Abstract
A total of 108 pregnant and lactation SD rats were divided into six groups, daily and orally dosed with Bacillus subtilis-copper (CuBs) and CuSO4 (CuS) both at three doses equivalent to 1.5 mg (Cu deficiency), 3 mg, and 6 mg Cu per kg diet, and the effects of the Cu source and dosage on the growth rate, immune status, duodenal development, and cecal microbial diversity were examined on 24-day-old offspring rats. The six offspring rats from each group were randomly selected for measuring the body weight gain and taking blood samples, and three rats were sacrificed for taking duodenum and cecum content samples. We found CuBs increased the body weight gain, development of duodenal villi, and survival rate of the offspring; increased the IgM content and lysozyme activity in serum; reduced the intestinal permeability; and increased the abundances of Lachnospiraceae, Ruminococcaceae, and Intestinibacter in the cecal content, when compared with CuS. We also found that Cu deficiency showed detrimental effects on the body weight gain and length, the survival rate of the offspring, and the immune indices in serum, as well as the increased intestinal permeability. We concluded that CuBs is better Cu source than CuSO4 for reproductive rats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Appropriate concentrations of copper (Cu) in diet can increase the gut microbial diversity, promote absorption of nutrients in the gut, and increase growth rate of poultry. To achieve high growth rates of livestock animals and poultry, it is a common practice to add high doses of Cu in diet, which could result in an increase of the amount of metal deposition in the product, a low utilization efficiency of Cu in the body, and excessive Cu excretion into farming environments, causing water pollution. Therefore, it is of great significance to search for novel sources of Cu, which can be utilized efficiently by farming animals, so dietary supplementation of Cu could be reduced without a loss of animal’s productivity.
Dietary addition of 1.5 g/kg of montmorillonite Cu could effectively reduce the number of Clostridium and Escherichia coli, and increase the villus height and the ratio of villus height to crypt depth in small intestinal mucosa in weaned piglets [1]. Montmorillonite Cu effectively alleviates diarrhea and inflammation, improved intestinal microbial diversity, and ensured intestinal mucosal integrity of weaned piglets [2]. Recent studies clearly showed that dextran nano-Cu was conducive to the stability of the intestinal flora and may be a potential substance for chlortetracycline replacement in diet for weaned piglets [3]. Dietary supplementation of probiotics could increase zinc utilization by converting inorganic zinc to organic zinc [4].
Bacillus subtilis has a very strong ability of binding metal elements (including Cu) [5], so Bacillus subtilis-copper (CuBs) is a good source of organic Cu, which can improve the balance between anions and cations in the intestine and improve the immunity and the growth rate of animal. Therefore, CuBs can be a potential novel source of organic Cu. Hence, this study supplied pregnant and lactating SD rats with CuBs or copper sulfate (CuSO4, CuS) both at three dosages, and compared the effectiveness of these two Cu sources on the growth rate, immune status, intestinal development, and cecal flora diversity in 24-day-old offspring rats.
Materials and Methods
Rats, Management, and Experimental Design
A total of 108 pregnant Sprague-Dawley (SD) rats were used in this experiment. The use of rats and the experimental protocol were approved by the Animal Ethics Committee of the College of Animal Science and Technology, Qingdao Agricultural University, China. All rats were given deionized water to drink and fed a basal diet (modified AIN-93G diet which contained Cu at 0.33 mg/kg). The animals were housed in stainless steel cages in a temperature- and humidity-controlled room with a 12:12-h light/dark cycle.
The rats were randomly divided into six groups with 18 rats in each group, and allocated to six treatments: daily drenched (0.2 mL per 10 g body weight) once with a CuS solution or CuBs suspension to provide the amounts of Cu equivalent to 1.5 mg, 3 mg, or 6 mg per kg of diet; thus, the experiment formed a 2 (CuS vs CuBs) × 3 (low (L), medium (M), and high (H) doses of Cu) factorial design, respectively, referring to CuS-L, CuS-M, CuS-H, CuBs-L, CuBs-M, and CuBs-H. The Bacillus subtilis-Cu was a product provided by the Institute of High Quality Waterfowl, Qingdao Agricultural University, which contains Bacillus subtilis 1.5 × 109 CFU/g and Cu 2906 mg/kg. Bacillus subtilis is domesticated by the National Waterfowl Industry Technology System Nutrition and Feed Function Laboratory, and collected by the General Microbiological Center of the China Microbial Culture Collection Management Committee (Accession No. CGMCC 15329). The treatments started from 5 days of the pregnancy until the end of lactation when the offspring rats were at 24 days of age.
Measurements and Sampling
On the last day of the experiment, the offspring rats were fasted overnight; then, six rats in each group were randomly selected, and the individual body weights and body lengths were recorded. Then, the rats were euthanatized with ether. Blood sample was taken from the eyeball. Serum was harvested by centrifugation of blood at 3000 rpm for 15 min, and stored at − 80 °C until analysis later on.
The duodenums of three rats randomly selected from the six offspring rats in each group were immediately excised. After removing the content, the lumen was rinsed with sterile saline, and then duodenum tissue was cut into small pieces of 1 × 1 mm, and fixed with 2.5% glutaraldehyde overnight.
The cecum was immediately removed, and the content was collected and stored at − 80 °C for analysis of microbial diversity later on.
Analyses
Duodenum Morphology by Scanning Electron Microscopy
The fixed duodenum tissue was dehydrated sequentially using 50%, 70%, 80%, 90%, 100%, and 100% (vol/vol) ethanol:normal saline, each for 10 min, followed with 50%, 100%, and 100% (vol/vol) isoamyl acetate:ethanol, each for 10 min. The dehydrated tissue was then dried on liquid CO2, pasted on the table, then sputter-coated lightly with gold and examined in scanning electron microscopy (SEM, JEOL Oxford 7500F, Japan).
Duodenum Permeability and Immune Indicators
The concentration of d-lactic acid (D-LA) and diamine oxidase (DAO) activity in serum were determined using commercial kits (Nanjing Jiancheng Institute of Biological Engineering, China) following the manufacturer’s instructions. These measurements were used as the indicators to the duodenum permeability.
The concentrations of mononuclear chemokinin 1 (MCP-1) and immune globulin M (IgM) in serum were determined using commercial kits (Nanjing Jiancheng Institute of Biological Engineering, China) following the manufacturer’s instructions.
Microbial Diversity in Cecum
Microbial DNA in the cecal content was extracted using E.Z.N.A.® Stool DNA Kit (Tiangen Biochemical Technology Co, China), following the manufacturer’s instructions. The quality and quantity of genomic DNA were assessed with a nanodrop spectrophotometer, with the A260/A280 ratio between 1.8 and 2.0 considered a criterion for the quality control. No obvious RNA banding was shown by gel electrophoresis, and genomic bands were clear and complete. DNA was frozen at − 80 °C prior to PCR amplification.
To amplify the V3–V4 region of 16S rRNA gene for Illumina deep sequencing, universal primers, 338F:5′-ACTCCTACGGGAGGCAGCA-3′ and 806R:5′-GGACTACHVGGGTWTCTAAT-3′, were used. The PCR was performed in a total reaction volume of 20 μL: H2O 13.25 μL, 10 × PCR ExTaq buffer 2.0 μL, DNA template (100 ng/mL) 0.5 μL, prime1 (10 mmol/L) 1.0 μL, prime2 (10 mmol/L) 1.0 μL, dNTP 2.0 μL, ExTaq (5 U/mL) 0.25 μL. After an initial denaturation at 95 °C for 5 min, an amplification was performed by 30 cycles of incubations for 30 s at 95 °C, 20 s at 58 °C, and 6 s at 72 °C, followed by a final extension at 72 °C for 7 min. Then, the amplified products were purified and recovered using 1.0% agarose gel electrophoresis method. Finally, the library construction and sequencing steps were performed by Beijing Biomarker Technologies Co. Ltd.
The bioinformatic analysis on the microbial in the cecal content was completed at the Biomarker Biocloud Platform. To obtain the raw tags, paired-end reads were merged by FLASH [6]. Then, raw tags were filtered and clustered in the next steps. The merged tags were compared with the primers, and the tags with more than six mismatches were discarded by FASTX-Toolkit [7]. Tags with an average quality score < 20 in a 50-bp sliding window were truncated using Trimmomatic [8] and tags shorter than 300 bp were removed. We identified possible chimeras by employing UCHIME, a tool included in mothur [9, 10]. The denoised sequences were clustered using Qiime UCLUST module and tags with similarity ≧ 97% were regarded as an OTU. Taxonomy was assigned to all OTUs by searching against the SILVA databases using the RDP classifier within QIIME [11,12,13].
Statistical Analysis
The differences of six treatments were examined in the two-way ANOVA procedure of GenStat (VSN International) with Cu source (CuS and CuBs) and their dosages as the factors in the model. The interaction of two factors was also examined. The least square means and standard error of the means are presented. Significance between the treatments was declared when P values were ≦ 0.05.
Results
The Growth and Development and Indices in Serum of Offspring Rats
There was no significant interaction between Cu source and dosage on the body weight, body length, the weigh to length ratio, and survival rate of offspring until 24 days old (P > 0.05) (Table 1). The rats on the CuBs treatments had higher body weight and survival rate than the offspring rats on the CuS treatments (P < 0.05). The dosage of Cu had significant influences on the body weight, body length, and survival rate (P < 0.05), which increased with the dosages. In addition, except for the lowest survival rate of the offspring rats on addition of 1.5 mg/kg of CuS, the other five groups had no significant difference (P > 0.05; Table 1).
As shown in Table 1, there was significant interaction between Cu source and dosage on serum concentration of D-LA (P < 0.05), and the concentration tended to decrease with the Cu dosage. On 1.5 mg/kg and 3.0 mg/kg Cu, the rats from CuBs had lower concentrations of D-LA as compared with the rats on CuS (P < 0.05), but there was no significant difference in D-LA concentration between CuBs and CuS at dose 6 mg/kg (P > 0.05). The DAO activity in serum decreased with the increasing Cu dosage (P < 0.05), but there was no significant difference between 1.5 mg/kg and 3.0 mg/kg doses (P > 0.05). The Cu sources had no significant effect on the DAO activity (P > 0.05), and no significant interaction between Cu source and dosage was found on the serum DAO activity (Table 1).
The serum concentration of monocyte chemokinin-1 increased with the increased Cu dosage, while there were no significant differences between 1.5 mg/kg and 3 mg/kg Cu dosages (Table 1). The Cu sources had no significant effect on the monocyte chemokinin-1 concentration (P > 0.05), and there was no significant interaction between Cu source and dosage (P > 0.05; Table 1).
There were significant interactions between Cu source and dosage on the serum concentrations of IgM and lysozyme activity of the offspring rats (P < 0.05; Table 1). The IgM concentration and lysozyme activity increased with the increased Cu dosage. At 1.5 mg/kg Cu dose, the rats on CuBs had higher IgM concentration than rats on CuS (P < 0.05), whereas there were no significant differences in between CuBs and CuS at doses of 3 mg/kg or 6 mg/kg respectively (P > 0.05; Table 1).
The significant difference in the lysozyme activity was found between Cu source and dosage (P < 0.05) as shown in Table 1. There was no significant difference in the lysozyme activity between CuBs and CuS at 1.5 mg/kg dose (P > 0.05); the rats had higher lysozyme activity on CuBs at 3 mg/kg dose (P < 0.05), but lower lysozyme activity on CuBs at 6 mg/kg dose when compared with the rats on the same doses of CuS (P < 0.05; Table 1).
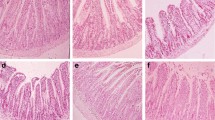
There was no significant difference in the duodenal villi between CuBs and CuS at the highest dose of Cu, and the intestinal villi appeared neatly and the gap between the villi was small (Fig. 1). At 1.5 mg/kg Cu dose, the villi of rats on CuBs appeared more neatly arranged compared with the CuS treatment, and the gap between the villi was smaller. The intestinal villus morphology appeared to be influenced by the Cu dosages, and with the increase of Cu dosage, the villi tended to be more neatly arranged, and the gap between the villi looked small.
Microbial Diversity Indices in Cecal Content
We used the diversity indexes as follows to reflect information on the abundance, coverage, and diversity of microbial community species in the cecal content. The indices indicating community richness are as follows: Chao1 estimator (Chao) and adaptive coherence estimator (ACE); the indices for community diversity include the following: Shannon, Simpson, and coverage. The coverage of each sample was greater than 0.99, which proved that the microbial species in the sample were almost fully identified and determined. As shown in Table 2, the Cu source and dosage had no significant effects on any of these indexes (P > 0.05).
Microbial Difference in the Cecal Content
Figure 2 shows LEfSe analysis of evolutionary branch diagram, in which brown, green, blue, purple, and brown dots represent the dominant species of each treatment, and yellow dots represent non-dominant species. The dot diameter is proportional to the relative abundance of the species. The dominant species for each treatment were different. Figure 3 is LDA analysis on the cecal content of the offspring. Under the condition that the LDA threshold is 4, a total of five groups of special bacteria were detected. The dominant species in the rats on 1.5 mg/kg CuS were Fusobacterium, Bacterium, and Clostridium innocuum, and were Bilophila, Parabacteroides, Porphyromonadaceae, Parabacteroides, and Roseburia on 6 mg/kg CuS. In comparison, the dominant species in the rats on 1.5 mg/kg CuBs was Lactonifactor; were Ruminococcaceae, Bacterium, and Lachnospiraceae on 3 mg/kg CuBs; and was Intestinibacter on 6 mg/kg CuBs.
LEfSe analysis on the cecal content of 24-day-old offspring of SD rats received CuSO4 or CuBs during pregnancy and lactation (n = 3). At the threshold of LDA = 4, the LEfSe analysis did not find that the CuS-M group had biomarkers, which indicates that there were no significant differences between this group and other groups, so there is no CuS-M group in the figure
LDA analysis on the cecal content of 24-day-old offspring of SD rats received CuSO4 or CuBs during pregnancy and lactation (n = 3). At the threshold of LDA = 4, the LDA analysis did not find that the CuS-M group had biomarkers, which indicates that there were no significant differences between this group and other groups, so there is no CuS-M group in the figure
Discussion
Effects of the Cu Source and Dosage on the Growth and Development of 24-Day-Old Offspring
This study found that drenching pregnant and lactation rats with CuBs increased the body weight and survival rate of their offspring compared with using the same dose of CuS. Zhang et al. [14] found that adding Bacillus subtilis and CuSO4 in the diet significantly increased the growth of Wulong goose compared with adding CuSO4 only. Wang et al. [15] and Zhao et al. [16] found that chelated Cu could promote the growth of broilers than inorganic Cu. Those reports are in agreement with our findings in this study. In addition, Cu deficiency in the body can limit animal’s growth [17], and our study confirmed that Cu deficiency (i.e., 1.5 mg/kg Cu per kg diet) inhibited the growth of body length and body weight gain, and reduced the survival rate of the offspring.
Effect of Bacillus subtilis-Cu on the Immunity of 24-Day-Old Offspring
Copper is a cofactor to many antioxidant enzymes and plays an important role in the immunity, which can stabilize cell permeability [18, 19]. Previous studies have found that the forms of Cu have different effects on immunity of the body [20, 21]. Huang et al. [22] reported that an addition of CuSO4 or copper methionine in diet had no effect on immune indicators such as IgG and lymphocytes. Liao et al. [21] found that at two concentrations (67 mg and 134 mg Cu/kg) of CuSO4 and copper methionine, there were no differences in IgG and lymphocytes. However, Senthilkumar et al. [23] showed that an addition of 14 mg Cu/kg protein-copper in diet could significantly improve the immune response of lambs than an addition of CuSO4. In addition, Bacillus subtilis can significantly increase immune competency of animals [24, 25]. The results of this study proved that CuBs was more beneficial to increase serum IgM and other immune indicators, and improve the immunity of the offspring than CuS. We also found in this study that copper deficiency could reduce the serum IgM content and lysozyme activity of the offspring, so impaired immune competency. This has been reported in other species of animals. For example, Prohaska et al. [26] reported that long-term copper deficiency caused severe deformation and damage to the structures of the lymph, spleen, and thymus in mice. The low concentration of copper in the body was related to abnormal function of mouse macrophages [26], and Bae [27] reported copper deficiency–related reduction of neutrophil immune function. These results are in agreement with our findings in this study.
Effect of Cu Source and Dosage on the Duodenum Permeability of 24-Day-Old Offspring
Plasma D-LA and DAO levels are important indicators for monitoring intestinal barrier function and damage. DAO is an enzyme mostly present in the cytoplasm of intestinal mucosa. Once the intestinal mucosa is damaged, intestinal mucosal cells can release a large amount of DAO into the blood circulation, so its activity in serum can reflect changes in the mucosal barrier function and intestinal permeability [28]. This study found that CuBs was more effective than CuS in reducing serum D-LA content, reducing intestinal permeability, and enhancing intestinal mucosal barrier. This may be related to the influence of phosphorylation of cytoskeleton and the strength of the tight junction protein on the intestinal mucosal cell interaction and cell stability, and enhance the barrier of the intestinal mucosa [29]. In addition, this study found that copper deficiency increased serum DAO and D-LA levels, and increased intestinal mucosal permeability in the offspring rats.
Copper absorption occurs mainly in the duodenum, so duodenum development could directly determine the absorption of copper [30]. Previous studies have shown that copper glycine as a copper supplement could improve the utilization of copper, but it caused exfoliation of a large amount of cells on the intestinal villi, which may result in intestinal diseases [31]. Dosing the rats with CuBs, however, increased the density of villi in the duodenum of the offspring, and the effect appeared stronger than dosing CuS in this study, suggesting that CuBs is more conducive to the development of duodenal villi.
Effects of Cu Source and Dosage on the Microbial Diversity in Cecum Contents of the Offspring
It is well known that pathogenic bacteria need to be colonized in the gastrointestinal tract to have harmful effects [32], and probiotic colonization in the gastrointestinal tract could prevent the establishment of pathogenic bacteria [33]. When the amount of copper added was 1.5 mg/kg, Fusobacterium and Clostridium innocuum were characteristic strains. The abundance of Fusobacterium was increased in gastric and colorectal cancer patients [34,35,36], and the abundances of Parabacteroides and Clostridium were increased in patients with irritable bowel syndrome [37], suggesting that an increase of these bacterial species in the intestinal content is risky to health. As strict anaerobic bacteria, Lachnospiraceae and Ruminococcaceae are the two most abundant Clostridiales families in mammalian gut, which play an important role in plant degradation and can maintain intestinal health [38]. Ruminococcaceae belongs to the Firmicutes, which produces butyric acid that exerts probiotic physiological functions in the gut [39, 40]. The increase of the Lachnospiraceae and Ruminococcaceae abundance in the rats on CuBs may be due to the accelerated consumption of trace oxygen by Bacillus subtilis towards anaerobic conditions in the intestinal tract, thereby promoting the growth of both species of bacteria. In addition, Intestinibacter was a dominant species in the rats on the high dose of CuBs, and this species can produce glutamate synthetase that is involved in ammonia detoxification, inter-organ nitrogen flux, acid-base homeostasis, and cell signaling [41]. We propose that Bacillus subtilis can accelerate the process of anaerobic environment, which alters microbial diversity, leading to the enrichment of beneficial bacteria [41, 42], reduction of the abundance of potential pathogenic bacteria [43, 44], changes of intestinal morphology, and growth of the body [45, 46].
Conclusion
This study found that dosing pregnant and lactation rats with Bacillus subtilis-Cu increased the body weight and survival rate of their offspring, promoted the development of duodenal villi, increased serum IgM content and lysozyme activity, reduced the intestinal permeability, and altered the microbial diversity in the cecal content with enrichments of beneficial bacterial species such as the Lachnospiraceae, Ruminococcaceae, and Intestinibacter. The results suggest that Bacillus subtilis-Cu is a good source of Cu for animals. Copper deficiency during pregnancy and lactation had detrimental effects on the growth and development, survival, immune functions, and intestinal permeability of the offspring.
References
Xia MS, Hu CH, Xu ZR (2005) Effects of copper bearing montmorillonite on the growth performance, intestinal microflora and morphology of weanling pigs. Anim Feed Sci Tech 118:307–317
Song J, Li YL, Hu CH (2013) Effects of copper-exchanged montmorillonite, as alternative to antibiotic, on diarrhea, intestinal permeability and proinflammatory cytokine of weanling pigs. Appl Clay Sci 77–78:52–55
Wang M, Du Y, Wang C, Tao W, He Y, Li H (2012) Effects of copper-loaded chitosan nanoparticles on intestinal microflora and morphology in weaned piglets. Biol Trace Elem Res 149:184–189
Liu D, Huang SM, Bi ZY, Wu C (2015) Optimization of zinc-riching conditions by probiotics in the form of mixing culture. J Anhui Agri Sci 43(20):259–262
Urrutia MM, Beveridge TJ (1994) Formation of fine-grained metal and silicate precipitates on a bacterial surface (Bacillus subtilis). Chem Geol 116:261–280
Magoc T, Salzberg SL (2011) Flash: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27:2957–2963
Pearson WR, Wood TC, Zhang Z, Miller W (1997) Comparison of DNA sequences with protein sequences. Genomics 46:24–36
Bolger A, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) Uchime improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microb 75:7537–7541
Caporaso G, Stombaugh J, Jesse K, Knights D, Koenig JE, Ley RE, Lozupone CA, Mcdonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) Qiime allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microb 73:5261–5267
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarza P, Peplies J, Glockner FO (2012) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:590–596
Zhang ZN, Wang BW, Ge WH, Zhang MA, Yue B, Zhang HW, Zhang YY (2016) Effects of Bacillus subtilis cooperate with copper on growth performance, slaughter performance, nutrient availability and meat quality of wulong geese aged from 5 to 16 weeks. Chinese J Anim Nutr 28:2830–2838
Wang Z, Cerrate S, Coto C, Yan F, Waldroup P (2007) Evaluation of MINTREX® copper as a source of copper in broiler diets. J Poult Sci 6:308–313
Zhao J, Shirley R, Vazquezanon M, Dibner J, Richards J (2010) Effects of chelated trace minerals on growth performance, breast meat yield, and footpad health in commercial meat broilers. J Appl Poult Res 19:365–372
Soetan K, Olaiya C, Oyewole O (2010) The importance of mineral elements for humans, domestic animals and plants - a review. African J Food Sci 4:200–222
Gaetke L, Chow CK (2003) Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology 189:147–163
Kim B, Nevitt T, Thiele D (2008) Mechanisms for copper acquisition, distribution and regulation. Nat Chem Biol 4:176–185
Creech B, Spears J, Flowers W, Hill G, Lloyd K (2004) Effect of dietary trace mineral concentration and source (inorganic vs. chelated) on performance, mineral status, and fecal mineral excretion in pigs from weaning through finishing. J Anim Sci 82:2140
Liao P, Li M, Li Y, Tan X, Zhao F, Shu X, Yin Y (2017) Effects of dietary supplementation with cupreous n-carbamylglutamate (NCG) chelate and copper sulfate on growth performance, serum biochemical profile and immune response, tissue mineral levels and fecal excretion of mineral in weaning piglets. Food Agri Immunol:1–15
Huang Y, Zhou T, Lee J, Jang H, Park J (2010) Effect of dietary copper sources (cupric sulfate and cupric methionate) and concentrations on performance and fecal characteristics in growing pigs. Asian-Aust J Ani Sci 23:757–761
Senthilkumar P, Nagalakshmi D, Reddy YR, Sudhakar K (2009) Effect of different level and source of copper supplementation on immune response and copper dependent enzyme activity in lambs. Trop Anim Health Pro 41:645–653
Molnar A, Podmaniczky B, Kurti P, Tenk I, Glavits R, Virag G, Szabo Z (2011) Effect of different concentrations of Bacillus subtilis on growth performance, carcase quality, gut microflora and immune response of broiler chickens. Brit Poultry Sci 52:658–665
Pabst O, Herbrand H, Friedrichsen M (2006) Adaptation of solitary intestinal lymphoid tissue in response to microbiota and chemokine receptor CCR7 signaling. J Immunol 177:6824–6832
Prohaska J, Downing S, Lukasewycz O (1983) Chronic dietary copper deficiency alters biochemical and morphological properties of mouse lymphoid tissues. J Nutr 113:1583–1590
Bae B, Percival SS (1993) Retinoic acid-induced hl-60 cell differentiation is augmented by copper supplementation. J Nutr 123:997–1002
Faries P, Simon R, Martella A, Lee M, Machiedo G (1999) Intestinal permeability correlates with severity of injury in trauma patients. Surg Clin N Am 79:1373–1383
Ng SC, Hart A, Kamm MA, Stagg AJ, Knight SC (2009) Mechanisms of action of probiotics: recent advances. Inflamm Bowel Dis 15:300–310
Linder MC, Hazeghazam M (1996) Copper biochemistry and molecular biology. Am J Clin Nutr 63:797–811
Tomaszewska E, Dobrowolski P, Kwiecien M (2016) Intestinal alterations, basal hematology, and biochemical parameters in adolescent rats fed different sources of dietary copper. Biol Trace Elem Res 171:185–191
Yirga H (2015) The use of probiotics in animal nutrition. J Prob & Health 3:1–10
Cho JH, Zhao PY, Kim IH (2011) Probiotics as a dietary additive for pigs: a review. J Anim Vet Adv 10:2127–2134
Hsieh YY, Tung SY, Pan HY, Yen CW, Xu HW, Lin YJ, Deng YF, Hsu WT, Wu CS, Li C (2008) Increased abundance of clostridium and fusobacterium in gastric microbiota of patients with gastric cancer in Taiwan. Sci Rep 8:158–169
Li YY, Ge QX, Cao J, Zhou YJ, Du YL, Shen B, Wan YJ, Nie YQ (2016) Association of fusobacterium nucleatum infection with colorectal cancer in Chinese patients. World J Gastroentero 22:3227–3233
Fukugaiti MH, Ignacio A, Fernandes MR, Ulysses Ribeiro J, Nakano V, Avila-Campos MJ (2015) High occurrence of fusobacterium nucleatum and clostridium difficile in the intestinal microbiota of colorectal carcinoma patients. Braz J Microbiol 46:1135–1140
Ohman L, Tornblom H, Simren M (2015) Crosstalk at the mucosal border: importance of the gut microenvironment in IBS. Nat Rev Gastro Hepat 12:36–49
Biddle A, Stewart L, Blanchard J, Leschine S (2013) Untangling the genetic basis of fibrolytic specialization by lachnospiraceae and ruminococcaceae in diverse gut communities. Diversity 5:627–640
Louis P, Flint HJ (2009) Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett 294:1–8
Huws S, Kim E, Lee M, Scott M, Tweed J, Pinloche E, Wallace R, Scollan N (2011) As yet uncultured bacteria phylogenetically classified as Prevotella, Lachnospiraceae incertae sedis and unclassified Bacteroidales, Clostridiales and Ruminococcaceae may play a predominant role in ruminal biohydrogenation. Environ Microbiol 13:1500–1512
Haberle J, Gorg B, Rutsch F, Schmidt E, Toutain A, Benoist J, Gelot A, Suc A, Hohne W, Schliess F, Haussinger D, Koch HG (2005) Congenital glutamine deficiency with glutamine synthetase mutations. New Engl J Med 353:1926–1933
Yirga H (2015) The use of probiotics in animal nutrition. Journal of Probiotics & Health 3:1–10
Parvez S, Malik KA, Kang A, Kim HY (2006) Probiotics and their fermented food products are beneficial for health. J Appl Microbiol 100:1171–1185
Patterson JA, Burkholder KM (2003) Application of prebiotics and probiotics in poultry production. Poultry Sci 82:627–631
Lei X, Piao X, Ru Y, Zhang H, Péron A, Zhang H (2015) Effect of bacillus amyloliquefaciens-based direct-fed microbial on performance, nutrient utilization, intestinal morphology and cecal microflora in broiler chickens. Asian-Aust J Anim Sci 28:239–246
Lescheid DW (2014) Probiotics as regulators of inflammation: a review. J Funct Foods 4:299–311
Funding
This study is supported by the grants from the National Waterfowl Industry Technology System Foundation of China (No. CARS-43-11) and the National Key R&D Program “Application and Demonstration of Green Waterfowl Efficient Safe Aquaculture Technology” (2018YFD0501501).
Author information
Authors and Affiliations
Contributions
G.L., B.W., W.G., and M.Z. conceived and designed the experiments; G.L., W.G., and B.Y. performed the experiments; G.L. and Y.H. analyzed the data; and G.L., K.L., and M.K. wrote the paper. All of the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
This study was approved by the Animal Ethics Committee of the College of Animal Science and Technology, Qingdao Agricultural University, China.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, G., Wang, B., Huang, Y. et al. The Growth Rate, Immune Status, Duodenal Development, and Cecal Microbial Diversity of 24-Day-Old Offspring of SD Rats Received Bacillus subtilis-Cu or CuSO4 During Pregnancy and Lactation Periods. Biol Trace Elem Res 191, 435–442 (2019). https://doi.org/10.1007/s12011-019-1638-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-1638-5