Abstract
This study evaluated the influence of cellulose nanofiber (CNF) incorporated layer-by-layer water-resistant coating on consumer acceptance of blueberries and cherries processed in light syrup thermally (TP, 97 °C for 7 min) or non-thermally (high hydrostatic pressure (HHP), 414 MPa for 10 min). The coating technique was validated on canned blueberries in a commercial canning operation. Consumers (n = 75 and 77 for blueberry and cherry study, respectively) scored overall, color, aroma, and texture likeness of coated and uncoated processed fruits using a 9-point hedonic scale and firmness intensity using a 5-point just about right (JAR) scale. Coating treatment significantly (P < 0.05) increased aroma and texture liking scores of TP and HHP blueberries compared with uncoated ones, but did not impact overall and color liking scores. Coated TP (2.67) and HHP (2.96) blueberries received closer to JAR score in firmness that uncoated ones (1.88 and 2.57, respectively). Informing consumers about the benefit of developed coatings altered consumers’ attitude from negative (~ 80 and ~ 57% gave “neutral” and “disliking” scores on TP and HHP samples, respectively) into positive status (“I would like the overall appearance”). Coating treatment resulted in higher aroma liking scores in HHP cherries, but decreased (P < 0.05) overall and color liking scores on both TP and HHP cherries due to the presence of visible coating substances onto fruit surface. In commercial canning process, coated blueberries showed great retention of bioactive compounds and firmness. This study demonstrated that CNF incorporated layer-by-layer coating is promising to produce high quality of processed fruits in light syrup.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Natural anthocyanin pigments represent attractive red, purple, and blue color in plants, fruits, and vegetables (Li et al. 2015; Yousuf et al. 2015). Some in vitro and in vivo studies have proved the potential health benefits of anthocyanins with antioxidant capacity, cardio-protective effect, anti-inflammatory property, reduction in the risk of diabetes, and inhibition of tumor cell growth (Flores et al. 2015; Norberto et al. 2013). Unfortunately, these bioactive compounds are heat, oxygen, pH sensitive, and water soluble, and the pigments easily leach from the fragile berry fruit when thermally processed in aqueous media (i.e., canned fruit) (Jung et al. 2015), which in turn resulted in the significant loss of fruit quality (i.e., color, flavor, and texture) and health benefit.
Previously, authors had developed a cellulose nanofiber (CNF) incorporated layer-by-layer (LBL) water-resistant coating technique for preventing anthocyanin pigments degradation and leaching from simulated canned blueberries in the light syrup (Jung et al. 2015; Zhao et al. 2014). The technique was based on the principle that by reinforcing CNF into chitosan matrix, it could reduce water absorption and solubility of the coatings through hydrogen bonds and/or electrostatic interactions between CNF and chitosan, thus preserving fruits against the surrounding aqueous medium. This coating technique helped retain color, flavor, and texture of thermally processed blueberries. For assessing the potential commercial application of the developed coating technique, it is important to investigate consumers’ acceptance towards the sensory quality of processed fruits and the developed technique. Furthermore, it is necessary to validate the technique in a scaled-up commercial canning operation.
In this study, blueberries and cherries were prepared using our developed coating technique, and then filled in light syrup and processed thermally or non-thermally. Thermal process (canning) is effective to kill microorganisms for ensuring food safety and extending product shelf-life, but it could significantly destroy fruit color, flavor, and texture (Cavender and Kerr 2011; Patras et al. 2010). Non-thermal process using high hydrostatic pressure (HHP) has created new opportunities to provide the balance between food safety and quality (Plaza et al. 2012; Raso and Barbosa-Cánovas 2003). It was reported that HHP at < 600 MPa was unable to dissociate covalent bonds of low molecular weight compounds, such as pigment and flavor compounds (Tauscher 1995). It was thus hypothesized that HHP would offer better product quality for fragile blueberries or cherries in comparison with the thermal process (TP).
Therefore, the objectives of this study were (1) to investigate consumer acceptance of TP and HHP processed blueberries and cherries that were prepared using CNF incorporated LBL coating technique, and (2) to validate the durability of developed coating technique on blueberries in a commercial canning facility. Based upon our best knowledge, this study was the first attempt to evaluate the consumer acceptance of processed fruits using CNF incorporated coating technique. Results generated from this study would provide valuable information for directing future research in utilizing CNF incorporated LBL water-resistant coating for improving quality of processed fruits.
Materials and Methods
Materials
CNF slurry (2.95 g/100 g wet basis) was obtained from the Process Development Center of the University of Maine (ME, USA). CNF was extracted from northern bleached softwood kraft pulp using the Masuko MKZB15-50J super mass collider creating a high shear zone, thus liberating nanofibers present in natural lignocellulosic fibers with dimensions of 20–50 nm in width and up to several hundred microns in length (The Process Development Center, University of Maine, 2016). The following chemical reagents were used for the experiments: Folin−Ciocalteu’s (FC) phenol reagent (MP Biomedicals, LLC, Santa Ana, CA, USA), gallic acid (Sigma-Aldrich Co., LLC, St. Louis, MO, USA), acetic acid (Avantor Performance Materials International, Inc., Center Valley, PA, USA), L-ascorbic acid (Avantor Performance Materials International, Inc., Center Valley, PA, USA), 2, 2-diphenyl-1-picrylhydrazyl (DPPH) (95%) (Alfa Aesar, Ward Hill, MA, USA), hydrochloric acid (HCl) (EMD Chemicals, Inc., Gibbstown, NJ), sodium acetate (EMD Chemicals, Inc. Gibbstown, NJ, USA), potassium chloride (KCl) (EMD Chemicals, Inc., Gibbstown, NJ, USA), sodium carbonate (EMD Chemicals, Inc., Gibbstown, NJ, USA), trifluoroacetic acid (EMD Chemicals, Inc., Gibbstown, NJ, USA), and Tween 80 (Amresco, OH, USA). Analytical-grade methanol, ethanol, and acetone were used for the extraction of anthocyanins. Methyl cellulose (MC) and sodium alginate (SA) were obtained from Alfa Aesar (viscosity 1600 cPs, MA, USA) and TIC Gums, Inc. (White Marsh, MD, USA), respectively.
Coating Treatment for Blueberries and Cherries
Fresh highbush blueberries (Vaccinium corymbosum L.) were donated by a local berry grower in Oregon, USA, and fresh Bing cherries (Prunus avium L.) were purchased from a local market. Fruits were washed using Environne (Consumer Health Research, Inc., OR, USA) and dried to evaporate surface water at ambient conditions. Uncoated samples were transferred to glass jars or polyethylene bags with light syrup for TP and HHP, respectively, as controls. For coated samples, fruits were pretreated by soaking in 1% CaCl2 and 1% Tween 80 for 30–60 min, washed, and dried at ambient temperature. The purpose of pretreatment was to improve interactions of CaCl2 with pectin in the cell wall of fruit throughout the epicuticular wax layer (Schlegel and Schönherr 2002; Schönherr 2000), thus improving the stability of anthocyanins bound to fruit cell wall (Padayachee et al. 2013; Padayachee et al. 2012). The layer-by-layer (LBL) coating technique including three steps was further applied as follows (Jung et al. 2015): (1) pre-coating using 0.5% (w/w wet basis) sodium alginate + 0.5% (w/w wet basis) methyl cellulose to induce close attachment of coating materials onto fruit surfaces and dried at the ambient conditions under a fan, (2) CNF reinforced water resistance coating using 2% (w/w wet basis) chitosan dissolved in 1% (w/w wet basis) acetic acid + 0.5% (w/w wet basis) CaCl2 + 0.2% (w/w wet basis) CNF to protect fruit against surrounding liquid and processed conditions and dried at the ambient conditions under a fan, and (3) hydrogel formation by dipping above treated fruit in 0.5% (w/w wet basis) sodium alginate bath for 10 min and 3% (w/w wet basis) CaCl2 bath for 10 min, respectively, to preserve coating onto fruit within liquid. Coated fruits were also transferred into glass jars or polyethylene bags with light syrup for TP and HHP, respectively. The quantity of each coating ingredient in 100 g of coated fruit samples was estimated at ~ 11.5 mg sodium alginate, ~ 5.8 mg methyl cellulose, ~ 23.2 mg chitosan, ~ 11.6 mg acetic acid, ~ 34.9 mg CaCl2, and ~ 2.3 mg CNF (total weight of ~ 89.3 mg/100 g fruit or ~ 0.089% fruit weight). These numbers were based upon the coating thickness of ~ 0.05 mm onto fruit surface (~ 1-cm diameter of blueberry, and ~ 74 blueberries for 100-g sample).
Thermal and High Hydrostatic Pressure Processes of Fruit
Uncoated and coated fruit were subjected to TP and HHP processes. For TP, 6–8 pieces of blueberry or cherry were placed into Mason jars (~ 120-mL jar for blueberries and ~ 470-mL jars for cherries owing to their larger size of cherries) filled with 150 or 100 mL of 20% sucrose solution, respectively, with formic acid adjusted to pH 4.0. The jars were then processed in a water bath at 97 °C for 7 min, followed by cooling in tap water. For HHP, 15–20 pieces of blueberry or 10–15 pieces of cherry were packed in a polyethylene bag filled with 100 mL of 20% sucrose solution at pH 4.0, and processed at 414 MPa for 10 min in a 22-L high EPSI (Haverhill, MA, USA) HHP unit in conjunction with a Flow International (South Kent, WA, USA) 40 hp. intensifier. A pressure of 414 MPa was selected because it has been widely applied for various food products with pH below 4.5 to suppress microbial growth (Ramaswamy, Balasubramaniam, and Kaletunç 2010). The polyethylene bags were placed into the water filled 22-L high pressure vessel with ice to prevent temperature increase of samples during pressurization. HHP processed fruit were then transferred to sterilized jars (~ 470 mL) for sensory evaluation.
Recruiting of Consumers
Permission to carry out the sensory study was approved by the Institutional Review Board for the Protection of Human Subjects at the Oregon State University (OSU) (Study ID: 6910 - Development of Innovative Technologies to Retain Naturally-Occurring Anthocyanin Pigments and Their Bioactivities in Processed Whole Fruits). Consumers over 18 years old were recruited by email using the Sensory Science Laboratory database (OSU, Corvallis, Oregon, USA) and screened using criteria questions, such as frequency of consumption or purchasing for blueberry, cherry, or other fruit (fresh or processed). Before participating in the evaluation, eligible consumers were asked to sign a consent form. In this study, two groups of consumers were recruited, one for blueberry study (n = 75) and another for cherry study (n = 77). Sensory evaluation test was conducted in the Sensory Science Laboratory, Oregon State University (Corvallis, OR, USA).
Consumer Sensory Evaluation
Fruit samples were prepared 1 day prior to the day of the sensory study, stored at a 4 °C refrigerator after processing, and then warmed up to ambient temperature at the day of test before serving to consumers. Fruit samples were either placed in Mason jars or plastic cups (see details below) labeled with 3-digit random numbers, and placed inside a tray when presenting to consumers in isolated sensory tasting booths under white fluorescent light. Note that while CNF is prepared from natural wood fibers using mechanical method and is not toxic and safe based on the literatures (Endes et al. 2016), it is not “generally recognized as safe (GRAS)” by FDA for direct food application yet. Hence, consumers were not asked to eat the fruit coated with CNF, but observe and touch the samples.
The steps applied on the sensory study of blueberries are illustrated in Fig. 1. Study background information “Driven by health and aesthetic demands, there is an increasing market demand for colorful foods. However, in many cases, the state of the art is not capable of both preserving color and simultaneously retaining the healthy, flavorful and appealing nature of fruits during food processing” was firstly provided to the consumers. The consumers were asked to observe samples in Mason jars and rank the overall appearance and color liking using a 9-point hedonic scale (1 = dislike extremely, 5 = neither like nor dislike, and 9 = like extremely). A branch question “You indicated that you neither liked nor disliked the overall appearance of this product. If you knew this product preserved natural color, flavor, and texture in processed fruits without the use of chemical color additives, how would this influence your liking or disliking of the overall appearance of the product” was then asked to the consumer who ranked 5 (neutral) or below (disliking) in the overall appearance liking for coated fruits. This branch question was intended to investigate how consumers might change their attitude towards the products after knowing the “healthy” benefit of the product. The consumers were then asked to open the lid of served jar, smell and touch the samples for scoring aroma and texture liking using the 9-point hedonic scale. Firmness intensity was ranked using JAR scale (1 = much too soft, 2 = somewhat too soft, 3 = just about right, 4 = somewhat too firm, 5 = much too firm).
Illustration of the steps applied in the consumer sensory study of blueberries after thermal process (TP) and high hydrostatic pressure (HHP) process, and cross-tabulation percentage results in 9-point hedonic scale; 1–4 was considered the “negative” and “dislike” part of the scale; 5 was considered the “neutral” part of the scale; 6–9 was considered the “positive” and “like” part of the scale
For the cherry study, consumers were firstly asked to observe samples in Mason jar with a question “Does this product meet your expectations for naturally processed cherries without color additives?” and then a branch question “You indicated that this naturally processed cherry product did not meet your expectations for a good naturally processed cherry product without color additives. Please explain.” was asked to those who answered “No.” After that, consumers were asked to observe the samples again and rank the overall appearance and color liking using a 9-point hedonic scale as mentioned above. Furthermore, sample cups with 2–3 cherries per cup were provided to the consumers and asked them to smell and touch the samples, and score aroma and texture liking using the same 9-point hedonic scale. Firmness intensity of fruits was also evaluated using same JAR scale. Both blueberry and cherry studies were done in a 30 min of time period per session.
Validation of Coating in Canned Blueberries at a Commercial Canning Operation
Uncoated and coated blueberries were filled into the #300 can (269–302 g of fruits in each can) with 30–34 °Brix dilute high fructose corn syrup (~ 140 g). Cans were sealed and a total of 24 prepared cans were thermally processed in a commercial fruit canning facility (Oregon, USA) that operated at ~ 91 °C for 5 min in vacuum pressure of 127–508 mmHg (the center temperature of cooked cans was ~ 91 °C), and then cooled down in a cold water tank for about 12–18 min to reach ~ 35 °C. Cans were stored in a 4 °C refrigerator and analyzed on the quality of both fruits and filling syrup after 1 week of storage.
Evaluation of Selected Quality Parameters of Fruit and Filling Syrup
Soluble solid content (TSS, °Brix) and titratable acidity (TA) of fruits and filling syrup were measured using the methods of Fisk et al. (2008). Briefly, fruits and syrup (~ 10 g) were weighed, mixed with distilled water at a 1:9 (w/w) ratio, and blended for 1 min using a kitchen blender (Osterizer, Jarden Corp., Mexico). The mixture was filtered through a filter paper (Whatman No. 1) to remove insoluble materials. TSS of the filtrates was measured using a refractometer (Brix RA-250 HE, Kyoto Electronics Manufacturing Co., Ltd., Kyoto, Japan). The filtrate was also titrated with a standardized 0.1 N aqueous NaOH solution up to an end point of pH 8.2 using a pH meter (Corning 125, Corning Science Products, Tewksbury, MA). TA value was calculated using citric acid as the predominant acid in blueberries. Firmness of fruits was determined using a texture analyzer (TA.XT2, Stable Micro Systems, Inc., UK). Individual fruit was compressed for a total deformation of 3 mm between two parallel plates at a speed of 0.5 mm/s. The stem−calyx axis of blueberries was placed in parallel onto the compression plates. The highest peak was marked as the firmness (N). Color (L*: lightness, a*: redness, and b*: yellowness) and haziness of syrup placed in a 5-mm length quartz cell were evaluated by a ColorQuest HunterLab Spectrophotometer (Hunter Associates Laboratories Inc., VA, USA). Chroma was calculated by \( \sqrt{a^{\ast 2}+{b}^{\ast 2}}. \)Light going through a solution containing large size soluble molecules can be scattered, which gave a “haze” perception of the solution (Ngo and Zhao 2009). Pour-plate method using plate count agar was used to determine the total plate counts in the blueberry sample (Duan et al. 2010). Briefly, fruits (~ 10 g) were put into a sterile sampling bag, diluted with 90 mL of sterilized 0.1% peptone water, and homogenized using a stomacher type pulverizer (Stomacher 400 Circulator, Seward, London, UK) at 230 rpm for 2 min. The serial dilutions of fruit homogenates were spread onto plate count agar (HiMedia Laboratories Pvt. Ltd., India) and incubated at 37 °C for 16–24 h, and the colonies were counted. All tests were run in triplicates.
Analysis of Total Phenolic Content in Fruit and Filling Syrup
Fruit samples were extracted using 60% methanol at a 1:10 (sample/methanol) ratio for 20 min using a sonication unit (B-200H, Branson, Danbury, CT, USA) in 50-mL centrifuge tubes based on the optimized extraction conditions developed by our laboratory (Wang et al. 2016). The obtained extracts were centrifuged (International Equipment Co., Boston, MA, USA) at 10,000×g and 4 °C for 10 min and filtrated through a Whatman No. 1 filter paper. Methanol was evaporated with a vacuum rotary evaporator (Brinkmann Instruments, Westbury, NY, USA) at 40 °C, and resulting extracts were re-dissolved in citric buffer (pH 3.5) in a 25-mL volumetric flask. The extracts were stored in a − 80 °C freezer (VWR International LLC, Radnor, PA, USA) until further analysis. Syrup samples (10 g) were also blended with 90 mL of distilled water and filtered using the Whatman No. 1 filter paper to get rid of insoluble suspended materials. Total phenolic content (TPC) was measured using the FC reagent-based colorimetric assay (Singleton and Rossi 1965). TPC was calculated as gallic acid equivalent (GAE) and reported as mg/100 g of fresh fruits. Total monomeric anthocyanin (TMA) content was determined using the pH-differential method (Lee et al. 2005) and expressed as mg cyanide-3-glucoside/100 g of fresh fruits with a molecular weight of 449.2 g/mol and a molar absorptivity of 26,900.
Experimental Design and Statistical Analysis
For the sensory study, the significant difference between uncoated and coated fruits within each process (TP and HHP, respectively) was analyzed by a paired t test (P < 0.05) (Microsoft Excel, Redmond, WA, USA).
For the commercial canning operation, a completely randomized design with two levels (uncoated vs. coated) was applied. The significant difference between uncoated and coated fruit was also analyzed by a paired t test (P < 0.05).
Results and Discussion
Consumer Acceptance on the Sensory Quality of Processed Blueberries
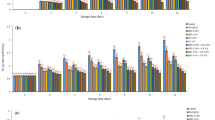
Consumer acceptance on the sensory quality of processed blueberries was compared between coated and uncoated fruits after TP and HHP process (Table 1). For TP samples, there was no difference in overall and coloring liking scores between coated and uncoated blueberries, but coated fruits showed significantly (P < 0.05) higher aroma and texture liking scores in comparison with uncoated one. In relation to texture, coated fruits (2.67) received close to JAR (3.0) score, compared with uncoated fruits (1.88). HHP samples received similar sensory score to that of TP samples, in which coated fruits had higher aroma and texture liking scores as well as close (2.96) to JAR score. After informing consumers about benefits of the coating (Fig. 1), among the 40% of the consumers who initially ranked overall liking scores within 1–5 (Fig. 2), ~ 28 and ~ 56% of them changed their attitude towards the products and selected “I would like the overall appearance much more” and “I would like the overall appearance somewhat more”, respectively. Only ~ 16% of them selected that “This statement would not influence my liking or disliking of overall appearance.” For HHP samples, among the 19% of consumers who initially ranked overall liking scores within 1–5, 7 and 50% of them changed their attitude by selecting “I would like the overall appearance much more” and “I would like the overall appearance somewhat more,” respectively. Hence, consumer education about the developed coating technique could potentially impact their purchase intent and acceptance towards new technology and products.
Changes of consumers’ ranking on overall appearance liking of coated and processed blueberries after informing them about the benefit of coatings; thermal process (TP) was conducted at 97 °C for 7 min; high hydrostatic pressure (HHP) process was carried at 414 MPa for 10 min; inset indicated the percentage of consumers ranked 1–5 scores as “neutral” and “dislike” in a 9-point hedonic scale of overall appearance liking for TP and HHP blueberries before informing them about the coating benefit
Consumer Acceptance on the Sensory Quality of Processed Cherries
Consumer acceptance on the sensory quality of uncoated and coated cherries subjected to TP or HHP process is reported in Table 2. For TP samples, uncoated fruits received higher scores in overall liking and color liking (P < 0.05) but had no difference in aroma and texture liking from uncoated fruits. In relation to texture, both coated (3.14) and uncoated (2.88) cherries received close to JAR score. For HHP samples, uncoated fruits retained higher (P < 0.05) overall liking and color liking scores than that of coated fruits, whereas coated fruits had higher aroma liking score than that of uncoated fruits. This result was similar to that of blueberries, demonstrating the benefit of coating for preserving fruit aroma. No difference in texture liking and texture JAR score was observed between coated and uncoated cherries. Regarding to consumers’ response to the branch question (Fig. 3), for TP cherries, ~ 55% consumers answered “Yes” (satisfied) on coated fruits. However, ~ 45% consumers answered “No” (not satisfied) on coated fruits because of “visible film or coating onto fruit surface,” “too light/pale syrup,” “greyish fruit surface,” “not appealing fruit and syrup color,” and “watery syrup.” For HHP cherries, ~ 72% consumers answered “Yes” to coated fruits, indicating that more consumers were satisfied with HHP coated cherries in comparison with TP ones. However, ~ 28% were still unsatisfied with coated cherries due to the following reasons: “visible film/coating,” “bubbles on fruit surface,” and “light liquid color.” These results demonstrated that visible coating substances onto cherry surface could impact consumers’ satisfaction.
The principal component analysis (PCA) was applied to further observe the trends in sensory data and to understand the coordination among eight different types of fruit product (coated and uncoated blueberries and cherries after TP or HHP process) and consumers’ acceptance on fruit sensory quality (Fig. 4). Figure 4a, b shows the projection of the samples on the plane defined by the first and second principal components and the corresponding loading plot, respectively. The first and second principal components described 88.4% of the variability (49.2 and 39.1%, respectively). It was seen that overall and texture liking scores were positively related to color liking and JAR, respectively, but aroma liking score was negatively related to overall liking score (Fig. 4a). These results indicated that the higher aroma liking of coated fruits had no positive impact on overall liking score. Three samples, including uncoated HHP cherries, coated HHP blueberries, and uncoated TP cherries, were characterized by higher overall liking and color liking scores, and coated HHP cherries, coated TP cherries, and coated TP blueberries were grouped by higher aroma liking scores (Fig. 4b). These trends were consistent with the results in consumer acceptances on sensory qualities of each product reported in Tables 1 and 2.
Quality Characteristics of Canned Blueberries Through the Commercial Canning Operation
Our previous study demonstrated that the developed LBL coating was not solubilized in filling syrup and stable during high temperature process (Jung et al. 2015), thus helping retain anthocyanin pigments and firmness of processed fruits. This study further validated the developed coating technique in a commercial canning operation by evaluating selected quality parameters of processed blueberries and filling syrup after 1 week of storage at 4 °C (Table 3). Based on the visual observation (photos in Table 3), more anthocyanin pigments (deep purple color observed in the stainless bowl) were leached to filling syrup from uncoated blueberries in comparison with that from coated fruits. TA (%) and firmness of coated blueberries were significantly (P < 0.05) higher than those of uncoated one, whereas TA of filling syrup with coated blueberries was significantly (P < 0.05) higher; TSS, TPC, and TMA were significantly lower than those in syrup with uncoated ones, meaning that coating reduced the leaching of soluble solids and bioactive compounds from coated fruits into filling syrup. Consistently, filling syrup with coated fruits had lower Chroma (i.e., color intensity) and haziness (i.e., impurities) values than that with uncoated fruits, again indicating less color leaching from fruits into the syrup. These results supported that coating effectively preserved bioactive compounds, color and firmness of canned blueberries. The higher TA value of both fruit and filling syrup in coated sample was probably because the acetic acid (1%) used for dissolving chitosan penetrated into fruits and released to the syrup after the thermal process. On the other hand, the coated samples contained lower TPC and TMA than that of uncoated fruits although the differences were not statistically (P > 0.05) significant. These results might be owning to two reasons: (1) the amount of extracted TMA from the coated fruits was underestimated since coating materials might be partially counted into the total weight of fruit sample and (2) the phenolic compounds was present as bound form with less dissociation during the thermal process in coated fruits (Lin et al. 2016). In addition, the commercial canning process was effective to suppress the bacterial growth in canned blueberries, showing no bacterial detection in all processed samples. Hence, this study validated that developed LBL water-resistant coating technique is applicable for preserving color, bioactive compounds, and firmness of fruits in light syrup subjected to thermal or high hydrostatic pressure process. It should be also noted that the production cost and consumer education are critical factors for transferring the developed technique to the commercial application.
Conclusions
Consumer acceptance and quality of processed blueberries or cherries subjected to our previously developed cellulose nanofiber (CNF) incorporated layer-by-layer (LBL) water-resistant coating were evaluated in this study. Consumer acceptance study demonstrated that coating improved texture and aroma liking scores of HHP or TP blueberries, and increased aroma liking of HHP cherries in comparison with uncoated fruits. It was also found that consumer’s education about the benefit of coating technique is important for improving their acceptability towards the new technique and fruit products. Consumers who were not satisfied with coated cherries mostly had concerns on the presence of visible coating substances on the fruit surface. Hence, the quality of coatings needs to be improved to be less visible and more homogenous onto fruit surface. Based on the results from the studies of consumer sensory evaluation and validation in commercial canning operation, CNF incorporated coating have a potential for preserving color, aroma, and texture of processed fruits, a technique challenge where processed fruit industry has been facing for long term. Currently, however, CNF is not generally recognized as safe (GRAS) yet, thus limiting its commercial applications in the food industry. More studies are also necessary to improve the current coating formulation on cherries for providing better appearance onto cherry surface and to validate the coating performance on cherries at commercial operation conditions.
References
Cavender, G. A., & Kerr, W. L. (2011). Inactivation of vegetative cells by continuous high-pressure processing: new insights on the contribution of thermal effects and release device. Journal of Food Science, 76(7), E525–E529.
Duan, J., Cherian, G., & Zhao, Y. (2010). Quality enhancement in fresh and frozen lingcod (Ophiodon elongates) fillets by employment of fish oil incorporated chitosan coatings. Food Chemistry, 119(2), 524–532.
Endes, C., Camarero-Espinosa, S., Mueller, S., Foster, E. J., Petri-Fink, A., Rothen-Rutishauser, B., Weder, C., & Clift, M. J. D. (2016). A critical review of the current knowledge regarding the biological impact of nanocellulose. Journal of Nanobiotechnology, 14(1), 78.
Fisk, C. L., Silver, A. M., Strik, B. C., & Zhao, Y. (2008). Postharvest quality of hardy kiwifruit (Actinidia arguta ‘Ananasnaya’) associated with packaging and storage conditions. Postharvest Biology and Technology, 47(3), 338–345.
Flores, G., Ruiz del Castillo, M. L., Costabile, A., Klee, A., Bigetti Guergoletto, K., & Gibson, G. R. (2015). In vitro fermentation of anthocyanins encapsulated with cyclodextrins: release, metabolism and influence on gut microbiota growth. Journal of Functional Foods, 16, 50–57.
Jung, J., Cavender, G., Simonsen, J., & Zhao, Y. (2015). Investigation of the mechanisms of using metal complexation and cellulose nanofiber/sodium alginate layer-by-layer coating for retaining anthocyanin pigments in thermally processed blueberries in aqueous media. Journal of Agricultural and Food Chemistry, 63(11), 3031–3038.
Lee, J., Durst, R. W., & Wrolstad, R. E. (2005). Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: collaborative study. Journal of AOAC International, 88(5), 1269–1278.
Li, D., Wang, P., Luo, Y., Zhao, M., & Chen, F. (2015). Health benefits of anthocyanins and molecular mechanisms: update from recent decade. Critical Reviews in Food Science and Nutrition, 57, 1729–1741.
Lin, Z., Fischer, J., & Wicker, L. (2016). Intermolecular binding of blueberry pectin-rich fractions and anthocyanin. Food Chemistry, 194, 986–993.
Ngo, T., & Zhao, Y. (2009). Stabilization of anthocyanins on thermally processed red D’Anjou pears through complexation and polymerization. LWT - Food Science and Technology, 42(6), 1144–1152.
Norberto, S., Silva, S., Meireles, M., Faria, A., Pintado, M., & Calhau, C. (2013). Blueberry anthocyanins in health promotion: a metabolic overview. Journal of Functional Foods, 5(4), 1518–1528.
Padayachee, A., Netzel, G., Netzel, M., Day, L., Mikkelsen, D., & Gidley, M. J. (2013). Lack of release of bound anthocyanins and phenolic acids from carrot plant cell walls and model composites during simulated gastric and small intestinal digestion. Food & Function, 4(6), 906–916.
Padayachee, A., Netzel, G., Netzel, M., Day, L., Zabaras, D., Mikkelsen, D., & Gidley, M. J. (2012). Binding of polyphenols to plant cell wall analogues—Part 1: anthocyanins. Food Chemistry, 134(1), 155–161.
Patras, A., Brunton, N. P., O'Donnell, C., & Tiwari, B. K. (2010). Effect of thermal processing on anthocyanin stability in foods; mechanisms and kinetics of degradation. Trends in Food Science & Technology, 21(1), 3–11.
Plaza, L., Colina, C., Ancos, B.d., Sánchez-Moreno, C., Pilar Cano, M., 2012. Influence of ripening and astringency on carotenoid content of high-pressure treated persimmon fruit (Diospyros kaki L.). Food Chemistry 130, 591–597, 3.
Ramaswamy, R., Balasubramaniam, V. M., & Kaletunç, G. (2010). Appendix 1: High pressure processing nonthermal processing technologies for food (pp. 593–598): Wiley-Blackwell.
Raso, J., & Barbosa-Cánovas, G. V. (2003). Nonthermal preservation of foods using combined processing techniques. Critical Reviews in Food Science and Nutrition, 43(3), 265–285.
Schlegel, T. K., & Schönherr, J. (2002). Stage of development affects penetration of calcium chloride into apple fruits. Journal of Plant Nutrition and Soil Science, 165(6), 738–745.
Schönherr, J. (2000). Calcium chloride penetrates plant cuticles via aqueous pores. Planta, 212(1), 112–118.
Singleton, V. L., & Rossi, J. A. (1965). Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture, 16, 144–158.
Tauscher, B. (1995). Pasteurization of food by hydrostatic high pressure: chemical aspects. Z Lebensm Unters Forch, 200(1), 3–13.
Wang, W., Jung, J., Tomasino, E., & Zhao, Y. (2016). Optimization of solvent and ultrasound-assisted extraction for different anthocyanin rich fruit and their effects on anthocyanin compositions. LWT - Food Science and Technology, 72, 229–238.
Yousuf, B., Gul, K., Wani, A. A., & Singh, P. (2015). Health benefits of anthocyanins and their encapsulation for potential use in food systems: a review. Critical Reviews in Food Science and Nutrition, (13), 2223–2230.
Zhao, Y., Simonsen, J., Cavender, G., Jung, J., Fuchigami, L.H., 2014. Patent US 9826750B2, Nano-cellulose coatings to prevent damage in foodstuffs.
Acknowledgements
The authors thank the Process Development Center at the University of Maine for providing CNF.
Funding
This study was funded by the Oregon State University Venture Development Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, J., Simonsen, J., Wang, W. et al. Evaluation of Consumer Acceptance and Quality of Thermally and High Hydrostatic Pressure Processed Blueberries and Cherries Subjected to Cellulose Nanofiber (CNF) Incorporated Water-Resistant Coating Treatment. Food Bioprocess Technol 11, 1412–1421 (2018). https://doi.org/10.1007/s11947-018-2114-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-018-2114-5