Abstract
The objective of this research was to examine and contrast the levels of cadmium (Cd), copper (Cu), zinc (Zn), and selenium (Se) in the muscle and hepatopancreas tissues of two species, namely pharaoh cuttlefish (Sepia pharaonis) and Indian squid (Uroteuthis duvauceli), from the Persian Gulf. A total of thirty individuals of each species were gathered in January 2009 from the northern waters of the Persian Gulf. The metal concentrations were significantly higher in muscle tissue (p < 0.05) than in other tissues. S. pharaonis had higher metal concentrations than U. duvauceli. In the muscle and hepatopancreas samples of S. pharaonis, the highest mean concentrations were found to be for Zn (58.45 ± 0.96 µg/g dw) and Cu (1541.47 ± 192.15 µg/g dw), respectively. In U. duvauceli, the highest concentration of measured elements was seen for Zn in both muscle (36.52 ± 0.56 µg/g dw) and hepatopancreas (60.94 ± 2.65 µg/g dw). Se had the lowest concentration among the elements measured in both species. There was a negative and significant correlation between Cu and biometrical factors (total body length and weight) in both muscle and hepatopancreas samples of S. pharaonic and only in the muscle samples of U. duvauceli (p < 0.01, R2 = − 052; p < 0.01, R2 = − 0.055). However, there was a strong correlation between Zn and biometrical factors in hepatopancreas samples of both species. The comparison of metal concentrations with standards revealed that only Cd levels in S. pharaonis exceeded the ESFA and WHO standards, whereas other metals were below the standards.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Covering nearly 70% of the Earth’s surface, marine ecosystems are vital to human survival as they provide a significant food source. Regrettably, the escalation of various human activities has resulted in the significant degradation of the marine ecosystem, primarily caused by the discharge of wastewater and inadequate waste management. Heavy metal pollution is one of the most significant environmental pollutants, particularly in water ecosystems [1]. The study of heavy metals in marine ecosystems has gained significant attention in recent years. Heavy metals are of particular interest due to their toxicity and potential to bioaccumulate in various aquatic species [2, 3]). Over time, the toxic metals accumulate irreversibly in the organs of marine organisms, resulting in the onset of various diseases that threaten the survival of aquatic life and pose significant health risks to humans [1, 4, 5].
Heavy metals typically enter water environments through various means, including atmospheric deposition, erosion, and human activities such as the discharge of industrial wastewater, municipal sewage, and leachate from mining activities [5, 6].
Today, in different parts of the world, different tissues of fish are used for environmental monitoring and also to assess the health risk of people who consume these fish [7,8,9,10,11]. Cephalopods are a significant part of marine ecosystems, serving as both prey and predators. They are a class of marine invertebrates found in a vast array of marine habitats, from coastal waters to deep oceans [12]. Cephalopod species with varying habitats and lifespans have the potential to accumulate both organic and inorganic pollutants, including metals [13,14,15,16,17]. Consequently, they serve as significant contributors to the transfer of contamination to higher levels of the food chain, such as birds and marine mammals [18]. In addition to consuming organisms that have a high ability to bioaccumulate essential and non-essential metals, cephalopods are also an important source of food for other large fish and various types of whales, so they can be an important carrier of these heavy metals to the upper levels of the food chain [14, 18, 19]. So far, various reports have been published regarding the high concentration of metals in different cephalopod species [14, 19,20,21,22]. It is noteworthy that most of the sites have focused on the ability of metals such as cadmium, copper, and zinc to accumulate in the digestive glands (hepatopancreas) of these organisms even in areas with low pollution [14, 19, 20, 23, 24].
The Persian Gulf holds special significance as one of Iran’s two main water areas. It has served as a crucial source for extracting abundant food resources but has unfortunately been increasingly impacted by various pollutants in recent years. The northern region of the Persian Gulf is characterized by shallow depths, limited water circulation, high temperature, and salinity, making it particularly vulnerable to the adverse effects of pollutants on the marine environment [25]. Additionally, the Persian Gulf has been significantly impacted by the entry of pollutants through its river systems, past wars, oil extraction, and the transportation of fuel materials by ships, among other factors [26]. Currently, there is a lack of information on heavy metals in these species in the Persian Gulf, despite their potential as useful bio indicators for biological monitoring in the region. The goals of this research are (1) to investigate the relationship between heavy metals and each other and (2) to compare different organs in terms of the accumulation of cadmium, copper, selenium, and zinc in them.
Materials and Methods
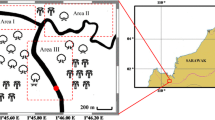
The Persian Gulf is a shallow and semi-enclosed sea with an average depth of less than 35 m, which is linked to the Oman Sea and the Indian Ocean through the Strait of Hormuz. Its average temperature ranges from 28 to 30 ℃ and can reach up to 35 ℃. The dissolved oxygen level in the water varies from 4 to 7 mg/L [26]. The study area in this research is situated on the coast of Hormozgan province, between Qeshm and Kish islands, and is located within the geographical coordinates of 27°N ′07 and 22°E ′12 to 27°N 19° and 52°E ′40 in the Persian Gulf region.
Collection of Samples and Biometry
In December 2017, two species of cephalopods, namely the pharaoh cuttlefish (Sepia pharaonis) and Indian squid (Uroteuthis duvauceli), were captured using the bottom trawling method from the waters in the northern part of the Persian Gulf between Qeshm Island and Kish (Fig. 1). The collection was carried out over a week by the Ferdous 1 research vessel, and 30 samples of each species were collected. The samples were immediately placed in coded bags and frozen at − 20℃ before being transported to the laboratory. In the laboratory, the samples were washed with distilled water and biometrically measured, and their mantle length (ML) and total length (TL) were calculated using a biometric board. The weight of each sample was calculated using a digital scale.
Extraction of Heavy Metals
To prepare the required tissue samples, first, the samples are dissected, and after determining the gender, the studied tissues, including muscle and hepatopancreas, were separated, and a certain amount of each tissue was weighed. Weighted samples were freeze-dried and homogenized and passed through 230-mesh sieve. The prepared powder was stored in closed polyethylene containers until further processing.
For digestion, 1 g of the sample was taken in a polyethylene beaker with 4 mL of 60% nitric acid (Merck, Darmstadt, Germany) and 1 mL of 70% perchloric acid (Merck, Darmstadt, Germany). The samples were then placed in a bain-marie bath at 100 ℃ for 2 h to perform digestion [27]. After the completion of the digestion process, the solution was filtered using Whatman 42 filter paper (Merck, Darmstadt, Germany), and the volume was made up to 10 mL using deionized water. Besides, in addition to the digested samples, in each series of eight, a procedural blank was digested.
Instrumental Analysis
Cu and Zn were quantified using Philips PU 9400 analytical atomic absorption spectrometer (Philips, Cambridge, Cambs), with a slotted tube atom trap (STAT), and aliquoting device, to optimize the sensitivity, and for Cd, Flame Atomic Absorption Spectrophotometer Model SensAA, GBC Scientific Equipment Australian made, provided with Cathode Lamps suitable for this chemical element was used. An air-acetylene mixture was also used to create a flame. After preparing the standards and applying the related settings to the device, the standards from low to a high concentration were used to prepare the calibration curve for each metal (R2 ≤ 0.99). The recovery range varied between 90 and 105. Selenium was measured by graphite furnace atomic absorption spectrometry (model AAnalyst 3030; PerkinElmer, Turku, Finland) using Zeeman background correction. At first, the desired range was determined by conducting a preliminary analysis, and then the required standards were prepared. After drawing the calibration curve using standards, we inject the original samples into the device and read the absorbance. Based on the calculations, the recovery of the device was 96.6%, which is within the acceptable range (85–110). The concentration of elements in the digested samples was calculated from the equation of the calibration curve for each metal.
Quality Control
In order to increase the accuracy of the test and reduce the error rate, the quality control method was performed as follows. All containers used in the sampling, digestion, and storage of samples were washed and dried with dilute nitric acid and distilled water. The instrument was calibrated with a NIST-traceable heavy metal standard solution (AccuTrace Single Element Standard; AccuStandard Inc., New Haven, CT, USA). Three standard reference materials (SRM, National Institute of Standards and Technology—NIST), namely SRM 1633b (ConstituentElements in coal fly ash), SRM 2709 (San Joaquin Soil Baseline Trace Element Concentrations), and SRM 2711 (Montana II soil), were each analyzed in seven replicates [28] and were used for checking the reliability of the analysis. All SRM fell within the recovery range (94.8–104.7%) of the expected values while replicating SRM and hair sample measurements had a relative standard deviation (RSD) < 10%. The calibration curve was drawn every day using a control sample (Blank) and four standard concentrations. Then the correctness of the method was confirmed by using the control solution and a standard sample close to the middle concentration range (approximately once every 10 samples). For every 15 samples, one sample was randomly re-measured. Also, a standard sample was selected and measured with each batch of sample injection to ensure the correctness of the device’s working process.
Statistical Analysis
SPSS 16 software was used for the statistical analysis of data, and Excel 2007 was used for drawing graphs. The Kolmogrov-Smirnov test was used to check the normality of the data. The correlation between the two metals was investigated with Pearson’s normal data correlation test. Due to the normality of the data, paired T-test was used to compare the concentration of elements in the tissues of one species, and the Dunnett T3 test was used for several elements in the tissues of one species. An unpaired t-test was used to compare elements of two types. One-sample T-test was also used to compare the concentration of elements with international standards.
Results and Discussions
General Discussion
Table 1 presents the biometric data, including total weight, mantle length, and total length, for the pharaoh cuttlefish and Indian squid species. The concentrations of Cd, Cu, Zn, and Se in the muscle and hepatopancreas were measured and expressed in micrograms per gram of dry weight, as shown in Tables 2 and 3. The levels of all measured metals in the hepatopancreas were significantly higher than those in the muscle tissue (p < 0.01). Among the heavy metals, Cu and Zn had the highest average concentrations in hepatopancreas and muscle tissue, respectively. Conversely, Se had the lowest average concentrations in both species (Figs. 2 and 3). Negative and significant correlations were observed between Cu levels in muscle tissue and hepatopancreas and body weight and length in both species (p < 0.01 R2 = − 052, p > 0.01 R2 = − 0.055) (Figs. 4, 5, and 6). Zinc concentrations in the hepatopancreas had a significant positive correlation with body length and weight (p < 0.01 R2 = 0.41) (Fig. 7). No significant correlations were observed regarding the other investigated elements. Strong and significant correlations were observed between Cu and Zn in both muscle and hepatopancreas samples, as well as between Cd and Se in muscle only, for Sepia pharaonis (R2 = 0.51, p < 0.01 and R2 = 0.74, p < 0.01, and R2 = 0.72, p < 0.01, respectively) (Figs. 8, 9, and 10). In contrast, in Indian squid, Cd with Cu (R2 = 0.43, p < 0.05) and Cd with Zn (R2 = 0.52, p < 0.01) showed a significant and strong correlation in muscle, while Cu with Cd (R2 = 0.81, p < 0.01) showed a significant and strong correlation in hepatopancreas (Figs. 11, 12, and 13) (Table 4).
Additionally, a strong and significant correlation was observed between the molar concentration of selenium and cadmium in tiger squid muscle (Fig. 14). The concentrations of all investigated elements, except Se, in both muscle and hepatopancreas tissues of the two species showed significant differences (p < 0.01) (Figs. 15 and 16). The results indicated that only the concentration of cadmium metal in Indian squid exceeded the standard limit of [29] WHO and EFSA, while the concentration of other metals was below the standard limit (Table 5).
Levels and Comparison of Heavy Metals
Upon examining the tissues separately, significant differences in the concentration of elements were observed in each tissue of the studied species, except for the concentration of cadmium and zinc in the hepatopancreas of pharaoh cuttlefish and cadmium and copper in the hepatopancreas of Indian squid. All other elements measured in the tissues of the studied species exhibited significant differences. Moreover, a comparison of the two tissues in terms of element accumulation revealed that the concentration of all metals in the hepatopancreas tissue was significantly higher than that in the muscle tissue for both species.
Cadmium
The results of this study indicate that both species had high levels of Cd in their hepatopancreas samples, with pharaoh cuttlefish exhibiting a significantly higher concentration than Indian squid. The accumulation of Cd in the hepatopancreas may be attributed to the high metal-detoxifying ability of cephalopods, particularly in this organ [19]. Furthermore, factors such as the proximity to bed sediments, the carnivorous diet of these species, and potential cannibalism may also contribute to the observed increase in Cd concentration in both species [30]. Iron deficiency can also impact cadmium toxicity as it increases the absorption of cadmium in the body through iron reduction. Previous studies on cephalopods by Bustamante et al. (2008) and Pereira et al. (2009) have reported that the highest concentration of cadmium is found in the hepatopancreas, which is in line with the findings of this study.
Copper and Zinc
The results of this study show that copper and zinc concentrations in the tissues of both species are higher than the other metals measured. This finding is consistent with the results of a study by Craig and Overnell (2003) who investigated the levels of heavy metals in different tissues of a squid species (Loligo forbesi) on the southern coast of Scotland. Among the metals measured by them, the concentrations of zinc and copper in all tissues were significantly higher than other elements. Similarly, Prafulla et al. (2001) measured essential and non-essential metals in two species of squid (Uroteuthiis duvauceli and Doryteuthis sibogae) on the southwest coast of India, and their results showed high levels of copper and zinc in the tissues of these species, consistent with the findings of this study. Zinc is typically found in higher concentrations than other metals in marine organisms’ tissues due to its involvement in various enzymatic reactions in living organisms. Both copper and zinc are involved in essential metabolic processes and serve as catalysts for enzymes that produce heat and are vital for energy production and the transport of iron in the body [31]. Cephalopods require copper for the production of respiratory pigments, hemocyanin, and other essential functions in their bodies. Hemocyanin, a protein containing soluble copper, is used by cephalopods as a respiratory pigment to transport oxygen [32]. Consequently, the high demand for copper in cephalopods results in a high concentration of this element in these species, making copper the second most abundant essential metal in cephalopods after iron [33].
Selenium
In all the samples, significantly low levels of Se were observed. Se is an element that by forming bonds with some toxic metals such as Hg and Cd can reduce their toxicity. So far, little research has been done on the detoxification role of selenium in cephalopods.
Selenium Detoxification Effects Against Cadmium
The molar ratio of Se/Cd is a good indicator to predict the availability of these two elements and the ability to detoxify Cd. Molar ratios > 1 indicate an excess of Se over cadmium and the activation of the selenide detoxification mechanism [34] and the formation of a bond between selenium and cadmium metallothioneins (Cd-Se-selenoprotein) [27]. The molar ratio < 1 indicates a deficiency of Se in Cd detoxification and the occurrence of synergistic effects between the two elements, which in addition to causing cadmium poisoning due to the involvement of selenium in the body with cadmium causes a hidden lack of selenium in the species and consequently in consumers [22].
The molar ratio of Se/Cd was less than 1 in both muscle tissue and hepatopancreas of both species, with values ranging from 0.007 (Indian squid hepatopancreas) to 0.544 (Indian squid muscle) (Table 5). Moreover, a significant and strong correlation was observed only between the molar concentrations of Se and Cd in the tissues of the two species, indicating the potential formation of a Cd-Se compound as part of the selenium detoxification process against cadmium (Fig. 14).
Correlation of Heavy Metals with Biometric Parameters
A significant negative correlation was observed between Cu levels and biometric parameters (total body length and weight) in both the muscle (R2 = 0.52; p < 0.01) and hepatopancreas (R2 = 0.55; p < 0.01) samples of S. pharaonic and only in the muscle samples of U. duvauceli (R2 = 0.57; p < 0.01) (Figs. 4 and 5). However, a strong correlation was found between Zn and biometric factors in the hepatopancreas samples of both species (R2 = 0.41; p < 0.01). This relationship is consistent with a study by Kim et al. (2008) which investigated the accumulation of heavy metals (cadmium, zinc, and copper) in squid (Todarodes pacificus) samples collected from Korean coastal waters. The results showed no significant relationship between the size and gender of squids and the concentration of metals in different organs, which may be due to the species’ ability to regulate the concentration of these elements in the body. Additionally, a similar relationship was observed for Cu levels with biometric parameters in Sepia pharaonic in two different lagoons in Portugal, one of which had a history of anthropogenic and industrial pollution and the other receiving urban effluents [19].
Correlation of Heavy Metals in Textures
In both muscle tissue and hepatopancreas of Sepia pharaonic, a significant positive correlation was observed between Cu and Zn (R2 = 0.51; p < 0.01 and R2 = 0.72; p < 0.01, respectively) and between Cd and Se (R2 = 0.72; p < 0.01) in muscle samples (Fig. 6). This strong correlation between Cu and Zn may indicate their affinity towards metallothioneins, as well as Cu’s propensity to bind to other proteins that regulate Zn concentration in the tissues [35]. In Indian squid, a strong and significant positive correlation was observed between Cd and Cu in both muscle (R2 = 0.43; p < 0.05) and hepatopancreas (R2 = 0.52; p < 0.05) samples. Positive correlations between Cd and essential elements like Cu and Zn have been observed in most cephalopod studies. The positive correlation of Cd with Cu and Zn may be due to the presence of metallothioneins, as Cd can compete with Zn and Cu in binding with these proteins and transfer with them, like other toxic elements [19, 24, 36].
Previous studies conducted by Seixas et al. in 2005 and Preira et al. in 2009 have shown a significant positive relationship between the concentrations of cadmium, zinc, and copper in the species examined. This is consistent with the findings of the current study. Furthermore, the correlation between the concentration of selenium and cadmium in muscle tissue suggests the formation of the Cd-Se compound, which is part of the protective mechanism of selenium against cadmium. Although enzymes that require cadmium for their function have not been identified, the amount of cadmium accumulation in the body is generally dependent on the levels of zinc and copper. Metal-related proteins such as metallothioneins have been found to play a small role in binding to cadmium and other heavy metals in most marine mollusks [32].
Comparison of the Concentration of Elements in Two Species
In general, the concentration of heavy metals in the hepatopancreas and muscle of pharaoh cuttlefish was much higher than that of Indian squid. Except for Se, all the other elements had a significant difference in the muscle tissue of the two species (p < 0.001) (Fig. 15). Furthermore, levels of all the elements except Se, between the species, were significantly different (p < 0.01) (Fig. 16), from each other in both muscle and hepatopancreas tissue samples. The difference in the concentration of elements in different species may be due to many factors such as diet, habitat, metabolic activity, and behavior of the species, or due to the difference in size and age of the species [37]. However, given the fact that the two studied species have a similarity in food habits (pharaoh cuttlefish: benthic; Indian squid: benthopelagic), higher exposure of benthic organisms to heavy metals might be the reason for elevated levels of heavy metals in pharaoh cuttlefish [38, 39]. Among 7 species of fish from the coasts of Uruguay, benthic organisms displayed the highest levels of heavy metals due to their close association with bottom sediment [40]. In addition, the greater average length and weight of pharaoh cuttlefish compared to Indian squid can be another reason for this difference in concentration.
Comparing the Concentration of Metals in the Studied Species with the Standard
Comparing the concentration of metals with international standards showed that only the concentration of Cd in Indian squid was significantly higher (p < 0.01) than the standard limit of WHO and EFSA, and the concentration of other metals was lower than the standard limit. The pollution of the Persian Gulf region and the high ability of cephalopod species to accumulate cadmium in their hepatopancreas can be one of the reasons for the high concentration of cadmium in this species. In most of the studies conducted on various cephalopod species, especially squid, very high concentrations of cadmium have been reported in these species [14, 19, 32].
Conclusion
This study analyzed the concentrations of Cd, Cu, Zn, and Se in the muscle and hepatopancreas tissues of two dominant cephalopod species in the Persian Gulf. The results revealed that the concentrations of these elements in the hepatopancreas tissue were significantly higher than in the muscle tissue of both species. Surprisingly, the increased concentration of essential metals did not result in a decrease in cadmium absorption. Additionally, there was a significant difference in the metal concentrations between the two species, with the benthic pharaoh cuttlefish species having higher levels of measured metals than the pelagic Indian squid species. The high concentrations of metals, particularly cadmium, in these species, indicate contamination of the Persian Gulf with heavy metals, posing a threat to aquatic animals and consumers. Cephalopods are ecologically important species in marine ecosystems and play a crucial role in the food chain as both predators and prey. Thus, their ability to accumulate metals can lead to the transfer of pollution to higher levels of the food chain.
References
Zehra I, Kauser T, Zahir E, Naqvi II (2003) Determination of Cu, Cd, Pb and Zn concentration in edible marine fish Acanthopagurus berda (DANDYA) along Baluchistan coast-Pakistan. Int J Agric Biol 5:80–82
Blevins RD, Pancorbo OC (1986) Metal concentrations in muscle of fish from aquatic systems in East Tennessee, USA. Water Air Soil Pollut 29:361–371
Duruibe JO, Ogwuegbu MOC, Egwurugwu JN et al (2007) Heavy metal pollution and human biotoxic effects. Int J Phys Sci 2:112–118
Mansour SA, Sidky MM (2002) Ecotoxicological studies. 3. Heavy metals contaminating water and fish from Fayoum Governorate, Egypt. Food Chem 78:15–22
Prasath P, Khan TH (2008) Impact of tsunami on the heavy metal accumulation in water, sediments and fish at Poompuhar coast, Southeast Coast of India. E-J Chem 5:16–22
Alam MGM, Tanaka A, Allinson G et al (2002) A comparison of trace element concentrations in cultured and wild carp (Cyprinus carpio) of Lake Kasumigaura, Japan. Ecotoxicol Environ Saf 53:348–354
Türkmen M, Dura N (2016) Assessment of heavy metal concentrations in fish from south western black sea. Indian J Geo-Marine Sci 45:1552–1559
Türkmen M, Akaydin A (2017) Metal levels in tissues of commercially important fish species from Southeastern Black Sea Coasts. Indian J Geo Mar Sci 46:2357–2360
Türkmen M, Pınar EO (2018) Bioaccumulation of metals in economically important fish species from antalya bay, northeastern mediterranean sea. Indian J Geo-Marine Sci 47:180–184
Turkmen M (2023) Novel perspectives of geography, Environment and Earth Sciences Vol. 2
Turkmen M (2023) Novel perspectives of geography, Environment and Earth Sciences Vol 5
Boyle P, Rodhouse P (2008) Cephalopods: ecology and fisheries. Wiley
Yamada H, Takayanagi K, Tateishi M et al (1997) Organotin compounds and polychlorinated biphenyls of livers in squid collected from coastal waters and open oceans. Environ Pollut 96:217–226
Bustamante P, Caurant F, Fowler SW, Miramand P (1998) Cephalopods as a vector for the transfer of cadmium to top marine predators in the north-east Atlantic Ocean. Sci Total Environ 220:71–80
Ueno D, Inoue S, Ikeda K et al (2003) Specific accumulation of polychlorinated biphenyls and organochlorine pesticides in Japanese common squid as a bioindicator. Environ Pollut 125:227–235
Storelli MM, Giacominelli-Stuffler R, Storelli A, Marcotrigiano GO (2006) Cadmium and mercury in cephalopod molluscs: estimated weekly intake. Food Addit Contam 23:25–30
Miramand P, Bustamante P, Bentley D, Kouéta N (2006) Variation of heavy metal concentrations (Ag, Cd Co, Cu, Fe, Pb, V, and Zn) during the life cycle of the common cuttlefish Sepia officinalis. Sci Total Environ 361:132–143
Lahaye V, Bustamante P, Spitz J et al (2005) Long-term dietary preferences of common dolphins in the Bay of Biscay using a metallic tracer. Mar Ecol Prog Ser 305:275–285
Pereira P, Raimundo J, Vale C, Kadar E (2009) Metal concentrations in digestive gland and mantle of Sepia officinalis from two coastal lagoons of Portugal. Sci Total Environ 407:1080–1088
Miramand P, Bentley D (1992) Concentration and distribution of heavy metals in tissues of two cephalopods, Eledone cirrhosa and Sepia officinalis, from the French coast of the English Channel. Mar Biol 114:407–414
Raimundo J, Vale C, Duarte R, Moura I (2008) Sub-cellular partitioning of Zn, Cu, Cd and Pb in the digestive gland of native Octopus vulgaris exposed to different metal concentrations (Portugal). Sci Total Environ 390:410–416
Seixas S, Bustamante P, Pierce GJ (2005) Interannual patterns of variation in concentrations of trace elements in arms of Octopus vulgaris. Chemosphere 59:1113–1124
Smith JD, Plues L, Heyraud M, Cherry RD (1984) Concentrations of the elements Ag, Al, Ca, Cd, Cu, Fe, Mg, Mn, Pb and Zn, and the radionuclides 210Pb and 210Po in the digestive gland of the squid Nototodarus gouldi. Mar Environ Res 13:55–68
Bustamante P, Cosson RP, Gallien I et al (2002) Cadmium detoxification processes in the digestive gland of cephalopods in relation to accumulated cadmium concentrations. Mar Environ Res 53:227–241
Pourang N, Nikouyan A, Dennis JH (2005) Trace element concentrations in fish, surficial sediments and water from northern part of the Persian Gulf. Environ Monit Assess 109:293–316
Agah H, Leermakers M, Elskens M et al (2007) Total mercury and methyl mercury concentrations in fish from the Persian Gulf and the Caspian Sea. Water Air Soil Pollut 181:95–105
Endo T, Hisamichi Y, Kimura O et al (2008) Contamination levels of mercury and cadmium in melon-headed whales (Peponocephala electra) from a mass stranding on the Japanese coast. Sci Total Environ 401:73–80
Behrooz RD, Sahebi S, Majnoni F et al (2013) Mercury contamination in commercial fresh and salt water fish of the Zabol Chahnimeh reservoirs and the Gulf of Oman (Iran). Food Addit Contam Part B 6:175–180. https://doi.org/10.1080/19393210.2013.779322
(EFSA) EFSA (2009) Cadmium in food-scientific opinion of the panel on contaminants in the food chain. EFSA 7:980
Bustamante P, González AF, Rocha F et al (2008) Metal and metalloid concentrations in the giant squid Architeuthis dux from Iberian waters. Mar Environ Res 66:278–287
Linder MC, Wooten L, Cerveza P et al (1998) Copper transport. Am J Clin Nutr 67:965S-971S
Craig S, Overnell J (2003) Metals in squid, Loligo forbesi, adults, eggs and hatchlings. No evidence for a role for Cu-or Zn-metallothionein. Comp Biochem Physiol Part C Toxicol Pharmacol 134:311–317
Prafulla V, Francis L, Lakshmanan PT (2001) Concentrations of trace metals in the squids, Loligo duvauceli and Doryteuthis sibogae caught from the Southwest Coast of India popular. Asian Fish Sci 14:399–410
Yang D, Chen Q, Xu S (2007) Synthesis of CdSe/CdS with a simple non-TOP-based route. J Lumin 126:853–858
Viarengo A, Ponzano E, Dondero F, Fabbri R (1997) A simple spectrophotometric method for metallothionein evaluation in marine organisms: an application to Mediterranean and Antarctic molluscs. Mar Environ Res 44:69–84
Manso M, Carvalho ML, Nunes ML (2007) Characterization of essential and toxic elements in cephalopod tissues by EDXRF and AAS. X-Ray Spectrom An Int J 36:413–418
Burger J, Gochfeld M (2005) Heavy metals in commercial fish in New Jersey. Environ Res 99:403–412
Roméo M, Siau Y, Sidoumou Z, Gnassia-Barelli M (1999) Heavy metal distribution in different fish species from the Mauritania coast. Sci Total Environ 232:169–175
Amin B, Ismail A, Arshad A et al (2009) Gastropod assemblages as indicators of sediment metal contamination in mangroves of Dumai, Sumatra, Indonesia. Water Air Soil Pollut 201:9–18
Viana F, Huertas R, Danulat E (2005) Heavy metal levels in fish from coastal waters of Uruguay. Arch Environ Contam Toxicol 48:530–537
Acknowledgements
We are very grateful to the laboratory manager of Tarbiat Modares University, Mrs. Haghdost, for her cooperation.
Funding
This article was supported by the University of Zabol (Iran) with grant code (IR-UOZ-GR-0088).
Author information
Authors and Affiliations
Contributions
RA; validation, AE; software, RDB; conceptualization, methodology, writing original draft preparation, FR; investigation, TV; resources, PC; writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asvad, S.R., Esmaili-Sari, A., Behrooz, R.D. et al. Comparison of Cd, Cu, Se, and Zn Concentration in the Muscle and Hepatopancreas of Sepia pharaonis and Uroteuthis duvauceli in the North of Persian Gulf (Iran). Biol Trace Elem Res 202, 743–753 (2024). https://doi.org/10.1007/s12011-023-03712-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03712-1