Abstract
Particle-induced X-ray emission (PIXE) method was used in this present research to identify the elements present in selected vegetable samples to show the possible influence in the metal absorption by the vegetables grown in a saline region of Rampal area of Munshiganj District, Bangladesh. The data acquisition setup is calibrated using a 2.5-MeV proton beam in the current ranges of 5nA to 15nA. The detector was used to measure the X-rays emitted during the irradiation. Data acquisition system MAESTRO-32 was used to measure the spectrum picks, and concentration calculation has been done by GUPIX/DAN-32 software. The purpose of this study is to determine the concentration of heavy and trace elements in these samples and to give current information on their safety for consumption. The result shows that K, Ca, and Fe have the highest concentrations, while lead exhibits the lowest but alarming rates compared to reference materials. The findings were likened to IAEA-V-10, IAEA-359, SRM 1515 (apple leaf), and SRM 1573a (tomato leaf). The outcomes of the present investigation demonstrate that these samples are not devoid of health risks in intake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Food plants that contain minerals are of particular importance because they are the primary source of vital elements in human nutrition [1]. Vegetables provide essential bioavailable trace elements to the human body, and a continuous basis of these various elements is required and highly recommended during daily activities. Abundance or deficiency of mineral nutrients in vegetables can cause multiple health problems, and a variety of ailments can be suffered by people. Excessive consumption of different vegetables can enhance mineral balance, lower cardiovascular disease risk, and reduce several cancer risks. Trace elements do not produce calories but maintain osmotic body pH consistency and are used as coenzymes that control metabolic activities [2]. Trace element levels in vegetables can be influenced by various aspects, including inheritances, soil chemistry, and agricultural system trends [3]. Minerals are classified into three types, major elements (Na, K, Ca, Mg, Cl, etc.), trace elements (Co, Cr, Cu, Fe, I, Mn, Mo, Ni, S, Se, Zn, etc.), and minor elements (As, Al, Br, Ba, Bi, Cs, Cd, etc.) [4]. Pb, Sb, Cd, As, Se, and Hg are nonhazardous nutritional elements widely considered to have poisonous effects even in small amounts (less than a hundred ppm) [5]. With this purpose, determining both biological and trace amounts of food items is essential for food safety, effectiveness, and dietary values [6].
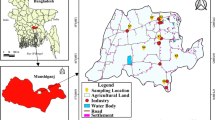
Several nuclear methods, such as X-ray fluorescence (XRF), atomic absorption spectrophotometer (AAS), proton-induced X-ray emission spectroscopy, and neutron activation analysis (NAA), have been widely used in the natural sciences, especially in the ecological field, on account of their ability to detect a wide range of elements with sufficient sensitivity [7,8,9,10, 47,48,49,50]. In the present study, the PIXE method in the Accelerator Facilities Laboratory at the Dhaka Atomic Energy Centre has been used to identify the elements present in some environmental samples, vegetable (Table 1) grown in the saline region in Rampal, Munshiganj District, Bangladesh. PIXE has a benefit due to the high X-ray cross section and low background contribution from bremsstrahlung. The most significant trace and minor components can be calculated in a single run in a limited period. Furthermore, samples can be fit easily for PIXE analysis. In the study of vegetable materials, several researchers used PIXE technology previously [1, 2, 11]. In this research, the pellets have been irradiated by using a 2.5-MeV proton beam. For data collection and spectrum analysis, the application MAESTRO-32 software with GUPIX/DAN-32 was used. The PIXE system has been calibrated and standardized using an X-ray source.
Material and Methods
Particle-Induced X-ray Emission Technique (PIXE)
PIXE spectroscopy with atomic fluorescence is furthermost widespread besides commonly used analytical techniques for 3-MeV accelerators and is done with characteristic X-rays. Once charged particles with enough energy strike a sample, a vacancy may be formed in an atom’s inner shells; when the velocity of the incoming ions equals the acceleration of the inner shell electrons, the chance of finding a vacancy increase. This chance (cross section) for ejecting electrons from the inner shell is very high for MeV ions. This vacancy may be filled in various forms, each of which may produce X-rays with the energy associated with the particular atomic number.
The typical X-rays are identified in the PIXE technology by the [Si (Li)] detector. The disseminative energy study of the detector indicators will show the characteristics of the several elements present in the sample, and, especially, determining the charge or the number of incoming particles can be used to precisely quantify the elemental concentrations.
Experimental Setup
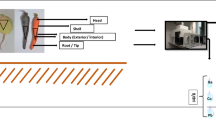
The elemental concentration evolution was executed with 2.5-MeV proton beam energy collection from the 3-MV Van de Graaff accelerators using PIXE in the present study. The vacuum chamber was used to do the overall research. In the chamber, a vacuum of about 2.0 × 10−5 mbar is often achieved. Several turbo-pumping systems are used to vacant the beamline from the accelerator tank. For vacuum operations, the IBA scattering system makes use of a small turbopump. When charged particles with sufficient energy hit a sample, a vacancy in the inner shells of an atom may be formed. The possibility of creating a vacancy is higher when the velocity of the incoming ions matches the velocity of the inner shell electrons. For MeV, this probability for ejecting inner shell electrons is quite high. Such a vacancy can be occupied in a number of ways, and one of the processes may emit X-rays with energy characteristic of that specific atomic number. These emitting typical X-rays are detected by semiconductor detector, specifically Si (Li) detector used to evaluate typical X-rays in the beamline. The detector was connected to a multichannel analyzer to convert the analog data into digital (MCA). For two purposes, the 170-m-thick Mylar absorber has been utilized in front of the X-ray detector. The first is to prevent radiation damage from the sample’s scattered high-energy particles. The second is to diminution the count rate of the X-ray detector, which is crucial to decrement the pulse pile and dead time during the data processing period [1]. The most critical factors considered while charge studies were the subtraction of secondary electrons, the loss of scattered ions, and charge formation in specimen protection. The charge was determined in the Faraday cup, located in the chamber afterward at the beamline’s end. PIXE detected high Z elements, and it provides high-accuracy concentrations of the mainstream of sample components.
Quantitative Analysis
Calibration in PIXE research requires considering the projectile energy loss in the sample and the impacts of the emitted X-rays on absorption and fluorescence. The output of each X-ray peak can be determined from physical factors like the cross-sectional ionization of the specific particle energy and the features that regulate X-ray creation for a thin, uniform, and homogenized sample with minor energy loss from the bombardment particle and small absorption of the X-rays. Particle current may be used to express and calculate the total number of incident particles. For dense homogeneous targets where the bombarding particles are fully stopped and the sample heavily absorbs the emitted X-rays, the yield of each X-ray peak may be obtained using reiterative procedures like those used for electron and X-ray excitation. [12]. The simplest calibration techniques for a PIXE device focus on determining sensitivity factors, which convert exact concentration data to counts in X-ray peaks. However, a system contrast of the theoretical and investigational calibration procedures favorable conditions and homogeneous samples shows that the assessment of elemental concentrations can be done with a precision level of 3–5% [12, 13].
Sampling and Sample Preparation
Seven vegetable samples (Table 1) have been picked up from several rural and industrial zones of Rampal. The collected vegetable samples are made into slices separately and then dried to make them moisture-free. Every type of sample was divided into two for two separate drying systems. Electric oven drying system (max. 200 °C) and freeze-drying system (− 85 °C) were applied to dry the samples separately. In the oven drying system, a maximum 60 °C temperature was used for burning prevention. All the samples were dried continuously until their constant weight. Then the dried samples are ground into fine powder separately. Two pellets were made from each of the samples and preserved for irradiation. “Hand Pellet-Maker” and “Hydraulic Pellet-Maker” (3 tons pressure) were applied to make pellets. Electronic balance was used for sample measurement [14,15,16,17]. Then 0.20-gm dried vegetable powder was pressed by a hydraulic pellet maker. Each pellet was made 7-mm diameter and 1.0-mm thickness in size. Pellet of different samples was preserved separately in vacuum desiccators for the protection of contamination and moisture absorption.
Irradiation and Data Acquisition
At a time, seven different sample slides, quartz, and two standard samples were set with the wheeler and put in the scattering chamber. A 2.5-MeV proton beam with a beam current of 10 to 15 nA was used to irradiate the samples. The data acquisitions were done with the well-established PIXE technique and by the use of the [Si (Li)] detector (Model: SL30165). The detector window material is beryllium, thickness 0.025 mm, active diameter 6.2 mm, and active area 30 mm. The detector depletion and bias voltage are ( −) 100 V DC and ( −)700 V DC, respectively. Quad bias supply unit model: 710 (ORTEC, 0–1000 V) is used. A 170-µm Mylar absorber has been used to save the detector from a high level of X-rays. The spectroscopy amplifier model: 671 and MCB model: 919E (ETHERNIM) are used in the data acquisition setup. The distance between the X-ray detector and the sample ladder is 140 mm. Charges are collected by Faraday cup and set behind the sample wheeler, and for data acquisition, MAESTRO-32 was used. The spectrum data collected by PIXE experiments have been analyzed by GUPIX/DAN-32 commercial software with the help of Eq. 1, putting the values of the standard sample’s concentration, Cst, Ss; the sample’s stopping power, Ss; the standard’s stopping power, Sst; the sample’s yield, Ys; and the standard’s yield, Yst. We calculated the concentrations of the elements in the following equation using Yst [18, 19]. The analyzed spectrums obtained from sample irradiation are shown in Fig. 1(Basella alba).
Results and Discussion
In this present study, 24 elements have been detected. The observed data in Table 2 indicates that all irradiated samples contain essential elements such as S, Cl, Ca, Fe, K, Mn, and Zn; interestingly, their concentrations are in different ranges. Alarmingly toxic elements, Cr, V, and Pb, have also been detected in Colocasia esculenta (Kachu) and Ipomoea aquatica (kolmi shak), respectively, but their concentrations are in alarming range. In general, the concentration of any food product reaches the essential limits; it is unhealthy and toxic to life. Detailed documentation on the nature and severity of the real and possible contaminants in food and proper care should also increase food safety. Ipomoea aquatica had the highest Al concentration (409 ± 4.63 mg/kg), while Spinacia oleracea had the lowest concentration (154 ± 3.84 mg/kg) (Table 2). Aluminum is a crucial chemical factor for good stomach bones and stimulates phosphate absorption in the stomach [20, 21], restricted by IAEA-V-10 and IAEA-359 (Table 3). The mean value for Al is accomplished 243.4286 mg/kg by determining the mean value to SRM 1515 (apple leaf) 284.5 mg/kg and in SRM 1573a (tomato leaf) 598.4 mg/kg. Sulfur in cysteine-protein groups such as insulin is commonly spread all over the body as sulfhydryl and disulfide links. According to the National Research Council, in 1968, RDA per S day is 2–3gm for a standardized male of 22.8 BioMed study 1 year of age [22]. Colocasia esculenta was recorded in the highest percentages of S (298 ± 2.73 mg/kg) and Amaranthus dubius (Table 2) in the lowest proportions (167 ± 4.35 mg/kg). Chloride is considered an essential mineral in the body [23], requiring a daily consumption of 2.3 g. The body must act as an essential nutrient. The comparatively high amounts of Cl (Fig. 2) were 1312 ± 3.16 mg/kg in Colocasia esculenta and 496 ± 4.00 mg/kg in Amaranthus dubius. The elemental report for chlorine in Standard Reference Materials (SRM 1515) (apple leaf) was 582 mg/kg, which was slightly lower than our calculated mean value (369.5714 mg/kg) (Table 3) for all vegetables. The consequences of basic asphyxiant argon gasses are proportional to how the air volume in the air is decreased (partial pressure). Until significant effects occur [24], oxygen may be decreased to 75% of the natural air percentage. In exchange, this involves a quick asphyxiant in the air and gas mixture at a concentration of 33%. If the clear asphyxiant crosses 50%, there will be marked signs. In just a few minutes, a 75% concentration is deadly. The estimated value in Ar is 827.8571 mg/kg when averaging SRM 1515 (apple leaf) NCV and SRM 1573a (tomato leaf) are calculated. Amaranthus dubius recorded the highest percentage (19,982 ± 4.33 mg per kg) (Table 2), and in Raphanus sativus, the lowest value (12,297 ± 4.14 mg/kg) 2000 mg/day is the recommended prescription for potassium for adults in decent physical health. Potassium is the intracellular m eye made in the blood [25], leading to the so-called “hyperkalemia” sequence where the kidneys do not participate enough. Table 3 reveals potassium for all samples calculated for IAEA-V-10 to be daily suited for humans at 21,000 mg/kg and 32,500 mg/kg for IAEA-359. K deficiency causes muscle cramps, a loss of appetite, and an erratic heartbeat [26], so improving heart function is critical. Basella alba contains the highest calcium concentration, 86,404 ± 0.31 mg/kg (Table 2, Fig. 2), which is an essential mineral for maintaining healthy teeth and bones. During the first 2 months of development, calcium consumption for children consuming calcium is approximately a parameter of the body’s water balance. Calcium intake for children fed with calcium is about 33.7 ± 2.0 mg/100 kcal. The calcium fence is calculated to be 68 ± 38 mg/day for most of these beginners [27, 28], based on urinary calcium and normal excretion numbers. The best result in both samples was accomplished 34,754.29 mg/kg (Table 3) by determining the mean value to Standard Reference Material (SRM 1515) (apple leaf) and Certified Mass Fraction Values for Elements in SRM 1573a (tomato leaf) (Fig. 4). Spinacia oleracea has the highest concentrations of scandium (25,461 ± 5.33 mg/kg). However, the concentration of Abelmoschus esculentus (L.) is the lowest of absorption (1559 ± 2.82 mg/kg) (Table 2), while some of its compounds may be carcinogenic. This can cause pulmonary embolism, especially when exposed over a long period. When scandium builds up in the human body [29], it can threaten the liver. Ipomoea aquatica has elevated levels of titanium (1647 ± 4.12 mg/kg), while Basella alba and Raphanus sativus levels are not detectable. The human body contains measurable amounts of titanium (Table 2), and it is estimated that we breathe in about 0.8 mg/day [30]. Still, the majority of it passes through us without being absorbed. Titanium is not a toxic metal, and the human body can withstand massive amounts of it. In a comparison of Standard Reference Content (SRM 1515) (apple leaf) and Approved Mass Fraction Values for Elements in SRM 1573a (tomato leaf), the greatest proportion of vanadium element was found 523 ± 3.84 mg/kg in Amaranthus dubius, and the cheapest amount was found 313 ± 3.09 mg/kg in Abelmoschus esculentus (L.) (Fig. 3). Exposure to elevated levels of vanadium oxides in the breathing air of commercial vanadium mining enterprises is the primary indicator of possible adverse effects produced by vanadium.
Figure 4 indicates that the total concentration of Cr in vegetable specimens was 766 mg/kg, which is greater than the appropriate Certified Mass Fraction Values for Elements in SRM 1573a (tomato leaf). Chromium has been shown to improve glucose resistance in people with diabetes, older adults, and malnourished children [31]. Cr RDA for adults and children above the age of 4 is 50–200 g/day. This highest degree of Cr in Colocasia esculenta can be affected by the vegetable plots’ local soil and water situations. Oligovanadates occur at higher levels [32, 33], particularly by decavanadate thermodynamically stable at 2.3–6.3 pH, and can be further stabilized by contact with proteins at higher pH. The total permitted amounts of Mn are 47 mg/kg and 31.9 mg/kg (Table 3, Fig. 4), according to IAEA-V-10 and IAEA-359, respectively, which is similar to our human intake. The average concentration for Fe 3466.286 mg/kg is greater than all reference materials in Table 3. Heme iron is present in proteins that transport oxygen, such as hemoglobin and myoglobin [34, 35]. Non-heme iron is present in oxidative phosphorylation proteins and iron storage factors such as transferrin and ferritin. The reasonably high amounts of Co were 733 ± 5.43 mg/kg in Spinacia oleracea and 430 ± 3.57 mg/kg (Table 2) in Abelmoschus esculentus (L.), respectively. Cobalt is used in the body to assist in the synthesis and digestion of vitamin B12.
Furthermore, cobalt aids in the treatment of illnesses such as anemia and some infectious diseases. Cobalt also helps in myelin regeneration [36], which wraps and supports nerve cells. The mean value of registered samples for cobalt is 36,466 mg/kg (Table 3), which is greater than 0.577 mg/kg compared to Certified Mass Fraction Values for Elements in SRM 1573a (tomato leaf). The bulk of nickel in food is not absorbed from the intestine, and less than 10% of the nickel consumed is usually taken up in the food [37]. The maximum permitted Ni concentration is 0.5 mg/kg in vegetables and fruits. The value of Ni in Abelmoschus esculentus (L.) contained in the current review in 762 ± 3.03 mg/kg (Table 2) contravened the permissible Ni intake cap as set out in International Chemical Safety Program (IPCS) in its Health and Safety Guide No 62.
In comparison to Standard Reference Material (SRM 1515) (apple leaf) and Certified Mass Fraction Values for Elements in SRM 1573a (tomato leaf), the highest copper factor was found 3581 ± 4.42 mg/kg in Amaranthus dubius and the lowest quantity 222 ± 4.69 mg/kg in Spinacia oleracea. Copper is an important trace mineral that cannot be produced by the human body and must thus be obtained from nutritional sources on a day-to-day basis [38]. According to the World Health Organization, 1–3 mg of copper a day is needed to avoid any deficit symptoms. Various health and nutrition associations worldwide have developed nutritional reference values stressing the importance of copper as part of a safe diet. Amaranthus dubius contained 6746 ± 7.82 mg/kg of Zn, and Abelmoschus esculentus (L.) contained 1466 ± 10.19 mg/kg of Zn (Table 2, Fig. 2). According to IAEA-V-10, the actual allowable concentration of Zn is 24 mg/kg. The mean volume measured (744.2857 mg/kg) is greater than the recommended appropriate human consumption level of IAEA-359 (Table 3, Fig. 4) when this value is above IAEA-359’s healthy limits. Organic bromines are commonly used as insecticides and other poisons. They are, however, toxic not only to the animals against which they are used [39] but also to larger animals. In certain cases, they are also harmful to humans. The most severe health consequences that bromine-containing organic pollutants can induce are nervous system dysfunction and genetic material destruction. Almost all samples contained quantities ranging from 1981 ± 3.57 mg/kg in Amaranthus dubius to 22,060 ± 3.39 mg/kg in Ipomoea aquatica (Table 2).
The highest concentration of Sr in Raphanus sativus is 8701 ± 5.96 mg/kg, while the lowest concentration, 1437 ± 6.89 mg/kg, in Colocasia esculenta, which can kill cancer cells in the human body (detected mean value 5203.286 mg/kg). This strontium is not present in dietary supplements. There is some concern in using strontium to treat osteoarthritis since recent research indicates it may improve collagen production and cartilage in joints [40]. The systemic toxicity of zirconium and its salts is usually limited. The dietary consumption is estimated to be about 50 µg [41]. The majority of what enters the intestine is not absorbed, and what is adsorbed accumulates slightly more in the skeleton than in the bone. Except for Amaranthus dubius and Raphanus sativus, all other samples contain zirconium in concentrations ranging from 185 ± 4.00 mg/kg in Ipomoea aquatica to 3362 ± 4.24 mg/kg (Table 2) in Abelmoschus esculentus. For all sources of silver, the Occupational Safety and Health Administration and the Mine Safety and Health Administration prescribe an allowable exposure limit (PEL) of 0.01 mg/m3, and the National Institute for Occupational Safety and Health recommends a recommended exposure limit [42] of 0.01 mg/m3. The minimum silver concentration was determined 305 ± 7.00 mg/kg in Spinacia oleracea, and the strongest was found in Abelmoschus esculentus (L.) at 1285 ± 3.34 mg/kg. Fortunately, most individuals only come into contact with tellurium compounds on a limited occasion. They are teratogenic and can only be treated by qualified chemists [43] because even minimal concentrations produce foul-smelling breath and revolting poor hygiene. Normally, reference materials IAEA-V-10 (hay powder) and IAEA-359 (cabbage) (Table 3) are not recommended for Te element. Iodine deficiency affects various conditions that may have a long-term impact on your health, most notably goiter [44]. Any of these conditions present themselves in the mother’s womb before the baby is conceived. Raphanus sativus had the lowest iodine concentration of 131 ± 3.63 mg/kg, and Spinacia oleracea had the highest at 829 ± 4.69 mg/kg. Lead reaches the body mainly by inhalation or swallowing, where it is consumed, transmitted [45], and excreted directly. The gastrointestinal and respiratory tracts are the main sources of inorganic lead absorption. But for Ipomoea aquatica (172 ± 4.94 mg/kg), no other vegetables display Pb concentration (Fig. 3).
Conclusion
In this research section on trace elemental evaluation of specific segments of some vegetables using the PIXE technique, the findings identified 24 elements and compared them (mean value) to reference materials. They provided valuable data about the metal content of vegetables grown in the saline region of Rampal. As a potential preventive mechanism suitable for human well-being, trace mineral concentrations have also been proposed. The current outcomes justify using these vegetables because they contain large quantities of the elements K, Ca, Cr, Mn, Cu, and Zn, all of which contribute to insulin’s action. These vegetables also have toxic elements such as Cr, V, and Pb, which can be caused by fertilizers and metal-based pesticides in cultivation, increasing vulnerability to health problems. The present investigation demonstrates that these vegetables are not devoid of health risks in intake. So, the government should strictly monitor the environmental condition of this studied area. Further analysis is also required as the concentration level of vegetables was higher than that in reference materials.
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Fahad, S. M., et al. "Determination of the elemental composition of Malabar spinach, lettuce, spinach, hyacinth bean, and cauliflower vegetables using proton-induced X-ray emission technique at Savar subdistrict in Bangladesh." BioMed Res Int 2015 (2015). https://doi.org/10.1155/2015/128256.
Rahman MR et al (2014) Studies of essential and trace elements in some fruits and vegetables of southwestern Bangladesh by PIXE technique. Pak J Nutr 13(2):62–66
Beattie JK, Quoc TN (2000) Manganese in pineapple juices. Food Chem 68(1):37–39. https://doi.org/10.1016/S0308-8146(99)00149-1
Williams AB, Ayejuyo OO, Ogunyale AF (2009) Trace metal levels in fruit juices and carbonated beverages in Nigeria. Environ Monit Assess 156(1):303–306. https://doi.org/10.1007/s10661-008-0485-1
Fraga CG (2005) Relevance, essentiality and toxicity of trace elements in human health. Mol Aspects Med 26(4–5):235–244. https://doi.org/10.1016/j.mam.2005.07.013
Medaković, Davorin, et al. "Trace metals in fish biominerals as environmental indicators: handheld XRF analyses." Key Engineering Materials. Vol. 672. Trans Tech Publications Ltd, 2015. https://doi.org/10.4028/www.scientific.net/KEM.672.328.
Tuzen M (2009) Toxic and essential trace elemental contents in fish species from the Black Sea, Turkey. Food Chem Toxicol 47(8):1785–1790. https://doi.org/10.1016/j.fct.2009.04.029
Vickers NJ (2017) Animal communication: when I’m calling you, will you answer too? Curr Biol 27(14):R713–R715. https://doi.org/10.1016/j.cub.2017.05.064
Alexander ME et al (1974) Trace element analysis of seawater and fish samples by proton induced X-ray emission spectroscopy. Int J Appl Radiat Isot 25(5):229–233. https://doi.org/10.1016/0020-708X(74)90032-5
Pantelica A et al (2011) PIXE analysis of some vegetable species. Romanian Rep Phys 63(4):997–8
Winefordner, J D. Particle-induced X-ray emission spectrometry (PIXE). Vol. 184. John Wiley & Sons, 1995.
Zeisler, R, and Vincent P. G. Nuclear analytical methods in the life sciences. Springer Science & Business Media, 2012.
Taylor Jr, F. G., et al. "Environmental effects of chromium and zinc in cooling water drift." (1974).
Perry, H. M. "Hypertension and true geochemical environments in relation to health and diseases." (1972).
Arinola OG et al (2008) Status in Nigerian cassava processors. Pak J Nutr 7(6):770–772
Song, K, et al. "Determination of trace cobalt in fruit samples by resonance ionization mass spectrometry." Microchem J 75.2 (2003): 87–96. https://doi.org/10.1016/S0026-265X(03)00052-3.
Campbell JL et al (1986) An intercomparison of spectral data processing techniques in PIXE. Nucl Instrum Methods Phys Res, Sect B 14(2):204–220. https://doi.org/10.1016/0168-583X(86)90045-5
Bandhu HK et al (2000) Elemental composition and sources of air pollution in the city of Chandigarh, India, using EDXRF and PIXE techniques. Nucl Instrum Methods Phys Res, Sect B 160(1):126–138. https://doi.org/10.1016/S0168-583X(99)00574-1
Fahad SM et al (2014) Study of elemental profile of some medicinal plants of Bangladesh. J Nuc Particle Physics 4(1):1–6. https://doi.org/10.5923/j.jnpp.20140401.01
DeVoto E, Yokel RA (1994) The biological speciation and toxicokinetics of aluminum. Environ Health Perspect 102(11):940–951. https://doi.org/10.1289/ehp.94102940
Sharifi-Rad, Mehdi, et al. "Lifestyle, oxidative stress, and antioxidants: back and forth in the pathophysiology of chronic diseases." Front physiol 11 (2020): 694. https://doi.org/10.3389/fphys.2020.00694.
Siqwepu O, Salie K, Goosen N (2020) Evaluation of potassium diformate and potassium chloride in the diet of the African catfish, Clarias gariepinus in a recirculating aquaculture system. Aquaculture 526:735414. https://doi.org/10.1016/j.aquaculture.2020.735414
Soltani, Mahmoud, et al. "Modified atmosphere packaging: a progressive technology for shelf-life extension of fruits and vegetables." J Appl Packag Res 7.3 (2015): 2.
Inger-Lise et al S (2018) Benefit and risk assessment of increasing potassium intake by replacement of sodium chloride with potassium chloride in industrial food products in Norway. Food Chem Toxicol 111:329–340. https://doi.org/10.1016/j.fct.2017.11.044
EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). "Scientific opinion on dietary reference values for carbohydrates and dietary fibre." EFSA J 8.3 (2010): 1462.
Committee on Nutrition (1999) Calcium requirements of infants, children, and adolescents. Pediatrics 104(5):1152–1157
Heaney RP et al (1982) Calcium nutrition and bone health in the elderly. Am J Clin Nutr 36(5):986–1013. https://doi.org/10.1093/ajcn/36.5.986
George R, Walsh LJ (2010) Thermal effects from modified endodontic laser tips used in the apical third of root canals with erbium-doped yttrium aluminium garnet and erbium, chromium–doped yttrium scandium gallium garnet lasers. Photomed Laser Surg 28(2):161–165. https://doi.org/10.1089/pho.2008.2423
Balazic, Matej, et al. "Titanium and titanium alloy applications in medicine." Int J Nano Biomater 1.1 (2007): 3–34. https://doi.org/10.1504/IJNBM.2007.016517.
Anderson RA (1998) Chromium, glucose intolerance and diabetes. J Am Coll Nutr 17(6):548–555
Gundhla, Isaac Zvikomborero. A PH-metric speciation and anti-diabetic study of oxovanadium (IV) amino acid derivatives. Diss. Rhodes University, 2011.
Sánchez-Lara, Eduardo, et al. "Decavanadate salts of cytosine and metformin: a combined experimental-theoretical study of potential metallodrugs against diabetes and cancer." Front Chem 6 (2018): 402. https://doi.org/10.3389/fchem.2018.00402.
Zeng, Guangming, et al. "Spatial analysis of human health risk associated with ingesting manganese in Huangxing Town, Middle China." Chemosphere 77.3 (2009): 368–375. https://doi.org/10.1016/j.chemosphere.2009.07.020.
Abbaspour N, Hurrell R, Kelishadi R (2014) Review on iron and its importance for human health. J Res Med Sci 19(2):164
Marquez JE et al (2018) Effect of cadmium, copper and lead on the growth of rice in the coal mining region of Quang Ninh, Cam-Pha (Vietnam). Sustainability 10(6):1758. https://doi.org/10.3390/su10061758
Fratesi R et al (2002) Contemporary use of Ni and Bi in hot-dip galvanizing. Surf Coat Technol 157(1):34–39. https://doi.org/10.1016/S0257-8972(02)00137-8
Loland JØ, Singh BR (2004) Copper contamination of soil and vegetation in coffee orchards after long-term use of Cu fungicides. Nutr Cycl Agroecosyst 69(3):203–211. https://doi.org/10.1023/B:FRES.0000035175.74199.9a
Prasad AS (2008) Zinc in human health: effect of zinc on immune cells. Mol Med 14(5):353–357. https://doi.org/10.2119/2008-00033.Prasad
Singh N, Kumar D, Sahu AP (2007) Arsenic in the environment: effects on human health and possible prevention. J Environ Biol 28(2):359
David BNL et al (2010) Zirconium: biomedical and nephrological applications. ASAIO J 56(6):550–556. https://doi.org/10.1097/MAT.0b013e3181e73f20
Wang, Bin, et al. "A stable zirconium-based metal-organic framework for specific recognition of representative polychlorinated dibenzo-p-dioxin molecules." Nat commun 10.1 (2019): 1–8.
Panyala, Nagender Reddy, Eladia María Peña-Méndez, and Josef Havel. "Silver or silver nanoparticles: a hazardous threat to the environment and human health?" J Appl Biomed 6.3 (2008).
van der Reijden OL, Zimmermann MB, Galetti V (2017) Iodine in dairy milk: sources, concentrations and importance to human health. Best Pract Res Clin Endocrinol Metab 31(4):385–395
Tsuji LJS et al (2009) Determining tissue-lead levels in large game mammals harvested with lead bullets: human health concerns. Bull Environ Contam Toxicol 82(4):435–439. https://doi.org/10.1007/s00128-009-9647-2
Ravisankar R et al (2006) Determination and distribution of rare earth elements in beach rock samples using instrumental neutron activation analysis (INAA). Nucl Instrum Methods Phys Res, Sect B 251(2):496–500. https://doi.org/10.1016/j.nimb.2006.07.021
Gowrishankar R, Kumar M, Menon V et al (2010) Trace element studies on Tinospora cordifolia (Menispermaceae), Ocimum sanctum (Lamiaceae), Moringa oleifera (Moringaceae), and Phyllanthus niruri (Euphorbiaceae) using PIXE. Biol Trace Elem Res 133:357–363. https://doi.org/10.1007/s12011-009-8439-1
PRITHIVIRAJ B et al (2011) Elemental accumulation patterns of the lichen species Physcia tribacoides Nyl., Heterodermia dissecta and Bacidia beckhausii Körber from the Walayar Rf Region, Tamil Nadu. India. International Journal of PIXE 21(03n04):133–144. https://doi.org/10.1142/S0129083511002252
Ravisankar R et al (2007) Gamma-ray spectroscopic and PIXE analysis of beach rock samples of south east coast of Tamilnadu, India. Int J PIXE 17(03n04):193–203. https://doi.org/10.1142/S0129083507001253
Manikandan E et al (2011) 2 MeV-PIXE Technique for coastal material analysis. Int J of PIXE 21(03n04):75–86. https://doi.org/10.1142/S0129083511002215
Elayaperumal M, Vedachalam Y, Loganathan D, Kumaravelu TA, Anusuya GS, Kennedy J (2021) Ion beam analysis of proton-induced X-ray emission (PIXE) techniques for elemental investigation of young stage neem leaf of southern India, Tamil Nadu. Biol Trace Elem Res 199(9):3540–3546. https://doi.org/10.1007/s12011-020-02443-x
Acknowledgements
The authors are very grateful for the Dhaka Atomic Energy Center (AECD) research done by Bangladesh.
Author information
Authors and Affiliations
Contributions
Shirin Akter, designed and performed experiments; SM Fahad, sample collection, analyzed data, wrote the paper; Shah Sultan Ashrafi, statistical analysis and drew all figures; MJ Abedin, supervised the research, performed experiments; YN Jolly co-wrote the paper; MJ Kabir, designed experiments, M Safiur Rahman, analyzed data; Bilkis A Begum, performed experiments, KM Mamun, designed and performed experiments, Md. Hazrat Ali, checked and designed the paper.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akter, S., Fahad, S.M., Ashrafi, S.S. et al. Elemental Analysis of Basella alba, Spinacia oleracea, Abelmoschus esculentus (L.), Ipomoea aquatica, Colocasia esculenta, Amaranthus dubius, and Raphanus sativus Vegetables Using the PIXE Technique in a Saline Region of Bangladesh, Rampal Area. Biol Trace Elem Res 200, 2999–3008 (2022). https://doi.org/10.1007/s12011-021-02866-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02866-0