf Crops Foods 15(1):139–151
Wang L et al (2024) Quality assessment of black ginseng materials utilizing chemometrics and modeling inflammation in Zebrafish. Qual Assur Saf Crops Foods 16(1):23–37
Xu J et al (2023) Flexible sensing enabled packaging performance optimization system (FS-PPOS) for lamb loss reduction control in E-commerce supply chain. Food Control 145:109394
Zhang H et al (2023) Fuzzy-PID-based atmosphere packaging gas distribution system for fresh food. Appl Sci 13(4):2674
Xu J et al (2022) Intelligent dynamic quality prediction of chilled chicken with integrated IoT flexible sensing and knowledge rules extraction. Foods 11(6):836
Zamanpour S et al (2023) A systematic review to introduce the most effective postbiotics derived from probiotics for aflatoxin detoxification in vitro. Ital J Food Saf 35(4):31–49
Basso ABG et al (2023) Individual and combined decontamination effect of fermentation and ultrasound on aflatoxin B1 in wheat-based doughs: A preliminary study. Qual Assur Saf Crops Foods 15(3):96–103
Tenge BN et al (2022) Verification of the Accuscan gold reader and RIDA smart phone application rapid test kits in detection and quantification of aflatoxin levels in maize from selected regions in Kenya. Qual Assur Saf Crops Foods 14(4):125–135
Thakaew R, Chaiklangmuang S (2023) Aflatoxin B1 elimination in low-grade maize by co-influence of heat and chemical treatment. Qual Assur Saf Crops Foods 15(3):55–67
Elhelaly AE et al (2022) Residual contents of the toxic metals (lead and cadmium), and the trace elements (copper and zinc) in the bovine meat and dairy products: residues, dietary intakes, and their health risk assessment. Toxin Rev 41(3):968–975
Song W et al (2023) Tectorigenin suppresses the viability of gastric cancer cells in vivo and in vitro. Qual Assur Saf Crops Foods 15(3):117–125
Jiang M, Zheng S (2022) Geniposide inhibits non-small cell lung cancer cell migration and angiogenesis by regulating PPARγ/VEGF-A pathway. Qual Assur Saf Crops Foods 14(1):46–54
Geyik ÖG et al (2023) Effects of phenolic compounds of colored wheats on colorectal cancer cell lines. Qual Assur Saf Crops Foods 15(4):21–31
Wang M et al (2022) Brusatol inhibits the growth of prostate cancer cells and reduces HIF-1α/VEGF expression and glycolysis under hypoxia. Qual Assur Saf Crops Foods 14(4):13–22
Bahardoust M et al (2023) Effect of metformin use on survival and recurrence rate of gastric cancer after gastrectomy in diabetic patients: a systematic review and meta-analysis of observational studies. J Gastrointest Cancer 4(1):1–12
Bahardoust M et al (2024) Vitamin B12 deficiency after total gastrectomy for gastric cancer, prevalence, and symptoms: a systematic review and meta-analysis. Eur J Cancer Prev 33(3):208–216
Abbas MM et al (2023) Prevalence of anterior nares colonization of Palestinian diabetic patients with Staphylococcus aureus or methicillin-resistant Staphylococcus aureus. Qual Assur Saf Crops Foods 15(4):32–41
Zhu BJ et al (2023) Combining network pharmacology and bioinformatics to identify bioactive compounds and potential mechanisms of action of Sedum aizoon L in the treatment of atherosclerosis. Qual Assur Saf Crops Foods 15(3):104–116
Nartea A et al (2022) Is coffee powder extract a possible functional ingredient useful in food and nutraceutical industries? Ital J Food Saf 34(1):140–148
Ghahraman MA, Farahani S, Tavanai E (2022) A comprehensive review of the effects of caffeine on the auditory and vestibular systems. Nutr Neurosci 25(10):2181–2194
de Melo Pereira GV et al (2020) Chapter Three - Chemical composition and health properties of coffee and coffee by-products. In: Toldrá F (ed.) Advances in food and nutrition research, vol 2. Academic Press, pp 65–96
Winiarska-Mieczan A et al (2021) Assessment of the risk of exposure to cadmium and lead as a result of the consumption of coffee infusions. Biol Trace Elem Res 199(6):2420–2428
Noël L et al (2012) Li, Cr, Mn Co, Ni, Cu, Zn, Se and Mo levels in foodstuffs from the Second French TDS. Food Chem 132(3):1502–1513
Czarniecka-Skubina E et al (2021) Consumer choices and habits related to coffee consumption by poles. Int J Environ Res Public Health 18(8):3948
Chudy S (2014) Development of coffee market and changes in coffee consumption among Poles. J Agribus Rural Dev 34(4):25–31
Winiarska-Mieczan A et al (2023) Cadmium and lead concentration in drinking instant coffee, instant coffee drinks and coffee substitutes: safety and health risk assessment. Biol Trace Elem Res 201(1):425–434
Webb A et al (2020) Sources of elevated heavy metal concentrations in sediments and benthic marine invertebrates of the western Antarctic Peninsula. Sci Total Environ 698:134268
Heshmati A et al (2020) The concentration and health risk of potentially toxic elements in black and green tea—both bagged and loose-leaf. Qual Assur Saf Crops Foods 12(3):140–150
Kazemi A et al (2022) Alterations and health risk assessment of the environmental concentration of heavy metals in the edible tissue of marine fish (Thunnus tonggol) consumed by different cooking methods. Reg Stud Mar Sci 53:102361
Armbruster DA, Pry T (2008) Limit of blank, limit of detection and limit of quantitation. Clin Biochem Rev 29(Suppl 1):S49
EPA U (2000) Risk-based concentration table, vol 2. United States Environmental Protection Agency, Philadelphia, Washington DC, pp 12–16
Mohammadpour A et al (2023) The concentration of potentially toxic elements (PTEs) in drinking water from Shiraz, Iran: a health risk assessment of samples. Environ Sci Pollut Res 30(9):23295–23311
Xiong J et al (2022) Occurrence of aflatoxin M1 in three types of milk from Xinjiang, China, and the risk of exposure for milk consumers in different age-sex groups. Foods 11(23):3922
Özlü H (2024) Occurrence, dietary exposure and risk assessment to aflatoxins in red pepper flakes from Southeast of Türkiye. Qual Assur Saf Crops Foods 16(1):69–77
EPA (2011) Exposure factors handbook: vol 2, 2011 edn. EPA/600/R-09, pp 3–6
Mohammadpour A et al (2022) Assessment of potentially toxic elements (PTEs) in fruits from Iranian market (Shiraz): A health risk assessment study. J Food Compos Anal 114:104826
Xiong J et al (2022) Occurrence of aflatoxin M1 in yogurt and milk in central-eastern China and the risk of exposure in milk consumers. Food Control 137:108928
Einolghozati M et al (2023) The level of heavy metal in fresh and processed fruits: a study meta-analysis, systematic review, and health risk assessment. Biol Trace Elem Res 201(5):2582–2596
EPA (2016) USEPA, 2016. Integrated risk information system, vol 5, pp 6–12. https://www.epa.gov/iris/
Harrison RL (2010) Introduction to monte carlo simulation. In: AIP conference proceedings. Am Inst Phys 11:25–29
Zheng K et al (2023) Epidemiological evidence for the effect of environmental heavy metal exposure on the immune system in children. Sci Total Environ 868:13–16
Jan AT et al (2015) Heavy metals and human health: mechanistic insight into toxicity and counter defense system of antioxidants. Int J Mol Sci 16(12):29592–29630
Hu L et al (2022) Effects of inorganic and organic selenium intervention on resistance of radish to arsenic stress. Ital J Food Saf 34(1):44–58
Butt MS, Sultan MT (2011) Coffee and its consumption: benefits and risks. Crit Rev Food Sci Nutr 51(4):363–373
Albals D et al (2021) Multi-element determination of essential and toxic metals in green and roasted coffee beans: a comparative study among different origins using ICP-MS. Sci Prog 104(2):12–17
Jaishankar M et al (2014) Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol 7(2):60
Ashu R, Chandravanshi BS (2011) Concentration levels of metals in commercially available Ethiopian roasted coffee powders and their infusions. Bull Chem Soc Ethiop 25(1):12–19
Ludwig IA et al (2014) Coffee: biochemistry and potential impact on health. Food Funct 5(8):1695–1717
Gökcen BB, Şanlier N (2019) Coffee consumption and disease correlations. Crit Rev Food Sci Nutr 59(2):336–348
De Toni L et al (2017) Phthalates and heavy metals as endocrine disruptors in food: a study on pre-packed coffee products. Toxicol Rep 4:234–239
Gogoasa I et al (2013) The mineral content of different coffee brands. J Hortic For Biotechnol 17(4):68–71
Alkherraz AM et al (2021) A Comparative Study of Some Nuts And Seeds Constituents Available in Local Market at Misurata City-Libya. Biquarterly Iran J Anal Chem 8(1):1–9
Kowalska G (2021) The safety assessment of toxic metals in commonly used herbs, spices, tea, and coffee in Poland. Int J Environ Res Public Health 18(11):5779
Winiarska-Mieczan A et al (2021) Assessment of the risk of exposure to cadmium and lead as a result of the consumption of coffee infusions. Biol Trace Elem Res 199:2420–2428
Persad AS, Cooper GS (2008) Use of epidemiologic data in Integrated Risk Information System (IRIS) assessments. Toxicol Appl Pharmacol 233(1):137–145
Khunlert P et al (2021) Pyrethroid and metal residues in different coffee bean preparing processes and their human health risk assessments via consumption. bioRxiv 13:24–30
Taghizadeh SF et al (2023) Assessment of Carcinogenic and Non-carcinogenic Risk of Exposure to Metals via Consumption of Coffee, Tea, and Herbal Tea in Iranians. Biol Trace Elem Res 201(3):1520–1537
Funding
This project was supported by Hormozgan University of Medical Sciences, Bandar Abbas, Iran (IR.HUMS.REC.1399.448).
Author information
Authors and Affiliations
Contributions
Study design was conducted by Vali Alipour, Yadolah Fakhri and Analysis of data by Iman Mahmoudi, Mohammad Borzoei, Freshtehm Mehri, Maryam Sarkhosh, and prepares manuscript by Vali Alipour, Freshtehm Mehri, Maryam Sarkhosh, Intissar limam, Rasul Nasiri, Yadolah Fakhri.
Corresponding authors
Ethics declarations
Ethical Approval
This project was approved by the ethics committee of Hormozgan University of Medical Sciences, Bandar Abbas, Iran (IR.HUMS.REC.1399.448).
Consent to Participate
The authors declare their Consent to Participate in this article.
Consent to Publish
The authors declare their Consent to Publish this article.
Competing Interests
The authors declare no competing interests.
Disclosure Statement
None to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alipour, V., Mahmoudi, I., Borzoei, M. et al. Concentration of Potentially Toxic Elements (PTEs) in Rapid Coffee Products in Bandar Abbas, Iran: Probabilistic Non-Carcinogenic and Carcinogenic Risk Assessment. Biol Trace Elem Res (2024). https://doi.org/10.1007/s12011-024-04228-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12011-024-04228-y