Abstract
Selenium levels can regulate the function of T cells, macrophages, B cells, natural killer cells and other immune cells. However, the effect of selenium on the immune function of dendritic cells (DCs) isolated from selenium-supplemented mice is unknown. In this study, C57BL/6J mice were randomly divided into three groups and fed diets containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium levels for 8 weeks. Immature (imDCs) and mature (mDCs) dendritic cells were then isolated from the bone marrow. Next, the migration, phagocytic capacity and mixed lymphocyte reaction (MLR) for imDCs and mDCs were detected by transwell and flow cytometry. The levels of C-C chemokine receptor type 7 (CCR7), major histocompatibility complex II (MHCII) and reactive oxygen species (ROS) were assayed by flow cytometry. F-actin and superoxide dismutase (SOD) activity was detected by fluorescence microscopy and SOD assay kit, respectively. In addition, the extracellular signal-regulated kinase (ERK), Akt, Ras homolog gene family member A/Rho-associated protein kinase (RhoA/ROCK) signalling, selenoprotein K (SELENOK) and glutathione peroxidase 1 (GPX1) levels were measured by western blot analysis. The results indicated that selenium deficiency enhanced the migration of imDCs by ROS and SELENOK-mediated ERK, Akt and RhoA/ROCK pathways but impaired the antigen uptake of imDCs. Although a high selenium level inhibited the migration of imDCs, it had no effect on phagocytic capacity. For mDCs, low selenium levels impaired free migration, and high levels inhibited the chemotactic migration involved in F-actin and CCR7, respectively. Low and high selenium levels impaired the MLR by inhibiting MHCII surface localisation, which might be related to ROS- and SELENOK-mediated ERK, Akt and RhoA/ROCK signalling pathways. In summary, selenium may regulate the immune function of mouse DCs through the ROS- and SELENOK-mediated ERK, Akt and RhoA/ROCK signalling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dendritic cells (DCs) can be divided into plasmacytoid DCs, conventional DCs, monocyte-derived DCs (MoDCs) and Langerhans cells and play an important role in the innate and adaptive immune response [1, 2]. Functionally, DCs include immature (imDCs) and mature (mDCs) subsets. DCs are present in almost all tissues, including cancerous tissue, in which their function may be inhibited. As a result, DC vaccines for cancer immunotherapy have shown disappointing results because of the disruption of robust antigen presentation and migration [3,4,5]. Previous studies have demonstrated that selenium is an essential trace element that performs important functions in innate and adaptive immune cells [6,7,8,9]. Selenium supplementation can enhance T cell proliferation, natural killer cell activity and lymphocyte function in humans [6, 9]. The levels of selenium can also influence the differentiation of T cells and macrophages, the numbers of B cells and antibody production in mice [8]. Recently, it was reported that the differentiation and immune function of chicken DCs can be regulated by selenium [10, 11]. However, the immune function of DCs from selenium-supplemented mice and the detailed underlying mechanism remain unclear.
Numerous studies have suggested that redox signalling, including reactive oxygen species (ROS) and glutathione (GSH), is essential for the activation and function of immune cells. Selenium levels can regulate the production of ROS, which is the primary mechanism of selenium’s effect on the immune system [6, 8, 12]. Studies have shown that oxidative stress can regulate DC maturation and cytokine production under conditions of stress [13, 14] and ROS can promote the maturation and antigen presentation of DCs [15,16,17]. Intracellular GSH in human DCs can also regulate the production of interleukin (IL)-27 and T cell polarisation [18]. It was also reported that selenium deficiency can promote oxidative stress-induced immune damage in chicken spleens [19].
A previous study demonstrated that different stimuli can induce the maturation of human MoDCs through the mitogen-activated protein kinase (MAPK) pathway, including extracellular signal-regulated kinase (ERK) signalling, which can regulate antigen uptake and DCs migration [20]. The Akt signalling pathway also plays an important role in the function of DCs [21]; however, it is unclear whether the function of Akt in DCs is influenced by selenium levels. Recently, Bros et al. suggested that the migration of DCs can be regulated by the Ras homolog gene family member A/Rho-associated protein kinases (RhoA/ROCK) signalling and that the interaction between DCs and T cells was influenced by RhoA [22]. Taken together, the role of the ERK, Akt and RhoA/ROCK pathways in the function of mouse DCs regulated by selenium is unclear.
The immunologic function of selenoproteins and the biological effect of selenium in T cells, B cells and macrophages have been thoroughly studied. The immune-related selenoproteins include selenoprotein K (SELENOK), selenoprotein R, selenoprotein S and selenoprotein 15 [6,7,8,9]. SELENOK is the best example indicating that immune function, such as T cell migration and macrophage phagocytosis, can be regulated by calcium flux and redox signalling. SELENOK can regulate the immune function through post-translational modifications of proteins as a non-enzymatic selenoprotein [7, 23]. In addition, SELENOK may also contribute to the development of cancer [23] and cancer therapy in the context of DC activity [4, 5]. However, the immune function of SELENOK in mouse DCs remains unknown.
In this study, we evaluated the immune function and redox balance of DCs from selenium-supplemented mice. imDCs and mDCs were prepared from mice with low, medium and high selenium diets, and the migration and phagocytic capacity of imDCs and the migration ability and mixed lymphocyte reaction (MLR) for mDCs were assessed. In addition, ROS levels, superoxide dismutase (SOD) activity and glutathione peroxidase 1 (GPX1) levels were determined. Finally, the potential underlying mechanisms were evaluated, including F-actin levels and SELENOK, as well as the ERK, Akt and RhoA/ROCK signalling pathways.
Materials and Methods
Preparation of Mice and Sample Collection
Animal protocols were approved by the Institutional Animal Care and Use Committee at the Guizhou Medical University. Male C57BL/6J mice (3 weeks old) were obtained and housed in the Guizhou Laboratory Animal Engineering Technology Center (Experimental Facilities certification: SCXK (qian) 2018-0001). After 1 week of standard diets containing 0.15 ppm selenium (Jiangsu Xietong Organism, China), the mice were randomly divided into three groups. They were subsequently provided diets (Beijing Keao Xieli Feed Co., Ltd.) containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium levels for 8 weeks, as previously described [8, 24]. The diets were formulated with purified ingredients (Supplemental Table 1), and sodium selenite was added to achieve the targeted selenium levels. The mice were anaesthetised and sacrificed after 8 weeks of feeding, and the femurs were collected for the preparation of DCs.
Preparation of DCs from Mouse Bone Marrow
DCs were prepared from mouse bone marrow according to a previously described method [25]. Briefly, the femoral bone marrow suspensions (with red cells discarded) isolated from mice fed with low, medium and high selenium diets were cultured in RPMI 1640 medium containing 10% foetal bovine serum (HyClone, USA), 20 ng/mL recombinant mouse granulocyte-macrophage CSF (rmGM-CSF) and 10 ng/mL recombinant mouse interleukin-4 (rmIL-4) (PeproTech, Germany) in 6-well plates at a density of 2 × 106 cells/mL. Half of the medium was replaced once every 2 days, and the imDCs were obtained on day 7 of the culture process. The imDCs were transformed into mDCs by adding 100 ng/mL lipopolysaccharides (LPS) (Sigma-Aldrich, Germany) for 2 additional days.
Migration Ability of imDCs and mDCs
The imDCs and mDCs, prepared from mice fed with low, medium and high selenium diets, were seeded into the upper compartment of Transwell chambers (5 μm, Millipore), which contained serum-free RPMI 1640 medium. For free migration of imDCs and mDCs, the lower chamber contained complete RPMI 1640 medium. To induce chemotactic migration of mDCs, 100 ng/mL of chemokine (C-C motif) ligand 19 (PeproTech, Germany) was added to the lower chamber. The cells were incubated at 37 °C for 36 h, and the cell numbers in the upper and lower chambers were counted separately with a cell counter (Cellometer Mini, Nexcelom, USA). The cell migration rate was analysed and normalised to that in the low selenium diet group [26].
Phagocytic Capacity of imDCs
The phagocytic capacity of imDCs prepared from mice fed with low, medium and high selenium diets was determined by flow cytometry. Briefly, imDCs were resuspended and cultured with FITC-dextran (40KD, Sigma-Aldrich) in phosphate-buffered saline (PBS), followed by fixation with 3.7% paraformaldehyde. After washing with PBS, the cells were resuspended at a density of 1 × 106 cells/mL in PBS and analysed by flow cytometry (NovoCyte, ACEA Biosciences). The fluorescence intensity was analysed and normalised to that in the low selenium diet group [25].
Mixed Lymphocyte Reaction
The mouse T cells were isolated from the spleen homogenate of wild mice and separated using a Nylon Wool Fiber Syringe (Gamma Irradiated; Polysciences, England). The T cells were then incubated at 37 °C for 2 h and co-cultured with mDCs isolated from mice fed with low, medium and high selenium diets for 48 h. The ratios of mDCs to T cells were 1:1, 1:10 and 1:100. Subsequently, T cell proliferation was assayed using the CCK8 assay according to the manufacturer’s instructions [26].
C-C Chemokine Receptor Type 7 and Major Histocompatibility Complex II Detection
PE anti-mouse CCR7 and MHCII antibodies were obtained from BD Pharmingen. The mDCs were collected and washed twice with PBS, followed by fixation with 4% paraformaldehyde. After washing with PBS, the cells were incubated with fluorescent antibodies at the recommended concentration at 4 °C for 30 min. The cells were then washed twice with ice-cold PBS, resuspended in 300 μL PBS at a density of 1 × 106 cells/mL and analysed using flow cytometry [25].
F-Actin Observation
Mouse DCs were collected and seeded into 6-well plates containing 0.1 mg/mL polylysine-coated coverslips. After fixation with 4% paraformaldehyde at 37 °C for 30 min, the cells were permeabilised with 0.1% Triton X-100 in PBS for 30 min. The coverslips were then washed twice with PBS and blocked in 1% BSA for 30 min. F-actin was stained with FITC-Phalloidin (Invitrogen, USA) at 4 °C overnight. The cells were then washed and stained with DAPI (Invitrogen, USA) in a fluorescent quencher. The plates were imaged with a fluorescence microscope (Olympus, Japan), and the average optical (AO) data were analysed using Image J software (National Institutes of Health) [27].
Measurement of ROS
ROS content in mouse DCs was measured using a flow cytometry kit according to the manufacturer’s protocol. Briefly, DCs were collected and incubated with 2′,7′-dichlorodihydrofluorescein diacetate (Solarbio, China) for 20 min at 37 °C. After washing twice with PBS, the cells were resuspended in PBS at a density of 1 × 106 cells/mL, and ROS levels were determined by flow cytometry [28].
SOD Activity
The SOD activity of DCs was determined using a total SOD assay kit with WST-8 (Beyotime, China), according to the manufacturer’s protocol. Briefly, the cells were washed twice with cold PBS and then homogenised in ice-cold PBS. After incubation with WST-8 at 37 °C for 30 min, the absorbance of the homogenate supernatant at 450 nm was measured using a microplate reader (Cytation 5, BioTek).
Western Blot Analysis
Cell lysates were prepared in RIPA buffer containing a protease inhibitor cocktail, and western blot analysis was performed as described by Jia et al. [27]. The antibodies included SELENOK (ab139949) and GPX1 (ab108427) purchased from Abcam; ERK (AF0155), pERK (AF1015), Akt (AF6261), pAkt (AF0016) and GAPDH (AF0911) obtained from Affinity; and RhoA (2117) and ROCK (4035), which were acquired from Cell Signalling Technology.
Statistical Analysis
All data are presented as the mean ± SD. Analysis of variance was used to determine the statistically significant differences between groups, followed by Tukey’s test for multiple comparisons. P values less than 0.05 were considered statistically significant.
Results
Selenium Can Regulate the Immune Function of imDCs
To verify the effect of selenium on DCs in vivo, we performed experiments on the separation and induction of DCs from the bone marrow of mice provided with low, medium and high selenium-containing diets for 8 weeks. First, immune function was investigated in imDCs, including migration and phagocytic capacity. The results indicated that the free migration ability of imDCs gradually decreased with increased selenium concentration (P < 0.05) (Fig. 1a). Compared with the medium selenium diet group, low selenium levels increased the migration of imDCs (P < 0.001), whereas high selenium levels inhibited the migration ability of imDCs (P < 0.05). The phagocytic capacity of imDCs was significantly inhibited in the low selenium diet group compared with the medium and high selenium diet groups (P < 0.001), whereas there was no difference between the medium and high groups (Fig. 1b and c).
Effect of selenium on the immune function of imDCs. Migration ability (a) and phagocytic capacity (b, c) of imDCs isolated from mice fed with diets containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium for 8 weeks. Data are presented as the mean ± SD (n = 3). *P < 0.05, ***P < 0.001, compared with Medium; ###P < 0.001, compared with low; low, medium and high respectively represent DCs isolated from mice fed with different concentrations of selenium
Selenium Can Regulate the Immune Function of mDCs
Next, we assessed the migration and MLR of mDCs prepared from mice fed with low, medium and high selenium diets. The results indicated that low selenium inhibited the free migration of mDCs (P < 0.01), but had no effect on chemotactic migration (Fig. 2a). In contrast, high selenium inhibited the chemotactic migration ability of mDCs (P < 0.01), but had no effect on free migration. The chemotactic migration ability of mDCs was then verified by measuring the expression of CCR7 by flow cytometry. The results (Fig. 2b) indicated that low selenium had no effect on CCR7 expression, whereas high selenium significantly inhibited the expression of CCR7 (P < 0.05), consistent with the results of chemotactic migration. The results for the MLR of mDCs showed that it was significantly inhibited in the low and high selenium diet groups compared with the medium selenium diet group (P < 0.05) (Fig. 2c). This result was consistent with the MHCII expression levels (Fig. 2d and e).
Effect of selenium on the immune function of mDCs. Free and chemotactic migration ability (a), CCR7 levels (b), mixed lymphocyte reaction (c) and MHCII levels (d, e) of mDCs isolated from mice fed with diets containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium for 8 weeks. Data are presented as the mean ± SD (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001, compared with medium; #P < 0.05, compared with low; low, medium and high respectively represent DCs isolated from mice fed with different concentrations of selenium
Redox Balance in DCs Was Regulated by Selenium
We also studied the potential role of redox balance when immune function was altered by selenium. Our results indicated that ROS levels in imDCs gradually decreased with increased concentrations of selenium (P < 0.001) (Fig. 3a and b). ROS levels in mDCs were significantly decreased in the low and high selenium diet groups compared with the medium selenium diet group (P < 0.05), and there was no difference between the low and high selenium diet groups (Fig. 3c and d). The SOD activity in imDCs and mDCs underwent similar changes (Fig. 3e and f). SOD activity was significantly increased in the medium and high selenium diet groups compared with the low selenium diet group (P < 0.05). GPX1 levels in imDCs and mDCs gradually increased with increased selenium concentration (P < 0.01) (Fig. 3g, h and i).
Effect of selenium on the ROS levels in imDCs (a, b) and mDCs (c, d), SOD activity in imDCs (e) and mDCs (f) and GPX1 levels in imDCs and mDCs (g, h, i) isolated from mice fed with diets containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium for 8 weeks. Data are presented as the mean ± SD (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001, compared with medium; #P < 0.05, ##P < 0.01, ###P < 0.001, compared with low; low, medium and high respectively represent DCs isolated from mice fed with different concentrations of selenium
Changes in Selenium-Induced Migration Capacity Were Related to F-Actin
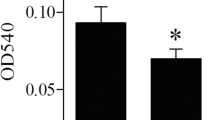
The F-actin levels related to migration ability are shown in Fig. 4. Our results indicated that the F-actin level in imDCs was increased in the low selenium diet group compared with the medium and high selenium diet groups (P < 0.05) and that there was no difference between the medium and high selenium diet groups. The levels in mDCs were significantly increased in the low and high selenium diet groups compared with the medium selenium diet group (P < 0.01). These results indicated that high levels of F-actin were associated with high mobility in imDCs and low levels of F-actin were conducive to mDCs migration.
Effect of selenium on the F-actin levels in imDCs (a, b) and mDCs (a, c) isolated from mice fed with diets containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium for 8 weeks. Data are presented as mean ± SD (n = 3). *P < 0.05, **P < 0.01, compared with medium; #P < 0.05, compared with low; low, medium and high respectively represent DCs isolated from mice fed with different concentrations of selenium
ERK, Akt and RhoA/ROCK May Be Involved in the Immune Function of DCs by Selenium
The signalling pathways affected by selenium were examined. The results indicated that the ERK, Akt and RhoA/ROCK pathways were influenced in the low, medium and high selenium diet groups. In imDCs, the protein levels of SELENOK, pERK, pAkt, RhoA and ROCK gradually decreased (P < 0.05) (Fig. 5a and b). In mDCs, the levels of SELENOK, RhoA and ROCK in the medium group were significantly increased (P < 0.05) (Fig. 5c and d). Compared with the low and high selenium diet groups, the levels of pERK and pAkt in the medium selenium diet group were significantly decreased (P < 0.01).
Effect of selenium on the ERK, Akt and RhoA/ROCK signalling pathway activity and SELENOK protein levels in imDCs (a, b) and mDCs (c, d) isolated from mice fed with diets containing low (0.08 ppm), medium (0.25 ppm) or high (1 ppm) selenium for 8 weeks. Data are presented as mean ± SD (n = 3). *P < 0.05; **P < 0.01; ***P < 0.001; low, medium and high respectively represent DCs isolated from mice fed with different concentrations of selenium
Discussion
Numerous studies have found that selenium levels can regulate the function of immune cells [6,7,8, 29], such as the differentiation of T cells and macrophages, the numbers of B cells and antibody production in mice and natural killer cell activity and lymphocyte function in humans. Studies have also demonstrated that selenium levels can regulate the differentiation and immune function of chicken DCs in vivo [10, 11] and DC-stimulated T cell proliferation can be inhibited by high levels of ebselen in vitro [17]. We evaluated the role of selenium in human DCs in vitro (unpublished); however, the role of selenium in mouse DCs in vivo was unclear. Therefore, in the present study, we investigated the immune function of DCs isolated from mice provided with a low, medium and high selenium diet. It was suggested that selenium deficiency (low selenium) enhances imDCs migration but the antigen phagocytosis ability was significantly inhibited. Conversely, high selenium inhibited the migration ability of imDCs with no effect on phagocytic capacity (Fig. 1). For mDCs, high selenium inhibited the chemotactic migration ability by down-regulating CCR7 expression, and both low and high selenium impaired the MLR by down-regulating MHCII expression (Fig. 2). The results indicated that both low and high selenium levels were detrimental to the immune function of DCs. Blood inflammatory cytokines and spleen proteomics in mice also showed that the immune function is indeed affected by selenium (data not shown). Previous studies have shown that selenium deficiency can impair the innate and adaptive immune responses, including DCs in chickens [6,7,8,9, 11, 29]. In our study, selenium also impaired antigen phagocytosis and MLR of mouse DCs. Consistent with the findings of Matsue et al. [17], higher selenium levels could impair the proliferation of T cells and reduce the antigen uptake and presentation efficiency of DCs by inhibiting the free migration of imDCs and chemotactic migration of mDCs.
Redox balance, such as ROS levels, is known to be important in the activation and function of immune cells [6,7,8,9, 12,13,14,15,16,17, 30, 31]. We evaluated the ROS levels in DCs isolated from mice provided with low, medium and high selenium diets. The results indicated that selenium deficiency increased ROS content in imDCs because of lower SOD activity and GPX1 protein levels or activity [29] compared with mice fed a medium selenium diet. Higher selenium levels can reduce ROS levels in mDCs without a change in the SOD activity. This result may be related to increased protein levels or activity of GPX1 [29] compared with mice in the medium selenium group. It has been demonstrated that ROS can promote the maturation and antigen presentation of DCs [15,16,17]. With respect to our finding, it appears that appropriate levels of ROS are important for the immune function of DCs. Our study also revealed that reduced antigen uptake due to selenium deficiency was associated with higher ROS levels in imDCs. The damage to MLR caused by both selenium deficiency and high selenium levels was related to lower ROS content in mDCs.
Studies have shown that F-actin is involved in cell migration [32,33,34,35,36]. Specifically, F-actin levels were shown to decrease when monocytes differentiated into imDCs and mDCs [36]. In our study, the enhanced migration ability of imDCs induced by selenium deficiency was associated with increased F-actin levels compared with that in the medium selenium group (Figs. 1 and 4). Also, the increase of mDCs migration ability caused by medium selenium levels was associated with decreased F-actin levels compared with the low and high selenium groups (Figs. 2 and 4). Additionally, it has been demonstrated that the migration of DCs can be regulated by RhoA/ROCK signalling [22]. This study also revealed that the effect of selenium on the migration ability of DCs was related to the RhoA/ROCK signalling pathway. The gradual decrease in the migration ability of imDCs in the low, medium and high selenium groups may be the result of decreased RhoA and ROCK expression (Figs. 1 and 5). Furthermore, the decreased migration ability of mDCs in the low and high selenium groups was also related to decreased RhoA and ROCK levels (Figs. 2 and 5). Reports have also suggested that the interaction between DCs and T cells is influenced by RhoA and that RhoA can regulate the MHCII surface localisation in DCs [22, 37, 38]. In our study, T cell activation was inhibited by a RhoA-induced reduction in MHCII surface localisation in the low and high selenium groups (Figs. 2 and 5). Taken together, selenium may regulate the immune function of DCs through the RhoA/ROCK pathway.
The ERK pathway is also involved in the maturation of human DCs by different stimuli and, to some degree, exhibits a negative regulatory effect on the maturation of DCs [17, 39,40,41]. Similarly, our results showed that increased ERK phosphorylation induced by selenium deficiency led to decreased antigen uptake in imDCs, and decreased ERK phosphorylation levels induced by medium selenium levels resulted in increased CCR7, MHCII and MLR in mDCs (Figs. 1, 2 and 5). ERK activation may promote the migration of imDCs [42, 43] but inhibit the migration of mDCs [44, 45]. Furthermore, the Akt pathway is also involved in the migration and maturation of DCs under different stimulation conditions [21, 46, 47]. In our study, Akt activation induced by selenium was consistent with ERK phosphorylation; however, additional studies are needed to confirm the role of the ERK and Akt signalling pathways in the selenium-induced differentiation and maturation of DCs.
Because of the important function of selenoproteins in the immune system [6,7,8,9, 23], we determined SELENOK levels in DCs isolated from mice provided with low, medium and high selenium diets. The results indicated that the expression level of SELENOK was associated with the migration of DCs. The overexpression of SELENOK enhanced the migration ability of microglia cells [48], whereas SELENOK knockout resulted in decreased migration of T cells and neutrophils [24]. In addition, SELENOK also mediated the migration of choriocarcinoma cells [49] and BGC 823 cells [50]. These results suggest that SELENOK is related to the migration of DCs. Previous studies have shown that SELENOK-dependent palmitoylation of Arf-GAP with SH3 domain, ANK repeat and PH domain-containing protein 2 (ASAP2) was required for FcγR-mediated phagocytosis of macrophages [51], and the ERK and Akt pathways were involved in SELENOK-mediated cell migration [49]. We hypothesise that SELENOK may have a protein-modifying effect in DCs similar to that of macrophages. In addition, SELENOK may regulate the immune function of DCs through the ERK, Akt and RhoA/ROCK signalling pathways.
In this study, we investigated the immune function of DCs isolated from mice fed with a low, medium or high selenium diet (summarised in Fig. 6). The results indicated that selenium deficiency enhances the migration of imDCs by ROS- and SELENOK-mediated ERK, Akt and RhoA/ROCK pathways but inhibits antigen uptake by imDCs. Although a high selenium level inhibited the migration of imDCs, it had no effect on phagocytic capacity. For mDCs, low selenium levels impaired free migration, and high selenium levels inhibited chemotactic migration associated with F-actin and CCR7, respectively. Low and high selenium levels impaired MLR by inhibiting MHCII surface localisation, which may be associated with ROS- and SELENOK-mediated ERK, Akt and RhoA/ROCK signalling pathways. In summary, selenium may regulate the immune function of mouse DCs through the ROS- and SELENOK-mediated ERK, Akt and RhoA/ROCK signalling pathways.
Abbreviations
- AO:
-
Average optical
- CCR7:
-
C-C chemokine receptor type 7
- DCs:
-
Dendritic cells
- ERK:
-
Extracellular signal-regulated kinase
- GPX1:
-
Glutathione peroxidase 1
- GSH:
-
Glutathione
- IL:
-
Interleukin
- imDCs:
-
Immature dendritic cells
- LPS:
-
Lipopolysaccharides
- MAPK:
-
Mitogen-activated protein kinases
- mDCs:
-
Mature dendritic cells
- MHCII:
-
Major histocompatibility complex II
- MLR:
-
Mixed lymphocyte reaction
- MoDCs:
-
Monocyte-derived DCs
- PBS:
-
Phosphate-buffered saline
- RhoA:
-
Ras homolog gene family member A
- ROCK:
-
Rho-associated protein kinases
- ROS:
-
Reactive oxygen species
- rmGM-CSF:
-
Recombinant mouse granulocyte-macrophage CSF
- rmIL-4:
-
Recombinant mouse interleukin-4
- SELENOK:
-
Selenoprotein K
- SOD:
-
Superoxide dismutase
References
Eisenbarth SC (2019) Dendritic cell subsets in T cell programming: location dictates function. Nat Rev Immunol 19(2):89–103
Collin M, Bigley V (2018) Human dendritic cell subsets: an update. Immunology 154(1):3–20
Segura E, Amigorena S (2015) Cross-presentation in mouse and human dendritic cells. Adv Immunol. Academic Press 127:1–31
Perez CR, De Palma M (2019) Engineering dendritic cell vaccines to improve cancer immunotherapy. Nat Commun 10(1):1–10
Bryant CE, Sutherland S, Kong B, Papadimitrious MS, Hart DNJ, Fromm PD (2019) Dendritic cells as cancer therapeutics. Semin Cell Dev Biol 86:77–88
Rayman MP (2012) Selenium and human health. Lancet 79(9822):1256–1268
Avery JC, Hoffmann PR (2018) Selenium, selenoproteins, and immunity. Nutrients 10(9):E1203
Huang Z, Rose AH, Hoffmann PR (2012) The role of selenium in inflammation and immunity: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal 16(7):705–743
Hawkes WC, Kelley DS, Taylor PC (2001) The effects of dietary selenium on the immune system in healthy men. Biol Trace Elem Res 81:189–213
Sun Z, Liu C, Pan T, Yao H, Li S (2017) Selenium accelerates chicken dendritic cells differentiation and affects selenoproteins expression. Dev Comp Immunol 77:30–37
Sun Z, Xu Z, Wang D, Yao H, Li S (2018) Selenium deficiency inhibits dendritic cells differentiation and immune function, imbalance the Th1/Th2 of dendritic cells. Metallomics 10(5):759–767
Gostner JM, Becker K, Fuchs D, Sucher R (2013) Redox regulation of the immune response. Redox Rep 18(3):88–94
Götz A, Ty MC, Rodriguez A (2019) Oxidative stress enhances dendritic cell responses to plasmodium falciparum. Immunohorizons 3(11):511–518
Kroening PR, Barnes TW, Pease L, Limper A, Kita H, Vassallo R (2008) Cigarette smoke-induced oxidative stress suppresses generation of dendritic cell IL-12 and IL-23 through ERK-dependent pathways. J Immunol 181(2):1536–1547
Zaccagnino P, Saltarella M, Maiorano S, Gaballo A, Santoro G, Nico B, Lorusso M, Del Prete A (2012) An active mitochondrial biogenesis occurs during dendritic cell differentiation. Int J Biochem Cell Biol 44(11):1962–1969
Kantengwa S, Jornot L, Devenoges C, Nicod LP (2003) Superoxide anions induce the maturation of human dendritic cells. Am J Respir Crit Care Med 167(3):431–437
Matsue H, Edelbaum D, Shalhevet D, Mizumoto N, Yang C, Mummert ME, Oeda J, Masayasu H, Takashima A (2003) Generation and function of reactive oxygen species in dendritic cells during antigen presentation. J Immunol 171(6):3010–3018
Kamide Y, Utsugi M, Dobashi K, Ono A, Ishizuka T, Hisada T, Koga Y, Uno K, Hamuro J, Mori M (2011) Intracellular glutathione redox status in human dendritic cells regulates IL-27 production and T-cell polarization. Allergy 66(9):1183–1192
Zhang Y, Liu Q, Yin H, Min Y, Li S (2020) Selenium deficiency causes immune damage by activating the DUSP1/NF-κB pathway and endoplasmic reticulum stress in chicken spleen. Food Funct 11:6467–6475
Nakahara T, Moroi Y, Uchi H, Furue M (2006) Differential role of MAPK signaling in human dendritic cell maturation and Th1/Th2 engagement. J Dermatol Sci 42(1):1–11
Kim JH, Kang TH, Noh KH, Kim SH, Lee YH, Kim KW, Bae HC, Ahn YH, Choi EY, Kim JS et al (2010) Enhancement of DC vaccine potency by activating the PI3K/AKT pathway with a small interfering RNA targeting PTEN. Immunol Lett 134(1):47–54
Bros M, Haas K, Moll L, Grabbe S (2019) RhoA as a key regulator of innate and adaptive immunity. Cells 8(7):733
Marciel MP, Hoffmann PR (2019) Molecular mechanisms by which selenoprotein K regulates immunity and cancer. Biol Trace Elem Res 192(7):60–68
Verma S, Hoffmann FKW, Kumar M, Huang Z, Roe K, Nguyen-Wu E, Hashimoto AS, Hoffmann PR (2011) Selenoprotein K knockout mice exhibit deficient calcium flux in immune cells and impaired immune responses. J Immunol 186(4):2127–2137
Zhang M, Tang H, Guo Z, An H, Zhu X, Song W, Guo J, Huang X, Chen T, Wang J, Cao X (2004) Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat Immunol 5(11):1124–1133
Zeng Z, Yao W, Xu X, Xu G, Long J, Wang X, Wen Z, Chien S (2009) Hepatocellular carcinoma cells deteriorate the biophysical properties of dendritic cells. Cell Biochem Biophys 55(1):33–43
Jia Y, Zhou J, Liu H, Huang K (2014) Effect of methionine sulfoxide reductase B1 (SelR) gene silencing on peroxynitrite-induced F-actin disruption in human lens epithelial cells. Biochem Biophys Res Commun 443(3):876–881
Jia Y, Li Y, Du S, Huang K (2012) Involvement of MsrB1 in the regulation of redox balance and inhibition of peroxynitrite-induced apoptosis in human lens epithelial cells. Exp Eye Res 100:7–16
Hoffmann FW, Hashimoto AC, Shafer LA, Dow S, Berry MJ, Hoffmann PR (2010) Dietary selenium modulates activation and differentiation of CD4+ T cells in mice through a mechanism involving cellular free thiols. J Nutr 140(6):1155–1161
Matos TJ, Duarte CB, Gonçalo M, Lopes MC (2005) Role of oxidative stress in ERK and p38 MAPK activation induced by the chemical sensitizer DNFB in a fetal skin dendritic cell line. Immunol Cell Biol 83(6):607–614
Mizuashi M, Ohtani T, Nakagawa S, Aiba S (2005) Redox imbalance induced by contact sensitizers triggers the maturation of dendritic cells. J Invest Dermatol 124(3):579–586
Pearce G, Audzevich T, Jessberger R (2011) SYK regulates B-cell migration by phosphorylation of the F-actin interacting protein SWAP-70. Blood 117(5):1574–1584
Tak H, Jang E, Kim SB, Park J, Suk J, Yoon YS, Ahn JK, Lee JH, Joe CO (2007) 14-3-3epsilon inhibits MK5-mediated cell migration by disrupting F-actin polymerization. Cell Signal 19(11):2379–2387
Wang LH, Xiang J, Yan M, Zhang Y, Zhao Y, Yue CF, Xu J, Zheng FM, Chen JN, Kang Z, Chen TS, Xing D, Liu Q (2010) The mitotic kinase Aurora-A induces mammary cell migration and breast cancer metastasis by activating the Cofilin-F-actin pathway. Cancer Res 70(22):9118–9128
Schachtner H, Weimershaus M, Stache V, Plewa N, Legler DF, Höpken UE, Maritzen T (2015) Loss of Gadkin affects dendritic cell migration in vitro. PLoS One 10(12):e0143883
Zeng Z, Liu X, Jiang Y, Wang G, Zhan J, Guo J, Yao W, Sun D, Ka W, Tang Y, Tang J, Wen Z, Chien S (2006) Biophysical studies on the differentiation of human CD14+ monocytes into dendritic cells. Cell Biochem Biophys 45(1):19–30
Ocana-Morgner C, Wahren C, Jessberger R (2009) SWAP-70 regulates RhoA/RhoB-dependent MHCII surface localization in dendritic cells. Blood 113(7):1474–1482
Seul HJ, Ahn YR, Song HM, Ha YJ, Lee JR (2012) Over-expression of a RhoA-specific guanine nucleotide exchange factor, p190RhoGEF, in mouse dendritic cells negatively regulates cellular responses to bacterial lipopolysaccharide. Mol Cell 34(2):159–164
Puig-Kröger A, Relloso M, Fernández-Capetillo O, Zubiaga A, Silva A, Bernabéu C, Corbí AL (2001) Extracellular signal-regulated protein kinase signaling pathway negatively regulates the phenotypic and functional maturation of monocyte-derived human dendritic cells. Blood 98(7):2175–2182
Nakagawa S, Ohtani T, Mizuashi M, Mollah ZU, Ito Y, Tagami H, Aiba S (2004) p38 mitogen-activated protein kinase mediates dual role of ultraviolet B radiation in induction of maturation and apoptosis of monocyte-derived dendritic cells. J Invest Dermatol 123(2):361–370
Aiba S, Manome H, Nakagawa S, Mollah ZU, Mizuashi M, Ohtani T, Yoshino Y, Tagami H (2003) p38 mitogen-activated protein kinase and extracellular signal-regulated kinases play distinct roles in the activation of dendritic cells by two representative haptens, NiCl2 and 2,4-dinitrochlorobenzene. J Invest Dermatol 120(3):390–399
Rajalingam K, Wunder C, Brinkmann V, Churin Y, Hekman M, Sievers C, Rapp UR, Rudel T (2005) Prohibitin is required for Ras-induced Raf–MEK–ERK activation and epithelial cell migration. Nat Cell Biol 7(8):837–843
Bąbolewska E, Pietrzak A, Brzezińska-Błaszczyk E (2014) Cathelicidin rCRAMP stimulates rat mast cells to generate cysteinyl leukotrienes, synthesize TNF and migrate: involvement of PLC/A2, PI3K and MAPK signaling pathways. Int Immunol 26(11):637–646
Katagiri A, Nakayama K, Rahman MT, Rahman M, Yeasmin S, Ishikawa M, Iida K, Nakayama N, Miyazaki K (2010) MEK inhibition suppresses cell invasion and migration in ovarian cancers with activation of ERK1/2. Exp Ther Med 1(4):591–596
Liao YC, Shih YW, Chao CH, Lee XY, Chiang TA (2009) Involvement of the ERK signaling pathway in fisetin reduces invasion and migration in the human lung cancer cell line A549. J Agric Food Chem 57(19):8933–8941
Filippi I, Morena E, Aldinucci C, Carraro F, Sozzani S, Naldini A (2014) Short-term hypoxia enhances the migratory capability of dendritic cell through HIF-1α and PI3K/Akt pathway. J Cell Physiol 229(12):2067–2076
Bhattacharyya S, Sen P, Wallet M, Long B, Baldwin AS Jr, Tisch R (2004) Immunoregulation of dendritic cells by IL-10 is mediated through suppression of the PI3K/Akt pathway and of IkappaB kinase activity. Blood 104(4):1100–1109
Meng XL, Chen CL, Liu YY, Su SJ, Gou JM, Huan FN, Wang D, Liu HS, Ben SB, Lu J (2019) Selenoprotein SELENOK enhances the migration and phagocytosis of microglial cells by increasing the cytosolic free Ca2+ level resulted from the up-regulation of IP3R. Neuroscience 406:38–49
Li M, Cheng W, Nie T, Lai H, Hu X, Luo J, Li F, Li H (2018) Selenoprotein K mediates the proliferation, migration, and invasion of human choriocarcinoma cells by negatively regulating human chorionic gonadotropin expression via ERK, p38 MAPK, and Akt signaling pathway. Biol Trace Elem Res 184(1):47–59
Ben SB, Peng B, Wang GC, Li C, Gu HF, Jiang H, Meng XL, Lee BJ, Chen CL (2015) Overexpression of selenoprotein SelK in BGC-823 cells inhibits cell adhesion and migration. Biochemistry (Mosc) 80(10):1344–1353
Norton RL, Fredericks GJ, Huang Z, Fay JD, Hoffmann FW, Hoffmann PR (2017) Selenoprotein K regulation of palmitoylation and calpain cleavage of ASAP2 is required for efficient FcγR-mediated phagocytosis. J Leukoc Biol 101(2):439–448
Funding
This research was funded by the National Natural Science Foundation of China (No. 21561006 and No. 21867007), Guizhou Provincial Natural Science Foundation (No. [2019]1258 and No. LH[2016]7372) and Opening fund of Hubei Key Laboratory of Bioinorganic Chemistry & Materia Medica (No. BCMM202002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Liangliang Zhang and Huan Xia contributed to the work equally and should be regarded as co-first authors
Electronic Supplementary Material
ESM 1
(DOCX 22 kb).
Rights and permissions
About this article
Cite this article
Zhang, L., Xia, H., Xia, K. et al. Selenium Regulation of the Immune Function of Dendritic Cells in Mice Through the ERK, Akt and RhoA/ROCK Pathways. Biol Trace Elem Res 199, 3360–3370 (2021). https://doi.org/10.1007/s12011-020-02449-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02449-5