Abstract
Purposeof Review
Female sex hormones have systemic effects unrelated to their reproductive function. We describe experiences of different research groups and our own, on aspects related to the importance of female sex hormones on blood pressure (BP) regulation and salt-sensitivity-mediated BP response and salt sensitivity without alterations in BP, as well as renal sodium handling and interactions with the immune system.
Recent Findings
Changes in sodium intake in normotensive premenopausal women cause more BP variations than in men. After menopause, women often develop arterial hypertension (HT) with a profile of sodium sensitivity. Besides, experimental results have shown that in adult rat models resembling the postmenopausal hormonal state induced by ovariectomy, controlling BP is not enough to avoid renal and other tissue infiltration with immune cells, which does not occur when sodium intake is low or normal. Therefore, excess sodium promotes an inflammatory state with the involvement of immune cells. The evidence of activation of adaptive immunity, besides changes in T cell subpopulations, includes changes in sodium transporters and receptors.
Summary
More studies are needed to evaluate the particular sodium sensitivity of women and its meaning. Changes in lifestyle and sodium intake reduction are the main therapeutic steps. However, to face the actual burden of salt-sensitive HT in postmenopausal women and its associated inflammatory/immune changes, it seems reasonable to work on immune cell activity by considering the peripheral blood mononuclear cell phenotypes of molecules and transport proteins related to sodium handle, both to screen for and treat cell activation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
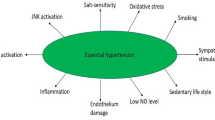
Female sex hormones have multiple systemic and organ-specific effects that are not related to their reproductive function. In this article, we describe the experiences of different research groups and our own, concerning aspects of blood pressure (BP) regulation, sodium intake/balance, salt-sensitivity-mediated BP responsiveness, and salt-sensitive activity other than BP, as well as renal sodium handling and interaction with the immune system. Female sex hormones regulate immune system activity, and immune response can intermediate between sex hormones and specific organ effects. In both reproductive-age women and young female animals, female sex hormones play a key role in the regulation of blood pressure, both by their renal protective effects and by their modulation of the immune system. The response of blood pressure to changes in salt intake may vary according to the presence or absence of female sex hormones, through the regulation of renal sodium handling, and to the activation of immune system cells (Fig. 1).
Regarding BP, the development of arterial hypertension (HT) in postmenopausal women often displays a pattern of salt sensitivity [1•, 2, 3•] and over the last years, an immune component linked to innate and adaptive immunity has been observed [4]. Although in this article we highlight the importance of excessive sodium intake, we must also take into account that this behavior is frequently accompanied by other eating habits and inappropriate physical activity such as excessive consumption of carbohydrates and a sedentary lifestyle, respectively. The combination of these factors leads to the development of metabolic syndrome [5•].
Sex Hormones and Salt-Sensitive Hypertension
Clinical and experimental studies have shown that HT is associated with an increase in sodium intake [6•]. Excess sodium can exacerbate ongoing HT or promote HT development in susceptible individuals or animals, so-called salt sensitive hypertension (SSHT) [7]. In fact, HT and high sodium (HS) consumption are nowadays worldwide health problems, especially in Western countries [5••]. These reports, however, do not distinguish salt-sensitive from saltresistant HT. Salt sensitivity, independent of BP, is a risk factor for cardiovascular (CV) morbidity and mortality, as well as other diseases [8••, 9, 10••]
The profile of sensitivity to salt or SSHT is observed when increasing the intake of NaCl or table salt increases BP. This is the predominant concept, but it is also defined as a decrease in BP through reduced NaCl intake. The BP response in SSHT is more marked in some individuals that are afterward named salt-sensitive patients. However, there is not yet a clear definition of SSHT [11•]. The people most affected by it are the elderly since 80% of hypertensive patients over 60 years of age suffer from SSHT, while only 50% of patients under 40 years of age present it [8•]. Sensitivity to salt is associated with increased CV disease morbidity and mortality including renal disease [5••], such as that occurring in other HT conditions.
Once the CV response pattern is identified, it could be avoided by decreasing NaCl intake. In clinical practice, this occurs both in patients with SSHT and those with HT with other etiologies who improve when NaCl intake is decreased. In addition, it is interesting to note that a yet undetermined number of people classified within the normotensive ranges also exhibit the phenomenon of salt sensitivity: their BP changes more than in other normotensive individuals when salt intake changes, without reaching the HT levels. This response pattern is also associated with a higher prevalence of CV disease and other diseases such as neoplasms [9].
When comparable groups of women and men, as well as female and male experimental animals from hypertensive strains, are studied [12, 13], consistently lower BP in fertile women/females than in men/males is observed [14•, 15]. In normotensive rat strains, males and females have similar levels of BP [16•]. Studies in experimental animals show that females can excrete sodium load faster and more efficiently than males [17•, 18]. Along these lines, CV events are also lower in women than in men, unless other factors such as smoking, obesity, or drug use concur. In addition, it is observed that if kidney disease or injury occurs, women of reproductive age have a better and slower evolution than men [19••]. Similarly, experimental studies of acute kidney injury also indicate a pattern of recovery or deterioration of the renal function according to age and sex, with young female rats resisting acute injury better and recovering much faster [20]. This profile changes in postmenopausal women. This change in the CV response profile and sodium intake is associated with a decrease in ovarian hormone levels, mainly estrogens [19••], a feature also described in several experimental animal models [13]. A decade after menopause, the pattern of CV and renal protection decreases and approaches the biological or systemic response of the men counterpart [21, 22•]. It is common for postmenopausal women to develop HT with a response pattern of salt sensitivity [22•]. However, a careful review of recent information shows that women have more frequency of CV and renal disease during their fertile age than previously thought. Therefore, the protection that female hormones give should not be taken as a paradigm, although the general trend of protection, as stated above, and the decline after menopause are still sustained [23••].
Female Sex Hormones and Renal Regulation of Sodium and Blood Pressure
Gonadal hormones and, in particular, female hormones play an important role in the maintenance of body fluid homeostasis and BP regulation [19••]. In young adult normotensive humans, the renin-angiotensin-aldosterone system, extracellular fluid volume (ECFV), and BP differ according to sex. Men have more systolic BP, plasma aldosterone, and ECFV than women. This is particularly observed under conditions of moderate-high sodium intake versus low sodium intake [24•]. Thus, variables related to ECFV, as stated here and below, are under the control of sex hormones.
Another particularly interesting study due to the number of participants and a design with a clear division of results into women and men under a dietary intervention is the GenSalt study [25••]. The authors showed that sex, age, and previous levels of BP are important to determine the intensity of BP response or variation when individuals change from low sodium to moderate-high sodium intake and to moderate-high sodium intake plus potassium. The importance of sex lies in the fact that women have lower basal BP but greater variation in BP levels with both types of sodium intake than men.
Researchers have traditionally shown that the presence or alterations of ovary hormones seem to play a relevant role in the regulation of vascular relaxation, and hence BP, even in aging female spontaneously hypertensive rats (SHR) [26]. Other BP regulatory mediators such as the renal kallikrein-kinin system also play an important role in SHR and show sexual and gonadal dimorphism [12]. Premenopausal women and adult female rats are protected against hemodynamic stress such as sodium loading, but this condition changes in the postmenopausal condition. The lack of estrogens may lead to a maladaptive response to sodium load [22•, 27•]. In addition, the role of progesterone seems to be more complex. Progesterone could interact with aldosterone depending on the sodium status [28]. Progesterone regulation of ECFV is more evident in circumstances such as pregnancy where a marked fluid retention is observed without an increase in BP [29]. Thus, progesterone could stimulate mineralocorticoid receptors during low aldosterone conditions and phosphorylate and activate the neutral sodium chloride cotransporter (NCC). Both effects could result in BP increments or sodium retention, as observed in some postmenopausal women receiving hormone replacement [29].
Classical experimental studies were performed on male or female animals with normal renal function and receiving mineralocorticoids and deoxycorticosterone acetate salt (DOCA-salt) to evaluate the systemic CV effect [30••] or with the chronic infusion of angiotensin II (Ang II) [31, 32•]. These traditional studies combine SSHT with an excess of salt-retaining hormones. Thus, HT ensues as a consequence of factors that retain sodium or increase the arterial vascular tone, such as Ang II and norepinephrine.
This early hypothesis of work has been expanded over the years because other studies have extended the knowledge on this topic, showing that the low availability of natriuretic and vasodilator factors, usually described as modulators, may predispose to HT. Several lines of evidence suggest that renal dopamine [33•, 34] and renal kallikrein in intact and gonadectomized SHR [12] play an important role in regulating sodium excretion, BP, and vascular tone. Other vasodilator and natriuretic factors, such as atrial natriuretic factor and intrarenal dopamine, interact to regulate sodium metabolism [35], while nitric oxide and prostaglandins also play a leading role in the pathophysiology of HT.
In most studies, men/males have been taken as the standard sex to be studied. However, as previously stated, it is increasingly evident that women/females must be included in this type of research, since findings and behavior regarding BP regulation and related issues like sodium handling or interaction with immune cells differ between the sexes.
In this conceptual framework, our group has developed an experimental model of SSHT in adult ovariectomized (oVx) Wistar rats. The studies were performed in female Wistar rats, a normotensive rat strain. Rats were bilaterally oVx at 60 days of life. Later, 90 days after oVx, rats received normal salt (NS, 0.24% NaCl) or HS intake (1% NaCl in drinking water) for 5–7 days. After this period, we conducted several studies to evaluate the profile of BP response and transporters or mediators linked to sodium balance in the kidney and its behavior under NS or HS intake.
The first investigations were conducted in oVx rats under NS intake. It was found that the activity of the renal kallikrein-kinin system, urinary kallikrein, and kallikrein gene expression in kidney tissue were increased in oVx rats. In this NS model, BP remained normal or even decreased [36]. However, upon treatment with the kallikrein-kinin system blocker, aprotinin, BP only increased in oVx rats. Therefore, even under normal conditions of sodium intake and BP, a vasodilator renal system was active to keep BP normal.
In subsequent studies, we analyzed the BP response and sodium transporters in the kidney, by comparing a baseline situation with NS intake and another one with HS intake [37•]. Importantly, oVx NS rats were normotensive, but HS intake uncovered the propensity to develop HT. Upon sodium challenge, oVx rats excreted less sodium than intact female rats and developed salt-sensitive HT. In this study, we found that oVx rats had an overexpressed renal Na+, K+-ATPase (NKA) pump. Even in normotensive rats under NS intake, NKA in renal tubular epithelial cells in the cortex and medulla was overexpressed. Another important hemodynamic finding was that under NS intake, oVx rats had decreased renal vascular resistance. Instead, under HS, while intact female (IF) rats increased renal plasma flow, oVx did not, resembling the characteristics of non-responding hypertensive humans [38]. The finding of an overexpressed renal NKA, even under NS intake, prompted us to investigate the possible cause. NKA expression/activity in renal tubular epithelial cells is regulated, among other mechanisms, by posttranslational phosphorylation/dephosphorylation reactions at Ser 23 residue. Phosphorylation of NKA reduces its activity while dephosphorylation of NKA increases it [39•, 40]. Therefore, we studied whether a defect in NKA phosphorylation could be responsible for decreased natriuresis and sodium retention, together with HT. For this purpose, we examined the regulation of NKA by renal dopamine [41•]. Although renal dopamine synthesis was equal in IF and oVx rats, several steps of the dopamine signaling pathway in renal tubular epithelial cells were altered. There was a lower expression of D1-like dopamine receptors (D1DR) in oVx rats. Besides, upon sodium load, oVx rats were unable to increase the expression of cytochrome P-4504A (CYP4A), which is the enzyme that increases 20-hydroxyeicosatetraenoic acid (20-HETE) production. The 20-HETE is a natriuretic factor that stimulates protein kinase C (PKC) alpha. PKC alpha phosphorylates NKA alpha-1 subunit at Ser 23 and diminishes NKA activity, which promotes sodium excretion. PKC alpha was also reduced. Therefore, in oVx rats, an altered renal dopaminergic pathway causes a failure in handling sodium load.
Three observations about this SSHT model are noteworthy. First, although the decrease in female sex hormones was abrupt (surgically induced), it was performed in adult rats, thus resembling the hormonal conditions of postmenopausal women or decline of ovary hormones for causes other than menopause [42]; second, the amount of sodium used as sodium loading was similar to that observed in office consulting patients and was administered for a short period of time (5–7 days); finally, the model was not Ang II or aldosterone-dependent. Aldosterone levels were reduced after sodium loading. Therefore, experimental evidence corroborates that ovarian hormones, through presence or absence, play a key role in integrating homeostatic aspects related to kidney function, ECFV, and BP.
Estrogens, Sodium, Immune Cells, and Sodium Sensitivity with or Without Hypertension
Estrogens and Immune Cells
Female sex hormones interact with innate and adaptive immune mechanisms. Given that sex hormones can regulate immune system physiology, that sodium intake influences immune cell activity, and that sex hormones and immune cells can induce and release, respectively, different types of cytokines, female hormones, and immune cells should be expected to have several points at which they can interact. Therefore, the interaction of estrogens with the immune system is both rich and complex [43••]. First, estrogens act by activating different estrogen receptors (ER) such as ER alpha, ER beta, and membrane receptor G protein-coupled estrogen receptor 1 (GPER-1). According to the presence or absence of these receptors, estrogen can promote different and, sometimes, opposite responses in tissues. All mononuclear immune cells in peripheral blood, like monocytes and B and T cells, express ER, but CD4+ T cells express more ER alpha than the other cell types while B cells express more ER beta [44].
As an overall estrogenic activity, exogenous administration of estrogen avoids T cell maturation and decreases the abundance of the thymus T cell subset. Estrogen also causes thymus atrophy [45]. Through the stimulation of receptors, estrogens can regulate T cell maturation, proliferation, and polarization, affecting different subsets depending on the interaction with specific immune cells, tissue physiology, pathological conditions, or cytokines in the environment. Thus, this is a complex network of possible interactions [46•]. For example, estradiol affects the cytotoxicity of natural killer (NK) cells, activation and differentiation of monocytes, macrophages, and dendritic cells, in a dose-dependent manner [47,48,49,50,51]. However, otherwise stated, we will focus on adaptive immunity. Specifically, estradiol decreases the differentiation of T lymphocytes towards proinflammatory subpopulations like T helper 17 (Th17) and increases regulatory lymphocytes or anti-inflammatory regulatory (Tregs) T lymphocytes [52•, 53••]. Low concentrations of estradiol promote a skew to a Th1 immune response, while at high concentrations (pregnancy as an extreme condition) trigger a Th2 immune response [54]. In addition, other physiological conditions, besides pregnancy, with normal-high levels of estrogens increase regulatory Treg lymphocytes [55]. Thus, in general terms, it can be stated that ER stimulation by estradiol in T cells favors an anti-inflammatory immune response by decreasing Th17 polarization, and importantly, by expanding the regulatory T cell compartment. On the other hand, if female sex hormones induce this kind of immune response, the absence or decrease of estrogens could be expected to promote an abnormal immune response with a bias of immune response towards inflammation in pathological conditions like HT (Fig. 2) [27, 42, 56••].
The scheme shows that at high sodium intake, women of reproductive age, and adult female rats may not develop salt-sensitive hypertension due to the protective effect of female sex hormones. However, in postmenopausal women and ovariectomized rats, the protective effect is lost. Therefore, in the absence of female sex hormones, an impaired renal sodium handling and activation of the immune system may occur, leading to the development of salt-sensitive hypertension
Although progesterone is not usually regarded as an important player in pathophysiological conditions such as immune-mediated hypertension, progesterone also acts on immunity by inducing an anti-inflammatory response. Progesterone, like estradiol, also has receptors expressed in different types of immune cells [57]. Progesterone shifts the immune response from Th1 to Th2 or Treg cells [58] and suppresses the differentiation to Th17 cells.
Female Hormones, Sodium, BP, and Adaptive Immunity
It has been well established in experimental settings that innate and adaptive immunity participate in the pathogenesis of arterial HT [59••, 60]. Guzik et al. [59••] were the first to show in male rats that T lymphocytes participate in the hypertensive response to chronic infusion of Ang II. Subsequently, it was observed that in female rats the model did not behave in the same way as it did in males [61••]. In other words, the response of T cells is specific to the sex in which it is studied, highlighting the importance of female hormones. However, regardless of sex, it is still unclear whether the immune cells found in renal tissue, blood vessels, or skin [62,63,64••] may induce HT development or whether the presence of immune cells is secondary to the increase in BP [61••]. Investigations into the role of the immune system in HT have analyzed the presence of CD4+ T helper cells or CD8+ T cytotoxic cells. In general, CD4+ T helper cells and their different subsets have been described as the predominant cells in the pathophysiology of HT in the kidney and other organs [43••, 61••, 65••].
Female sex hormones and sodium intake are also important players that can modulate the lymphocyte physiology and thus the direction the adaptive immune response may take. As stated above, the interplay between the immune system and female hormones is complex [43••, 61••, 66] since female sex hormones have a regulatory role in cells from both the innate and adaptive immune systems [53••]. Estrogen-mediated immunomodulation causes a reduced activation of effector T cells [52•] and potentiation of Treg cells [3•, 53••, 67]. On the other hand, the absence of female sex hormones may be expected to cause a different immune response in comparison with intact females [27•]. In addition, HS intake may induce changes in immune T cell subsets, like activation and polarization of CD4+ Th cells towards the proinflammatory Th17 subset [68, 69•]. To be activated and proliferating and also during migration, lymphocytes require several transporters and intracellular signals or receptors that mainly participate in the ion flux increase upon T cell activation [70, 71••, 72]. The increase in ion fluxes is the motor that drives lymphocyte activation, expansion, and migration. To perform this task, lymphocytes are endowed with sodium and chloride transporters, as well as sodium channels, specific receptors, and intracellular mediators also present in other cells. The sodium-dependent serum- and glucocorticoid-induced kinase 1 (SGK1) is expressed in lymphocytes. In the kidney, SGK1 increases the activity of ion transporters such as electroneutral NCC, type 2 Na+–K+–2Cl− cotransporter (NKCC2), epithelial sodium channel (ENaC), and NKA pump [73].
Thus, T lymphocytes CD4+ and CD8+ subsets express NKA, type 1 Na+–K+–2Cl− cotransporter (NKCC1), SGK1, dopamine receptors, mainly D1DR, lysine-deficient protein kinase 1 (WNK1), and other sodium and chloride transporters or channels as well [27•, 69••, 72, 74]. Then, T cells are good candidates to study the response of sodium transporters when facing changes in the surrounding environment, since, as mentioned above, immune cells could be part of the regulatory mechanisms of BP, compromised with HT and under the individual or interrelated influence of female sex hormones, and sodium intake. In this way, a study on T lymphocytes showed the importance of SGK1 and NKCC1 during the development of HT by the chronic infusion of Ang II or by DOCA-salt treatment, in which sgkl gene deletion or NKCC1 inhibition blunted BP increase [69••].
As previously described, the model of SSHT in adult oVx Wistar rats, besides deranged sodium handling by renal tubules with sodium retention and high BP, is associated with an overexpressed and stimulated NKA. To explore whether this is a specific alteration in kidney function or whether other systems/tissues that express sodium transport proteins could also be involved in this pathophysiological scenario, we extended studies to the immune response by exploring the behavior of sodium transporters in peripheral blood mononuclear cells (PBMC) from IF and oVx rats under normal or high salt intake. The results of this study demonstrate that the response of PBMC to HS differs according to the hormonal background. PBMC in oVx rats sensed sodium intake in a completely different way, compared with IF rats. Sodium transporters NKA and NKCC1 increased their expression in PBMC from oVx rats under HS intake. Besides, CD4+-specific protein and D1DR also increased in oVx HS, in comparison with oVx NS rats. oVx under NS intake did not develop HT. Therefore, high sodium was required to increase BP. In contrast, IF rats upon HS did not become hypertensive, and CD4 + protein and NKA were decreased by HS, as compared with IF NS rats and oVx HS rats.
We then explored whether findings suggesting immune cell activation in oVx HS rats were only present in PBMC or in renal tissues as well. In fact, inflammatory/immune CD45+ cells were found at the basolateral peritubular space. One of the main findings of these studies was that PBMC and their ion transport proteins are not neutral to variations in sodium intake and react very differently to HS intake in oVx in comparison with IF rats. Thus, in the absence of female sex hormones, HS intake and HT are variables that PBMC must face in their environment, showing patterns of different sensitivities to these stimuli.
Findings in renal tissue by immunohistochemistry show that in oVx HS rats the kidney cortex begins to be infiltrated by inflammatory/immune cells as part of this complex scenario. Activated immune cells release local Ang II, inflammatory cytokines, and oxidative stress intermediaries, and aggravate ongoing HT and cause kidney injury [56••] Hypothetically, a similar scenario probably develops in the basolateral space of intestinal cells and in the interstitial lymphatic compartment. In the absence of female sex hormones and with increased BP, HS intake at these interfaces could be processed as a neoantigen to which inflammatory/ immune cells react. Therefore, neoantigens [75, 76] or direct stimulation by Na+ [68, 69••] can be behind immune cell stimulation in this model of SSHT.
However, if immune cells are responsible for HT development or if once high BP ensues, somehow immune cells could be recruited, remains to be elucidated. Another question is whether this alteration is caused by HS intake, BP, or both, in SSHT models with infiltration of renal tissue by immune cells. In this regard, several interesting articles have described different findings. Rudemiller et al. [77] reported that CD247 T lymphocytes participate in the hypertension mechanism in Dahl SS rats, since genetically modified CD247 -/- rats do not show the magnitude of renal infiltration and HT that CD247 + / + rats develop. A particular finding of this study was that peripheral blood T lymphocytes do not develop the level of inflammatory cytokine mediators that renal lymphocytes have. The authors interpreted it as an indication that renal immune cells are active while PBMC are not. In addition, while Evans et al. [78] found that a reduction in renal perfusion pressure by a servo control mechanism in the left kidney of Dahl salt-sensitive rats decreases renal T cell infiltration, Pai et al. [79] observed that the pharmacological reduction of BP in Envigo rats is not as effective in reducing renal infiltration with immune cells. Thus, it seems clear that, at least in these attractive experimental works, the involvement of immune cells in SSHT is evident but the final pathophysiological mechanism is still under debate.
Our results have shown that a BP increase in oVx rats under HS intake is accompanied by activation of PBMC and renal infiltration with inflammatory/immune cells CD45+ leukocytes. These findings are all associated with the absence of female hormones. Recently, we also explored in oVx HS rats whether the changes observed in overexpressed NKA in PBMC, renal epithelial tubular cells, and kidney infiltration with CD45+ leukocytes were secondary to HS intake, high BP, or both (Vlachovsky et al., Communication at the Joint Meeting SAIC SAI&FAIC SAFIS 2022. Abstract published in: Medicina, Buenos Aires) [80]. All experimental animals under NS or HS intake, even IF rats, received hydralazine (HDZ) to reduce BP. HDZ decreased BP in all groups including IF NS rats. Despite effective BP reduction, HDZ did not cause any of the observed changes in oVx and IF rats. Therefore, the main changes are ascribed to high sodium, rather than high BP.
Two findings are relevant to this review. One is that NKA is already overexpressed in the kidney of oVx NS rats that are normotensive and become hypertensive upon HS intake, and the other one is that the location of CD45+ renal infiltrates is basolateral, like NKA overexpression, which opens the possibility of high sodium fluxes in the peritubular basolateral space of renal epithelial tubular cells. Potentially, this fact could create micro-domains of high sodium where immune cells can be activated. Therefore, expressed in terms of homeostasis or altered regulation, overexpressed NKA is the primary event and HS is the perturbation to the system.
Effects of Estrogens at the Arterial and Cardiovascular Levels
As stated above, sodium salt sensitivity, independent of BP, causes several organs and systems of the body to suffer pathological consequences either because of an excess in sodium intake or an abnormal response to sodium. This kind of damage depends on the environment in which sodium interacts with other mediators, including sex hormones. For instance, the endothelial function is impaired in rodents receiving a HS diet with no alterations in BP [81, 82]. However, it should be taken into account that in those studies, BP was measured in anesthetized rats [81], which may alter BP values, or by tail cuff [82], which may be inappropriate for detecting small changes in BP, in evaluating pathologic changes in high salt intake.
Humans’ arterial function is also affected during HS intake [83]. Oxidative stress seems to play an important role in this response. Regarding arterial dysfunction, women seem to have relative protection, since female sex hormones, particularly estrogens, have a positive action on nitric oxide generation, in comparison with men [84].
Various groups of researchers have focused on the vascular aspects of HT that occur in postmenopausal women. In some hypertensive postmenopausal women and in old female mice, 17-β estradiol treatment favors the synthesis of vascular nitric oxide, which reduces the salt sensitivity of BP and restores vasodilatory response, while its absence causes an increase in the response to Ang II followed by vasoconstriction and HT [85,86,87].
However, the consideration of the role of female hormones in protecting against CV, metabolic, and kidney diseases has changed drastically over the last years. Some time ago, the protective role of female sex hormones was taken for granted although it was considered to depend on age and the onset of menopause [3•, 19••]. Nevertheless, current studies and observational descriptions have shown that women are not so protected against CV diseases and renal dysfunction [23••], at least not as protected as they were thought to be before. Perhaps, women require a new definition of adequate systolic BP to avoid CV events, like myocardial infarction or heart failure [3•, 15]. Despite these observations, women are still considered to be more protected against CV injuries than men, this effect being mainly due to the protection offered by estrogens [88], or in the case of other injuries, like renal injuries, the progression to end-stage renal disease at a given reduced glomerular filtration rate is slower in females [23••]. In clinical studies, supplement or treatment with estrogens reduces risk factors for CV disease [89] or stress-related hypertension [90]. However, in a recent vascular ex vivo study using human samples, other researchers do not agree, at least partially, that estrogen supplementation is so effective in improving vascular dysfunction [91]. Therefore, here we have new issues for the ongoing discussion about estrogens as entirely protective, and perhaps may contribute to finding a new optimal threshold for women’s BP at different stages of life [3, 92].
Sex Hormones, Skin, Sodium, and Lymphatic Response
In the context of SSHT or HT, it was observed that sodium can induce the activation of immune cells in the kidney and skin [56, 93]. The sodium complexed with anionic molecules in the skin and the relationship with innate immunity and the lymphatic system has gained importance over the last years. Although still under debate [94••], these findings have changed the model of two compartments for sodium distribution and have possibly added part of the skin structures, the hypodermis, as a third compartment that regulates systemic sodium handling under the form of osmotically non-compromised sodium. This means that Na+ binds to glycosaminoglycans (GAGs), especially with carboxyl and sulfated anionic GAGs, which can facilitate the non-osmotic storage of sodium.
Skin approximately represents 6% of body weight and contributes to a great portion of the interstitium [95•]. Previous investigations have shown a positive correlation between the amount of sodium, GAGs, and NKA density in the extracellular matrix of bovine cartilage [96], and in rats under sodium loading, storage in the dermis correlated with an increment in GAGs [97••]. This reasoning comes from observations that both salt-sensitive and salt-resistant individuals without HT showed similar degrees of body sodium retention after salt loadings, and that sodium retention occurred without an increase in BP in salt-resistant individuals [98, 99]. Indeed, sodium retention was associated with weight gain in salt-sensitive individuals, while no difference in water balance or weight was observed in salt resistant participants. Then, another compartment could play an important role in sodium balance in the salt-resistant response.
Titze et al. [100, 101] proposed that osmotically inactive sodium is deposited in the skin and that sodium-resistant rats have more sodium in the skin than salt-sensitive animals. In these experiments, oVx Sprague–Dawley had a smaller capacity to store sodium in the skin than intact female rats [102•]. Later, these observations were extended to humans participating in a space flight simulation experiment [103]. In addition, other researchers working on hemodialysis patients have shown that patients with end-stage renal disease had more sodium in the skin than usual [104].
Wiig et al. [105] and others have reported the importance of innate immunity in this kind of sodium balance by describing the involvement of macrophages, TonEBP (NFAT5) tonicity protein, and vascular endothelial growth factor C (VEGF-C) and its receptor. According to this description, there is a linkage between high sodium, these osmosensitive intermediary proteins, and growth factors, which finally, under a sodium load, would induce an increase in GAGs storing sodium. At the same time, lymphangiogenesis through a signal generated in macrophages and mediated by TonEBP induced an increase in VEGF-C. According to this proposal, these events induce a higher amount of lymphatic vessels, which, after sodium is buffered by GAGs, could release the accumulated amount of the ion into the general circulation without harming uncontrolled volume expansion.
Nevertheless, Thowsen et al. [106] worked on male rats and found that under very high sodium load or sodium plus DOCA, sulfated and unsulfated GAGs were not increased. They also observed that sodium excess was redistributed to the intracellular compartment, mainly to skeletal and cardiac muscle cells. However, it is still under debate whether this mechanism plays a role in sodium balance and whether skin accumulation is hypertonic as proposed in initial studies [101]. Even if sodium is stored in the interstitium of the skin or other tissues, it is still unclear whether it is isotonic instead of hypertonic, as proposed by Rossitto et al. [107••].
The other discussion is whether this kind of storage prevents HT or, on the contrary, whether it promotes HT by vascular inflammation and endothelial/glycocalyx damage [95•]. This also creates the opportunity to establish the concept of skin sodium as a potential “double-edged sword” [95•]. Is it good or bad to accumulate sodium in the skin? This concept, in which female hormones could play an important role, is yet to be resolved. Lymphatic endothelial cells express estrogen receptors and females have higher serum levels of VEGF-C and VEGF-D [108].
High salt intake expands and increases the number of lymphatic vessels in the skin, as well as lymph flow in the skin and skeletal muscle [109]. These results are from studies in male rats with a high amount of sodium in the diet. In fact, rats developed HT only with high salt in the order of 8% intake. Then, when macrophages were inhibited, there was a reduction in lymphatic vessels and in lymph flow in skin and muscle. This demonstrates the importance of lymph flow regulated by macrophages to handle a HS intake.
However, it has also been shown that high sodium can induce an inflammatory pattern response in adaptive immune system cells [65•, 68, 69••], vascular inflammation, and endothelial/glycocalyx damage [95•], which finally cause arterial HT. However, in another report [110••] working with a much lower amount of sodium (1%), which did not induce hypertension in male rats, hypertension ensued when macrophages were inhibited.
Therefore, by regulating lymph flow probably via NFAT 5/TonBEP and VEGF-C, macrophages may work as buffers of sodium load mainly under conditions of moderate sodium intake. When sodium intake is massive, the operation of the system seems to be out of range to keep BP normal, probably by generating an inflammatory response. A sexual dimorphic pattern appears when plasma levels of VEGF-C are measured. Women have more angiogenic circulating factors than men [111]. As stated above, VEGF-C participates in lymphatic growth. In gonadectomized animals, lymph flow is decreased in comparison with intact females [112]. In a scenario in which lymph flow is decreased, female rats have a lower capacity of sodium storage in the skin, and the absence of estradiol reduces the arterial ability to increase nitric oxide synthesis upon sodium load creating a favorable scenario for HT development.
Similarly, Selvarajah et al. [113••] described a response to sodium with certain sex specificity since skin in men can buffer sodium intake in normotensive participants, maintaining stable BP, while women do not accumulate salt in the skin and their BP in a normotensive range, as stated above, is more unstable. Besides, researchers have reported that no change occurred in plasma VEGF-C between the placebo and sodium-loaded groups, which did not show a clear involvement of TonEBP or VEGF-C activation. Compromise of sex hormones, by presence or absence, agrees with and extends the concept that the immunomodulation exerted by sodium depends on the local microenvironment [93].
In research on humans, protocols often differ in the stimulus to find salt sensitivity. Therefore, some studies use an intravenous sodium load while others use different amounts of oral sodium intake for days. Another important fact is that until very recently, the sex of the involved participants was not taken into account. Experimental individuals were either all men or women and men pooled together. In the study by Barnett et al. [114•], a crossover design involving young people, normotensives, with oral placebo vs. sodium, found an increase in GFR and NGAL in urine, no change in BP, but an increase in body weight and plasma volume. Toering et al. [24•] reported on women’s and men’s responsiveness to sodium at intakes of 50 to 200 mmol/day. The basal BP and renin-angiotensin-aldosterone system responses differed greatly between the sexes. Men had a significantly higher plasma aldosterone, extracellular volume, and systolic BP than women during HS intake. Other studies, which also differentiated between women and men and included a good amount of participants, reported that BP behavior and body weight differed according to sex. BP, even within the normal range, is more unstable in women than in men [25••] and women tend to gain body weight during changes from low to HS intake [113••]. However, the fractional excretion of sodium seems to be sharper in women.
More standardized studies are probably needed to elucidate physiological and pathological characteristics and their importance in this field. Therefore, more studies to directly assess the relationship between BP and HT development with skin sodium and lymphatic response in females and males are required. So far, it has been difficult to determine what is abnormal or not when no precision exists about the “normal” amount of sodium that humans should consume. This is especially so because SSHT is not easy to study and it is currently restricted to research work and only clinically available by observation [4]. With no intent to extrapolate experimental findings to humans, if the model of SSHT in Wistar rats resembles that of menopausal women, a word of caution should be given to this population regarding sodium intake. Some reports state that patient groups with CV risk and a good response to HT treatment are not entirely free of potential CV events [115]. Therefore, it seems that factors other than HT, like inflammation and immune activation, are active.
Many postmenopausal hypertensive women receive BP-reducing therapy, which often fails to properly control high BP, as shown in the excellent study by Mill et al. [116••]. Besides, this population has an increased risk of developing chronic renal disease and chronic renal failure [23••]. Information and control in terms of why and how to reduce sodium intake should be reinforced in postmenopausal women, since HS intake may well be behind treatment failure and progression of CV and renal disease.
Conclusions and Perspectives
In this review, we describe the relationship among female sex hormones, sodium handling, salt-sensitive hypertension, and characteristics of sodium sensitiveness other than increased arterial pressure, the potential of a new putative compartment in the skin to buffer sodium body balance, and the possible role of immune mechanisms compromised with BP regulation. Although they look like a sum of different items, they are parts of a continuum that is described according to which piece is analyzed. Altogether, when deranged, it is a systemic pathophysiological alteration triggered on the homeostasis of the biological system (Fig. 3). This occurs in the context of the presence, absence, or low levels of female sex hormones.
Animal model of salt-sensitive hypertension. The scheme shows how blood pressure varies in intact and ovariectomized rats with normal or high sodium intake. Under high sodium intake, intact rats maintain the normal blood pressure, while ovariectomized rats increase their blood pressure, activate peripheral blood mononuclear cells, and show immune cell infiltration in their renal tissues. With the administration of hydralazine (HDZ), a vasodilator drug that reduces blood pressure, ovariectomized rats with high sodium intake show normal blood pressure values but maintain the leukocyte infiltration
Taking into account that, in middle- or high-income societies, menopause starts at age 47–52 years and that a great number of women with this condition lead an active life, with a life expectancy of roughly 30 more years, it is evident that health issues in this group are meaningful [118]. According to experimental and clinical studies, HS intake may be behind the failure in achieving appropriate results with anti-hypertensive therapies. Even reaching a good level of BP with therapy, inflammatory/immune cells can still be present in different tissues, especially in the kidneys and vessels. Therefore, tissue damage may continue regardless of BP control if HS intake is not controlled. In addition, adequate control of BP does not completely prevent CV events. The first step in treating this disorder is to control sodium intake. It requires public policies related to community education and commitments to the food industry, since 75% of ingested sodium comes in ultra-processed foods. It is also possible to administer synthetic female hormones, but sometimes it could be dangerous due to adverse side effects. Another possible treatment to solve this problem is the use of immunosuppressive or immunomodulation medications. However, this option of using current drugs is not viable because the consequences could be even worse. However, a point to work on concerning immune cell activity is to consider PBMC phenotypes of molecules and transporters as a new therapeutic target [117••]. In fact, transporters or receptors expressed in PBMC, such as NKA, NKCC1, SGK1, and D1DR, may play a dual role as screening molecules [27•, 69••, 71••] for sodium sensitivity and as targets to reduce immune cell activation and could thus be added to current therapies aimed at lowering BP. This is a new field in which sex differences are considered. The study of female physiology and pathophysiology will help to uncover current failures in our understanding of such specific issues.
Abbreviations
- Ang II:
-
Angiotensin II
- BP:
-
Blood pressure
- CV:
-
Cardiovascular
- CYP4A:
-
Cytochrome P-4504A
- D1DR:
-
D1-like dopamine receptors
- DOCA:
-
Deoxycorticosterone acetate
- ECFV:
-
Extracellular fluid volume
- ENaC:
-
Epithelial sodium channel
- ER:
-
Estrogen receptors
- GAGs:
-
Glycosaminoglycans
- GPER-1:
-
Membrane receptor G protein-coupled estrogen receptor 1
- HDZ:
-
Hydralazine
- 20-HETE:
-
20-hydroxyeicosatetraenoic acid
- HS:
-
High sodium
- HT:
-
Arterial hypertension
- IF rats:
-
Intact female rats
- NCC:
-
Neutral sodium chloride cotransporter
- NK:
-
Natural killer
- NKA:
-
Na+, K+-ATPase pump
- NKCC1:
-
Type 1 Na+–K+–2Cl− cotransporter
- NKCC2:
-
Type 2 Na+–K+–2Cl− cotransporter
- NS:
-
Normal salt or normal sodium
- oVx:
-
Ovariectomized
- oVx HS:
-
Ovariectomized hypersodic
- oVx NS:
-
Ovariectomized normosodic
- PKC:
-
Protein kinase C
- PBMC:
-
Peripheral blood mononuclear cells
- sgkl gene:
-
Serum- and glucocorticoid-induced kinase 1 gene
- SGK1:
-
Serum- and glucocorticoid-induced kinase 1
- SHR:
-
Spontaneously hypertensive rats
- SSHT:
-
Salt-sensitive hypertension
- Th:
-
T helper lymphocyte
- Tregs:
-
Regulatory T lymphocytes
- VEGF-C:
-
Vascular endothelial growth factor C
- WNK 1:
-
Lysine-deficient protein kinase 1 [with no lysine (K)]
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
• Barris CT, Faulkner JL, Belin de Chantemèle EJ. Salt sensitivity of blood pressure in women. Hypertension. 2023 (2):268–278. This review highlights the phenomenon of salt sensitivity and blood pressure in women introducing new perspectives.
Faulkner JL, Belin de Chantemèle EJ. Female sex, a major risk factor for salt-sensitive hypertension. Curr Hypertens Rep. 2020;22(12):99.
• Ghazi L, Annabathula RV, Bello NA, Zhou L, Stacey RB, Upadhya B. Hypertension across a woman’s life cycle. Curr Hypertens Rep. 2022;24(12):723–733. This review describes and analyzes the behavior of blood pressure in women’s life span, and emphasizes hypertension and pregnancy.
Sahinoz M, Elijovich F, Ertuglu LA, Ishimwe J, Pitzer A, Saleem M, Mwesigwa N, Kleyman TR, Laffer CL, Kirabo A. Salt sensitivity of blood pressure in blacks and women: a role of inflammation, oxidative stress, and epithelial Na+ channel. Antioxid Redox Signal. 2021;35(18):1477–93.
•• WHO, Recommendation. (2020). Available online: https://www.who.int/news-room/fact-sheets/detail/salt-reduction. Available data from hypertension, cardiovascular disease, and metabolic syndrome around the world.
• Appel LJ, Frohlich ED, Hall JE, Pearson TA, Sacco RL, Seals DR, Sacks FM, Smith SC Jr, Vafiadis DK, Van Horn LV. The importance of population-wide sodium reduction as a means to prevent cardiovascular disease and stroke: a call to action from the American Heart Association. Circulation. 2011 15;123(10):1138–43. Recommendations from AHA about the importance to reduce sodium consumption.
Matthews EL, Brian MS, Ramick MG, Lennon-Edwards S, Edwards DG, Farquhar WB. High dietary sodium reduces brachial artery flow-mediated dilation in humans with salt-sensitive and salt-resistant blood pressure. J Appl Physiol (1985). 2015 15;118(12):1510–5.
• Weinberger MH, Fineberg NS, Fineberg SE, Weinberger M. Salt sensitivity, pulse pressure, and death in normal and hypertensive humans. Hypertension. 2001;37(2 Pt 2):429–32. Importance of salt sensitivity beyond hypertension.
de Wardener HE, MacGregor GA. Harmful effects of dietary salt in addition to hypertension. J Hum Hypertens. 2002;16(4):213–23.
•• Rossitto G, Delles C. Does excess tissue sodium storage regulate blood pressure? Curr Hypertens Rep. 2022;24(5):115–122. Revision and discussion about the new role proposed for space in the skin to buffer excess sodium intake.
• Elijovich F, Weinberger MH, Anderson CA, Appel LJ, Bursztyn M, Cook NR, Dart RA, Newton-Cheh CH, Sacks FM, Laffer CL. American Heart Association Professional and Public Education Committee of the Council on Hypertension; Council on Functional Genomics and Translational Biology; and Stroke Council. Salt sensitivity of blood pressure: a scientific statement from the American Heart Association. Hypertension. 2016;68(3):e7-e46. New statements from AHA about the importance of salt sensitivity and hypertension.
Azurmendi PJ, Toro AR, Celía AF, Guevara D, Solerno MR, Di Ciano LA, Toledo JE, Ibarra FR, Arrizurieta EE, Oddo EM. Behavior of the renal kallikrein in spontaneously hypertensive rats: influence of sexual hormones and aldosterone-sensitive distal nephron ion channels. Peptides. 2023;160:170925.
Reckelhoff JF. Gender differences in the regulation of blood pressure. Hypertension. 2001;37(5):1199–208.
• Maranon R, Reckelhoff JF. Sex and gender differences in control of blood pressure. Clin Sci (Lond). 2013;125(7):311–8. Revision of epidemiological and experimental evidence on gender differences in blood pressure regulation.
Ji H, Niiranen TJ, Rader F, Henglin M, Kim A, Ebinger JE, Claggett B, Merz CNB, Cheng S. Sex differences in blood pressure associations with cardiovascular outcomes. Circulation. 2021;143(7):761–3.
• Munger K, Baylis C. Sex differences in renal hemodynamics in rats. Am J Physiol. 1988;254(2 Pt 2):F223–31. This paper reports one of the first experimental observations about sex differences between renal hemodynamics and blood pressure in rats.
• Veiras LC, Girardi ACC, Curry J, Pei L, Ralph DL, Tran A, Castelo-Branco RC, Pastor-Soler N, Arranz CT, Yu ASL, McDonough AA. Sexual dimorphic pattern of renal transporters and electrolyte homeostasis. J Am Soc Nephrol. 2017;28(12):3504–17. This paper describes experimental findings about sexual differences in nephron sodium transporters in rats.
Di Ciano LA, Azurmendi PJ, Vlachovsky SG, Celía AF, Oddo, EM, Arrizurieta EE, Silberstein CM, Ibarra FR. Diferencias de género en presión arterial, función renal y respuesta a la dieta hipersódica en ratas Wistar. Revista de Nefrología Diálisis y Trasplante. 2018;38(1):15–27. Available from https://www.revistarenal.org.ar/index.php/rndt/article/view/294.
•• Pechere-Bertschi A, Burnier M. Gonadal steroids, salt-sensitivity and renal function. Curr Opin Nephrol Hypertens. 2007;16(1):16–21. One of the most authoritative and cited reviews about sex hormones, renal physiology, and blood pressure in women.
Boddu R, Fan C, Rangarajan S, Sunil B, Bolisetty S, Curtis LM. Unique sex- and age-dependent effects in protective pathways in acute kidney injury. Am J Physiol Renal Physiol. 2017;313(3):F740–55.
Akahoshi M, Soda M, Nakashima E, Shimaoka K, Seto S, Yano K. Effects of menopause on trends of serum cholesterol, blood pressure, and body mass index. Circulation. 1996;94(1):61–6.
• Kim JM, Kim TH, Lee HH, Lee SH, Wang T. Postmenopausal hypertension and sodium sensitivity. J Menopausal Med. 2014;20(1):1–6. This review focuses on female sex hormones decline and salt-sensitive hypertension development.
•• Cobo G, Hecking M, Port FK, Exner I, Lindholm B, Stenvinkel P, Carrero JJ. Sex and gender differences in chronic kidney disease: progression to end-stage renal disease and haemodialysis. Clin Sci (Lond). 2016;130(14):1147–63. This work describes epidemiological findings about sex differences in renal disease progression. A call of attention to the concept of female hormone protection.
• Toering TJ, Gant CM, Visser FW, van der Graaf AM, Laverman GD, Danser AHJ, Faas MM, Navis G, Lely AT. Sex differences in renin-angiotensin-aldosterone system affect extracellular volume in healthy subjects. Am J Physiol Renal Physiol. 2018;314(5):F873-F78. This paper analyzes women’s and men’s extracellular volume regulation upon changes in sodium intake.
•• He J, Gu D, Chen J, Jaquish CE, Rao DC, Hixson JE, Chen JC, Duan X, Huang JF, Chen CS, Kelly TN, Bazzano LA, Whelton PK; GenSalt Collaborative Research Group. Gender difference in blood pressure responses to dietary sodium intervention in the GenSalt study. J Hypertens. 2009;27(1):48–54. Observational-interventional study that analyzes gender differences in the behavior of blood pressure and salt sensitivity to changes in sodium intake in a great number of experimental individuals.
Wynne FL, Payne JA, Cain AE, Reckelhoff JF, Khalil RA. Age-related reduction in estrogen receptor-mediated mechanisms of vascular relaxation in female spontaneously hypertensive rats. Hypertension. 2004;43(2):405–12. https://doi.org/10.1161/01.HYP.0000111833.82664.0c.
• Vlachovsky SG, Di Ciano LA, Oddo EM, Azurmendi PJ, Goette NP, Arrizurieta EE, Silberstein C, Ibarra FR. Ovariectomy and high salt increase blood pressure and alter sodium transport proteins in peripheral blood mononuclear cells of adult Wistar rats. Exp Physiol. 2021;106(10):2107–2123. https://doi.org/10.1113/EP089553. This experimental work shows the simultaneous activation of peripheral immune cells and kidney infiltration in ovariectomized Wistar rats that develop salt-sensitive hypertension.
Szmuilowicz ED, Adler GK, Williams JS, Green DE, Yao TM, Hopkins PN, Seely EW. Relationship between aldosterone and progesterone in the human menstrual cycle. J Clin Endocrinol Metab. 2006;91(10):3981–7. https://doi.org/10.1210/jc.2006-1154.
Rojas-Vega L, Gamba G. Mini-review: regulation of the renal NaCl cotransporter by hormones. Am J Physiol Renal Physiol. 2016;310(1):F10–4. https://doi.org/10.1152/ajprenal.00354.2015.
•• Titze J, Luft FC, Bauer K, Dietsch P, Lang R, Veelken R, Wagner H, Eckardt KU, Hilgers KF. Extrarenal Na+ balance, volume, and blood pressure homeostasis in intact and ovariectomized deoxycorticosterone-acetate salt rats. Hypertension. 2006;47:1101–7 https://doi.org/10.1161/01.HYP.0000221039.17735.1a. This paper shows findings about a third compartment to buffer body sodium.
Lara LS, McCormack M, Semprum-Prieto LC, Shenouda S, Majid DS, Kobori H, Navar LG, Prieto MC. AT1 receptor-mediated augmentation of angiotensinogen, oxidative stress, and inflammation in ANG II-salt hypertension. Am J Physiol Renal Physiol. 2012;302(1):F85-94. https://doi.org/10.1152/ajprenal.00351.2011.
• Reckelhoff JF, Fortepiani LA. Novel mechanisms responsible for postmenopausal hypertension. Hypertension. 2004;43(5):918–23. https://doi.org/10.1161/01.HYP.0000124670.03674.15. This review discussed possible mechanisms that could play a role in postmenopausal hypertension and proposed the aging female spontaneously hypertensive rat as a postmenopausal women model.
• Felder RA, Gildea JJ, Xu P, Yue W, Armando I, Carey RM, Jose PA. Inverse salt sensitivity of blood pressure: mechanisms and potential relevance for prevention of cardiovascular disease. Curr Hypertens Rep. 2022;24(9):361–74. https://doi.org/10.1007/s11906-022-01201-9. This review describes another face ofsodium regulation: when salt intake is low.
Banday AA, Lokhandwala MF.Renal dopamine oxidation and inflammation in high salt fed rats. J Am Heart Assoc. 2020;9(1):e014977. doi: https://doi.org/10.1161/JAHA.119.014977.
Brismar H, Holtbäck U, Aperia A. Mechanisms by which intrarenal dopamine and ANP interact to regulate sodium metabolism. Clin Exp Hypertens. 2000;22(3):303–7.
Azurmendi PJ, Oddo EM, Obika LF, Corbera NL, Martín RS, Ibarra FR, Arrizurieta EE. Gonadectomy influences blood pressure through the kallikrein-kinin system. Kidney Blood Press Res. 2009;32(5):342–8.
• Di Ciano LA, Azurmendi PJ, Toledo JE, Oddo EM, Zotta E, Ochoa F, Arrizurieta EE, Ibarra FR. Ovariectomy causes overexpression of renal Na(+), K(+)-ATPase and sodium-sensitive hypertension in adult Wistar rats. Clin Exp Hypertens. 2013;35(7):475–83. https://doi.org/10.3109/10641963.2012.758273. This research shows a first description about a salt-sensitive hypertension experimental model developed in ovariectomized adult Wistar rats.
Campese VM, Karubian F, Bigazzi R. Hemodynamic alterations and urinary albumin excretion in patients with essential hypertension. Am J Kidney Dis. 1993;21(5 Suppl 2):15–21.
• Li D, Cheng SX, Fisone G, Caplan MJ, Ohtomo Y, Aperia A. Effects of okadaic acid, calyculin A, and PDBu on state of phosphorylation of rat renal Na+-K+-ATPase. Am J Physiol. 1998;275(6):F863–9. This research shows the posttranslational regulation of renal sodium pump by phosphorylation/dephosphorylation.
Ibarra FR, Cheng SX, Agrén M, Svensson LB, Aizman O, Aperia A. Intracellular sodium modulates the state of protein kinase C phosphorylation of rat proximal tubule Na+, K+-ATPase. Acta Physiol Scand. 2002;175(2):165–71.
• Di Ciano LA, Azurmendi PJ, Colombero C, Levin G, Oddo EM, Arrizurieta EE, Nowicki S, Ibarra FR. Defective renal dopamine function and sodium-sensitive hypertension in adult ovariectomized Wistar rats: role of the cytochrome P-450 pathway. Am J Physiol Renal Physiol. 2015;308(12):F1358–68. This research demonstrated the defective phosphorylation of the renal sodium pump in the ovariectomized salt-sensitive Wistar rats experimental model.
Sandberg K, Ji H, Einstein G, Au A, Hay M. Is immune system-related hypertension associated with ovarian hormone deficiency? Exp Physiol. 2016;101(3):368–74.
•• Rosenzweig R, Gupta S, Kumar V, Gumina RJ, Bansal SS. Estrogenic bias in T-lymphocyte biology: implications for cardiovascular disease. Pharmacol Res. 2021;170:105606. This review overviews the estrogen effects on T–cell regulation and cardiovascular disease.
Phiel KL, Henderson RA, Adelman SJ, Elloso MM. Differential estrogen receptor gene expression in human peripheral blood mononuclear cell populations. Immunol Lett. 2005;97(1):107–13.
Grossman CJ, Roselle GA. The interrelationship of the HPG-thymic axis and immune system regulation. J Steroid Biochem. 1983;19(1B):461–7.
• Jacobsen H, Klein SL. Sex differences in immunity to viral infections. Front Immunol. 2021;12:720952. Interesting review which focuses on sex-specific differences in immunity and outcomes during SARS-CoV-2 infection.
Hao S, Zhao J, Zhou J, Zhao S, Hu Y, Hou Y. Modulation of 17beta-estradiol on the number and cytotoxicity of NK cells in vivo related to MCM and activating receptors. Int Immunopharmacol. 2007;7(13):1765–75.
Bengtsson AK, Ryan EJ, Giordano D, Magaletti DM, Clark EA. 17beta-estradiol (E2) modulates cytokine and chemokine expression in human monocyte-derived dendritic cells. Blood. 2004;104(5):1404–10.
Knöferl MW, Angele MK, Diodato MD, Schwacha MG, Ayala A, Cioffi WG, Bland KI, Chaudry IH. Female sex hormones regulate macrophage function after trauma-hemorrhage and prevent increased death rate from subsequent sepsis. Ann Surg. 2002;235(1):105–12.
Adachi A, Honda T, Egawa G, Kanameishi S, Takimoto R, Miyake T, Hossain MR, Komine M, Ohtsuki M, Gunzer M, Ikuta K, Kabashima K. Estradiol suppresses psoriatic inflammation in mice by regulating neutrophil and macrophage functions. J Allergy Clin Immunol. 2022;150(4):909-919.e8.
Mateus D, Sebastião AI, Carrascal MA, Carmo AD, Matos AM, Cruz MT. Crosstalk between estrogen, dendritic cells, and SARS-CoV-2 infection. Rev Med Virol. 2022;32(3): e2290.
• Chen RY, Fan YM, Zhang Q, Liu S, Li Q, Ke GL, Li C, You Z. Estradiol inhibits Th17 cell differentiation through inhibition of RORγT transcription by recruiting the ERα/REA complex to estrogen response elements of the RORγT promoter. J Immunol. 2015;194(8):4019–28. This research shows the pathway used by estrogen to inhibit Th17 cell polarization.
•• Polanczyk MJ, Hopke C, Vandenbark AA, Offner H. Estrogen-mediated immunomodulation involves reduced activation of effector T cells, potentiation of Treg cells, and enhanced expression of the PD-1 costimulatory pathway. J Neurosci Res. 2006;84(2):370–8. This research shows findings related to estrogen modulation of T cell subpopulations and involved pathways.
Straub RH. The complex role of estrogens in inflammation. Endocr Rev. 2007;28:521–74. https://doi.org/10.1210/er.2007-0001.
Polanczyk MJ, Carson BD, Subramanian S, Afentoulis M, Vandenbark AA, Ziegler SF, Offner H. Cutting edge: estrogen drives expansion of the CD4+CD25+ regulatory T cell compartment. J Immunol. 2004 15;173(4):2227–30.
•• Mattson DL. Immune mechanisms of salt-sensitive hypertension and renal end-organ damage. Nat Rev Nephrol. 2019;15(5):290–300. Interesting review of the relationship among salt sensitivity, immune mechanisms, and hypertension. A hypothesis regarding the onset and evolution of those variables is proposed.
Butts CL, Shukair SA, Duncan KM, Bowers E, Horn C, Belyavskaya E, et al. Progesterone inhibits mature rat dendritic cells in a receptor-mediated fashion. Int Immunol. 2007;19:287–96.
Lee JH, Ulrich B, Cho J, Park J, Kim CH. Progesterone promotes differentiation of human cord blood fetal T cells into T regulatory cells but suppresses their differentiation into Th17 cells. J Immunol. 2011;187:1778–87.
•• Guzik TJ, Hoch NE, Brown KA, McCann LA, Rahman A, Dikalov S, Goronzy J, Weyand C, Harrison DG. Role of the T cell in the genesis of angiotensin II induced hypertension and vascular dysfunction. J Exp Med. 2007;204(10):2449–60. First work describing the importance of immunity in arterial hypertension in male mice.
Norlander AE, Madhur MS, Harrison DG. The immunology of hypertension. J Exp Med. 2018;215(1):21–33. Erratum in: J Exp Med. 2018
•• Pai AV, Maddox T, Sandberg K. T Cells and hypertension: solved and unsolved mysteries regarding the female rat. Physiology (Bethesda). 2018;33(4):254–260. This review analyzes the role of T cells in female rats as hypertension promoters.
Xiao L, Kirabo A, Wu J, Saleh MA, Zhu L, Wang F, Takahashi T, Loperena R, Foss JD, Mernaugh RL, Chen W, Roberts J 2nd, Osborn JW, Itani HA, Harrison DG. Renal denervation prevents immune cell activation and renal inflammation in angiotensin ii-induced hypertension. Circ Res. 2015;117(6):547–57.
Mikolajczyk TP, Nosalski R, Szczepaniak P, Budzyn K, Osmenda G, Skiba D, Sagan A, Wu J, Vinh A, Marvar PJ, Guzik B, Podolec J, Drummond G, Lob HE, Harrison DG, Guzik TJ. Role of chemokine RANTES in the regulation of perivascular inflammation, T-cell accumulation, and vascular dysfunction in hypertension. FASEB J. 2016;30(5):1987–99.
•• Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, Park JK, Beck FX, Müller DN, Derer W, Goss J, Ziomber A, Dietsch P, Wagner H, van Rooijen N, Kurtz A, Hilgers KF, Alitalo K, Eckardt KU, Luft FC, Kerjaschki D, Titze J. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat Med. 2009;15(5):545–52. This research paper describes different strategies to demonstrate the role of interstitial and macrophages to regulate salt balance. The work proposes a hypertonic sodium accumulation in the skin of male rats and mice.
• Ren J, Crowley SD. Role of T-cell activation in salt-sensitive hypertension. Am J Physiol Heart Circ Physiol. 2019;316(6):H1345-H1353. Interesting review with clear concepts concerning T cells, sodium, and hypertension.
Pernis AB. Estrogen and CD4+ T cells. Curr Opin Rheumatol. 2007;19(5):414–20.
Mirandola L, Wade R, Verma R, Pena C, Hosiriluck N, Figueroa JA, Cobos E, Jenkins MR, Chiriva-Internati M. Sex-driven differences in immunological responses: challenges and opportunities for the immunotherapies of the third millennium. Int Rev Immunol. 2015;34(2):134–42.
Kleinewietfeld M, Manzel A, Titze J, Kvakan H, Yosef N, Linker RA, Muller DN, Hafler DA. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature. 2013;496(7446):518–22.
•• Norlander AE, Saleh MA, Pandey AK, Itani HA, Wu J, Xiao L, Kang J, Dale BL, Goleva SB, Laroumanie F, Du L, Harrison DG, Madhur MS. A salt-sensing kinase in T lymphocytes, SGK1, drives hypertension and hypertensive end-organ damage. JCI Insight. 2017;2(13):e92801. This research demonstrated the phenotypic changes in T lymphocytes in mice exposed to high sodium levels associated with hypertension.
Karitskaya I, Aksenov N, Vassilieva I, Zenin V, Marakhova I. Long-term regulation of Na, K-ATPase pump during T-cell proliferation. Pflugers Arch. 2010;460(4):777–89.
•• Vereninov AA, Vassilieva IO, Yurinskaya VE, Matveev VV, Glushankova LN, Lang F, Matskevitch JA. Differential transcription of ion transporters, NHE1, ATP1B1, NKCC1 in human peripheral blood lymphocytes activated to proliferation. Cell Physiol Biochem. 2001;11(1):19–26. This article shows part of the machinery that human lymphocytes use to activate their proliferation.
Köchl R, Thelen F, Vanes L, Brazão TF, Fountain K, Xie J, Huang CL, Lyck R, Stein JV, Tybulewicz VL. WNK1 kinase balances T cell adhesion versus migration in vivo. Nat Immunol. 2016;17(9):1075–83. Erratum in: Nat Immunol. 2017;18(2):246.
Lang F, Artunc F, Vallon V. The physiological impact of the serum and glucocorticoid-inducible kinase SGK1. Curr Opin Nephrol Hypertens. 2009;18(5):439–48.
Buttarelli FR, Fanciulli A, Pellicano C, Pontieri FE. The dopaminergic system in peripheral blood lymphocytes: from physiology to pharmacology and potential applications to neuropsychiatric disorders. Curr Neuropharmacol. 2011;9(2):278–88.
Barbaro NR, Foss JD, Kryshtal DO, Tsyba N, Kumaresan S, Xiao L, Mernaugh RL, Itani HA, Loperena R, Chen W, Dikalov S, Titze JM, Knollmann BC, Harrison DG, Kirabo A. Dendritic cell amiloride-sensitive channels mediate sodium-induced inflammation and hypertension. Cell Rep. 2017;21(4):1009–20.
Fehrenbach DJ, Mattson DL. Inflammatory macrophages in the kidney contribute to salt-sensitive hypertension. Am J Physiol Renal Physiol. 2020;318(3):F544–8. https://doi.org/10.1152/ajprenal.00454.2019.
Rudemiller N, Lund H, Jacob HJ, Geurts AM, Mattson DL. PhysGen Knockout Program. CD247 modulates blood pressure by altering T-lymphocyte infiltration in the kidney. Hypertension. 2014;63(3):559–64.
Evans LC, Petrova G, Kurth T, Yang C, Bukowy JD, Mattson DL, Cowley AW Jr. Increased perfusion pressure drives renal T-cell infiltration in the Dahl salt-sensitive rat. Hypertension. 2017;70(3):543–51.
Pai AV, West CA, de Souza AMA, Kadam PS, Pollner EJ, West DA Jr, Li J, Ji H, Wu X, Zhu MJ, Baylis C, Sandberg K. Renal T cell infiltration occurs despite attenuation of development of hypertension with hydralazine in Envigo’s female Dahl rat maintained on a low-Na+ diet. Am J Physiol Renal Physiol. 2019;317(3):F572–83.
Vlachovsky SG, Azurmendi PJ, Oddo EM, Di Ciano LA, Rodriguez SR, Goette NP, Paz LA, Silberstein C, Ibarra FR. Immune response after lowering-blood pressure in salt-sensitive hypertension rat model. Joint Meeting SAIC SAI&FAIC SAFIS 2022. Abstract published in: Medicina, Buenos Aires. 2022;82 (Suppl V):208.URL: https://medicinabuenosaires.com/revistas/vol82-22/s5/1s5.pdf.
Lenda DM, Boegehold MA. Effect of a high-salt diet on oxidant enzyme activity in skeletal muscle microcirculation. Am J Physiol Heart Circ Physiol. 2002;282(2):H395-402.
Guers JJ, Kasecky-Lardner L, Farquhar WB, Edwards DG, Lennon SL. Voluntary wheel running prevents salt-induced endothelial dysfunction: role of oxidative stress. J Appl Physiol (1985). 2019 1;126(2):502–510.
Greaney JL, DuPont JJ, Lennon-Edwards SL, Sanders PW, Edwards DG, Farquhar WB. Dietary sodium loading impairs microvascular function independent of blood pressure in humans: role of oxidative stress. J Physiol. 2012;590(21):5519–28.
Eisenach JH, Gullixson LR, Kost SL, Joyner MJ, Turner ST, Nicholson WT. Sex differences in salt sensitivity to nitric oxide dependent vasodilation in healthy young adults. J Appl Physiol (1985). 2012;112(6):1049–53.
Scuteri A, Stuehlinger MC, Cooke JP, Wright JG, Lakatta EG, Anderson DE, Fleg JL. Nitric oxide inhibition as a mechanism for blood pressure increase during salt loading in normotensive postmenopausal women. J Hypertens. 2003;21(7):1339–46.
Costa-Fraga FP, Goncalves GK, Souza-Neto FP, Reis AM, Capettini LA, Santos RA, Fraga-Silva RA, Stergiopulos N, da Silva RF. Age-related changes in vascular responses to angiotensin-(1–7) in female mice. J Renin Angiotensin Aldosterone Syst. 2018;19(3):1470320318789332.
Harrison-Bernard LM, Schulman IH, Raij L. Postovariectomy hypertension is linked to increased renal AT1 receptor and salt sensitivity. Hypertension. 2003;42(6):1157–63.
Meyer MR, Haas E, Barton M. Gender differences of cardiovascular disease: new perspectives for estrogen receptor signaling. Hypertension. 2006;47(6):1019–26.
Ylikorkala O, Orpana A, Puolakka J, Pyörälä T, Viinikka L. Postmenopausal hormonal replacement decreases plasma levels of endothelin-1. J Clin Endocrinol Metab. 1995;80(11):3384–7.
Komesaroff PA, Esler MD, Sudhir K. Estrogen supplementation attenuates glucocorticoid and catecholamine responses to mental stress in perimenopausal women. J Clin Endocrinol Metab. 1999;84(2):606–10.
SenthilKumar G, Katunaric B, Bordas-Murphy H, Young M, Doren EL, Schulz ME, Widlansky ME, Freed JK. 17β-Estradiol promotes sex-specific dysfunction in isolated human arterioles. Am J Physiol Heart Circ Physiol. 2023;324(3):H330–7.
Ji H, Kim A, Ebinger JE, Niiranen TJ, Claggett BL, Bairey Merz CN, Cheng S. Sex differences in blood pressure trajectories over the life course. JAMA Cardiol. 2020;5(3):19–26.
Jobin K, Müller DN, Jantsch J, Kurts C. Sodium and its manifold impact on our immune system. Trends Immunol. 2021;42(6):469–79.
•• Bie P. Mechanisms of sodium balance: total body sodium, surrogate variables, and renal sodium excretion. Am J Physiol Regul Integr Comp Physiol. 2018;315(5):R945-R962. This outstanding analytical review about body sodium balance revises and explains classic and recent findings.
• Jhee JH, Park HC, Choi HY. Skin sodium and blood pressure regulation. Electrolyte Blood Press. 2022;20(1):1–9. This review debates the question of whether skin sodium accumulation is “good or evil.” The potential double edge sword of skin-high sodium.
Mobasheri A. Correlation between [Na+], [glycosaminoglycan] and Na+/K+ pump density in the extracellular matrix of bovine articular cartilage. Physiol Res. 1998;47(1):47–52.
•• Wiig H, Luft FC, Titze JM. The interstitium conducts extrarenal storage of sodium and represents a third compartment essential for extracellular volume and blood pressure homeostasis. Acta Physiol (Oxf). 2018;222(3). This review describes the findings that support the theory about the interstitium as a third sodium compartment and analyzes many many excellent works performed by this research group.
Schmidlin O, Sebastian AF, Morris RC Jr. What initiates the pressor effect of salt in salt-sensitive humans? Observations in normotensive blacks Hypertension. 2007;49(5):1032–9.
Laffer CL, Scott RC 3rd, Titze JM, Luft FC, Elijovich F. Hemodynamics and salt-and-water balance link sodium storage and vascular dysfunction in salt-sensitive subjects. Hypertension. 2016;68(1):195–203.
Titze J, Krause H, Hecht H, Dietsch P, Rittweger J, Lang R, Kirsch KA, Hilgers KF. Reduced osmotically inactive Na storage capacity and hypertension in the Dahl model. Am J Physiol Renal Physiol. 2002;283(1):F134–41.
Titze J, Shakibaei M, Schafflhuber M, Schulze-Tanzil G, Porst M, Schwind KH, Dietsch P, Hilgers KF. Glycosaminoglycan polymerization may enable osmotically inactive Na+ storage in the skin. Am J Physiol Heart Circ Physiol. 2004;287(1):H203–8.
• Titze J, Lang R, Ilies C, Schwind KH, Kirsch KA, Dietsch P, Luft FC, Hilgers KF. Osmotically inactive skin Na+ storage in rats. Am J Physiol Renal Physiol. 2003;285(6):F1108–17. This article is one of the first original works from the group. Studies were conducted in male, fertile female, and ovariectomized female Sprague Dawley (SD) rats, to demonstrate the sodium storage in the skin.
Rakova N, Jüttner K, Dahlmann A, Schröder A, Linz P, Kopp C, Rauh M, Goller U, Beck L, Agureev A, Vassilieva G, Lenkova L, Johannes B, Wabel P, Moissl U, Vienken J, Gerzer R, Eckardt KU, Müller DN, Kirsch K, Morukov B, Luft FC, Titze J. Long-term space flight simulation reveals infradian rhythmicity in human Na(+) balance. Cell Metab. 2013 8;17(1):125–31.
Dahlmann A, Dörfelt K, Eicher F, Linz P, Kopp C, Mössinger I, Horn S, Büschges-Seraphin B, Wabel P, Hammon M, Cavallaro A, Eckardt KU, Kotanko P, Levin NW, Johannes B, Uder M, Luft FC, Müller DN, Titze JM. Magnetic resonance-determined sodium removal from tissue stores in hemodialysis patients. Kidney Int. 2015;87(2):434–41.
Wiig H, Schröder A, Neuhofer W, Jantsch J, Kopp C, Karlsen TV, Boschmann M, Goss J, Bry M, Rakova N, Dahlmann A, Brenner S, Tenstad O, Nurmi H, Mervaala E, Wagner H, Beck FX, Müller DN, Kerjaschki D, Luft FC, Harrison DG, Alitalo K, Titze J. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J Clin Invest. 2013;123(7):2803–15.
Thowsen IM, Karlsen TV, Nikpey E, Haslene-Hox H, Skogstrand T, Randolph GJ, Zinselmeyer BH, Tenstad O, Wiig H. Na+ is shifted from the extracellular to the intracellular compartment and is not inactivated by glycosaminoglycans during high salt conditions in rats. J Physiol. 2022;600(10):2293–309.
•• Rossitto G, Mary S, Chen JY, Boder P, Chew KS, Neves KB, Alves RL, Montezano AC, Welsh P, Petrie MC, Graham D, Touyz RM, Delles C. Tissue sodium excess is not hypertonic and reflects extracellular volume expansion. Nat Commun. 2020;11(1):4222. This work demonstrated that isotonic sodium accumulates not only in the skin, and debates recent findings regarding skin sodium accumulation. The article shows results in male and female rats, and in women and men.
Trincot CE, Caron KM. Lymphatic function and dysfunction in the context of sex differences. ACS Pharmacol Transl Sci. 2019;2(5):311–24.
Karlsen TV, Nikpey E, Han J, Reikvam T, Rakova N, Castorena-Gonzalez JA, Davis MJ, Titze JM, Tenstad O, Wiig H. High-salt diet causes expansion of the lymphatic network and increased lymph flow in skin and muscle of rats. Arterioscler Thromb Vasc Biol. 2018;38(9):2054–64.
•• Machnik A, Dahlmann A, Kopp C, Goss J, Wagner H, van Rooijen N, Eckardt KU, Müller DN, Park JK, Luft FC, Kerjaschki D, Titze J. Mononuclear phagocyte system depletion blocks interstitial tonicity-responsive enhancer binding protein/vascular endothelial growth factor C expression and induces salt-sensitive hypertension in rats. Hypertension. 2010;55(3):755–61. This investigation demonstrates that mononuclear phagocyte system depletion in the skin induces hypertension in male rats treated with DOCA and moderate amounts of sodium loading.
Silha JV, Krsek M, Sucharda P, Murphy LJ. Angiogenic factors are elevated in overweight and obese individuals. Int J Obes (Lond). 2005;29(11):1308–14.
Valenzuela GJ, Brace RA, Longo LD. Lymphatic and vascular responses to fluid infusion in castrated and noncastrated sheep. Am J Physiol. 1987;252(6 Pt 2):R1114–8.
•• Selvarajah V, Mäki-Petäjä KM, Pedro L, Bruggraber SFA, Burling K, Goodhart AK, Brown MJ, McEniery CM, Wilkinson IB. Novel mechanism for buffering dietary salt in humans: effects of salt loading on skin sodium, vascular endothelial growth factor C, and blood pressure. Hypertension. 2017;70(5):930–937. This paper describes the behavior of BP in women and men, and its relationship with subcutaneous electrolytes.
• Barnett AM, Babcock MC, Watso JC, Migdal KU, Gutiérrez OM, Farquhar WB, Robinson AT. High dietary salt intake increases urinary NGAL excretion and creatinine clearance in healthy young adults. Am J Physiol Renal Physiol. 2022 Apr 1;322(4):F392-F402. This research shows a crossover design from low to moderately high sodium and demonstrates that sodium increases urinary NGAL and GFR in women and men pooled together.
Madhur MS, Elijovich F, Alexander MR, Pitzer A, Ishimwe J, Van Beusecum JP, Patrick DM, Smart CD, Kleyman TR, Kingery J, Peck RN, Laffer CL, Kirabo A. Hypertension: do inflammation and immunity hold the key to solving this epidemic? Circ Res. 2021;128(7):908–33.
•• Mill JG, Baldo MP, Molina MDCB, Schmidt MI, Barreto SM, Chor D, Griep RH, Matos SM, Ribeiro ALP, Duncan BB, Aquino EM, Lotufo PA, Bensenor I. Sex-specific patterns in the association between salt intake and blood pressure: the ELSA-Brasil study. J Clin Hypertens (Greenwich). 2019; 21(4):502–509. Excellent study with a large number of adult participants, women and men. Variables are corrected by sex, age, medication, and blood pressure response to sodium intake. This study shows that women have a higher responsiveness of BP according to salt intake than men.
•• Fehrenbach DJ, Nguyen B, Alexander MR, Madhur MS. Modulating T cell phenotype and function to treat hypertension. Kidney360. 2023;4(4):e534-e543. This review highlights the importance of changes in T cell phenotype as potential targets to treat hypertension.
El Khoudary SR. Age at menopause onset and risk of cardiovascular disease around the world. Maturitas. 2020;141:33–8.
Author information
Authors and Affiliations
Contributions
Fernando R. Ibarra, Claudia Silberstein, and Sandra Vlachovsky contributed to the conception and design of the manuscript. Fernando R. Ibarra wrote the first draft of the manuscript. The work was critically revised by Claudia Silberstein, Sandra Vlachovsky, Luis Di Ciano, Elisabet Oddo, and Pablo Azurmendi. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Ethical approval was not required as our study did not involve humans or animals.
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vlachovsky, S.G., Di Ciano, L.A., Oddo, E.M. et al. Role of Female Sex Hormones and Immune Response in Salt-Sensitive Hypertension Development: Evidence from Experimental Models. Curr Hypertens Rep 25, 405–419 (2023). https://doi.org/10.1007/s11906-023-01257-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11906-023-01257-1