Abstract
In this work, we studied the impact of chronic iron exposure, in the form of iron sulfate (FeSo4), on affective and cognitive disorders and oxidative stress in the male Wistar rat. The treatment was carried out for 8 weeks, the rats received an intraperitoneal injection of iron at different doses: 0.25, 0.5, and 1 mg/kg. Affective and cognitive disorders are assessed in open field test (OFT), elevated plus maze (EPM), forced swimming test (FST), Morris water maze (MWM), and Y-maze. The hippocampus and prefrontal cortex of each animal were taken for biochemical examination. Our results show that iron exerts anxiogenic and depressogenic effects, which were observed first at the dose of 0.5 mg/kg and continued in a dose-dependent manner up to the maximum tested dose of 1 mg/kg. According to results from the MWM and Y-maze tests, continuous exposure to iron induces cognitive disorders that are defined by the disturbance of working memory and influences spatial learning performance causing a deficit of spatial memory retention. We noted that chronic exposure to iron can be associated with the appearance of a state of oxidative stress in the hippocampus and the prefrontal cortex demonstrated by an increase in lipid peroxidation, an increase in nitric oxide, and also by disturbances in the antioxidant defense systems following a determination of the concentrations of catalase. In conclusion, we can deduce from this work that chronic iron exposure can be related to the induction of cognitive and affective disorders and oxidative stress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Environmental pollutants have attracted the attention of researchers in recent decades because of their association with psycho-emotional disorders, especially heavy metals, whose ubiquity and neurotoxicity are well known [1]. Moreover, research including previous papers written by our team shows a positive correlation between exposure to certain heavy metals such as (Al, Cr, Cd, Ni…) [2,3,4,5,6] and affective and cognitive disorders, in particular during early and chronic exposure. Several studies using animal models have shown that iron overload leads to significant cognitive and affective impairments [7,8,9,10,11,12,13,14,15,16,17,18,19,20].
There are many sources for iron exposure as food coloring, in the identification of mushrooms, mining, writing inks, blue pigments, dyeing industries, photography, pharmaceuticals, deodorizers, disinfectants, fungicides, molluscicides, water treatment, and supplementary ferrous sulfate (as a source of iron), among other sources, all contain iron in the form of ferrous sulfate [21].
Iron plays a crucial part in physiological processes due to its ability to donate and receive electrons, but it may also cause serious oxidative damage by producing free radicals in the brain when there is an excess of iron in the body [22].
Numerous neurodegenerative disorders, including Parkinson’s (PD), Alzheimer’s (AD), and Huntington's diseases, among others, have been associated with iron deposition in certain brain regions [23,24,25,26].
Evidence that patients with neurodegenerative disorders have high brain iron concentrations and the identification of mutations in genes related to iron metabolism in the brain suggest that iron dysregulation and iron-induced oxidative stress (OS) in the brain may contribute to neuronal death associated with brain disorders [27].
More and more evidence points to an association between OS and iron-induced neurotoxicity. Through the Fenton reaction, free iron can cause oxidative stress [28]. Numerous neurodegenerative disorders have been linked to OS as one of their causative components. Studies on patients and experimental models show that oxidative damage occurs before pathogenic alterations in AD [29,30,31].
To our knowledge, few studies have examined the effects of iron on anxiety and depression in Wistar rats. In addition, no study has yet examined the effect of low-dose iron on affective and cognitive disorder and oxidative stress.
Based on these considerations, our research focuses on the effects of chronic intraperitoneal administration of iron at various doses (0.25, 0.5, and 1 mg/kg) in male Wistar rats for 8 weeks to assess the affective and cognitive behaviors using neurobehavioral tests and OS parameters such as nitric oxide (NO), and lipid peroxidation (LPO), and catalase (CAT) activity in the hippocampus and the prefrontal cortex.
Material and Methods
Animals and Experimental Conditions
Male Wistar strain rats weighing 120 ± 20 g, that were born and grown in the animal inventory of the Kenitra Faculty of the Ibn Tofail University were used in our investigation. The rats were housed in experimental cages (6 rats in each cage) that underwent regular cleaning, including the replacement of the bedding consisting of wood chips three times per week. They are maintained in a ventilated room, at a temperature of 21 °C ± 1, and with artificial lighting (a photoperiod: LD 12/12: 12 h of light, 12 h of darkness). Throughout the experiments, the rats had free access to tap water for drinking and dry food (standard diet provided by the Alf Sahel compound feed company) the feeding of animals contains: dry corn, barley, wheat bran, soybean, and sunflower oilseed cakes; protein 15%; fat 2%; phosphorus 0.3%; minerals 9%; cellulose: 15%; calcium 1%; vitamins per 100 g: vit A 1 000,000 UI, vit D3 15,000, vit E 2000. Rats are marked to facilitate individual identification and are kept in their cages to acclimate them to the conditions in the animal facility for 2 weeks before starting the experiment. All experimental procedures were approved by the University Ethics Committee for Animal Experiments.
Daily intraperitoneal injections of iron sulfate are administered to the rats (FeSo4) obtained from Sigma-Aldrich, St. Louis, MO, USA) at 4 pm at different doses, for 8 successive weeks. They are divided into 4 batches of 6 animals as follows:
-
1st group (control): male rats receiving an intraperitoneal injection of NaCl
-
2nd group (dose 1): male rats receiving a dose of 0.25 mg/kg FeSo4
-
3rd group (dose 2): male rats receiving a dose of 0.5 mg/kg FeSo4
-
4th group (dose 3): male rats receiving a dose of 1 mg/kg FeSo4
Note that the volume to be injected changes in relation to the weight of the rat according to the rule of three:

Experimental Design
Rules and Ethics of Animal Experimentation
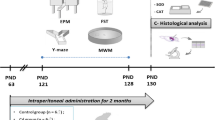
In accordance with the regulations of the Guide for the Care and Use of Laboratory Animals (National Research Council, revised 1996), every effort has been made to minimize animal suffering and reduce the number of animals used (Fig. 1).
Experimental design. Male “Wistar” rats weighing (120 ± 20), daily received an intraperitoneal injection of 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron for 2 months. The Open Field Test (OFT), Elevated Plus Maze Test (EPMT), Forced Swimming Test (FST), Y-Maze Test, and Morris Water Maze Test (MWMT) were used to assess the behavior and cognitive states of rats. The oxidative stress state in the brain was assessed by lipid peroxidation (LPO); nitrite assay (NO); catalase activity (CAT)
All procedures were performed to prevent potential pain or distress to the animals. The manipulations were carried out with the best possible respect for anesthesia and asepsis control measures, noise was kept to a minimum and all team members were provided with appropriate protective equipment. At the end of the last test, a final weight measurement of the animals was performed the day before their sacrifice in order to calculate the doses of anesthetic to be injected. The anesthetic product used was chloral at a concentration of 7%, dissolved in an adequate volume of 0.9% NaCl, used at a dose of 0.5 ml/100 g of animal weight.
As soon as the animals were anesthetized, they were sacrificed by decapitation and the brains of these animals were extracted from the skull box on ice, using previously sterilized dissection equipment.
Neurobehavioral Tests
Five tests allow the evaluation of the affective and cognitive state of the rodents through the measurement of several parameters. The behavior of the animals in the various devices was captured on camera and recorded. We have identified the most notable parameters in the recordings that can be used to identify affective and cognitive disorders.
Anxiety-Like Measurement
Open Field Test
The OFT is used to measure anxiety-like behavior in rodents [32]. The OFT is realized thanks to a device of square form, of 100 cm of side, and 40 cm of height. The white field platform (25 equal tiles) is subdivided into two zones, the periphery (16 equal tiles) bordering the partitions, considered as a relatively secure refuge zone, and the central zone (9 equal tiles) by definition open and reputed to be anxiogenic. The animal is placed in the center of the field for a period of 10 min during which its behavior is monitored and recorded with a video camera connected to a computer. The device is cleaned with ethanol 70% after the passage of each rat. The level of anxiety is estimated by referring to the movements in the two surfaces, the measured parameters are.
-The time spent in the central area of the Open Field (TCA) and the number of returns to the center (NRC), Central perimeter residence time is used as a measure of anxiety. The number of returns to the central area is also an indicator of emotional reactivity.
- Number of total squares (NTS). The total number of entries in the central and peripheral tiles is a reliable index of general locomotor activity [33].
Test of the Elevated Plus Maze (EPM)
The EPM test used in the present study is consistent with that described by (Pellow et al. 1985) [34] based on neophobia in rats. The maze consists of four branches 50 cm long and 10 cm wide kept 60 cm from the ground. The two closed arms are surrounded by 50 cm high walls, the edges are 1 cm high to prevent rats from falling. The animal is placed in the center of the device for 5 min. The whole of its behavior is filmed by a video camera linked to a computer. Entries into open arms (EOA) (an entry is defined when both of the rat's forepaws are on the arm), Total number of arms entries (TAE), and the time spent in open arms (TOA) are the parameters revealed by this test that assess locomotion and anxiety behaviors in animals [35]. The decrease in anxiety-like behavior is illustrated by a statistically significant increase in parameters in the open arms (time, inputs, or both). The total number of entries in all arms gives a general hyperactivity. To remove any lingering olfactory cues, the apparatus was cleaned between each examination with 70% ethyl alcohol.
Depression-Like Measurement
Forced Swimming Test (FST)
This test assesses the depressive state of rats [36], the animals are subjected to a 5-min forced swimming test. However, they are placed individually inside a cylinder of 30 cm diameter and 50 cm height, in water at 22 ◦C ± 1. The length of time the animal actively swims or floats only to keep its head above water is measured. After struggling in the water, the animal becomes almost motionless, moving its legs occasionally to stay afloat or regain its balance. This time of immobility (TIM) is interpreted as a reflection of “behavioral despair,” which occurs when the animal realizes that it could not escape. The presence of a depressive disorder is characterized by a significant reduction in Struggling Time (TST) and an increase in the TIM.
Cognitive Measurement
Y-Maze Test
This is a test to evaluate the natural tendency of a rat to alternate its choice after exploring a branch of a maze [37]. Indeed, in most cases, it will spontaneously explore the other branches that are unknown to it. The Y-labyrinth is composed of three identical lanes arranged along the medians of an equilateral triangle. These lanes are 13 cm long, 4.5 cm wide and 5.5 cm high. In our procedure, the rat is placed in one of the three branches of the maze, with its head directed towards the point of intersection of the 3 branches, and then left for 8 min to explore freely. It is considered to be in a branch if its 4 legs are inside. The order of the visits is recorded, from which the total number of visits is extracted as an index of general activity, as well as the distribution of the visits in the three branches. The data presented are the result of an analysis of this distribution to count the alternations and are expressed in percentage of alternation. This is calculated according to the formula:
Morris Water Maze Test
The water maze task used was an adaptation of the hidden escape paradigm described by Morris [38]. The experimental device is an opaque basin of 110 cm in diameter, whose walls are 33 cm high. A 13.5-cm-high platform (9 cm diameter) was placed in the pool, which was filled to a depth of 14 cm by water at a temperature of 22 °C ± 1. A non-toxic white paint (milk powder) is dissolved in water to make it opaque. The basin is installed in a room on the walls of which are hung various visual clues. The experimenter is hidden from the sight of the animal whose movements are followed by a video camera. The pool is virtually divided into 4 quadrants: northeast (NE), southeast (SE), southwest (SW), and northwest (NW).
-
The acquisition phase lasts 4 days with 4 trials per day. The platform is placed in the NW quadrant at 2 cm below the water surface. The rat is placed in the pool with its head against the wall at one of the four cardinal points. The time taken by the animal to reach the platform is recorded. Each trial lasts 60 s. If the rat has not found the platform at the end of the trial, it is placed on it by the experimenter for 20 s.
-
The probe test is performed the day after the last day of training. The platform is removed from the pelvis and the animal is placed in S. The time spent in the quadrant where the platform was located during the acquisition phase (NW) is measured in a single 60-s trial.
-
After 2 h of the probe test, the visible platform phase takes place. The platform is placed in the middle of the NW quadrant and made visible. The purpose of this visual discrimination task is to verify the absence of sensorimotor or visual bias. It consists of 4 trials of 60 s, during which the platform is made visible (1 cm above the water surface). The time taken to reach the platform is measured (latency) [39].
Biochemical Examination
Preparation of the Homogenate
The brains of the different groups studied were used. After grinding and homogenization in TBS (Tris 50 mM, NaCl 150 mM, pH 7.4), centrifugation (1500 rpm, 4 °C, 15 min) was performed for 10 min, and the resulting supernatant was aliquoted into Eppendorf tubes and stored at – 20 °C while waiting for the assays of the oxidative stress parameters.
Determination of Nitric Oxide
The following elements were mixed in a spectrophotometer tank: 100 μL of Griess reagent (Griess solution A = 0.1% naphthylethylene diamine chloralhydrate diluted in water + Griess solution B = 1% sulfanilamide diluted in 5% H3PO4) + 300 μl of the sample containing nitrite + 2.6 mL of distilled water. Incubation of the mixture was performed for 30 min at room temperature. Then, the optical density reading was taken at 548 nm [40]. Similarly, a standard range was prepared by using the different concentrations of nitrates (10, 20, 40, 60, 80, and 100 μM). The NO concentration of the samples is deduced from the calibration curve (standard range) established under the same conditions. NO levels are expressed as µmol/g tissue.
Lipid Peroxidation Assay
The rate of lipid peroxidation was indirectly determined by measuring the level of Malondialdehyde (MDA) in seahorse homogenates by the method of Draper and Hadley (1990) [41]. The principle of the assay is to react thiobarbituric acid (TBA) with one of the end products of lipid peroxidation: MDA to form a pink pigment with an absorbance at 532 nm. The optical density of the TBA-MDA complex (TBARS) is proportional to the MDA concentration. To conical tubes containing 500 µl of hippocampal homogenate, 0.5 ml of trichloroacetic acid (TCA) (20%) and 1 ml of TBA (0.67%) were added successively. The mixture was heated to 100 °C for 15 min and immediately cooled before the addition of 4 ml of n-butanol. Then the tubes were centrifuged for 15 min at 3000 rpm. Finally, the supernatant was collected from each tube and the absorbance was read by a spectrophotometer at a wavelength of 530 nm [42]. Similarly, a standard range was prepared by using the different concentrations of MDA (0.1, 0.2, 0.3, 0.3, 0.4 0.6, 0.8 μmol/l) to determine the MDA concentration of the samples. The amount of MDA in the sample is expressed as nmol/gram of tissue.
Determination of Catalase (CAT) Activity
The measurement of CAT activity in homogenates was determined according to the method of Aebi (1984) [43]. It is based on the change in optical density following the disappearance of H2O2. 0.05 ml of the sample (tissue extract or phosphate buffer for the blank), was added to 1.95 ml of phosphate buffer (0.05 M, pH 7.4) contained in a quartz cuvette. The reaction was triggered by the addition of 1 ml of H2O2 (0.05 M) and the decrease in absorbance was recorded for 2 min (readings every 30 s) in cuvettes at a wavelength of 240 nm. CAT activity is expressed as IU/min/g tissue (μmoles of H2O2 destroyed/min/g tissue, at 25 °C).
Statistical Analysis
The values of the parameters collected from the recordings were first entered in Excel. The statistical analysis of the different results is obtained thanks to the analysis of variances (ANOVA) 1st/2nd order with the software SPSS version 22. A repeated measures ANOVA is used for the MWM. The results are presented in the form of mean ± standard error of the mean (S.E.M.) and illustrated by figures produced by the GraphPad Prism 6 software. Intergroup differences are considered significant when p < 0.05, highly significant when p < 0.01, and very highly significant when p < 0.001.
Results
Effect of Iron on the Levels of Anxiety-Like Measured in the OFT
Time Spent in the Central Area (TCA)
The results, grouped in (Fig. 2a), show that iron in males increases the TCA parameter at low dose 0.25 mg/kg (Fe-0.25), (Cont/Fe-0.25: p < 0.05). While from the dose of 0.5 mg/kg iron decreases the TCA parameter. Animals in the groups treated with 0.5 mg/kg and 1 mg/kg of iron spend significantly less time in the central zone compared to the group treated with 0.25 mg/kg of iron: [(Fe-0.25/Fe-0.5: p < 0.01); Fe-0.25/Fe-1: p < 0.001). In addition, the effects of iron at 0.5 and 1 mg/kg are significantly lower than that obtained with 0.25 mg/kg.
a Total amount of time spent in the center (TCA). b Number of returns into the center area of the arena in the open-field behavior apparatus (NRC). c Number of total squares (NTS) in the open field in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are expressed as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
Number of Returns to the Center (NRC)
The iron-dependent dose effect is also observed in the NRC parameter (Fig. 2b). At doses of 0.5 and 1 mg/kg, Iron decreases the NRC parameter. While at the 0.25 mg/kg dose iron increases the NRC parameter. With a very significant difference, the rats treated with 1 mg/kg of iron and those treated with 0.5 mg/kg visit the central zone significantly less than the rats treated with 0.25 mg/kg of iron, [(Fe-0.25/ Fe-0.5: p < 0.05); Fe-0.25/ Fe-1: p < 0.01)]. However, no statistically significant difference was found when comparing the iron-treated groups with the controls [(Cont/Fe-0.25: p > 0.05); Cont/Fe-0.5: p > 0.05) et (Cont/Fe-1: p > 0.05)].
Number of Total Squares (NTS)
Unlike the TCA and NRC parameters, iron has no significant effect on the NTS parameter, whatever the dose considered [(Cont/Fe-0.25: p > 0.05); (Cont/Fe-0.5: p > 0.05); (Cont/Fe-1: p > 0.05)] (Fig. 2).
Effect of Iron on Anxiety Levels Measured in Elevated Plus Maze Test (EPM)
Time Spent in Open Arms (TOA)
The results grouped in (Fig. 3a) show that Iron does not induce any significant modification of the TOA parameter [(Cont/Fe-0.25: p > 0.05); (Cont/Fe-0.5: p > 0.05); (Cont/Fe-1: p > 0.05)]. In contrast, a statistically very significant difference is noted by comparing the different groups treated with iron between them (Fe-0.25/Fe-1: p < 0.01). In addition, groups treated with iron 1 mg/kg spend less time in open arms compared to rats treated with iron at 0.25 mg/kg.
a The total amount of time spent in exposed arms (TOA). b The number of entries in exposed arms (EOA). c The total number of arms entries (TEA) in elevated plus maze in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are expressed as mean ± SEM. The significance level is 0.05. * p < 0.05, ** p < 0.01, *** p < 0.001
Entry to Open Arms (EOA)
The dose-dependent effect of iron is not observed when considering the EOA parameter (Fig. 3b). The doses of 0.25, 0.5, and 1 mg/kg do not induce any significant modification of the parameter in comparison with the controls [(Cont/Fe-0.25: p > 0.05); (Cont/Fe-0.5: p > 0.05); (Cont/Fe-1: p > 0.05)]. A statistically significant difference is noted by comparing the different groups treated with iron between them. Rats treated with 1 mg/kg of iron visited significantly less the open arms compared to groups treated with 0.25 and 0.5 mg/kg [(Fe-0.25/Fe-1: p < 0.05); (Fe-0.5/Fe-1: p < 0.05)].
Total Entries in Arms (TEA)
Unlike the results observed for the TOA and EOA parameters, iron has no statistically significant effect on the TEA parameter, regardless of the dose considered (p > 0.05) (Fig. 3c).
Effect of Iron on Depressive-Like Performances Measured by Forced Swimming Test (FST)
Immobility Time (TIM)
In the FST test, iron considerably increases the TIM parameter, with superiority in the groups treated with 1 mg/kg with a very significant difference compared to the Control Cont / Fe-1: p < 0.01). At the dose of 0.25 mg/kg, iron decreases considerably with a significant difference compared to the Control TIM parameter (Cont/Fe-0.25: p < 0.05), while the 0.5 mg/kg dose does not induce any significant change in the parameter (Cont/Fe-0.5: p > 0.05). A statistically very highly significant difference is noted by comparing the different groups treated with iron between them [(Fe-0.25/Fe-0.5: p < 0.001); Fe-0.25/Fe-1: p < 0.001)] (Fig. 4a).
a Immobility time expressed in seconds (s) (TIM). b Struggling time (STS) in Forced swimming test expressed in seconds (s) in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. * p < 0.05, ** p < 0.01, *** p < 0.001
Struggling Time (TST)
Iron also decreases the dose-dependent parameter TST (Fig. 4b). Compared to the control, the TST is reduced significantly in the group treated with iron to 0.25 mg/kg and very significantly in the group treated with iron at 0.5 mg/kg and highly significantly in the group treated with iron at 1 mg/kg [(Cont/Fe-0.25: p < 0.05); Cont/Fe-0.5: p < 0.01) and (Cont/Fe-1: p < 0.001)]. In contrast, in the previous parameter, no statistically significant difference was observed when comparing the different groups treated with iron between them [(Fe-0.25/Fe-0.5: p > 0.05); Fe-0.5/Fe-1: p > 0.05); Fe-0.25/Fe-1: p > 0.05)].
Iron Effect on Memory
Y Maze Test
The results grouped in Fig. 5 show that iron affects the parameter of the percentage of alternation in the alternation test in the Y labyrinth. From 1 mg/kg, iron begins to affect this parameter, while the doses of 0.25 and 0.5 mg/kg does not induce any significant modification of the parameter [(Cont/Fe-0.25: p > 0.05); Cont/Fe-0.5: p > 0.05)]. At a dose of 1 mg/kg, iron reduces this parameter with a significant difference compared to the control group (Cont/Fe-1: p < 0.05). A statistically very significant difference is noted by comparing the different groups treated with iron between them [(Fe-0.25/Fe-1: p < 0.01); Fe-0.5/Fe-1: p < 0.05)] (Fig. 5).
Spontaneous alternation percentage measured in Y-maze test in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
Morris Water Maze
Acquisition phase
In the Morris Water Maze test, iron considerably increases the latency parameter during the 4 days of the acquisition phase with superiority in the group treated with 1 mg/kg with a significant difference compared to the control group (Cont/Fe-1 p < 0.05), while the doses of 0.25 and 0.5 mg/kg do not affect this parameter [(Cont/Fe-0.25: p > 0.05); Cont/Fe-0.5: p > 0.05)]. A statistically very significant difference is noted by comparing the different groups treated with iron between them [(Fe-0.25/Fe-1: p < 0.01); Fe-0.5/ Fe-1: p < 0.05)] (Fig. 6).
Latency to reach the hidden platform on each of the 4 days of the learning phase in the Morris water maze, in males after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. * p < 0.05
Percentage time spent in the correct quadrant during the probe trial
The results grouped in Fig. 7 show that iron affects the parameter of the percentage of time spent in the correct quadrant (NO). From 0.25 mg/kg, iron increases this parameter with a very significant difference compared to the control group (Cont/Fe-0.25: p < 0.01). In contrast, at the dose of 0.5 and 1 mg/kg the iron reduces this parameter with a significant difference in comparison with the control group [(Cont/Fe-0.5: p < 0.05); Cont/Fe-1: p < 0.05)]. A statistically very highly significant difference is noted by comparing the different groups treated with iron between them [(Fe-0.25/Fe-1: p < 0.001); Fe-0.5/Fe-1: p < 0.001)] (Fig. 7).
Percentage of time spent in the correct quadrant in the probe trial of the Morris water maze expressed as % in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
Visible platform test
Unlike the results observed for the Acquisition phase and Percentage time spent in the correct quadrant, iron has no statistically significant effect on the latency parameter to find the visible platform, regardless of the dose considered (p > 0.05) (Fig. 8).
Latency to reach the visible platform for each strain in the MWM expressed in second (S), in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
Iron Effect on Oxidative Stress
NO Concentrations in the Hipp and the PFC
The results grouped in Fig. 9 show that treatment of rats with iron significantly increases (NO) levels in the groups treated with 1 mg/kg compared with the control group in both HIPP: [Hipp. (Cont/Fe-1: p < 0.01)] and PFC: [PFC (Cont/Fe-1): p < 0.001)].
Determination of the nitric oxide (NO) levels in hippocampus (a) and in prefrontal cortex (b), expressed in µmol/g of tissue in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25) 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
In addition, there is a statistically significant difference between groups in the two structures studied, in HIPP Fe-1/Fe-0.25 (p < 0.01), and in PFC Fe-1/Fe-0.25 and Fe-1/Fe-0.5 (p < 0.001 and p < 0.001 respectively).
Catalase (CAT) Activity in the HIPP and the PFC
The results obtained show that treatment of rats with iron causes a significant decrease in (CAT) enzyme activity in the brain compared to control rats. In HIPP and PFC, CAT enzyme activities are significantly decreased in the groups treated with 0.5 mg/kg and 1 mg/kg of iron compared to the control group. [PFC (Cont/Fe-0.5: p < 0.05); (Cont/Fe-1): p < 0.01)]; [Hipp. (Cont/Fe-0.5: p < 0.05); Cont/Fe-1: p < 0.01)].
On the other hand, there is a statistically significant difference between the treated groups in HIPP Fe-1/Fe-0.25 (p < 0.01), and in PFC Fe-1/Fe-0.25 (p < 0.05). No difference was noted between these groups; Fe-0.5/Fe-1 (p > 0.05) in both structures studied (Fig. 10).
Changes in catalase (CAT) activity in hippocampus (a) and in prefrontal cortex (b), expressed as U/g of tissue in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25), 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
LPOin the HIPP and the PFC
According to the results obtained, there was a significant increase in the level of TBARS in the brains of the treated rats and those in HIPP and PFC compared to the control rats. The animals of the groups treated with 0.5 mg/kg and 1 mg/kg of iron, present significantly a higher level of TBARS compared to the control in PFC [(Cont/Fe-0.5: p < 0.05); Cont/Fe-1: p < 0.01)], and in HIPP the parameter TBARS is significantly affected by the factor treatment with 1 mg/kg of iron [(Cont/Fe-1: p < 0.001)].
In addition, there was a statistically significant difference between the treated groups in the two structures studied, in HIPP Fe-1/Fe-0.25 and Fe-1/Fe-0.5 (p < 0.001 and p < 0.001 respectively), and in PFC Fe-1/Fe-0.25 (p < 0.01). No difference was noted between these groups: Fe-0.5/Fe-1 (p > 0.05) in PFC (Fig. 11).
Determination of the lipid peroxidation levels in hippocampus (A) and in prefrontal cortex (B), TBARS levels expressed in nmol/g of tissue in male rats after 2 months of treatment with 0.9% of NaCl (control), 0.25 mg/kg (Fe-0.25) 0.5 mg/kg (Fe-0.5), and 1 mg/kg (Fe-1) of iron. Results are represented as mean ± SEM. The significance level is 0.05. *p < 0.05, **p < 0.01, ***p < 0.001
Discussion
The assessment of anxiety, depression, and memory behaviors in our study is based on the use of validated behavioral tests. OFT and EPM tests have been adopted for determining anxiety levels, FST for assessment of depressive states, and Y-maze and MWM tests for assessing working memory and spatial memory in rodents. Oxidative stress was determined by measuring NO, CAT, and TBARS levels.
Iron and Anxiety
In the present study, the assessment of anxiety levels obtained in the OFT is still confirmed by the use of EPM, one of the most widely used anxiety models in recent years [44]. Like OFT, EPM is based on the natural conflict between the desire to explore a new environment and the tendency to avoid a potentially dangerous area. All of the behaviors measured in EPM depend, directly or indirectly, on locomotor activity, which is probably the main confounding factor.
Based on the behavioral study of rats in OFT and EPM tests, the present study was able to show that iron, administered chronically, exerts an anxiety-inducing effect in rats. In OFT, this anxiogenic effect of iron is based on the fact that it decreases TCA and NRC parameters without altering locomotor activity. Its anxiogenic action begins at 0.5 mg/kg and reaches a maximum at a dose of 1 mg/kg. The locomotor activity measured in the OFT is not affected by this treatment, suggesting that the behavioral differences in the Porsolt test are due to changes in the affective state.
The anxiogenic action of iron, shown in this study, is consistent with the observation of Maaroufi (2009), which were reported in rats for 5 consecutive days, Rats were treated with moderate doses of iron (1.5 or 3.0 mg/kg) [17, 45]. Thus, the study by Chtourou (2015) was reported on the Wistar rat for 28 days and 5 days per week, in this case, the rats were treated with iron intraperitoneally at 50 mg of iron [46]. and also the study of Elferchichi which was reported in the Wistar rat for 5 consecutive days, the rats were treated with a dose of 3.0 mg/kg of iron sulfate [45].
Compared to these studies, our work has the advantage of administering very small amounts of iron chronically over a long period and obtaining an effect at lower doses, since the efficacy of iron appears at from 0.5 mg/kg.
The majority of research carried out in various animal species confirms the ability of iron to increase anxiety levels, regardless of the anxiety-inducing situations and behavioral tests adopted. Maaroufi (2009), and Chtourou (2015), evaluating the effects that iron could have on the anxiety-related behavior of adult Wistar rats, were able to show that iron produces more potent anxiety-inducing effects which results in increase in the time spent crossing the closed arms and increasing the average time spent in the closed arms, but a reduction in time (less time) spent in the open arm (TOA) of the maze by compared to the control group.
In conclusion, all the data mentioned above, associated with our results clearly show that iron has real anxiogenic properties.
Iron and Depression
In the present work, the behavior of depression was assessed by the FST. In this paradigm, the animal develops the characteristic behavior denoted by immobility commonly described as a symptom of hopeless behavior. Forced swimming is a suitably stressful situation both physically and emotionally in rats [47].
The present study also showed that chronic administration of iron at doses of 0.25 to 1 mg/kg in rats caused an increase in TIM and a reduction in the wrestling behavior represented by TST.
The observation that iron increases TIM and reduces TST in rats provides a strong argument for the depressive effect of iron. These results clearly suggest that chronic exposure to iron causes a state of despair in the treated animal which manifests itself in a reduction in swimming and the installation of a state of despair which is probably the consequence of the deterioration of dopaminergic and serotonergic pathways in the brain mainly striatum, hippocampus and the hypothalamic-pituitary axis [48, 49]. The production of free radicals which will be responsible for neurotoxic oxidative stress may be the result of abnormal distribution of iron in cells or tissues with regional accumulation. This behavior of rats could be reflected by an iron interaction with serotonergic and dopaminergic neurons in the hippocampus [48]. These systems appear to be involved in the regulation of CRF (corticotropin releasing factor) which plays an important role in anxiety behavior [48]. In addition, the serotonergic system has a central role in the modulation of anxiety [50]. These results demonstrate increased anxiety/emotional responsiveness to iron overload, which is similar to the behavioral phenotype of iron deficiency [51,52,53].
In conclusion, the results of the research shown above in conjunction with our own demonstrate that iron exposure induces depressive properties.
Iron and Cognitive Impairment
Regarding cognitive abilities, the present study also showed that chronic administration of iron in doses of 0.25 to 1 mg/kg in male rats causes a reduction in working memory, short-term memory and spatial learning performance under our experimental conditions. Iron is effective at just 1 mg/kg. In the Y-maze test, the working memory reduction effect is that it decreases the percent alternation parameter. In the Morris Pool, the effect that iron affects spatial learning performance is that it increases the latency parameter to reach the platform (the first 4 days). The effect that iron causes spatial memory retention deficit is based on the fact that it reduces the time spent in the correct quadrant during the probe test.
The results present indicate that chronic iron exposure causes cognitive disruption in male rats. These observations agree with those of some authors who demonstrate an impairment of learning performance in rats exposed to iron [17, 54, 55]. Chronic administration of iron intraperitoneally in male rats has been reported to cause an increase in the latency to find the platform submerged during the learning phase in the MWM as well as a decrease in time spent in the northwest quadrant during the probe test. These observations agree with those of Maaroufi (2009) [17] who showed that the administration of 3 mg/kg of iron increases the latency time to find the platform and the time spent in the correct quadrant was lower than that of the control group. Furthermore, during the visible platform phase, we do not observe any significant difference among any of the groups, which means that the deficits of the rats are not due to visual disturbances.
When combined, the findings from animal models demonstrate that iron overload causes memory deficits in a variety of learning and memory paradigms that are intended to evaluate memory types that are known to rely on specific memory systems. Aversive memory [18], recognition memory [15, 16], and spatial memory [10, 11, 17, 20] in rodents have all been demonstrated to be impaired by iron exposure at various stages of their lives. These memories are known to depend on hippocampal-cortical circuitry [56, 57]. Because cholinergic pathways play a crucial role in cognitive processes including learning and memory, its disruption can have negative effects on various learning and memory models [58]. Bohnen et al. (2005) results suggests that reduced acetyl-cholinesterase (AChE) activity, which is associated with impaired cognitive performance, is evidence of the loss of cholinergic pathways, a characteristic of aging and AD [59].
Other results found that Excess iron in the brain is involved in the development and pathogenesis of neurodegenerative disorders [60]. It has been clearly shown that iron overload disrupts the homeostasis of neurotransmitters. Infusions of iron in the substantia nigra disrupt monoaminergic systems, in particular the dopaminergic pathway [49, 61, 62]. Excessive iron deposits have been reported in the central nervous system (CNS) in several neurodegenerative conditions such as Alzheimer’s disease [54]. In addition, iron has also been shown to accumulate in both senile plaques and amyloid deposits in the AβPP2576 transgenic mouse model of Alzheimer’s disease [54].
Iron and Oxidative Stress
For the past two decades, toxicological research has concentrated on the development of (OS) following iron exposure as a potential neurotoxic mechanism [63,64,65,66,67,68,69].
Iron exposure can induce a state of oxidative stress by increased production of free radicals that accumulate in the brain, alteration of antioxidant defense mechanisms, or increased lipid peroxidation.
The oxidative stress caused by iron in the hippocampus and prefrontal cortex may be connected to the behavioral abnormalities seen in our investigation following iron administration. Knowing that the hippocampus and the prefrontal cortex play an important role in the learning and memorization process and could thus be the target structure of the neurotoxic effect caused by iron intoxication. In this sense, we have shown that chronic exposure to iron induces an increase in OS measured by LPO, NO, and catalase activity in the hippocampus as well as in the prefrontal cortex of rats. In the present study, the exposure of rats to iron resulted in a significant increase in LPO, indicated by the increase in TBARS levels (affected by the factor treatment with 1 mg/kg of iron) in the hippocampus and prefrontal cortex. These results are in agreement with the work undertaken by Chtourou et al. (2015) where they showed an increase of LPO in the hippocampus of iron-treated rats [46, 64]. In another research, rats were given oral doses of 0.0, 7.5, or 15.0 mg Fe2 + /kg of body weight on postnatal days 10–12 to investigate if iron exposure during the neonatal period had any long-term oxidative consequences. At 3 months of age, the activities of protein carbonyl, SOD, and TBARS were assessed. In the substantia nigra of iron-treated rats, they observed an increase in TBARS and protein carbonylation as well as a reduction in SOD activity [70].
LPO is the process of oxidative degradation of polyunsaturated fatty acids and its occurrence in biological membranes causes changes in membrane structure and function and leads to decreased membrane fluidity and the inactivation of several membrane enzymes [71]. In accordance with this, iron-induced LPO found in this study was linked to elevated NO, which may cause different harmful consequences [67]. Iron's oxidizing qualities are thought to be responsible for several of its hypothesized impacts. In the hippocampus and prefrontal brain, antioxidant enzymes like catalase (CAT) are shown to have decreased activity after iron treatment. An earlier study demonstrated that continuous iron treatment reduces the brain's SOD and CAT activity [64, 67]. Through the Fenton reaction, free iron can cause oxidative stress [28].
Conclusion
In conclusion, the current study showed that chronic iron administration caused affective and cognitive deficits which were observed first at the dose of 0.5 mg/kg and continued in a dose-dependent manner up to the maximum tested dose of 1 mg/kg, as well as brain neurotoxicity indicated by an increase in LPO and NO levels in the hippocampus and prefrontal cortex and a decrease in CAT activities in male rats.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ (2012) Heavy metal toxicity and the environment. Mol Clin Environ Toxicol 101:133–164. https://doi.org/10.1007/978-3-7643-8340-4_6
Lamtai M, Ouakki S, Zghari O, Mesfioui A, El Hessni A, Ouichou A (2019) Affective behavior dysregulation was induced by chronic administration of copper in Wistar rats. Neurosci Med 10(02):134–149. https://doi.org/10.4236/nm.2019.102009
Zghari O et al (2018) Effect of chronic aluminum administration on affective and cognitive behavior in male and female rats. J Behav Brain Sci 08(04):179–196. https://doi.org/10.4236/jbbs.2018.84012
Lamtai M et al (2018) Effect of chronic administration of nickel on affective and cognitive behavior in male and female rats: Possible implication of oxidative stress pathway. Brain Sci 8:8. https://doi.org/10.3390/brainsci8080141
Lamtai M et al (2018) Effect of chronic administration of cadmium on anxiety-like, depression-like and memory deficits in male and female rats: possible involvement of oxidative stress mechanism. J Behav Brain Sci 08(05):240–268. https://doi.org/10.4236/jbbs.2018.85016
El Brouzi MY et al (2021) Intrahippocampal effects of nickel injection on the affective and cognitive response in Wistar rat: potential role of oxidative stress. Biol Trace Elem Res 199(9):3382–3392. https://doi.org/10.1007/s12011-020-02457-5
de Lima MNM et al (2007) Desferoxamine reverses neonatal iron-induced recognition memory impairment in rats. Eur J Pharmacol 570(1–3):111–114. https://doi.org/10.1016/j.ejphar.2007.06.002
da Silva PF et al (2012) Memory impairment induced by brain iron overload is accompanied by reduced H3K9 acetylation and ameliorated by sodium butyrate. Neuroscience 200:42–49. https://doi.org/10.1016/j.neuroscience.2011.10.038
Lozoff B, Castillo M, Clark KM, Smith JB (2012) Iron-fortified vs low-iron infant formula: Developmental outcome at 10 years. Arch Pediatr Adolesc Med 166(3):208–215. https://doi.org/10.1001/archpediatrics.2011.197
Railey AM, Groeber CM, Flinn JM (2011) The effect of metals on spatial memory in a transgenic mouse model of alzheimer’s disease. J Alzheimer’s Dis 24(2):375–381. https://doi.org/10.3233/JAD-2011-101452
Guo C, Wang T, Zheng W, Shan ZY, Teng WP, Wang ZY (2013) Intranasal deferoxamine reverses iron-induced memory deficits and inhibits amyloidogenic APP processing in a transgenic mouse model of Alzheimer’s disease. Neurobiol Aging 34(2):562–575. https://doi.org/10.1016/j.neurobiolaging.2012.05.009
Rech RL et al (2010) Reversal of age-associated memory impairment by rosuvastatin in rats. Exp Gerontol 45(5):351–356. https://doi.org/10.1016/j.exger.2010.02.001
Perez V et al (2010) Iron leads to memory impairment that is associated with a decrease in acetylcholinesterase pathways. Curr Neurovasc Res 7(1):15–22. https://doi.org/10.2174/156720210790820172
De Lima MNM et al (2005) Selegiline protects against recognition memory impairment induced by neonatal iron treatment. Exp Neurol 196(1):177–183. https://doi.org/10.1016/j.expneurol.2005.07.017
de Lima MN et al (2008) Amelioration of recognition memory impairment associated with iron loading or aging by the type 4-specific phosphodiesterase inhibitor rolipram in rats. Neuropharmacology 55(5):788–792. https://doi.org/10.1016/j.neuropharm.2008.06.025
De Lima MNM et al (2005) Recognition memory impairment and brain oxidative stress induced by postnatal iron administration. Eur J Neurosci 21(9):2521–2528. https://doi.org/10.1111/j.1460-9568.2005.04083.x
Maaroufi K, Ammari M, Jeljeli M, Roy V, Sakly M, Abdelmelek H (2009) Impairment of emotional behavior and spatial learning in adult Wistar rats by ferrous sulfate. Physiol Behav 96(2):343–349. https://doi.org/10.1016/j.physbeh.2008.10.019
Schröder N, Fredriksson A, Vianna MRM, Roesler R, Izquierdo I, Archer T (2001) Memory deficits in adult rats following postnatal iron administration. Behav Brain Res 124(1):77–85. https://doi.org/10.1016/S0166-4328(01)00236-4
Fredriksson A, Schröder N, Eriksson P, Izquierdo I, Archer T (2000) Maze learning and motor activity deficits in adult mice induced by iron exposure during a critical postnatal period. Dev Brain Res 119(1):65–74. https://doi.org/10.1016/S0165-3806(99)00160-1
Fredriksson A, Schröder N, Eriksson P, Izquierdo I, Archer T (1999) Neonatal iron exposure induces neurobehavioural dysfunctions in adult mice. Toxicol Appl Pharmacol 159(1):25–30. https://doi.org/10.1006/taap.1999.8711
Nunes ES, Desai SN, Desai PV (2010) Effect of ferrous sulphate on aspartate and alanine aminotransferases of brain of Tilapia mossambica. Food Chem Toxicol 48(2):490–494. https://doi.org/10.1016/j.fct.2009.10.045
Gaasch JA, Lockman PR, Geldenhuys WJ, Allen DD, Van Der Schyf CJ (2007) Brain iron toxicity: Differential responses of astrocytes, neurons, and endothelial cells. Neurochem Res 32(7):1196–1208. https://doi.org/10.1007/S11064-007-9290-4
Hirsch EC, Brandel J-P, Galle P, Javoy-Agid F, Agid Y (1991) Iron and aluminum increase in the substantia nigra of patients with Parkinson’s disease: an X-ray microanalysis. J Neurochem 56(2):446–451. https://doi.org/10.1111/J.1471-4159.1991.TB08170.X
Dexter DT, Jenner P, Schapira AHV, Marsden CD (1992) Alterations in levels of iron, ferritin, and other trace metals in neurodegenerative diseases affecting the basal ganglia. Ann Neurol 32(1):S94–S100. https://doi.org/10.1002/ana.410320716
Riederer P et al (1989) Transition metals, ferritin, glutathione, and ascorbic acid in Parkinsonian brains. J Neurochem 52(2):515–520. https://doi.org/10.1111/J.1471-4159.1989.TB09150.X
Dexter DT et al (1987) Increased nigral iron content in postmortem parkinsonian brain. The Lancet 330(8569):1219–1220. https://doi.org/10.1016/S0140-6736(87)91361-4
Figueiredo LS, De Lima MNM (2013) Role of brain iron accumulation in cognitive dysfunction: Evidence from animal models and human studies. J Alzheimer’s Dis 34(4):797–812. https://doi.org/10.3233/JAD-121996
Berg D et al (2001) Brain iron pathways and their relevance to Parkinson’s disease. J Neurochem 79(2):225–236. https://doi.org/10.1046/j.1471-4159.2001.00608.x
Belkacemi A, Ramassamy C (2012) Time sequence of oxidative stress in the brain from transgenic mouse models of Alzheimer’s disease related to the amyloid-β cascade. Free Radic Biol Med 52(3):593–600. https://doi.org/10.1016/j.freeradbiomed.2011.11.020
Milward EA et al (2010) A cross-sectional community study of serum iron measures and cognitive status in older adults. J Alzheimer’s Dis 20(2):617–623. https://doi.org/10.3233/JAD-2010-1402
Smith MA et al (2010) Increased iron and free radical generation in preclinical Alzheimer disease and mild cognitive impairment. J Alzheimer’s Dis 19(1):353–372. https://doi.org/10.3233/JAD-2010-1239
Carola V, D’Olimpio F, Brunamonti E, Mangia F, Renzi P (2002) Evaluation of the elevated plus-maze and open-field tests for the assessment of anxiety-related behaviour in inbred mice. Behav Brain Res 134(1–2):49–57. https://doi.org/10.1016/S0166-4328(01)00452-1
Durand M et al (1999) Effects of repeated fluoxetine on anxiety-related behaviours, central serotonergic systems, and the corticotropic axis in SHR and WKY rats. Neuropharmacology 38(6):893–907. https://doi.org/10.1016/S0028-3908(99)00009-X
Pellow S, Chopin P, File SE, Briley M (1985) Validation of open : closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Methods 14(3):149–167. https://doi.org/10.1016/0165-0270(85)90031-7
Brown NS et al (2000) Thyroid hormone resistance and increased metabolic rate in the RXR-γ- deficient mouse. J Clin Invest 106(1):73–79. https://doi.org/10.1172/JCI9422
Porsolt RD (2000) Animal models of depression: utility for transgenic research. Rev Neurosci 11(1):53–58. https://doi.org/10.1515/REVNEURO.2000.11.1.53
Hughes RN (2004) The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neurosci Biobehav Rev 28(5):497–505. https://doi.org/10.1016/j.neubiorev.2004.06.006
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods 11(1):47–60. https://doi.org/10.1016/0165-0270(84)90007-4
Kahloula K, Adli DEH, Slimani M, Terras H, Achour S (2014) Effet de l’exposition chronique au nickel sur les fonctions neurocomportementales chez les rats Wistar pendant la période de développement. Toxicol Anal Clin 26(4):186–192. https://doi.org/10.1016/j.toxac.2014.09.056
Chao CC, Anderson WR, Hu S, Gekker G, Martella A, Peterson PK (1993) Activated microgila inhibit multiplication of toxoplasma gondii via a nitric oxide mechanism. Clin Immunol Immunopathol 67(2):178–183. https://doi.org/10.1006/clin.1993.1062
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 186:421–431. https://doi.org/10.1016/0076-6879(90)86135-I
Freitas RM, Sousa FCF, Vasconcelos SMM, Viana GSB, Fonteles MMF (2004) Pilocarpine-induced status epilepticus in rats: lipid peroxidation level, nitrite formation, GABAergic and glutamatergic receptor alterations in the hippocampus, striatum and frontal cortex. Pharmacol Biochem Behav 78(2):327–332. https://doi.org/10.1016/J.PBB.2004.04.004
Aebi H (1984) Catalase in vitro. Methods Enzymol. 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Lister RG (1987) The use of a plus-maze to measure anxiety in the mouse. Psychopharmacology 92(2):180–185. https://doi.org/10.1007/BF00177912
Elferchichi M, Maaroufi K, Ammari M, Sakly M, Abdelmelek H (2015) Effects of combined ferrous sulfate administration and exposure to static magnetic field on brain oxidative stress and emotional behavior. Arch Ital Biol 153(1):37–45. https://doi.org/10.4449/aib.v153i1.1481
Chtourou Y, Ben Slima A, Gdoura R, Fetoui H (2015) Naringenin mitigates iron-induced anxiety-like behavioral impairment, mitochondrial dysfunctions, ectonucleotidases and acetylcholinesterase alteration activities in rat hippocampus. Neurochem Res 40(8):1563–1575. https://doi.org/10.1007/s11064-015-1627-9
Abel EL (1994) A further analysis of physiological changes in rats in the forced swim test. Physiol Behav 56(4):795–800. https://doi.org/10.1016/0031-9384(94)90245-3
Kim J, Wessling-Resnick M (2014) Iron and mechanisms of emotional behavior. J Nutr Biochem 25(11):1101–1107. https://doi.org/10.1016/j.jnutbio.2014.07.003
Sengstock GJ, Olanow CW, Dunn AJ, Barone S, Arendash GW (1994) Progressive changes in striatal dopaminergic markers, nigral volume, and rotational behavior following iron infusion into the rat substantia Nigra. Exp Neurol 130(1):82–94. https://doi.org/10.1006/exnr.1994.1187
Voigt JP, Rex A, Sohr R, Fink H (1999) Hippocampal 5-HT and NE release in the transgenic rat TGR(mREN2)27 related to behavior on the elevated plus maze. Eur Neuropsychopharmacol 9(4):279–285. https://doi.org/10.1016/S0924-977X(98)00031-5
Felt BT et al (2006) Persistent neurochemical and behavioral abnormalities in adulthood despite early iron supplementation for perinatal iron deficiency anemia in rats. Behav Brain Res 171(2):261–270. https://doi.org/10.1016/j.bbr.2006.04.001
Burhans MS, Dailey C, Wiesinger J, Murray-Kolb LE, Jones BC, Beard JL (2006) Iron deficiency affects acoustic startle response and latency, but not prepulse inhibition in young adult rats. Physiol Behav 87(5):917–924. https://doi.org/10.1016/j.physbeh.2006.02.014
Beard JL, Erikson KM, Jones BC (2002) Neurobehavioral analysis of developmental iron deficiency in rats. Behav Brain Res 134(1–2):517–524. https://doi.org/10.1016/S0166-4328(02)00092-X
Salvador GA, Uranga RM and Giusto NM (2011) Iron and mechanisms of neurotoxicity. Int J Alzheimers Dis 2011. https://doi.org/10.4061/2011/720658.
Stankiewicz JM, Brass SD (2009) Role of iron in neurotoxicity: a cause for concern in the elderly? Curr Opin Clin Nutr Metab Care 12(1):22–29. https://doi.org/10.1097/MCO.0b013e32831ba07c
Squire LR (2004) Memory systems of the brain: a brief history and current perspective. Neurobiol Learn Mem 82(3):171–177. https://doi.org/10.1016/j.nlm.2004.06.005
Squire LR, Wixted JT, Clark RE (2007) Recognition memory and the medial temporal lobe: A new perspective. Nat Rev Neurosci 8(11):872–883. https://doi.org/10.1038/nrn2154
De-Mello N, Souza-Junior IQ, Carobrez AP (2005) Pilocarpine prevents age-related spatial learning impairments in rats. Behav Brain Res 158(2):263–268. https://doi.org/10.1016/j.bbr.2004.09.006
Bohnen NI et al (2005) Cognitive correlates of alterations in acetylcholinesterase in Alzheimer’s disease. Neurosci Lett 380(1–2):127–132. https://doi.org/10.1016/j.neulet.2005.01.031
Rouault TA, Cooperman S (2006) Brain iron metabolism. Semin Pediatr Neurolog 13(3):142–148. https://doi.org/10.1016/j.spen.2006.08.002
Ben-Shachar D, Youdim MBH (1991) Intranigral iron injection induces behavioral and biochemical ‘Parkinsonism’ in rats. J Neurochem 57(6):2133–2135. https://doi.org/10.1111/j.1471-4159.1991.tb06432.x
Sengstock GJ, Olanow CW, Menzies RA, Dunn AJ, Arendash GW (1993) Infusion of iron into the rat substantia nigra: nigral pathology and dose-dependent loss of striatal dopaminergic markers. J Neurosci Res 35(1):67–82. https://doi.org/10.1002/jnr.490350109
Viktorinova A (2018) Iron-mediated oxidative cell death is a potential contributor to neuronal dysfunction induced by neonatal hemolytic hyperbilirubinemia. Arch Biochem Biophys 654(April):185–193. https://doi.org/10.1016/j.abb.2018.07.022
Budni P, De Lima MNM, Polydoro M, Moreira JCF, Schroder N, Dal-Pizzol F (2007) Antioxidant effects of selegiline in oxidative stress induced by iron neonatal treatment in rats. Neurochem Res 32(6):965–972. https://doi.org/10.1007/s11064-006-9249-x
Glei DA, Goldman N, Chuang YL, Weinstein M (2007) Do chronic stressors lead to physiological dysregulation? Testing the theory of allostatic load. Psychosom Med 69(8):769–776. https://doi.org/10.1097/PSY.0b013e318157cba6
Chiueh CC (2001) Iron overload, oxidative stress, and axonal dystrophy in brain disorders. Pediatr Neurol 25(2):138–147. https://doi.org/10.1016/S0887-8994(01)00266-1
Dhakshinamoorthy V, Manickam V, Perumal E (2017) Neurobehavioural toxicity of iron oxide nanoparticles in mice. Neurotox Res 32(2):187–203. https://doi.org/10.1007/s12640-017-9721-1
Kokoszko A, Dabrowski J, Lewiński A, Karbownik-Lewińska M (2008) Protective effects of GH and IGF-I against iron-induced lipid peroxidation in vivo. Exp Toxicol Pathol 60(6):453–458. https://doi.org/10.1016/j.etp.2008.04.012
Valko M, Morris H, Cronin M (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12(10):1161–1208. https://doi.org/10.2174/0929867053764635
Dal-Pizzol F et al (2001) Neonatal iron exposure induces oxidative stress in adult Wistar rat. Dev Brain Res 130(1):109–114. https://doi.org/10.1016/S0165-3806(01)00218-8
Gutteridge JMC, Halliwell B (2000) Free radicals and antioxidants in the year 2000. A historical look to the future. Ann NY Acad Sci 899:136–147. https://doi.org/10.1111/J.1749-6632.2000.TB06182.X
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Rezqaoui Ayoub and Ibouzine-dineLaila. The first draft of the manuscript was written by Reqaoui Ayoub and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All animal experiments were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Animal Ethics Committee (Local Institutional Research Committee).
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rezqaoui, A., Ibouzine-dine, L., Elhamzaoui, A. et al. Potential Role of Oxidative Stress in the Effects of Chronic Administration of Iron on Affective and Cognitive Behavior on Male Wistar Rat. Biol Trace Elem Res 201, 4812–4826 (2023). https://doi.org/10.1007/s12011-023-03560-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03560-z