Abstract
Purpose of Review
With improvements in cancer treatment outcomes and an increase in cancer survivorship, understanding the importance of fertility preservation options prior to undergoing cancer treatment is essential. Therefore, we review herein the effect of cancer and its treatment on male fertility, the rationale for sperm cryopreservation, options for sperm retrieval, ART outcomes, and experimental options.
Recent Findings
Recent data update fertility outcomes with newer cancer therapies and provide longitudinal insight into survivor paternity with and without fertility preservation. Likewise, updated ART outcomes and future preservation options are discussed.
Summary
The effect of cancer and its treatment on spermatogenesis is well established. Sperm cryopreservation is the best pre-treatment insurance for the opportunity of future fertility. Post-therapy patients may also achieve fertility restoration with ART, using cryopreserved or freshly obtained sperm. Meanwhile, utilization of cryopreserved testicular stem cells for future transfer or for in vitro maturation represents exciting alternatives on the horizon.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past several decades, cancer survival rates in the United States (US) have significantly improved despite a rising incidence of all cancers diagnosed in those younger than 65 years of age [1]. Specifically, 9% of cancer diagnoses in the US are in those younger than 45 and 1% in those younger than 20 years of age [2]. Improvements in survival are attributable to advances in cancer therapies, giving rise to the concept of cancer survivorship [3]. Over the same period, the age of achieving paternity has grown 30% in men between ages 35 and 54, highlighting the significance of our society’s desire to maintain fertility across a much greater spectrum of age [4]. Therefore, an important co-endpoint of cancer survivorship is the ability to maintain fertility potential after treatment [5].
Prior to cancer treatment, some patients demonstrate subfertility due to direct effects of the disease [6]. Likewise, cancer treatments have a negative impact on fertility. In recent years, the advancement of assisted reproductive techniques (ART) has allowed conception for many patients with previously untreatable infertility conditions [7]. ARTs have been utilized to achieve conception for patients following cancer treatment with highly successful outcomes. But due to the cost, complexity and morbidity, clinicians should aim to only use ARTs when necessary.
The best insurance for post-treatment fertility potential is to attempt fertility preservation prior to initiation of treatment while not impeding access to treatment or overall survival [8]. Guidelines from the American Society of Clinical Oncology (ASCO) and other medical societies underscore such an approach [9,10,11]. The majority of surveyed oncologists (91%) agree with these guidelines, but less than half discuss fertility preservation options or fertility specialist referral with their patients prior to beginning cancer treatment [12]. Likewise, a patient’s desire for fertility preservation must not be assumed based upon age as 20% of surveyed men preparing for prostatectomy surgery expressed desire for fertility preservation [13].
Therefore, the purpose of this review is to discuss the complex impact of cancer and its treatment on male fertility, the methods available to preserve fertility and when to employ them, and the success of ARTs after cancer treatment and highlight potential future fertility preservation options.
The Effect of Cancer on Male Fertility
Malignant neoplasms can have a multi-factorial, negative impact on normal spermatogenesis long before the diagnosis of cancer is made [8, 14]. An intact hypothalamic-pituitary-gonadal (HPG) axis is essential to tightly regulate the production of hormones necessary for normal spermatogenesis. HPG axis disruption can occur with testis tumor secretion of beta human chorionic gonadotropin (beta-HCG) and/or alpha-fetoprotein (AFP), decreasing gonadotropin production and total motile sperm count [15, 16]. Likewise, excess prolactin sourced from prolactinomas, tumor paraneoplastic syndromes, or as a consequence of radiotherapy for head and neck malignancies can disrupt the HPG axis or lead to sexual dysfunction [17, 18]. Similarly, direct tumor invasion of the hypothalamus, pituitary gland, or testis and be detrimental to fertility [19]. Moreover, there is an association between baseline testicular dysfunction, as seen with cryptorchidism, testicular dysgenesis syndrome or non-malignancy-associated infertility, and an increased risk for cancer, leading to recommendations for increased cancer screening in these populations [20, 21].
Gonadal dysfunction may also be a consequence of systemic manifestations of malignancy including fever, malnutrition, and stress. Malignancies can induce a systemic inflammatory state mediated by secretion of metabolically active cytokines, manifesting in constitutional symptoms that may directly damage the germinal epithelium [22]. Gonadal temperature regulation is essential for normal spermatogenesis, and gonadal dysfunction can persist up to 3 months following an episode of high fever [23]. High fever and constitutional symptoms are a hallmark of lymphoma and several studies have shown a positive correlation between fever severity and derangement of semen parameters in lymphoma patients prior to initiating chemotherapy [24].
The Impact of Cancer on Sperm Characteristics
The negative effect of malignancy on baseline semen parameters prior to undergoing treatment has been well described across several cancer diagnoses [6, 25••, 26••, 27••, 28]. An initial report of 400 men diagnosed with various types of cancer seeking sperm cryopreservation found fertile and intermediate levels of sperm density and motility in most men respectively, except in those with testis cancer (TC) who demonstrated intermediate levels in both categories [6]. More recent series demonstrated lower sperm concentrations and fewer banked vials in those with TC, Hodgkin’s lymphoma (HL), and hematologic malignancies compared with other cancer diagnoses and healthy controls [25••, 26••]. No correlation between semen quality and cancer stage was observed [25••], and lower semen volume was noted in only one series of testis and hematopoietic malignancy patients [26••]. In the most recent series from 2017, similar decreases in pre-treatment sperm concentration and motility were demonstrated in those with TC but not hematopoietic malignancies [27••]. Furthermore, 16.9% of patients overall were azoospermic regardless of diagnostic groups, but older patients at diagnosis were less likely to be azoospermic [27••].
Qualitative sperm assays have also demonstrated the negative effects of cancer on sperm production. Several series incorporating various deoxyribonucleic acid (DNA) fragmentation assays have demonstrated worse DNA and chromatin integrity despite normal semen parameters in cancer patients compared to healthy controls [28,29,30]. However, other series failed to confirm these findings, leaving the issue unresolved [31]. Similarly, a prospective series from 2017 of lymphoma patients evaluated before treatment revealed significantly higher rates of sperm aneuploidy compared with healthy controls, most commonly hyperhaploid XY and chromosome 18 disomy [32••]. Yet the significance of these genetic abnormalities is unknown. Registry data of children born to fathers diagnosed with cancer has not demonstrated any increase in peri-natal complications, whereas a small but clinically insignificant increase in congenital abnormalities from 3.2 to 3.7 incidents per 100 offspring was noted, predominantly in those born within 2 years of the father’s cancer diagnosis [33].
Fertility Impairment with Cancer Treatment
Improved cancer treatment outcomes have largely been achieved by utilizing a combination of surgery, chemotherapy, and radiotherapy [3]. Each treatment can harm normal male fertility by causing derangements of the HPG axis, cytotoxic effects to the germinal epithelium, surgical anatomic or functional deficits, and induction of emotional or sexual dysfunction [8].
Surgery
A direct effect of surgery on fertility potential occurs with orchiectomy usually performed for TC. Several series have demonstrated unilateral orchiectomy to result in reduction of sperm concentration and total sperm count in up to 85% of patients with another 9% developing azoospermia; this effect is more pronounced in men with non-seminomatous germ cell tumors [34, 35]. Nonetheless, nearly 40% of men on surveillance after orchiectomy regardless of implementation of treatment for relapse ultimately achieve paternity [34, 35].
Any extirpative surgery within the retroperitoneum or pelvis can lead to damage of the parasympathetic pelvic nerve controlling erectile function, sympathetic nerves of the hypogastric and pelvic plexuses controlling emission and ejaculation, and/or the male ductal system through which sperm pass during ejaculation. Preservation of ejaculatory and erectile function has improved with utilization of nerve-sparing techniques when oncologically appropriate (Table 1). For example, retroperitoneal lymph node dissection (RPLND) was historically associated with a high rate of ejaculatory dysfunction, but implementation of nerve-sparing templates has resulted in ejaculatory preservation for 90–100% of post-chemotherapy or primary RPLND cases respectively, with similar results reported for robotic RPLND [36, 37•].
Pelvic surgery for prostate, bladder or colorectal cancers can also result in temporary or permanent ejaculatory dysfunction as well as obstruction of bilateral vasa deferentia and/or erectile dysfunction. As with RPLND, implementation of nerve-sparing techniques for various oncologic pelvic surgeries has resulted in significant improvements for recovery of postoperative erectile function (Table 1), but the risk of permanent impotence or anejaculation remains [38,39,40, 41•]. As men are increasingly delaying fatherhood, oncologists must recognize those who wish to achieve paternity after cancer treatment and provide appropriate counseling regarding fertility preservation options [42••].
Chemotherapy
Chemotherapy is a systemic treatment that targets actively dividing cells, which can include both normal and malignant cells. The effect of chemotherapy on male fertility largely depends upon the agent, dose, duration, and combination used [9]. Rapidly dividing spermatocytes are the highest risk cells within the germinal epithelium [43]. Slower dividing spermatogonia are divided into type A (pale) and (dark) cells, with the former experiencing apoptosis at low chemotherapeutic doses and the latter serving as a stem cell pool for repopulation [43]. However, higher drug doses can also damage the type A (dark) spermatogonia, resulting in a Sertoli-cell-only pattern [44]. The more mature spermatocytes and spermatids are less chemo-sensitive given their lack of cell division and may explain why some sperm initially remain in the ejaculate after chemotherapy [43]. Similarly, Leydig cells maintain a low proliferation rate and are more chemo-resistant but can succumb to higher chemotherapeutic doses, resulting in elevated luteinizing hormone (LH) levels despite low-normal testosterone levels [45].
Several classes of chemotherapy agents are gonadotoxic (Table 2). Alkylating agents are the most toxic and reliably induce azoospermia, persisting in 25% of men 5 years after treatment [46,47,48,49]. Alkylating agent dose independently predicts the ultimate recovery of spermatogenesis regardless of higher dose single agent or lower dose in combination with other agents as shown with cyclophosphamide doses over 19 or 7.5 g/m2, respectively [48]. Recent data with a 21-year follow-up revealed rates of 25, 28, and 48% for azoospermia, oligospermia, and normospermia, with worse recovery predicted by cumulative alkylating dose defined by each 1000 mg/m2 of cyclophosphamide equivalent dose [47]. Platinum-based cisplatin and carboplatin have demonstrated equivalent gonadotoxicity to alkylating agents with 20 to 47% of low versus high dose groups experiencing azoospermia after 5 years [50]. Temporary reductions in semen parameters are observed with other chemotherapeutic agents by avoiding damage to spermatogonial stem cells, although additive gonadotoxicity with alkylating or platinum agents can occur [43]. Newer biologic targeted and immunomodulating therapies are cytostatic and reduce semen parameters sometimes to azoospermic levels during active treatment but recovery is expected after cessation. However, these drugs may disrupt the HPG axis and cause hormonal abnormalities [51,52,53,54].
Combination Chemotherapy Regimens
Optimal outcomes for several malignancies have been achieved through the use of combination chemotherapy regimens. Optimal outcomes for hematopoietic malignancies were initially seen using MOPP/MVPP (mechlorethamine, oncovorin/vincristine, procarbazine, prednisone), but 85% of patients developed azoospermia, hypogonadism, and gynecomastia with persistence up to 15 years later when used during childhood [55, 56]. Substitution of mechlorethamine for cyclophosphamide (COPP) demonstrated similar rates of azoospermia that persisted 11 years after treatment [46]. ABVD (adriamycin, bleomycin, vinblastine, dacarbazine) is a contemporary regimen that excludes alkylating agents and demonstrated 90% recovery of semen parameters by 24 months with no azoospermia compared to alkylating regimens [57••]. Likewise, all regimens have shown worse rates of DNA fragmentation and sperm aneuploidy following treatment compared to controls except ABVD, which has not shown worse sperm aneuploidy [32••, 57••].
Contemporary testis cancer management has resulted in excellent survival rates. The first-line regimen, BEP (bleomycin, etoposide, cisplatin), has been shown in several series to negatively impact semen parameters [14]. The most recent series demonstrated a significant decrease in all semen parameters following treatment, with recovery of normospermia occurring in less than half of patients by 24 months and 2% remaining azoospermic predicted by greater than 2 cycles of BEP, use of radiotherapy, or abnormal baseline semen parameters [58]. The same series also found worse DNA fragmentation rates at 6 months with BEP but recovery to normal levels versus controls by 24 months [58]. The latter finding is in contrast to previous data showing persistence of abnormal levels of DNA fragmentation at 24 months following BEP treatment [59].
Radiotherapy
Radiotherapy (XRT) can be used to directly target the testes, but more commonly is used to target malignancies residing in the retroperitoneum or pelvis or in preparation for stem cell or bone marrow transplant, and can be gonadotoxic in men of all ages. Despite testicular shielding, a significant quantity of scatter can nonetheless affect the testicles and result in gonadotoxicity [60]. Akin to chemotherapy, the rapidly dividing spermatocytes are the most sensitive and earliest to be effected by radiation doses as low as 0.1 Gray (Gy) [61, 62]. Additional doses up to 0.65 Gy may result in oligospermia or azoospermia, which may become permanent at doses over 1.2 Gy [61, 62]. The implementation of fractionated protocols using lower individual doses of radiation in a prolonged schedule has resulted in similar or worse gonadotoxicity compared to single, equivalent doses [63]. Similarly, suppression of spermatogenesis by using gonadotropin releasing hormone (GnRH) agonists to protect spermatocyte DNA during XRT has also been unsuccessful [64]. Spontaneous recovery of spermatogenesis may occur and has been observed by 18 months with 1 Gy doses, 30 months with 2–3 Gy doses, and 5 years for 4 Gy or greater [62]. Moreover, XRT to the hypothalamus or pituitary can disrupt the HPG axis, causing secondary hypogonadism and infertility [65]. Lastly, Leydig cells are more resistant to the effects of XRT, requiring doses of 20–30 Gy to produce primary hypogonadism necessitating testosterone therapy [62, 66]. However, doses below 20 Gy can still disrupt Leydig cell function, resulting in subclinical hypogonadism with elevated LH levels [67].
Paternity Following Cancer Treatment
Taken as a whole, these data establish the negative effect of cancer and its treatment on semen quantity and quality, but a more important measure of fertility is the achievement of paternity. Initial data from the Childhood Cancer Survival Study (CCSS) with over 5 years follow-up in 2010 revealed that treatment with chemotherapy or XRT resulted in a significantly lower likelihood of paternity compared with sibling controls (HR 0.56; 95% CI, − 0.49 to 0.63). Predictors of worse outcomes included higher cumulative alkylating agent dose, XRT doses over 7.5 Gy, testicular XRT, and the use of cyclophosphamide or procarbazine agents. Even in those without a history of XRT or alkylating agent exposure, cancer survivors were less likely to achieve paternity versus controls (HR 0.91; 95% CI, 0.73 to 1.14) [5]. Persistent difficulty for cancer survivors with achieving paternity was found in updated CCSS data from 2016 with a 10-year follow-up demonstrating a 38% pregnancy rate of 83% reported one live birth versus 62% and 90% for sibling controls (HR 0.63; 95% CI 0.58 to 0.69). The study also confirmed predictors of worse pregnancy and live birth rates, including alkylating agent and cisplatin use although XRT use was not included in the analysis [68••]. In summary, these data validate the utmost importance of fertility preservation prior to undergoing any cancer treatment.
Male Fertility Preservation: Sperm Cryopreservation
The Rationale for Sperm Cryopreservation
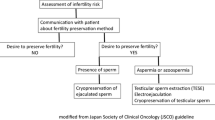
The need to preserve fertility prior to embarking upon cancer treatment is clear and supported by the ASCO guidelines. Obtaining ejaculated sperm for cryopreservation is a safe, non-invasive, and preferred method of male fertility preservation in all post-pubertal patients with success rates approaching 90% [69]. The initiation of puberty is critical to the success of sperm cryopreservation and often begins between the ages 12 and 14. Other correlates of spermatogenesis and an ability to ejaculate sperm include at least Tanner Stage II development, testis volume > 10 cm3 and nocturnal seminal emission [22]. Despite meeting these criteria, some patients may still have difficulty in producing an ejaculated specimen. Pre-pubertal patients not meeting these criteria generally are not candidates for sperm cryopreservation, but experimental alternatives could be considered after appropriate counseling.
Sperm cryopreservation is also possible in patients presenting with azoospermia in whom sperm can be subsequently identified in 22% using pellet analysis [70]. Indeed, only a single, motile sperm is needed for egg fertilization with ARTs and, therefore, one sample may be sufficient for cryopreservation; however, three samples collected 48 h apart are optimal [8]. Cryopreserved sperm can remain frozen for decades, with the longest duration of cryopreserved sperm resulting in a live birth reported at 28 years [71]. Furthermore, sperm cryopreservation can provide peace of mind. In a recent survey of patients given the option of sperm cryopreservation, 50% chose to bank sperm of which 80% felt encouraged or empowered by their decision. The study also revealed that 70% of patients post-chemotherapy desired paternity and 80% would recommend sperm cryopreservation to a friend [72].
In contrast to these compelling data, over 50% of surveyed oncologists do not routinely discuss sperm cryopreservation despite over 90% admitting sperm cryopreservation should be offered to all patients [12]. This was confirmed in a recent series where only 29% of cancer patients appropriate for fertility preservation were provided counseling and 11% underwent referral for sperm banking [73••]. Those with a lower median income, Medicaid insurance, or who were older in age were less likely to be provided fertility preservation counseling [73••]. Other similarly designed studies have confirmed that older patients and those already with children are less likely to bank, whereas only 24–30% choose to bank even if appropriately counseled [74, 75••, 76]. Other commonly cited reasons for not banking include lack of interest, anxiety, immaturity, embarrassment, sexual orientation, religious background, and cost; many of which can be mitigated through the implementation of a formalized oncofertility program [8, 22, 77].
ART Outcomes Using Cryopreserved Sperm
Despite the use of cryopreserved sperm resulting in ART success with reported paternity rates of 49–82%, only 6–18% of those choosing sperm cryopreservation ultimately utilize their sperm [75••, 78••]. Additionally, only 16% of patients discard their samples, implying that many couples may have not reached a point at which they are ready to pursue parenthood [78••]. More recent reports have revealed a 26% pregnancy rate and 36% live birth rate using cryopreserved sperm for ICSI [26••]. Indeed a recent systematic review demonstrated an aggregate pregnancy and live birth rate of 13 and 8% respectively with IUI versus 30 and 25% respectively with IVF/ICSI [78••]. These data collectively articulate the fertility success that can be achieved with sperm cryopreservation.
Sperm Retrieval in those with Ejaculatory Dysfunction
Retrograde Ejaculation
There are several scenarios in which patients may have difficulty or inability to ejaculate, and special techniques can be employed after careful consideration of the underlying etiology. In those with a low volume ejaculate, intact sensation of orgasm, history of pelvic or retroperitoneal surgery (RPLND) or certain medical conditions (diabetes), retrograde ejaculation may ensue and can be diagnosed with a post-ejaculate urine (PEU) analysis. For these patients, medical treatment with sympathomimetic medications (pseudoephedrine or imipramine) may improve bladder neck contraction and result in anterograde ejaculation. In more severe cases, a retrograde ejaculate can be obtained via PEU after urine alkalinization (sodium bicarbonate or potassium citrate) for 2–3 days. The quantity and quality of retrieved sperm is usually sufficient for intrauterine insemination (IUI) or in vitro fertilization (IVF) with intracytoplasmic sperm injection (ICSI) or can be cryopreserved for later use [3, 8].
Anejaculation
In peri- or post-pubertal patients with anorgasmia or anejaculation (no evidence of retrograde ejaculation on PEU) or those with a history of cancer treatment, pelvic or retroperitoneal surgery, or underlying medical or neurologic disorder, neurostimulatory methods can be implemented, including penile vibratory stimulation (PVS) or electroejaculation (EEJ). Surgical sperm retrieval (SR) is necessary in cases refractory to neurostimulatory methods.
PVS is simple, non-invasive, safe, and considered first-line as it can be performed at home or in the office without anesthesia, although use in peri-pubertal adolescents under anesthesia should be considered. PVS involves placement of one or two vibrating device(s) (FertiCare® Orion Medical Group, Silverado CA, USA) directly on the frenulum and/or dorsum of the penis until ejaculation occurs [3, 8, 79]. EEJ is utilized when PVS is not successful or appropriate and must be performed under general anesthesia. A Seager Electroejaculator® (Dalzell USA Medical Systems, The Plains, VA, USA) is used transrectally, providing direct electrical stimulation to the prostate and seminal vesicles, resulting in ejaculation in the majority of cases. When EEJ is not successful, concurrent testicular sperm extraction (TESE) can be performed concurrently under anesthesia [80].
The ejaculate retrieved via neurostimulatory methods typically demonstrates low volume, high sperm concentration, low motility, and low morphology. Nonetheless, such samples are often suitable for fresh or cryopreserved IUI or IVF/ICSI, often resulting in pregnancy success [3, 8, 80]. Specifically in young men with cancer who are unable to ejaculate or with non-obstructive azoospermia (NOA), EEJ has demonstrated higher SR rates (60%) for cryopreservation compared to TESE (33%) with no reported complications or delay in initiation of definitive cancer therapy [81].
Surgical Sperm Retrieval
Indications for surgical SR for fertility preservation include those patients with azoospermia, peri-pubertal patients with anejaculation or when neurostimulatory methods are not successful or available. As mentioned, cancer patients often demonstrate deranged semen parameters prior to initiating treatment. Although rare, obstructive azoospermia (OA) may be organic or due to previous cancer treatment and should be distinguished from NOA by an appropriate clinical evaluation demonstrating small/soft testes, normal volume fructose positive semen, and elevated gonadotropins in NOA patients.
Percutaneous Versus Open Approaches
Surgical SR options are categorized by percutaneous and open approaches. Percutaneous techniques are cheaper, less invasive, and easily performed in the office with local anesthetic; however, the blind nature by which samples are aspirated can lead to undue trauma or unsatisfactory results. A direct comparison of testicular sperm aspiration with TESE in NOA patients demonstrated significantly improved SR rates with TESE (11 versus 43%), confirmed in a 2015 meta-analysis illustrating SR rates two times more likely with TESE [82••, 83]. Similarly for OA patients, microscopic epididymal sperm aspiration (MESA) has demonstrated superiority over percutaneous approaches [7, 84]. In sum, open surgical techniques are considered the gold standard.
The ability to find sperm for IVF/ICSI in NOA patients using TESE was first described in the mid-1990s [85] and has evolved to include testis mapping or microdissection TESE (mTESE) techniques. Improvement in overall SR rates from 40 to 63% has been shown with mTESE, minimizing testicular tissue loss and bleeding complications [86, 87]. While mTESE is considered the gold standard for NOA patients, it does require an operative microscope and an experienced microsurgeon, potentially limiting generalizability.
ART Outcomes of Surgical Sperm Retrieval in Cancer Patients
For cancer patients presenting with NOA prior to treatment, the use of TESE has demonstrated SR rates ranging 33–45%; with higher rates observed in lymphoma (47%) and lower rates in late stage testis cancer (25%) groups [81, 88]. Others have utilized the time under anesthesia for orchiectomy to perform contralateral testis biopsy with cryopreservation of retrieved sperm called “onco-TESE” [88]. Onco-TESE has evolved to include a backtable dissection of the extirpated, tumor-containing testis, allowing identification of multiple areas of spermatogenesis in normal testicular tissue surrounding the tumor identified best using a mTESE over TESE technique [88,89,90]. Live births achieved using cryopreserved sperm from onco-TESE specimens have been reported up to 1 year following cancer treatment [89]. It is therefore important to attempt ejaculated sperm cryopreservation prior to undergoing orchiectomy to determine if onco-TESE needs to be performed during the same operative session [91].
For cancer survivors presenting with NOA following cancer treatment, the use of TESE up to 11 years after cancer treatment has demonstrated SR rates ranging 41.6–65%, with the largest series reporting 31% pregnancy and 30% live birth rates after several cycles of ICSI [44, 92]. More recently, the use of mTESE in 84 patients at a mean of 18 years after cancer treatment demonstrated a 43% SR rate, 57% fertilization rate, 50% pregnancy rate and 42% live birth rate using ICSI. Significant predictors of worse SR rates included Sertoli-cell-only pathology (38.2%) and alkylating agent exposure (21.4%) with hypospermatogenesis portending a 100% SR rate [93].
Despite the established ability to surgically obtain sperm for cryopreservation or for ART before or after cancer treatment, very few data have evaluated which approach is superior. The largest series to date evaluating 318 IVF/ICSI cycles (95% TESE, 5% MESA) performed using cryopreserved (88%) versus freshly obtained (12%) sperm reported a significantly improved fertilization rate with cryopreserved (60%) versus fresh (53.6%) sperm, and a non-statistically significant trend toward improved pregnancy rate with cryopreserved (23.6%) versus fresh (23%) sperm [94]. For those pursuing freshly obtained sperm, a waiting period of 2 years following cancer diagnosis and treatment should be implemented given the slightly increased risk of offspring congenital abnormalities [33]. Taken together, these data illustrate the importance of fertility preservation prior to undergoing treatment, as many unknowns exist, including the possibility of not surgically retrieving sperm in nearly half of NOA patients following cancer treatment.
Experimental Options for Fertility Preservation
Current methods for fertility preservation require mature spermatozoa for subsequent IVF/ICSI use. In peri-pubertal patients in whom sperm cannot be retrieved or pre-pubertal patients not having begun spermatogenesis, the only option is experimental use of immature testicular tissue (ITT) obtained via TESE [95••, 96]. Under approved research protocols and extensive patient counseling, some institutions allow the cryopreservation of ITT for later use, which has been chosen in over 90% of families given the option [96]. Current options for the use of cryopreserved ITT under investigation include isolation and autotransplant of germ cells, auto or xenotransplant of ITT, or perhaps the most promising, in vitro maturation of germ cells to produce spermatozoa for IVF/ICSI use [95••, 97]. Yet safety concerns exist over the co-transplant of malignant cells into a cured host, which has been reported with leukemia in animal models [98]. While these options are both exciting and promising, further research and clinical trials are needed. Meanwhile, the continued practice of ITT cryopreservation is recommended in the appropriate setting [99].
Conclusion
Patients with cancer often present with impaired spermatogenesis. Superimposed on this baseline defect are the iatrogenic effects of treatment via chemotherapy, radiotherapy, and surgery. Sperm cryopreservation is the best pre-treatment insurance for future fertility. However, post-therapy patients may also realize fertility restoration with ART when appropriate using cryopreserved or freshly obtained sperm. Meanwhile, utilization of cryopreserved testicular stem cells for future use via auto-transfer or in vitro maturation represents exciting alternatives on the horizon.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
SEER Cancer Statistics Review, 1975–2014 [database on the Internet]. National Cancer Institute. 2017. Available from: https://seer.cancer.gov/csr/1975_2014/. Accessed: April, 2017.
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62(1):10–29. https://doi.org/10.3322/caac.20138.
Kort JD, Eisenberg ML, Millheiser LS, Westphal LM. Fertility issues in cancer survivorship. CA Cancer J Clin. 2014;64(2):118–34. https://doi.org/10.3322/caac.21205.
Ventura SJ, Hamilton BE, Sutton PD. Revised birth and fertility rates for the United States, 2000 and 2001. Natl Vital Stat Rep. 2003;51(4):1–18.
Green DM, Kawashima T, Stovall M, Leisenring W, Sklar CA, Mertens AC, et al. Fertility of male survivors of childhood cancer: a report from the Childhood Cancer Survivor Study. J Clin Oncol. 2010;28(2):332–9. https://doi.org/10.1200/jco.2009.24.9037.
Williams DH, Karpman E, Sander JC, Spiess PE, Pisters LL, Lipshultz LI. Pretreatment semen parameters in men with cancer. J Urol. 2009;181(2):736–40. https://doi.org/10.1016/j.juro.2008.10.023.
Bernie AM, Ramasamy R, Stember DS, Stahl PJ. Microsurgical epididymal sperm aspiration: indications, techniques and outcomes. Asian J Androl. 2013;15(1):40–3. https://doi.org/10.1038/aja.2012.114.
Coward RM, Kovac JR, Smith RP, Lipshultz LI. Fertility preservation in young men treated for malignancies: options for precancer treatment. Sex Med Rev. 2013;1(3):123–34. https://doi.org/10.1002/smrj.13.
Lee SJ, Schover LR, Partridge AH, Patrizio P, Wallace WH, Hagerty K, et al. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J Clin Oncol. 2006;24(18):2917–31. https://doi.org/10.1200/jco.2006.06.5888.
Loren AW, Mangu PB, Beck LN, Brennan L, Magdalinski AJ, Partridge AH, et al. Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(19):2500–10. https://doi.org/10.1200/jco.2013.49.2678.
Munoz M, Santaballa A, Segui MA, Beato C, de la Cruz S, Espinosa J, et al. SEOM Clinical Guideline of fertility preservation and reproduction in cancer patients (2016). Clin Transl Oncol. 2016;18(12):1229–36. https://doi.org/10.1007/s12094-016-1587-9.
Schover LR, Brey K, Lichtin A, Lipshultz LI, Jeha S. Oncologists' attitudes and practices regarding banking sperm before cancer treatment. J Clin Oncol. 2002;20(7):1890–7. https://doi.org/10.1200/jco.2002.07.174.
Salonia A, Capogrosso P, Castiglione F, Russo A, Gallina A, Ferrari M, et al. Sperm banking is of key importance in patients with prostate cancer. Fertil Steril. 2013;100(2):367–72 e1. https://doi.org/10.1016/j.fertnstert.2013.03.049.
JW-C Cheng EK. Etiology of Cancer-Induced Male Infertility. In: A Majzoub AA, editor. The Complete Guide to Male Fertility Preservation. Springer International Publishing; 2018. p. 3–18.
Carroll PR, Whitmore WF Jr, Herr HW, Morse MJ, Sogani PC, Bajorunas D, et al. Endocrine and exocrine profiles of men with testicular tumors before orchiectomy. J Urol. 1987;137(3):420–3.
Hansen PV, Trykker H, Andersen J, Helkjaer PE. Germ cell function and hormonal status in patients with testicular cancer. Cancer. 1989;64(4):956–61.
El-Sakka AI, Hassoba HM, Sayed HM, Tayeb KA. Pattern of endocrinal changes in patients with sexual dysfunction. J Sex Med. 2005;2(4):551–8. https://doi.org/10.1111/j.1743-6109.2005.00082.x.
Ratnasingam J, Karim N, Paramasivam SS, Ibrahim L, Lim LL, Tan AT, et al. Hypothalamic pituitary dysfunction amongst nasopharyngeal cancer survivors. Pituitary. 2015;18(4):448–55. https://doi.org/10.1007/s11102-014-0593-6.
Sabanegh ES Jr, Ragheb AM. Male fertility after cancer. Urology. 2009;73(2):225–31. https://doi.org/10.1016/j.urology.2008.08.474.
Lip SZ, Murchison LE, Cullis PS, Govan L, Carachi R. A meta-analysis of the risk of boys with isolated cryptorchidism developing testicular cancer in later life. Arch Dis Child. 2013;98(1):20–6. https://doi.org/10.1136/archdischild-2012-302051.
Walsh TJ, Croughan MS, Schembri M, Chan JM, Turek PJ. Increased risk of testicular germ cell cancer among infertile men. Arch Intern Med. 2009;169(4):351–6. https://doi.org/10.1001/archinternmed.2008.562.
Trost LW, Brannigan RE. Oncofertility and the male cancer patient. Curr Treat Options in Oncol. 2012;13(2):146–60. https://doi.org/10.1007/s11864-012-0191-7.
Jung A, Schuppe HC. Influence of genital heat stress on semen quality in humans. Andrologia. 2007;39(6):203–15. https://doi.org/10.1111/j.1439-0272.2007.00794.x.
Marmor D, Elefant E, Dauchez C, Roux C. Semen analysis in Hodgkin's disease before the onset of treatment. Cancer. 1986;57(10):1986–7.
•• Caponecchia L, Cimino G, Sacchetto R, Fiori C, Sebastianelli A, Salacone P, et al. Do malignant diseases affect semen quality? Sperm parameters of men with cancers. Andrologia. 2016;48(3):333–40. https://doi.org/10.1111/and.12451. This is a large, single center, retrospective analysis of semen parameters in cancer patients presenting for sperm cryopreservation stratified by cancer diagnosis, demonstrating worse baseline parameters in testis cancer and lymphoma patients, confirming previous data.
•• Depalo R, Falagario D, Masciandaro P, Nardelli C, Vacca MP, Capuano P, et al. Fertility preservation in males with cancer: 16-year monocentric experience of sperm banking and post-thaw reproductive outcomes. Ther Adv Med Oncol. 2016;8(6):412–20. https://doi.org/10.1177/1758834016665078. This is the largest single center review of semen parameters in male cancer patients presenting for cryopreservation with ART outcome data using banked sperm for IVF/ICSI establishing fertilization, implantation, pregnancy and live birth outcomes.
•• DiNofia AM, Wang X, Yannekis G, Ogle S, Hobbie WL, Carlson CA, et al. Analysis of semen parameters in a young cohort of cancer patients. Pediatr Blood Cancer. 2017;64(2):381–6. https://doi.org/10.1002/pbc.26221. The largest review of sperm cryopreservation in peri-pubertal and post-pubertal male cancer patients demonstrating the feasibility and high success rate in this population.
O'Flaherty C, Vaisheva F, Hales BF, Chan P, Robaire B. Characterization of sperm chromatin quality in testicular cancer and Hodgkin's lymphoma patients prior to chemotherapy. Hum Reprod (Oxford, England). 2008;23(5):1044–52. https://doi.org/10.1093/humrep/den081.
Said TM, Tellez S, Evenson DP, Del Valle AP. Assessment of sperm quality, DNA integrity and cryopreservation protocols in men diagnosed with testicular and systemic malignancies. Andrologia. 2009;41(6):377–82. https://doi.org/10.1111/j.1439-0272.2009.00941.x.
Stahl O, Eberhard J, Cavallin-Stahl E, Jepson K, Friberg B, Tingsmark C, et al. Sperm DNA integrity in cancer patients: the effect of disease and treatment. Int J Androl. 2009;32(6):695–703. https://doi.org/10.1111/j.1365-2605.2008.00933.x.
McDowell S, Harrison K, Kroon B, Ford E, Yazdani A. Sperm DNA fragmentation in men with malignancy. Fertil Steril. 2013;99(7):1862–6. https://doi.org/10.1016/j.fertnstert.2013.02.015.
•• Martinez G, Walschaerts M, Le Mitouard M, Borye R, Thomas C, Auger J, et al. Impact of Hodgkin or non-Hodgkin lymphoma and their treatments on sperm aneuploidy: a prospective study by the French CECOS network. Fertil Steril. 2017;107(2):341–50 e5. https://doi.org/10.1016/j.fertnstert.2016.10.001. This study represents the largest, prospective cohort to date evaluating the impact of cancer and its treatment on sperm aneuploidy demonstrating worse aneuploidy rates with treatment lasting up to 2 years but returning to baseline beyond 2 years.
Stahl O, Boyd HA, Giwercman A, Lindholm M, Jensen A, Kjaer SK, et al. Risk of birth abnormalities in the offspring of men with a history of cancer: a cohort study using Danish and Swedish national registries. J Natl Cancer Inst. 2011;103(5):398–406. https://doi.org/10.1093/jnci/djq550.
Liguori G, Trombetta C, Bucci S, Benvenuto S, Amodeo A, Ocello G, et al. Semen quality before and after orchiectomy in men with testicular cancer. Arch Ital Urol Androl. 2008;80(3):99–102.
Petersen PM, Skakkebaek NE, Rorth M, Giwercman A. Semen quality and reproductive hormones before and after orchiectomy in men with testicular cancer. J Urol. 1999;161(3):822–6.
Masterson TA, Cary C, Rice KR, Foster RS. The evolution and technique of nerve-sparing retroperitoneal lymphadenectomy. Urol Clin N Am. 2015;42(3):311–20. https://doi.org/10.1016/j.ucl.2015.04.005.
• Abdul-Muhsin HM, L'Esperance JO, Fischer K, Woods ME, Porter JR, Castle EP. Robot-assisted retroperitoneal lymph node dissection in testicular cancer. J Surg Oncol. 2015;112(7):736–40. https://doi.org/10.1002/jso.24018. The authors present one of the largest multi-institutional series of primary and post-chemotherapy robotic RPLND demonstrating non-inferior outcomes to the open approach.
Asgari MA, Safarinejad MR, Shakhssalim N, Soleimani M, Shahabi A, Amini E. Sexual function after non-nerve-sparing radical cystoprostatectomy: a comparison between ileal conduit urinary diversion and orthotopic ileal neobladder substitution. Int Braz J Urol. 2013;39(4):474–83. https://doi.org/10.1590/s1677-5538.ibju.2013.04.04.
Capogrosso P, Salonia A, Briganti A, Montorsi F. Postprostatectomy erectile dysfunction: a review. World J Men's Health. 2016;34(2):73–88. https://doi.org/10.5534/wjmh.2016.34.2.73.
Haberman K, Wittig K, Yuh B, Ruel N, Lau C, Wilson TG, et al. The effect of nerve-sparing robot-assisted radical cystoprostatectomy on erectile function in a preoperatively potent population. J Endourol. 2014;28(11):1352–6. https://doi.org/10.1089/end.2014.0315.
• Liu Z, Huang M, Kang L, Wang L, Lan P, Cui J, et al. Prognosis and postoperative genital function of function-preservative surgery of pelvic autonomic nerve preservation for male rectal cancer patients. BMC Surg. 2016;16:12. https://doi.org/10.1186/s12893-016-0127-4. One of the largest rectal cancer series to date employing nerve sparing techniques demonstrating improved rate of erectile function recovery using nerve sparing techniques.
•• Conti SL, Eisenberg ML. Paternal aging and increased risk of congenital disease, psychiatric disorders, and cancer. Asian J Androl. 2016;18(3):420–4. https://doi.org/10.4103/1008-682x.175097. The authors present a concise review of the implications of advanced paternal age and overall increased risk for congenital abnormalities, cancer and psychiatric disorders.
Meistrich ML. Effects of chemotherapy and radiotherapy on spermatogenesis in humans. Fertil Steril. 2013;100(5):1180–6. https://doi.org/10.1016/j.fertnstert.2013.08.010.
Meseguer M, Garrido N, Remohi J, Pellicer A, Simon C, Martinez-Jabaloyas JM, et al. Testicular sperm extraction (TESE) and ICSI in patients with permanent azoospermia after chemotherapy. Hum Reprod (Oxford, England). 2003;18(6):1281–5.
Howell SJ, Shalet SM. Effect of cancer therapy on pituitary-testicular axis. Int J Androl. 2002;25(5):269–76.
Bokemeyer C, Schmoll HJ, van Rhee J, Kuczyk M, Schuppert F, Poliwoda H. Long-term gonadal toxicity after therapy for Hodgkin's and non-Hodgkin's lymphoma. Ann Hematol. 1994;68(3):105–10.
Green DM, Liu W, Kutteh WH, Ke RW, Shelton KC, Sklar CA, et al. Cumulative alkylating agent exposure and semen parameters in adult survivors of childhood cancer: a report from the St Jude Lifetime Cohort Study. Lancet Oncol. 2014;15(11):1215–23. https://doi.org/10.1016/s1470-2045(14)70408-5.
Meistrich ML, Wilson G, Brown BW, da Cunha MF, Lipshultz LI. Impact of cyclophosphamide on long-term reduction in sperm count in men treated with combination chemotherapy for Ewing and soft tissue sarcomas. Cancer. 1992;70(11):2703–12.
Waxman J, Terry Y, Rees LH, Lister TA. Gonadal function in men treated for acute leukaemia. Br Med J (Clin Res Ed). 1983;287(6399):1093–4.
Petersen PM, Hansen SW, Giwercman A, Rorth M, Skakkebaek NE. Dose-dependent impairment of testicular function in patients treated with cisplatin-based chemotherapy for germ cell cancer. Ann Oncol. 1994;5(4):355–8.
Huyghe E, Zairi A, Nohra J, Kamar N, Plante P, Rostaing L. Gonadal impact of target of rapamycin inhibitors (sirolimus and everolimus) in male patients: an overview. Transpl Int. 2007;20(4):305–11. https://doi.org/10.1111/j.1432-2277.2006.00423.x.
Longo I, Sanchez-Mateos P, Lazaro P, Longo N. Azoospermia in a patient receiving interferon alpha for a stage III melanoma. Acta Derm Venereol. 2002;82(5):389–90.
Shash E, Bassi S, Cocorocchio E, Colpi GM, Cinieri S, Peccatori FA. Fatherhood during imatinib. Acta Oncol (Stockholm, Sweden). 2011;50(5):734–5. https://doi.org/10.3109/0284186x.2011.577562.
Mariani S, Giona F, Basciani S, Brama M, Gnessi L. Low bone density and decreased inhibin-B/FSH ratio in a boy treated with imatinib during puberty. Lancet (London, England). 2008;372(9633):111–2. https://doi.org/10.1016/s0140-6736(08)61023-5.
van Beek RD, Smit M, van den Heuvel-Eibrink MM, de Jong FH, Hakvoort-Cammel FG, van den Bos C, et al. Inhibin B is superior to FSH as a serum marker for spermatogenesis in men treated for Hodgkin's lymphoma with chemotherapy during childhood. Hum Reprod (Oxford, England). 2007;22(12):3215–22. https://doi.org/10.1093/humrep/dem313.
Whitehead E, Shalet SM, Blackledge G, Todd I, Crowther D, Beardwell CG. The effects of Hodgkin's disease and combination chemotherapy on gonadal function in the adult male. Cancer. 1982;49(3):418–22.
•• Bujan L, Walschaerts M, Brugnon F, Daudin M, Berthaut I, Auger J, et al. Impact of lymphoma treatments on spermatogenesis and sperm deoxyribonucleic acid: a multicenter prospective study from the CECOS network. Fertil Steril. 2014;102(3):667–74 e3. https://doi.org/10.1016/j.fertnstert.2014.06.008. The authors present one of the largest and only prospective cohort of male cancer patients evaluated for sperm DNA damage demonstrating higher DNA fragmentation before and after cancer treatment.
Bujan L, Walschaerts M, Moinard N, Hennebicq S, Saias J, Brugnon F, et al. Impact of chemotherapy and radiotherapy for testicular germ cell tumors on spermatogenesis and sperm DNA: a multicenter prospective study from the CECOS network. Fertil Steril. 2013;100(3):673–80. https://doi.org/10.1016/j.fertnstert.2013.05.018.
O'Flaherty C, Hales BF, Chan P, Robaire B. Impact of chemotherapeutics and advanced testicular cancer or Hodgkin lymphoma on sperm deoxyribonucleic acid integrity. Fertil Steril. 2010;94(4):1374–9. https://doi.org/10.1016/j.fertnstert.2009.05.068.
Fraass BA, Kinsella TJ, Harrington FS, Glatstein E. Peripheral dose to the testes: the design and clinical use of a practical and effective gonadal shield. Int J Radiat Oncol Biol Phys. 1985;11(3):609–15.
Howell S, Shalet S. Gonadal damage from chemotherapy and radiotherapy. Endocrinol Metab Clin N Am. 1998;27(4):927–43.
Rowley MJ, Leach DR, Warner GA, Heller CG. Effect of graded doses of ionizing radiation on the human testis. Radiat Res. 1974;59(3):665–78.
Speiser B, Rubin P, Casarett G. Aspermia following lower truncal irradiation in Hodgkin's disease. Cancer. 1973;32(3):692–8.
Brennemann W, Brensing KA, Leipner N, Boldt I, Klingmuller D. Attempted protection of spermatogenesis from irradiation in patients with seminoma by D-Tryptophan-6 luteinizing hormone releasing hormone. Clin Investig. 1994;72(11):838–42.
Littley MD, Shalet SM, Beardwell CG, Ahmed SR, Applegate G, Sutton ML. Hypopituitarism following external radiotherapy for pituitary tumours in adults. Q J Med. 1989;70(262):145–60.
Shalet SM, Tsatsoulis A, Whitehead E, Read G. Vulnerability of the human Leydig cell to radiation damage is dependent upon age. J Endocrinol. 1989;120(1):161–5.
Sklar C. Reproductive physiology and treatment-related loss of sex hormone production. Med Pediatr Oncol. 1999;33(1):2–8.
•• Chow EJ, Stratton KL, Leisenring WM, Oeffinger KC, Sklar CA, Donaldson SS, et al. Pregnancy after chemotherapy in male and female survivors of childhood cancer treated between 1970 and 1999: a report from the Childhood Cancer Survivor Study cohort. Lancet Oncol. 2016;17(5):567–76. https://doi.org/10.1016/s1470-2045(16)00086-3. Very large retrospective cohort study evaluating the otehrwise poorly described outcome of pregnancy and live birth in male cancer survivors demonstrating lower rates overall and even worse rates in men treated with alkylating agents compared to sibling controls.
Menon S, Rives N, Mousset-Simeon N, Sibert L, Vannier JP, Mazurier S, et al. Fertility preservation in adolescent males: experience over 22 years at Rouen University Hospital. Hum Reprod (Oxford, England). 2009;24(1):37–44. https://doi.org/10.1093/humrep/den361.
Jaffe TM, Kim ED, Hoekstra TH, Lipshultz LI. Sperm pellet analysis: a technique to detect the presence of sperm in men considered to have azoospermia by routine semen analysis. J Urol. 1998;159(5):1548–50. https://doi.org/10.1097/00005392-199805000-00038.
Feldschuh J, Brassel J, Durso N, Levine A. Successful sperm storage for 28 years. Fertil Steril. 2005;84(4):1017. https://doi.org/10.1016/j.fertnstert.2005.05.015.
Saito K, Suzuki K, Iwasaki A, Yumura Y, Kubota Y. Sperm cryopreservation before cancer chemotherapy helps in the emotional battle against cancer. Cancer. 2005;104(3):521–4. https://doi.org/10.1002/cncr.21185.
•• Grover NS, Deal AM, Wood WA, Mersereau JE. Young men with cancer experience low referral rates for fertility counseling and sperm banking. J Oncol Pract. 2016;12(5):465–71. https://doi.org/10.1200/jop.2015.010579. The authors present the largest single institution evaluation of oncology counseling and referral rates for male fertility preservation, which confirmed previous findings of poor counseling and referral rates.
Hoshi M, Oebisu N, Takada J, Iwai T, Tsuruta R, Nakamura H. Pre-chemotherapy preservation of fertility in male patients with high-grade malignant bone and soft tissue tumors. Mol Clin Oncol. 2014;2(6):1111–4. https://doi.org/10.3892/mco.2014.367.
•• Sonnenburg DW, Brames MJ, Case-Eads S, Einhorn LH. Utilization of sperm banking and barriers to its use in testicular cancer patients. Support Care Cancer. 2015;23(9):2763–8. https://doi.org/10.1007/s00520-015-2641-9. One of the larger, more recent fertility preservation center review providing answers to how cancer survivors utilize their banked specimens specifically younger and childless men are more likely to bank, 18% of men utilized their banked specimens, and 82% of used specimens resulted in paternity success.
Girasole CR, Cookson MS, Smith JA Jr, Ivey BS, Roth BJ, Chang SS. Sperm banking: use and outcomes in patients treated for testicular cancer. BJU Int. 2007;99(1):33–6. https://doi.org/10.1111/j.1464-410X.2006.06537.x.
Sheth KR, Sharma V, Helfand BT, Cashy J, Smith K, Hedges JC, et al. Improved fertility preservation care for male patients with cancer after establishment of formalized oncofertility program. J Urol. 2012;187(3):979–86. https://doi.org/10.1016/j.juro.2011.10.154.
•• Ferrari S, Paffoni A, Filippi F, Busnelli A, Vegetti W, Somigliana E. Sperm cryopreservation and reproductive outcome in male cancer patients: a systematic review. Reprod Biomed Online. 2016;33(1):29–38. https://doi.org/10.1016/j.rbmo.2016.04.002. The most recent systematic review of utilization of cryopreserved sperm, highlighting only 16% of men choose to discard their samples over time.
Brackett NL, Kafetsoulis A, Ibrahim E, Aballa TC, Lynne CM. Application of 2 vibrators salvages ejaculatory failures to 1 vibrator during penile vibratory stimulation in men with spinal cord injuries. J Urol. 2007;177(2):660–3. https://doi.org/10.1016/j.juro.2006.09.044.
Schatte EC, Orejuela FJ, Lipshultz LI, Kim ED, Lamb DJ. Treatment of infertility due to anejaculation in the male with electroejaculation and intracytoplasmic sperm injection. J Urol. 2000;163(6):1717–20.
Berookhim BM, Mulhall JP. Outcomes of operative sperm retrieval strategies for fertility preservation among males scheduled to undergo cancer treatment. Fertil Steril. 2014;101(3):805–11. https://doi.org/10.1016/j.fertnstert.2013.11.122.
•• Bernie AM, Mata DA, Ramasamy R, Schlegel PN. Comparison of microdissection testicular sperm extraction, conventional testicular sperm extraction, and testicular sperm aspiration for nonobstructive azoospermia: a systematic review and meta-analysis. Fertil Steril. 2015;104(5):1099–103 e1–3. https://doi.org/10.1016/j.fertnstert.2015.07.1136. The authors present a concise review and meta-analysis of available data and demonstrated the superiority of micro-TESE for sperm retrieval in non-obstructive azoospermia patients.
Friedler S, Raziel A, Strassburger D, Soffer Y, Komarovsky D, Ron-El R. Testicular sperm retrieval by percutaneous fine needle sperm aspiration compared with testicular sperm extraction by open biopsy in men with non-obstructive azoospermia. Hum Reprod (Oxford, England). 1997;12(7):1488–93.
Sheynkin YR, Ye Z, Menendez S, Liotta D, Veeck LL, Schlegel P. Controlled comparison of percutaneous and microsurgical sperm retrieval in men with obstructive azoospermia. Hum Reprod (Oxford, England). 1998;13(11):3086–9.
Devroey P, Liu J, Nagy Z, Goossens A, Tournaye H, Camus M, et al. Pregnancies after testicular sperm extraction and intracytoplasmic sperm injection in non-obstructive azoospermia. Hum Reprod (Oxford, England). 1995;10(6):1457–60.
Shah R. Surgical sperm retrieval: techniques and their indications. Indian J Urol. 2011;27(1):102–9. https://doi.org/10.4103/0970-1591.78439.
Schlegel PN, et al. Hum Reprod (Oxford, England). 1999;14(1):131–5.
Schrader M, Muller M, Sofikitis N, Straub B, Krause H, Miller K. "Onco-tese": testicular sperm extraction in azoospermic cancer patients before chemotherapy-new guidelines? Urology. 2003;61(2):421–5.
Descombe L, Chauleur C, Gentil-Perret A, Aknin-Seifer I, Tostain J, Levy R. Testicular sperm extraction in a single cancerous testicle in patients with azoospermia: a case report. Fertil Steril. 2008;90(2):443 e1–4. https://doi.org/10.1016/j.fertnstert.2007.07.1308.
Haddad N, Al-Rabeeah K, Onerheim R, Zini A. Is ex vivo microdissection testicular sperm extraction indicated for infertile men undergoing radical orchiectomy for testicular cancer? Case report and literature review. Fertil Steril. 2014;101(4):956–9. https://doi.org/10.1016/j.fertnstert.2013.12.052.
Rives N, Perdrix A, Hennebicq S, Saias-Magnan J, Melin MC, Berthaut I, et al. The semen quality of 1158 men with testicular cancer at the time of cryopreservation: results of the French National CECOS Network. J Androl. 2012;33(6):1394–401. https://doi.org/10.2164/jandrol.112.016592.
Damani MN, Master V, Meng MV, Burgess C, Turek P, Oates RD. Postchemotherapy ejaculatory azoospermia: fatherhood with sperm from testis tissue with intracytoplasmic sperm injection. J Clin Oncol. 2002;20(4):930–6. https://doi.org/10.1200/jco.2002.20.4.930.
Hsiao W, Stahl PJ, Osterberg EC, Nejat E, Palermo GD, Rosenwaks Z, et al. Successful treatment of postchemotherapy azoospermia with microsurgical testicular sperm extraction: the Weill Cornell experience. J Clin Oncol. 2011;29(12):1607–11. https://doi.org/10.1200/jco.2010.33.7808.
Wald M, Ross LS, Prins GS, Cieslak-Janzen J, Wolf G, Niederberger CS. Analysis of outcomes of cryopreserved surgically retrieved sperm for IVF/ICSI. J Androl. 2006;27(1):60–5. https://doi.org/10.2164/jandrol.05076.
•• Del Vento F, Vermeulen M, de Michele F, Giudice MG, Poels J, des Rieux A, et al. Tissue engineering to improve immature testicular tissue and cell transplantation outcomes: one step closer to fertility restoration for prepubertal boys exposed to gonadotoxic treatments. Int J Mol Sci. 2018;19(1) https://doi.org/10.3390/ijms19010286. The authors present an excellent review of available data to date regarding the techniques and success using immature testicular tissue for transplantation, highlighting the bright future for this technology.
Wyns C, Curaba M, Petit S, Vanabelle B, Laurent P, Wese JF, et al. Management of fertility preservation in prepubertal patients: 5 years' experience at the Catholic University of Louvain. Hum Reprod (Oxford, England). 2011;26(4):737–47. https://doi.org/10.1093/humrep/deq387.
Sato T, Katagiri K, Gohbara A, Inoue K, Ogonuki N, Ogura A, et al. In vitro production of functional sperm in cultured neonatal mouse testes. Nature. 2011;471(7339):504–7. https://doi.org/10.1038/nature09850.
Jahnukainen K, Hou M, Petersen C, Setchell B, Soder O. Intratesticular transplantation of testicular cells from leukemic rats causes transmission of leukemia. Cancer Res. 2001;61(2):706–10.
Jurewicz M, Hillelsohn J, Mehta S, Gilbert BR. Fertility preservation in pubertal and pre-pubertal boys with cancer. Pediatr Endocrinol Rev. 2018;15(3):234–43. https://doi.org/10.17458/per.vol15.2018.jhmg.fertilitypubertalboys.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
J. Abram McBride, M.D. and Larry I. Lipshultz each declare no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Andrology and Infertility
Rights and permissions
About this article
Cite this article
Abram McBride, J., Lipshultz, L.I. Male Fertility Preservation. Curr Urol Rep 19, 49 (2018). https://doi.org/10.1007/s11934-018-0803-2
Published:
DOI: https://doi.org/10.1007/s11934-018-0803-2