Abstract
Cadmium is primarily utilized in the construction of particles known as quantum dots. Hepatotoxicity caused by microparticles of cadmium is very well known; however, toxicity of nanoparticles of cadmium is not well understood. The present study describes the toxicity of cadmium sulfide nanoparticles (CdSNPs) in the liver of rat. Adult Wistar rats were administered CdSNPs (10 mg/kg) on alternate days for 45 days. Serum enzymes (ALT, AST, ALP), biomarkers of lipid peroxidation (MDA, H2O2, and NO), and metallothionein concentration were determined. Histopathological and TEM observations were also made to record morphological changes. CdSNPs (10 mg/kg) induced significant changes in the structure and function of liver. Values of serum enzymes and reactive species increased significantly in rats treated with CdSNPs in comparison to CdS-treated rats. Histopathological observations showed extensive parenchymal degeneration. Ultrastructural studies exhibited proliferation of endoplasmic reticulum, microsomes, and lysosomes. It is concluded that NP-membrane interaction leads to the generation of reactive species that alter membrane integrity and induce oxidative stress. These events may activate cell death pathways in hepatocytes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recently engineered nanoparticles (ENPs) whose diameter is < 100 nm are now widely applied/used in electronics, chemical industry, environmental protection, biomedicine, and drug delivery systems. Exposure to these nanomaterials is likely to induce largely unknown toxicological effects in man/animals and environment. Inorganic nanoparticles include oxides of different metals viz. TiO2, ZnO, Al2O3, Fe3O4, CeO2 etc. Cadmium sulfide (CdS) and cadmium selenide (CdSe) are categorized as quantum dots (QDs). Due to their small size, they possess “substantial surface zone to volume” ratio that renders ENPs more biologically active [1, 2].
Nanoparticles can enter in the body through different routes viz. skin, lungs, and gastrointestinal tract [3,4,5]. For biomedical applications, they may be administered through intravenous, intradermal, and intraperitoneal routes [6, 7]. Toxicity of ENPs depends upon their size, shape, surface area, charge, persistence in the target tissue/system, depurination, and immune response from the host [8].
While metallic nanoparticles (MNPs) have been considered potentially important for drug delivery systems [9], cadmium is primarily utilized in the construction of particles known as quantum dots. These are semiconductor metalloid crystal structures of approximately 2–100 nm containing about 200–10,000 atoms [10, 11]. They have unique optical and electronic properties that give the particle a bright, highly stable, and “size tunable” florescence. Several reviews have described the application of quantum dots in cellular imaging, cancer detection, and treatment as radio and chemo-sensitizing agents and targeted drug delivery [10,11,12,13]. However, QDs contain substantial amount of cadmium in a highly reactive form.
Cadmium is known to cause hepatic and renal toxicity through oxidative stress, depletion of endogenous antioxidants, apoptosis, mitochondrial injury, and disturbance in intracellular calcium signaling [14,15,16,17]. Health risks, however, involved after the exposure to cadmium nanoparticles, by and large, remain unknown. The main objective of the present investigation was to determine the possible effects/mechanisms involved in liver injury caused by cadmium sulfide nanoparticles (CdSNPs) in rat. The selected parameters of this multiphase study include serum enzymes, lipid peroxidation, oxidative stress, and cadmium accumulation in liver, metallothionein induction, histopathological, and ultrastructural observations.
Materials and Methods
Reagents/Chemicals
Cadmium sulfide nanoparticles were purchased from a commercial supplier—M/S, Nanobeach, Delhi. Cadmium sulfide was supplied by Hi-Media (Mumbai). Thiobarbituric acid, 1,1,3′,3’tetramethoxy propane, bovine serum albumin, 5′,5′ dithiobis-2-nitrobenzoic acid, osmium tetraoxide, glutaraldehyde, and Epon 812 were purchased from Sigma Chemical Company (USA). Other chemicals/reagents were procured from Hi-media (Mumbai). Commercial kits for the estimation of serum enzymes were procured from M/S ARKRAY Health Care Pvt. Ltd., Mumbai.
Characterization of Nanoparticles
CdSNPs were purchased from commercial supplier M/S NanoBeach, New Delhi. The size, shape, and elemental analysis of CdSNPs were determined using standard method viz. transmission electron microscopy (TEM), field emission scanning electron microscope (FE-SEM), and energy-dispersive x-ray analysis (EDAX) facility at Sophisticated Analytical Instrument Center of Punjab University Chandigarh (India). Briefly, nanoparticles were dispersed in ethyl alcohol and ultrasonicated. A few milliliters of solution was put onto carbon-coated grid (400 mesh) and then dried at room temperature for analysis using TEM (Hitachi- H-7500) 120 KV equipped with detector (charged coupled device) and tungsten filament. For FE-SEM analysis, CdSNPs were dispersed in ethanol. After evaporation, samples were coated with gold and observed using FE-SEM with energy-dispersive X-ray analyzer (JEOL, Japan, Model- JSM 6100). Zeta potential and DLS (Malvern, Zetasizer Nano ZS90) of CdSNPs were performed at Indian Institute of technology, Roorkee, India.
Maintenance of Test Animals
Male Wistar rats (150 g ± 30 g) were procured from the animal facility of Jamia Hamdard University, Delhi. They were housed individually in polypropylene cages under standard laboratory conditions (room temperature—25 °C ± 5 °C and relative humidity—50% ± 10% and 12 h dark/light cycle) in the animal facility of Department of Zoology, Ch. Charan Singh University, Meerut. They were fed laboratory chow (Golden Feeds, Delhi) and tap water ad libitum.
Prior permission from Institutional Ethical Committee was sought before making these investigations.
Treatments
Rats were divided into three groups each containing five rats. Rats of group A were administered a predetermined sublethal dose of CdSNPs (10 mg/kg b.w.) mixed in saline by gavage on alternate days for 45 days as described by Rana et al. [18].
Similarly, rats of group B were administered a predetermined sublethal dose of CdS microparticles (10 mg/kg b.w) mixed in saline by gavage on alternate days for 45 days.
Rats of group C were administered saline only by gavage on alternate days for 45 days to serve as controls. No mortality occurred during these investigations. Record of their body weight gain or loss was maintained.
Liver Function Tests
Enzyme biomarkers of liver function were estimated following standard methods viz. aspartate transaminase (AST) and alanine transaminase (ALT) [19] and alkaline phosphatase (ALP) [20].
Bioaccumulation of Nanocadmium Concentration in Liver
Cadmium and nanocadmium concentration in liver were analyzed through atomic absorption spectrophotometry, (EC.Hyderabad, India). Small pieces (1 g) of liver from each rat were collected immediately after sacrifice and digested in 10 ml of concentrated nitric acid at 80°C for 16 h. After digestion, samples were diluted with double-distilled water to 100 ml. A 5-ml aliquot was analyzed for cadmium analysis using atomic absorption spectrophotometry. A hollow cathode lamp for cadmium was used, and absorption was recorded at 228.8 nm. Metallic cadmium (Hi-media) was used as the standard.
Cadmium Metallothionein (Cd-MT) Concentration
Cd-MT concentration in liver was determined through cadmium saturation method of Onosaka and Cherian [21]. Briefly, liver samples were perfused with saline, homogenized in 1.15% potassium chloride and centrifuged at 9000×g. The supernatant was mixed with Tris–HCl buffer (pH 8) and freshly prepared hemoglobin. Cd-MT (Sigma) was used as the standard. Finally, the supernatant was analyzed for Cd through atomic absorption spectrophotometry as suggested by Rana and Kumar [22].
Lipid Peroxidation
The formation of thiobarbituric acid reactive substances (TBARS) was measured following the method of Jordan and Schenkman [23]. 1-1-3-3-tetra methoxypropane was used as the standard. Absorbance was recorded at 532 nm. Proteins were analyzed following the method of Lowry et al. [24]. Bovine serum albumin (Sigma) was used as the standard.
Hydrogen Peroxide
The basic level of H2O2 in liver homogenates (5% w/v prepared in 0.25 M sucrose) was estimated by ferrithiocyanate method as described by Thurman et al. [25]. Presence of H2O2was measured at 480 nm using a spectrophotometer (Systronics, India).
Nitric Oxides
Greiss reagent was used to measure the nitric oxides in liver samples as described by Cortas and Wakid [26].
Reduced Glutathione
GSH in liver samples was measured using 5′-5′ dithiobis-2-nitrobenzoic acid (DTNB) as described by Ellman [27]. Absorbance was recorded at 412 nm using a spectrophotometer (Systronics, India).
Histopathology
Small pieces of liver (5 mm) were fixed in 10% neutral formaldehyde. After dehydration, the samples were embedded in paraffin wax. Six-micron-thick sections were stained with hematoxylin and eosin and examined under a research microscope (Nikon, Japan).
TEM Observations
Very small cubes (1 mm3) of liver were immersed in 2.5% glutaraldehyde, postfixed in 1.0% osmium tetraoxide, dehydrated through a graded series of ethanol, and embedded in Epon 812. After several changes in propylene oxide, ultrathin sections stained with uranyl acetate and lead nitrate were examined under a Philips, CMIO transmission electron microscope, at the Electron Microscope Facility, All India Institute of Medical Sciences, New Delhi.
Statistical Analyses
Student’s t test was applied to make multiple comparisons among different groups. Differences between groups with p values < 0.05 were considered significant.
Results
Characterization of Nanoparticles
Standard methods were performed to evaluate the physical properties of CdS. TEM observations showed that average size of these particles ranged between 5 and 9 nm (Fig. 1a). The FE-SEM image indicates the formation of nanoclusters (Fig. 1b). Using Scherrer’s formula, the crystallite size of CdS has been calculated which revealed that CdSNP mean crystallite size was < 9 nm. Figure 1 c shows the elemental composition of the sample analyzed by EDAX which confirms the peaks of cadmium and proves nanoparticles free from any impurities. Figure 1 d presents the intensity weighted particle-sized distribution of CdSNPs. The value of zeta potential was (− 15.7) mv (Fig. 1e).
a The size of CdSNPs is shown by transmission electron microscope. b Field emission scanning electron microscopy observations showing external morphology of CdSNPs. c EDAX showing elemental composition of CdSNPs. d Shows intensity weighed particle size distribution. e Zeta potential showing electrokinetic potential of CdSNPs
Biological Observations, Bioaccumulation of NPs, and Metallothionein Induction
Following a 45-day treatment schedule with CdSNPs and CdS bulk particles, respectively, their effects on body weight and liver/body weight ratio were examined. Whereas control rats exhibited a progressive growth in terms of body weight gain, rats treated with CdSNPs and CdS bulk particles showed a loss in body weight. Percent loss of body weight was higher in CdS-treated rats than CdSNP-treated rats (Table 1). Hepatosomatic index increased in CdS-treated rats but declined in CdSNP-treated rats (Table 1). Chemobiokinetics of CdSNPs showed that they were sequestered by the hepatic tissue. Hepatic concentration of CdSNPs was higher than bulk particles (Table 1). CdSNPs could induce the synthesis of metallothionein in liver. However, its concentration was higher in the liver of CdSNP-treated rats than those treated with CdS particles (Table 1).
Liver Function Studies
Present observations show that liver function is also affected after exposure to CdSNPs. It increases the efflux of transaminases into the blood showing disturbance in transamination reactions. Values for AST were higher in CdSNP-treated rats than for those treated with CdS bulk particles. However, values for ALT were higher in CdS-treated rats than in those treated with CdSNPs (Table 1). Another possible marker of liver function, i.e., alkaline phosphate, exhibited higher values in CdSNP-treated rats than in those treated with bulk CdS (Table 1). All these observations indicate significant liver function impairment in CdSNP-treated rats.
Biomarkers of Oxidative Stress (LPO, H2O2, NO, and SH Content)
Lipid peroxidation was assayed employing thiobarbituric acid. These TBA chromogens were measured as malondialdehyde. Results exhibited significant increase in LPO in the liver of CdSNP-treated rats. It was significantly higher than the liver of bulk CdS-treated rats. Based on these observations, it could be concluded that LPO plays a critical role in the hepatotoxic manifestations of CdSNPs (Table 1).
Another set of observations on H2O2, an important contributor in lipid peroxidation, supported the findings on malondialdehyde. It was higher in liver of CdSNP-treated rats than those administered CdS bulk particles (Table 1).
Although concentration of nitric oxides (an indicator of nitrosative stress) was also higher in the liver of CdSNP-treated rats than CdS-treated rats, they do not appear to play an important role in hepatotoxicity of CdSNPs. However, combined together, all the three biomarkers of LPO favor the conclusion that hepatotoxicity of CdSNPs is routed through oxidative damage (Table 1).
It is an established fact that Cd exhibits strong affinity with SH groups. It depletes glutathione (GSH) content of the hepatic cell. CdS and CdSNPs both inhibited thiols in the liver. However, disturbance was greater in CdS-treated rats than CdSNP-treated rats (Table 1).
Histopathological Observations
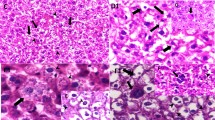
Ingestion of bulk and nanoparticles of CdS by rat induced distinct pathological lesions in the liver. The liver of CdS-treated rat exhibited extensive hydropic degeneration and ballooning of parenchymal cells. Formation of dysplastic tissue/nodules was another significant effect of CdS in the liver. Kupffer cell hyperplasia was also observed. Nuclei were found to contain dense chromatin and increase in size. However, no significant change in their shape was observed (Fig. 2a, b).
a Transverse section (TS) of the liver of rat treated with microparticles of CdS shows hydropic degeneration, ballooning of cells (BC) with dense nuclei.X200. b Dysplasia (DP) (nodule formation) signifies the toxicity of CdS in rat liver. × 200. c TS of the liver of rat treated with CdSNPs shows inflamed sinusoids (SNs), extensive parenchymal degeneration (PD), and a few binucleated cells. × 100. d A magnified view shows pyknotic nuclei eosinophilia and presence of erythrocytes in hepatic sinusoids (SNs) of CdSNP-treated rat. × 200. e TS of the liver of a control rat shows intact parenchyma (PC) and other cellular components. × 200
Liver of CdSN-treated rats showed extensive cytoplasmic degeneration/coagulation and sinusoidal inflammation. However, no formation of dysplasia was noticed. Many binucleated cells were observed. Intracytoplasmic inclusions were found uniformly distributed throughout the lobule. Portal changes were insignificant (Fig. 2c, d). TS of the liver of control rat showed intact parenchymal cells with round nuclei (Fig. 2e).
Ultrastructure Studies
Conspicuous changes in the ultrastructure of hepatic parenchymal cells were observed in CdS-treated rats. Prominent changes included the presence of vacuoles and proliferation of smooth endoplasmic reticulum. Mitochondria acquired elongated shape. Increase in the number of peroxisomes was also observed (Fig. 3a).
a Transmission electron microscopic observations (TEM) on the liver of CdS-treated rat show presence of vacuole (VC) proliferation of endoplasmic reticulum (ER) and elongated mitochondria (MT), × 2500. b T.E.M of the liver of CdSNP-treated rat shows mitochondrial (MT), microsomal (MIC), and peroxisomal (PER) proliferation (× 4000). c T.E.M of the liver of the control rat shows round nucleus (NU), normal mitochondria (MT), and endoplasmic reticulum. (ER), × 2500
In CdSNP-treated rats, the shape and size of mitochondria did not change but their number was increased. Cytoplasmic vacuoles were wanting; however, the number of microsomes and peroxisomes increased. Nuclear chromatin was uniformly distributed (Fig. 3b).
In liver of control rats, normal nuclei, round mitochondria, and endoplasmic reticulum were observed (Fig. 3c).
Discussion
Since the discovery of “itai itai” in Japan by Murata and Kobayashi [28, 29], possible mechanisms responsible for its system toxicity in target systems, i.e., kidney, liver, bones, and testes, have been investigated in a number of laboratories. Although Cd2+ is not a Fenton metal, there are several mechanisms by which Cd2+ can indirectly generate ROS [14, 16, 30]. It has been proposed that the mechanisms of acute Cd2+ toxicity involve a persistent rise in ROS and Ca2+ which disrupt cell function and trigger cell death [15]. Liver injury caused by Cd2+ has also been studied by a number of workers [31, 32]. Pathological lesions depend on liver cadmium concentration. It is sequestered by low-molecular-weight protein, i.e., metallothionein. The microparticles that fail to bind with metallothionein due to its saturation manifest toxicity.
Nonetheless, liver injury caused by nanoparticles remains to be a subject of further studies. Nanoparticles are classified on the basis of their chemical structure, morphology, applicability, and method of synthesis etc. Our microscopical observations showed that CdSNPs were crystalline and formed agglomerates. Their size ranged from 5 to 9 nm. NPs < 5 nm in diameter are known to be the most hazardous whereas NPs bigger than 40 nm are known to be less toxic [33].
Administration of these particles to rats on alternate days for a total duration of 45 days did not significantly affect the growth of rats. Contrarily, hepatosomatic index declined in CdSNP-treated rats and increased in CdS-treated rats. These observations are supported by observations on kidney reported earlier by K. Rana et al. [18]. Nevertheless, accumulation of nano cadmium was higher in liver as compared to bulk particles of cadmium. Bioaccumulation of nanoparticles depends on their interaction with serum proteins. Endocytosis of nanoparticles does occur after their binding with serum proteins [34]. Further, intracellular degradability might also contribute in their cytotoxicity [35, 36].
Both nano and microparticles of Cd2+ induced the synthesis of metallothionein in liver. However, the induction was higher in the liver of CdSNP-treated rats. These proteins offer protection against toxic metals and several pro-oxidants [37]. In mammals metallothionein (MT1 and MT2), genes are distributed in several tissues whereas metallothionein (MT3 and MT4) genes are restricted in their expression [38]. Cd2+ causes rapid transcriptional induction of MT1 and MT2 genes [39]. Moreover, gene expression changes observed by Chen et al. [40] showed that seven genes from metallothionein family viz. MT1F, MT1G, MTTH, MTTX, MT2A, and MT2E are upregulated by CdTe QDs and CdCl2 in HEK293 cells. Protective transcriptional effect of metallothionein(s) reciprocates with the bioaccumulation of Cd2+ in hepatic tissue. Nevertheless, we assume that hepatotoxicity of CdSNPs might not only arise from the release of Cd2+ ions but also from intracellular distribution of nanoparticles and related unknown molecular effects at nanoscale.
To assess these effects, liver function was examined through conventional biomarkers viz. serum transaminases. Elevated activities of aspartate transaminase (AST) and alanine transaminase (ALT) reflect specific hepatocyte destruction, whereas alkaline phosphatase (ALP) is a nonspecific indicator of liver function. Although several reports on the effects of Cd2+ on liver are available in the literature [41,42,43,44], effects of CdSNPs on serum enzymes have also been recently studied. These workers showed that hepatotoxicity of smaller CdSNPs is greater than of larger CdSNPs [45]. The present results show that level of these enzymes in the serum of CdSNPs treated rats increased significantly. The impact of other NPs on enzyme parameters of liver function is also not clear. Bedmarski et al. [46] studied these enzymes in rats orally administered with gold nanoparticles, and Rajan et al. [47] studied the same parameters in rats treated with iron oxide nanoparticles. As occasional rise in AST and ALT does not necessarily reflect liver toxicity, supporting histopathological observations are needed.
One of the reasons for impaired liver function might be the oxidative stress. The present results show that administration of CdS bulk particles and CdSNPs to rats induced oxidative stress in the liver as indicated by greater values for MDA, H2O2, and NO than control rats. It is an established fact that Cd toxicity is manifested through the generation of free radicals [48,49,50]. NP toxicity has also been attributed to enhanced generation of ROS [51, 52]. The large surface area and reactive nature of molecules enrich them with massive oxidizing capabilities. The mechanisms through which NPs can generate ROS have also been reviewed by Pisanic et al. [53]. NPs can generate ROS directly due to an exposure to acidic environment (lysosomes), from either the surface of NPs or leached ions [54, 55]. Secondly, NPs can interact with organelles such as mitochondria and generate ROS [56]. Further, NPs can interact with redox-active proteins such as NADPH oxidase. They can also interact with cell surface receptors and activate intracellular signaling pathways. These particles can also transfer energy to nearby oxygen molecules and lead to the formation of ROS which in turn leads to cell inflammation, injury, and cell death [57].
Cadmium possesses strong affinity towards sulfhydryl groups. It directly inhibits these proteins through conjugation mechanisms mediated by glutathione S-transferases [58, 59]. The present results also showed a significant decline in GSH in the liver of rats treated with bulk CdS particles. Earlier studies from our laboratory also support these observations [60, 61]. However, treatments of rats with CdSNPs for 45 days also declined GSH value in liver. Therefore, it may be considered as another mechanism of CdSNPs toxicity. Several reports indicate that MNPs (magnetic nanoparticles) are potent inducers of oxidative stress [59, 62, 63].
Finally, histopathological observations were made in the liver of CdS and CdSNP-treated rats. Cellular architecture of healthy rat liver comprises radially arranged arrays of hepatic parenchymal cells around the central vein, normal hepatic sinusoids, with no inflammatory cells or necrotic spaces. Hepatic cells possess large, round, and centrally placed nuclei. Our studies showed different pathological changes in the liver of CdS- and CdSNP-treated rats. Dysplastic, necrotic, and inflammatory changes were observed in CdS-treated rats. Inflamed sinusoidal cells were predominantly present. Several workers have reported that Cd2+ causes liver injury in different experimental animals [64,65,66,67]. However, a few workers have studied the effects of QDs on liver. The present results find support from earlier studies made by Wang et al. [68], who observed diffuse hydropic degeneration and ballooning of hepatic cells after 14 days of treatment of mice with cadmium chloride quantum dots. Binucleated regenerating cells were noticed after 28 days of exposure to QDs. However, dysplastic changes and edema were also recorded by us in rat liver after 45 days treatment with CdSNPs. In another study, Liu et al. [69] observed hepatocellular vacuolation or steatosis in the liver of mice treated with CdS nanodots. Although nanomaterial of different elemental composition can bind to common ligands in the liver, their toxicity can solely be predicted based on their elemental composition [70]. It is the shape, size, and extent of bioaccumulation that determine their hepatotoxicity.
Ultrastructural changes observed in the liver of Cd-treated rats corresponded with light microscopical observations. These include dilation of rough endoplasmic reticulum with concomitant loss of ribosomes. Other significant observation was the increase in the number of perichromatin granules in nuclei. Cd treatment caused mitochondrial changes as well. The present results are consistent with numerous reports available in the literature on adverse effects of Cd in liver [67, 71,72,73]. Though toxicity of CdSNPs to plasma membrane, ER, mitochondria, and nucleus of rat hepatocytes are not known, QDs have been reported to exhibit adverse effects upon different subcellular structures [49].
NPs possess strong penetrating ability into cells. Further, ligands attached to MNP modify their surface activities increasing their hydrophilicity and biocompatibility. These properties allow the NPs to penetrate in the cell and interact with membranes [74]. Since QD can induce cell death through lipid peroxidation, their effect on mitochondria is worth studying. Present investigations on mitochondrial structure in the liver of CdSNP-treated rats showed changes in their morphology and biogenesis. Mitochondrial swelling and loss of cristae were also observed. These findings agree with earlier reports of Cho et al. [75] who reported that Cys-CdTe-QD treatments to MCF-7 cells resulted into mitochondrial swelling and disorientation. Morphological changes in mitochondria reflect damage to mitochondrial integrity particularly the changes in mitochondrial membrane potential [76], increase in intracellular Ca2+, apoptosis [77, 78], cellular respiration, a depression of ATP synthesis, and inhibition of oxidative phosphorylation [79].
In conclusion, the present study reveals that CdSNPs are more toxic than CdS microparticles in rat liver. They bioaccumulate in the liver leading to synthesis of metallothionein and form ligands increasing their hydrophilicity. This facilitates their penetration into the membranes/cells. The NP-membrane interaction leads to the generation of ROS, alters membrane integrity, and induces oxidative stress that terminates into cell death. Based on the available information, it can be assumed that CdSNPs may directly interact with cell organelle and alter the structure and function of hepatic parenchyma.
References
Mamalis AG (2007) Recent advances in nanotechnology. J Mater Process Technol 181:52–58
Oberdorster G, Oberdorster E, Oberdorster J (2005) Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect 113:823–839. https://doi.org/10.1289/ehp.7339
Peter HH, Irene BH, Oleg VS (2004) Nanoparticles-known and unknown health risks. J.Nanobiotechnology. 2:12. https://doi.org/10.1186/1477-3155-2-12
Davoren M, Herzog E, Casey A, Benjamin C, Gordon C, Hugh JB, Fiona ML (2007) In vitro toxicity evaluation of single walled carbon nanotubes on human A549 lung cells. Toxicol in Vitro 21:438–448. https://doi.org/10.1016/j.tiv.2006.10.007
Hulderman LZ, Salmen T, Chapmen R, Stephen R, Shih-Houng SL, Shvedova Y, Luster MIA, Simeonuva PP (2007) Cardiovascular effects of pulmonary exposure to single wall carbon nanotubes. Environ Health Perspect 115(3):377–382
Stern ST, McNeil ES (2008) Nanotechnology safety concerns revisited. Toxicol Sci 101:4–21. https://doi.org/10.1093/toxsci/kfm169
De Jong WH, Hagens WI, Krystek P, Burger MC, Sips AJ, Geertsma RE (2008) Particle size dependent organ distribution of gold nanoparticles after intravenous administration. Biomater. 29:1912–1919. https://doi.org/10.1016/j.biomaterials
Jeffrey WC, Zeldin DC, Bonner JC, Nestmann RE (2008) Pulmonary applications and toxicity of engineered nanoparticles. Am J Physiol-Lung C 295:1–55
Sintov AC, Velasco-Agerirre C, Gallardo-Toledo E, Araya E, Kogan MJ (2016) Metal nanoparticles as targeted carriers circumventing the blood–brain barrier. Int Rev Neurobiol 130:199–227. https://doi.org/10.1016/bs.irn.2016.06.007
Smith AM, Duan H, Mobs AM, Nile S (2008) Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv Drug Deliv Rev 60:1226–1240. https://doi.org/10.1016/j.addr.2008.03.015
Juzenas P, Chen W, Sun YP, Codho MAN, Genralov R, Genraloves N, Christensen IL (2008) Quantum dots and nanoparticles for photodynamic and radiation therapies of cancer. Adv Drug Deliv Rev 60:1600–1614. https://doi.org/10.1016/j.addr.2008.08.004
Alivistas P (2004) The use of nanocrystals in biological detection. Nat.Biotechnol. 22:47–52. https://doi.org/10.1038/nbt927
Hardman RA (2006) Toxicologic review of quantum dots: toxicity depends upon physic-chemical and environmental factors. Environ.healthPerspect 114:165–172. https://doi.org/10.1289/ehp.8284
Paunovic J, Vucevic D, Radosavljevic T, Pantic S, Nikolovski D, Dugalic S, Pentavi I (2017) Effect of metallic nanoparticles on physiological liver functions. Rev Adv Mater Sci 49:123–128
Valko M, Morris H, Cronin MT (2005) Metals toxicity and oxidative stress. Curr Med Chem 12:1161–1208. https://doi.org/10.2174/0929867053764635
Thevenod F, Lee WK (2013) Cadmium and cellular signaling cascades: interaction between cell death and survival pathways. Arch.Toxicol. 87:1743–1786. https://doi.org/10.1007/s00204-013-1110-9
Thevenod F (2009) Cadmium and cellular signaling cascades: to be or not to be? Toxicol Appl Pharmacol 238:221–239. https://doi.org/10.1016/j.taap.2009.01.013
Rana K, Verma Y, Rani V, Rana SVS (2018) Renal toxicity of nanoparticles of cadmium sulphide in rat. Chemosphere. 193:142–150. https://doi.org/10.1016/j.chemosphere.2017.11.011
Reitman S, Frankel AS (1957) Colorimetric method for determination of serum glutamic oxaloacetic and glutamic pyruvic transaminases. Am J Clin Pathol 28:56–63. https://doi.org/10.1093/ajcp/28.1.56
King EJ, Armstrong AR (1934) A convenient method for determining serum and bile phosphatase activity. Can Med Assoc J 31:376–381
Onosaka S, Cherian MG (1982) The induced synthesis of metallothionein in various tissues of rats in response to metals. II. Influence of zinc status and specific effect on pancreatic metallothionein. Toxicology 23(1):11–20. https://doi.org/10.1016/0300-483x(82)90037-3
Rana SVS, Kumar A (2000) Metallothionein induced by cadmium or zinc inhibits lipid peroxidation in rats exposed to dimethylnitrosamine. Arh Hig Rada Toksikol 51(3):279–286
Jordan RA, Schenkman JB (1982) Relationship between malondialdehyde production and arachidonate consumption during NADPH-supported microsomal lipid peroxidation. Biochem Pharmacol 31(7):1393–1400. https://doi.org/10.1016/0006-2952(82)90034-X
O.H. Lowry, N.J. Rosebrough, A. L. Farr, RJ Randall, Protein measurement with the Folin phenol reagent, J Biol Chem 193 (1) (1951) 265–275
Thurman RG, Ley HG, Scholz R (1972) Hepatic microsomal ethanol oxidation, hydrogen peroxide formation and the role of catalase. Eur J Biochem 25:420–430. https://doi.org/10.1111/j.1432-1033.1972.tb01711.x
Cortas NK, Wakid NW (1990) Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clin Chem 36:1440–1443
Ellman GL (1959) Tissue sulphydryl groups. Arch Biochem Biophys 82:70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Murata I, Hirono T, Saeki Y, Nakagawa S (1970) Cadmium enteropathy, renal osteomalacia (“ItaiItai” disease in Japan). Bull Soc Int Chir 29(1):34–42
Kobayashi J, Nakahara H, Hasegawa T (1971) Accumulation of cadmium in organs of mice fed on cadmium-polluted rice. Jap J Hyg 26:401–407. https://doi.org/10.1265/jjh.26.401
Liu J, Qu W, Kadiiska MB (2009) Role of oxidative stress in cadmium toxicity and carcinogenesis. Toxicol Appl Pharmacol 238(3):209–214. https://doi.org/10.1016/j.taap.2009.01.029
Goering PL, Klassen CD (1984a) Tolerance to cadmium induced hepatotoxicity following cadmium pretreatment. Toxicol ApplPharmacol 74:308–313. https://doi.org/10.1016/0041-008X(84)90283-7
Friberg L, Kjellstrom T, Nordberg G, Piscator M (1979) “Cadmium” In handbook on the toxicology of metals (L.Friberg, G.F. Nordberg and V.B.Vonk Eds.), Elsevier/North Holland Biomedical Press. N.Y. 355–377
Soenen SJ, De M (2009) Cuyper, assessing cytotoxicity of (iron oxide based) nanoparticles: an overview of different methods exemplified with cationic magneto liposomes. Contrast Media Mol I 4(5):207–219. https://doi.org/10.1002/cmmi.282
Lynch I, Dawson KA (2008) Protein-nanoparticle interactions. Nano Today 3:40–47. https://doi.org/10.1016/S1748-0132(08)70014-8
Soenen SJ, Himmelreich U, Nuytten N, Pisanic TR, Ferrari A, De M (2010) Cuyper. Intracellular nanoparticle coating stability determines nanoparticle diagnostics efficacy and cell functionality. Small 6(19):2136–2145. https://doi.org/10.1002/smll.201000763
A.D. Lehmann, W.J. Parak, F. Zhang, Z. Ali, C. R€ocker, G.U. Nienhaus, P. Gehr, B. Rothen-Rutishause, Fluorescent magnetic hybrid nanoparticles induce a dose-dependent increase in proinflammatory response in lung cells in vitro correlated with intracellular localization, Small .6 (6) (2010) 753–762. https://doi.org/10.1002/smll.200901770
Waalkes MP, Wilson MJ, Poirier LA (1985) Reduced cadmium induced cytotoxicity in cultured liver cells following 5- azacytidine pretreatment. Toxicol Appl Pharmacol 81:250–257. https://doi.org/10.1016/0041-008x(85)90161-9
C.D, Klaassen, J. Liu, S. Choudhary, Metallothionein: an intracellular protein to protect against cadmium toxicity, Annu.Rev.Pharmacol. 39(1999) 267–294. https://doi.org/10.1146/annurev.pharmtox.39.1.267
Andrews GK (2000) Regulation of metallothionein gene expression by oxidative stress and metal ions. Biochem.Pharmacol. 59:95–104. https://doi.org/10.1016/S0006-2952(99)00301-9
Chen N, He Y, Su Y, Li X, Huang Q, Weng H, Zhang X, Tai R, Fan C (2012) The cytotoxicity of cadmium-based quantum dots. Biomaterials. 33:1238–1244
Rana SVS, Rastogi N (1998) Effect of cadmium on liver function in diabetic rats. Toxicol. Ind. Health 14:1–5. https://doi.org/10.1177/074823379801400306
Friberg L, Piscator M, Nordberg GF, Kjellstorm T (1974) Cadmium in the environment. CRC Press, Cleveland, Ohio
Dudley RE, Svoboda DJ, Klassen CD (1982) Acute exposure to cadmium causes severe liver injury in rats. Toxicol Appl Pharmacol 65:302–313. https://doi.org/10.1016/0041-008x(82)90013-8
Farber J (1979) Reactions of liver to injury: necrosis. In: Farber E, Fisher MM, eds. Toxic injury of the liver, Part A. New York: Marcel Dekker, 215–41
Lu L, Meiqing S, Qingzhao L, Hongmei Z, Alvarez PJJ, Huajie L, Wei C (2014) Genotoxicity and cytotoxicity of cadmium sulfide nanomaterials to mice: comparison between nanorods and nanodots. Environ Eng Sci 31(7):373–380. https://doi.org/10.1089/ees.2013.0417
M. Bednarski, , J. Dudek, L. Knustelska, J. Navinski, M. Sapa, G. Zygmunt, M Nowa K, , M. Luty-Bloch, M. Wojnicki, K. Fitzner, M. Tesiorowski, The influence of the route of administration of gold nanoparticles on their tissue distribution and basic biochemical parameters: in vivo studies,Pharmacol Rep. 67(2015) 405–409. https://doi.org/10.1016/j.pharep.2014.10.019
Rajan B, Satish S, Balakumar S, Devaki T (2015) Synthesis and dose interval dependent hepatotoxicity evaluation of intravenously administered polyethylene glycol-8000 coated ultra-small superparamagnetic iron oxide nanoparticle on Wistar rats. Environ Toxicol Pharmacol 39(2015):727–735. https://doi.org/10.1016/j.etap.2015.01.018
Gabor S, Anca Z, Bordas E (1978) Cadmium induced lipid peroxidation in kidney and testes. Effect of zinc and copper. Rev Roum Biochim 15:113–117
Waalkes MP, Wilson MJ, Poirier LA (1985) Reduced cadmium induced cytotoxicity in cultured liver cells following 5- azacytidine pretreatment. Toxicol Appl Pharmacol 81:250–257. https://doi.org/10.1016/0041-008x(85)90161-9
Sunderman FW Jr (1986) Metal and lipid peroxidation. Acta Pharmacol Toxicol 59(Suppl 7):248–255. https://doi.org/10.1111/j.1600-0773.1986.tb02755.x
Nel A, Xia T, Mädler L, Li N (2006) Toxic potential of materials at nanolevel. Science. 311:622–627
Xia T, Kovochich M, Liong M, Mädler L, Gilbert B, Shi H, Yeh JI, Zink JI, Nel AE (2008) Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS. Nano 2:2121–2134. https://doi.org/10.1021/nn800511k
T.R II. Pisanic, J. D. Blackwell, VI. Shubayev, R. R. Finones, S. Jin (2007) Nanotoxicity of iron oxide nanoparticle internalization in growing neurons, Biomaterials 28:2572–2581
Stroh A, Zimmer C, Gutzeit C, Jakstadt M, Marschinke F, Jung T, Pilgrimm H, Grune T (2004) Iron oxide particles for molecular magnetic resonance imaging cause transient oxidative stress in rat macrophages. Free Radic Biol Med 36(8):976–984. https://doi.org/10.1016/j.freeradbiomed.2004.01.016
Jain TK, Reddy MK, Morales MA, Leslie-Pelecky DL, Labhasetwar V (2008) Biodistribution, clearance and biocompatibility of iron oxide magnetic nanoparticles in rats. Mol Pharm 5(2):316–327. https://doi.org/10.1021/mp7001285
Soto KF, Carrasco A, Powell TG, Garza KM, Murr LE (2005) Comparative in vitro cytotoxicity assessment of some manufactured nanoparticulate materials characterized by transmission electron microscopy. Nanoparticle Res 7:145–169. https://doi.org/10.1007/s11051-005-3473-1
Choi AO, Cho SJ, Desbarats J, Lovric J, Maysinger D (2007) Quantum dot-induced cell death involves Fas upregulation and lipid peroxidation in human neuroblastoma cells. J Nanotechnology 5:1–2. https://doi.org/10.1186/1477-3155-5-1
Stohs SJ, Bagchi D, Hassoun E, Bagchi M (2000) Oxidative mechanisms in the toxicity of chromium and cadmium ions. J Environ Pathol Toxicol Oncol 20:77–88
Sharma B, Singh S, Siddiqi NJ (2014) Biomedical implications of heavy metals induced imbalances in redox systems, Biomed Res Int. 640754. https://doi.org/10.1155/2014/640754
S.V.S. Rana , R. Singh, Species differences in glutathione- dependent enzymes in the liverand kidney of two fresh water fishes and their implications for cadmium toxicity. Ichthyol. Res .43(1996) 223–229. https://doi.org/10.1007/BF02347594
S.V.S. Rana and K.Taketa Eds. Oxidative stress and liver injury by environmental xenobiotics, In “Liver and Environmental Xenobiotcs”. Springer Verlag.pp 114–134
Donaldson K, Brown D, Clouter A, Duffin R, MacNee W, Renwick L, Tran L, Stone V (2002) The pulmonary toxicology of ultrafine particles. J Aerosol Med 15:213–220. https://doi.org/10.1089/089426802320282338
Akhtar MJ, Ahamed M, Kumar S, Khan MM, Ahmed J, Alrokayan SA (2012) Zinc oxide nanoparticles selectively induce apoptosis in human cancer cells through reactive oxygen species. Int J Nanomedicine 7:845–857. https://doi.org/10.2147/ijn.s29129
Colucci AV, Winge D, Krasno MD (1975) Cadmium accumulation in rat liver. Arch Environ Health 30:153–157. https://doi.org/10.1080/00039896.1975.10666665
Stowe HD, Wilson M, Goyer RA (1972) Clinical and pharmacological effects of oral cadmium toxicity in rabbits. Arch.Pathol. 94:389–405
Hoffman EO, Cook JA, Diluzio NR, Coover JA (1975) The effects of acute cadmium administration in the liver and kidney of rat. Light and Electron microscopic studies. Lab Investig 32:655–664
Dudley RE, Svoboda DJ, Klassen CD (1984) Time course of cadmium induced ultrastructural changes in rat liver. Toxicol Appl Pharmacol 76(1):15–160. https://doi.org/10.1016/0041-008X(84)90038-3
M. Wang, J. Wang, H. Sun, S. Han, S. Feug, L. Shi, P. Meng, J. Li, P. Huang, , Z. Sun, Time dependent toxicity of cadmium chloride quantum dots on liver and kidney of mice: histopathological changes with elevated free cadmium ions and hydroxyl radicals, Int J Nanomedicine 11(2016), 2319–2328. https://doi.org/10.2147/IJN.S103489
Liu L, Sun M, Li Q, Zhang H, Alvarez PJJ, Liu H, Chen W (2014) Genotoxicity and cytotoxicity of cadmium sulfide nanomaterials to mice: comparison between nanoraods and nanodots. Environ Eng Sci 31:373–380. https://doi.org/10.1089/ees.2013.0417
Xiu ZM, Zhang QB, Puppala HL, Colvin VL, Alveraj PJ (2012) Negligible particle specific antibacterial activity of silver nanoparticles. Nano Lett 12:4271–4275. https://doi.org/10.1021/nl301934w
Sina JF, Chin B (1978) Cadmium modification of nucleolar ultrastructure and RNA synthesis in Physarumpolycephalum. Toxicol Appl Pharmacol 43:449–459. https://doi.org/10.1016/S0041-008X(78)80004-0
Puvian E, Lange M (1980) Functional significance of perichromatin granule accumulation induced by cadmium chloride in isolated liver cells. Exp Cell Res 128:47–58. https://doi.org/10.1016/0014-4827(80)90385-7
Gamulin S, Car N, Narancsik P (1982) Effects of cadmium on polyribosome structure and function in mouse liver. Experimenta. 33:1144–1145. https://doi.org/10.1007/bf01922292
Verma A, Stellacci F (2006) Effect of surface properties on nanoparticle cell interactions. Small 6(1):12–21. https://doi.org/10.1002/smll.200901158
Cho SJ, Maysinger D, Jain M, Roder B, Hackbarth S, Winnik FM (2007) Long term exposure to CdTe quantum dots causes functional impairments in live cells. Langmuir. 23:1974–1980. https://doi.org/10.1021/la060093j
P.X. Petit , H. Lecocur, E. Zorn, , C. Daughet, B. Mignotte, M. L.Gongeon, Alterations in mitochondrial structure and function are early events of dexamethasone induced thymocyte apoptosis, J Cell Biol 130(1995) 157–167. https://doi.org/10.1083/jcb.130.1.157
Armstrong JS (2006) Mitochondrial membrane permeabilization: the sine qua non for cell death. Bioassays. 28:253–260. https://doi.org/10.1002/bies.20370
Yeh JH, Huang CC, Yeh MV, Wang JS, Lee JK, Jan CR (2009) Cadmium induced cytosolic Ca2+ elevation and subsequent apoptosis in renal tubular cells. Basic Clin Pharmacol Toxicol 104:345–351
Brand MD, Nicholls DG (2011) Assessing mitochondrial dysfunction in cells. Biochem J 435:297–312. https://doi.org/10.1042/BJ20110162
Acknowledgments
The authors thank Director, Sophisticated Analytical Instrument Center, Punjab University, Chandigarh for FE-SEM, TEM, and EDAX. Director, Sophisticated Instrument facility of Indian Institute of Technology, Roorkeeis, is acknowledged for DLS and zeta potential. We are grateful to the In-charge, Electron Microscope Facility, All India Institute of Medical Sciences, New Delhi, for extending all possible help in TEM for ultrastructural studies of tissue.
Funding
The Department of Science and Technology, New Delhi, provided financial support (SR/FT/LS-46/2011) KR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rana, K., Verma, Y. & Rana, S.V.S. Possible Mechanisms of Liver Injury Induced by Cadmium Sulfide Nanoparticles in Rat. Biol Trace Elem Res 199, 216–226 (2021). https://doi.org/10.1007/s12011-020-02128-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02128-5