Abstract
Purpose of Review
Cardiovascular disease is the leading cause of death and allograft loss among kidney transplant recipients, and hypertension is an independent risk factor for cardiovascular morbidity of this patient population. The etiology of hypertension is multifactorial, including pre-transplant volume overload, post-transplant recipient and donor-associated variables, and transplant-specific causes (immunosuppressive medications, allograft dysfunction and surgical complications such as transplant artery stenosis).
Recent Findings
No randomized controlled trials have assessed the optimal blood pressure targets and explored the best antihypertensive regimen for kidney transplant recipients. According to the large observational studies, it is reasonable to achieve a blood pressure goal of equal to or less than 130/80 mmHg in the long-term follow-up for minimizing the cardiovascular morbidity. The selection of antihypertensive agents should be based on the patient’s co-morbidities; however, the initial choice could be calcium channel blockers especially in the first few months of transplantation. In patients with cardiovascular indications of renin-angiotensin-aldosterone system inhibition, given the well-described benefits in diabetic and proteinuric patients, it is reasonable to consider the use of renin-angiotensin-aldosterone system inhibitors.
Summary
There is a need for future prospective trials in the transplant population to define optimal blood pressure goals and therapies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypertension (HT) remains one of the most common clinical problems in kidney transplant recipients (KTRs) and is a well-recognized risk factor for cardiovascular morbidity and mortality [1, 2]. The overall prevalence varies depending on the transplant population studied and the hypertension definition used. It is associated with factors such as age, male gender, obesity, and smoking that affects the general population, and, also, it is associated with transplant-specific causes [3]. Allograft pathologies, post-transplant proteinuria, immunosuppression-related factors, donor-related factors, and transplant renal artery stenosis are important causes of HT [4].
Several studies have demonstrated the association between HT and allograft survival and cardiovascular morbidity in the literature [5,6,7]. The Collaborative Transplant Study, a cohort of 30.000 KTRs, showed a graded association between systolic and diastolic blood pressure levels and allograft failure [8]. Paoletti et al. showed that HT causes graft dysfunction and increases proteinuria in KTRs [9]. Rigatto et al. reported that high blood pressure increases left ventricular hypertrophy in individuals whose HT is uncontrolled in the post-transplantation period [10]. Despite major clinical concern, no randomized clinical trials are assessing the optimal target blood pressure level and defining the best antihypertensive regimen for this patient population.
Causes of Hypertension in Kidney Transplant Recipients
The pathogenesis of post-transplant HT is multifactorial. In addition to the traditional risk factors encountered in the general population, transplant-specific causes pertain to KTRs including pre-existing recipient, donor, and peritransplant factors such as immunosuppresive medications, delayed graft function, allograft pathologies, and surgical complications (Table 1) [4, 5]. Long standing HT before transplantation, due to the development of vascular stiffness with excess fluid during the dialysis period may produce post-transplant HT. Obesity and metabolic disorders which induce variable injury to the transplanted kidney are effective in the development of post-transplant HT [11, 12]. The development of HT in a transplant recipient can accelerate kidney dysfunction—especially if it is accompanied by proteinuria [6]. Furthermore, acute cellular and/or antibody-mediated rejection episodes, chronic transplant glomerulopathy, and recurrent glomerular disease may be associated with chronic allograft dysfunction and post-transplant HT [13]. The role of native kidneys in the pathogenesis of post-transplant HT has not been completely elucidated. Native kidneys can induce HT through activation of the sympathetic nervous system or increased renin secretion [14, 15]. A few small studies have reported that bilateral native nephrectomies before or after transplantation improve blood pressure control [16,17,18]. In contrast, Midvedt et al. showed no blood pressure improvement after bilateral nephrectomy in a population of 158 living donor transplant recipients [19].
There are numerous donor factors associated with HT in transplant recipients. These include pre-existing donor HT, older donor age, and poor allograft quality including donor kidney size. Guidi et al. reported that the transplantation of a “hypertensive” kidney from a hypertensive donor determines a tenfold increase in the requirement of antihypertensive therapy than the transplantation of a “normotensive” kidney to obtain a similar blood pressure control [20]. Similarly, Blanca et al. reported that post-transplant HT and cardiovascular disease have been observed more frequently in individuals transplanted of the kidneys from extended criteria donors (who have cerebrovascular disease and/or HT) [21]. In another study, el-Agroudy et al. reported that low donor/recipient body weight ratio decreases long-term allograft survival in KTRs [22]. They hypothesized that kidneys from small donors are at risk for graft loss if they are transplanted into large recipients, and they demonstrated that a low donor/recipient body weight contributes to inferior long-term renal allograft survival [22]. It is a well-known fact that the size of the donor kidney relative to the recipient plays a role in the development of post-transplant HT due to underdosing nephrons and subsequent maladaptive hyperfiltration. Even for this purpose, transplant programs have been created to target the fitting of kidneys suitable for the recipient’s height and length [23].
Calcineurin inhibitors and steroids are blamed for the development of HT among KTRs. Steroids are known to increase blood pressure by sodium retention and volume expansion. The development of post-transplant HT depends on the dose of steroids used [24, 25]. Early reduction or discontinuation of the steroid has been reported to have positive effects in maintaining HT control [25], but in today’s practice, discontinuation of steroids is not recommended because it shortens the survival of the transplanted kidney. Cyclosporin, one of the most used drugs in the 1980s in transplantation practice, increases the prevalence of post-transplant HT among KTRs [26]. Cyclosporin, the first-generation drug of calcineurin inhibitors, may impair renal perfusion and lead to HT by increasing renal vasoconstriction, which can lead to HT. Taler et al reported the prevalence of HT in KTRs to be as high as 7090% because of characteristic vascular changes of cyclosporin which causes renal and systemic vasoconstriction [27]. Registry data and observations from clinical trials have suggested lower rates of post-transplant HT with a tacrolimus-based regimen compared with a cyclosporin-based regimen [28, 29]. Eventually, tacrolimus have replaced cyclosporin in almost all immunosuppressive protocols in the current transplantation practice in recent decades. Given the importance of appropriate immunosuppression to avoid acute rejection, we suggest lifestyle modifications and antihypertensive medication (if necessary) instead of modifying the immunosuppression protocol from tacrolimus-based regimen to mTOR inhibitor-based regimen of the KTR.
Disruption of graft functions during acute cellular or antibody-mediated rejection can cause HT development. Activation of the renin-angiotensin-aldosterone system (RAAS) with impaired kidney function, fluid loading of the patient leads to the development and/or deterioration of HT. High-dose steroid therapy and intravenous immunoglobulin (IVIG) treatment during rejection therapies also facilitate HT worsening [30]. HT associated with acute rejection episodes responds well to treatment of rejection, whereas it often worsens with treatment protocols consisting of high-dose steroids and IVIG. HT associated with chronic allograft nephropathy is similar to that associated with chronic kidney disease (CKD). We usually advise lifestyle modifications and antihypertensive medication in addition to attempts to prolong allograft survival.
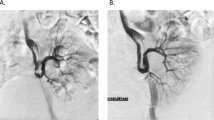
Transplant renal artery stenosis is a vascular complication that can be seen in 1–25% after transplantation [31]. It can be seen between 3 months and 2 years after transplantation and often develops in the surgical anastomosis line [31]. While it is a potentially treatable cause of post-transplant HT, failure to diagnose may compromise transplant kidney outcomes. When stenosis is treated interventionally, both the graft function of the KTR is maintained for a long time and blood pressure control becomes easier. Progression of stenosis in the transplant artery, worsening of graft functions, and/or uncontrolled HT development indicate the need for revascularization [32]. Revascularization options can be summarized as endovascular interventions (balloon angioplasty and stent application) and surgical revision of the renal artery. Chen et al. compared outcomes between angioplasty and stent placement methods in the treatment of transplant renal artery stenosis [33]. They showed that there was no difference between the 2 methods in terms of blood pressure, graft functions, and graft survival [33]. They also showed that stent placement resulted in fewer cases of re-stenosis compared with balloon angioplasty alone [33]. Surgical revision of the renal artery should be reserved for cases where angioplasty fails.
Which Blood Pressure Goal Should be Targeted in Transplant Recipients?
No randomized controlled trials are investigating the optimal blood pressure goal in transplant recipients. There is also no evidence to support the superiority of one treatment choice over another. Considering the various HT guidelines written in the last 10 years and the recently published “Turkish Hypertension Consensus Report 2019,” the comments and recommendations regarding optimal blood pressure level in KTRs are insufficient [34].
In several international guidelines, such as International Society of Hypertension Global Hypertension Practice Guideline from 2020, Joint National Committee (JNC) 8 Guideline from 2014, and European guidelines from 2013, recommend a blood pressure less than 140/90 mmHg for the general population, 130/80 mmHg for CKD population, and diabetics [35,36,37]. In the full text of these guidelines, although diabetics and CKD patients are evaluated, there are no specific recommendations for kidney transplantation.
One of the 2 guidelines to review the blood pressure targets of KTRs is the HT Guideline published in 2017 in partnership with the American Society of Cardiology and the American Heart Association [38•]. According to this guideline, the blood pressure goal in KTRs is suggested as equal to or less than 130/80 mmHg [38•]. This recommendation was based on the results of the SPRINT study which was published in 2015 [39•]. The blood pressure goal was extrapolated from CKD patients’ data and should be evaluated by randomized controlled trials, since no transplanted patient was included in the SPRINT trial.
Another recommendation to interpret the blood pressure goal in KTRs is found in the KDIGO (Kidney Disease: Improving Global Outcomes) 2012 guideline that interpret the blood pressure goal as equal to or less than 130/80 mmHg [40]. The level of evidence is classified as 2D which is based on expert opinion and is provided solely by epidemiologic data concerning long-term graft survival [40].
Although there is no recommendation regarding transplant recipients in the Canadian guideline published in 2017, blood pressure goal for non-diabetic CKD patients is interpreted as less than 140/90 mmHg [41]. Finally, according to the recently published national guideline, Turkish Hypertension Consensus Report, 2019, blood pressure goal is less than 140/80 mmHg in CKD patients whose age is > 65 years and equal to or less than 130/80 mmHg whose age is < 65 years. Transplant recipients are not specified separately in this guideline [34]. The blood pressure goals recommended by various guidelines in individuals with CKD, diabetics, and KTRs are summarized in Table 2.
Therefore, the optimal blood pressure values recommended for transplant recipients are the conclusions from epidemiological studies evaluating long-term graft survival and cardiovascular morbidity and are at the level of expert opinion (level of evidence 2D). According to a recently published meta-analysis, the recommended target blood pressure value should be less than 140/90 mmHg [42].
According to our opinion, while a KTR is considered as a stage 2–3 CKD patient even under the best conditions, it is necessary to optimize the patient’s cardiovascular risk factors. Blood pressure targets should not be set very tightly at the beginning of the transplantation (first 6 months), in which a blood pressure target should be planned to ensure adequate organ perfusion and reduce the risks of hypotension and graft thrombosis. This target can be considered as less than 140/90 mmHg in the first 6 months. This target should be considered as equal to or less than 130/80 mmHg in order to control cardiovascular risks, prevent target organ damage, and prolong graft survival after 6 months of transplantation.
Which Anti-hypertensive Drug Could be Selected in Transplant Recipients?
There are no randomized controlled trials that determine optimal antihypertensive therapy in KTRs. The choice of treatment should be individualized by evaluating the patient’s co-morbidities, drug to drug interactions, and efficacy/tolerability index of the drug.
Calcium channel blockers (CCB) are generally the first choice in a KTR with high blood pressure. They are used as the first option to neutralize the vasoconstrictive effects of calcineurin inhibitors, as they reduce renal vascular resistance. Kuypers et al. reported that transplant recipients using calcineurin inhibitors and CCBs had better graft functions in post-transplant 2 years than those who did not use CCBs [43]. Non-dihydropyridine CCBs (diltiazem and verapamil) should be used with caution in KTRs as they increase calcineurin inhibitor drug levels [44]. On the contrary, in Turkey, diltiazem is preferred by many transplant nephrologists at stage 1-2 HT, as it allows the liver cytochrome p450 (CYP3A4) enzyme inhibition to reduce the dose of the calcineurin inhibitor used [45].
Cross et al. published a Cochrane Database systematic review, which included 29 studies comparing CCBs to placebo in 2262 KTRs, 10 studies comparing RAAS blocker with placebo in 445 KTRs, and 7 studies comparing RAAS blockers with CCBs in 405 KTRs [46••]. They found that CCBs reduces graft loss (risk ratio [RR], 0.75, 95% CI, 0.57–0.99) and improves glomerular filtration rate (4.45 mL/min, 95% CI, 2.22–6.68); however RAAS blockers have inconclusive results for glomerular filtration rate (−8.07 mL/min, 95% CI, −18.57 −2.43) and graft loss [46••]. They also reported that in direct comparison with CCBs, although RAAS blockers decrease proteinuria (−0.28 g/24 h, 95% CI, −0.47 −0.10), the data of graft loss was inconclusive (RR, 7.37, 95% CI, 0.39–140.35). The authors concluded that CCBs may be preferred as first-line antihypertensive treatment in KTRs [46••].
There is no evidence-based difference between dihydropyridine and non-dihydropyridine CCB use in transplantation practice. There is not any trial examining the possible beneficial effects—allograft survival, graft loss, decreasing proteinuria—of different sub-classes of CCBs in the literature. While we have been using diltiazem as first-line antihypertensive therapy in the first 6 months in KTRs in our transplantation unit to reduce the dose of calcineurin inhibitor used, however in recent years, we are using lercanidipine as the first option because of more prominent antihypertensive effect in hypertensive KTRs. Furthermore, lercanidipine dilates both afferent and efferent glomerular arteries and improves renal resistive index which is important in long-term allograft survival [47, 48]. Prospective, randomized controlled studies are needed to add crucial insight into this subject.
Renin-angiotensin-aldosterone system (RAAS) blockers, on the other hand, are a group that is withdrawn in the early post-transplant period but helps maintain graft functions by reducing intra-glomerular pressure and proteinuria in the long term. In the early period, the clinician would disapprove treating with RAAS blockers because of hyperkalemia, while graft function has just started. Moreover, treatment with RAAS blockade may impair patient’s anemia [49]. Additionally, RAAS blockade may disrupt graft functions, which create confusion in terms of acute rejection. According to our opinion, because of these reasons, it is an uncomfortable treatment choice for both the transplantation team and the patient in the first several months. However, in the long term, the progression-slowing effect of RAAS inhibitors especially in proteinuric recipients cannot be neglected.
In an observational study by Shin et al., RAAS blockade has been shown to prolong graft survival in proteinuric transplant recipients [50]. In this observational study, Shin et al. analyzed 2680 KTRs who had a functioning graft at 6 months after transplantation. They assessed the association of RAAS blockers with patient and graft survival using time-dependent Cox analysis [50]. They found that RAAS blockade is positively associated with lower risk of graft loss [50].
In a retrospective study conducted by Heinze et al., RAAS blockers have been reported to show better graft survival in the post-transplant 10th year when compared with other antihypertensive drugs [51]. The authors reported that the 10-year patient survival rate was 74% in the RAAS blockade group, but only 53% in the non-RAAS blockade group [51]. Similarly, 10-year graft survival was better in the RAAS blockade group when compared with the non-RAAS blockage group (59% vs 41%, p = 0.002). They concluded that the use of RAAS blockers was associated with longer patient and graft survival and more frequent use of these medications may reduce the incidence of renal allograft failure in KTRs [51].
A prospective, double-blinded, randomized controlled trial of 213 KTRs evaluated the effectiveness of ramipril versus placebo on an endpoint of worsening allograft function, development of end-stage kidney disease, or death [52]. The authors found that treatment with ramipril compared with placebo did not lead to a significant reduction in doubling of serum creatinine, end-stage kidney disease, or death in KTRs with proteinuria. They concluded that their results do not support the use of RAAS blockers for improving clinical outcomes in transplant recipients [52].
The largest study on this subject—which was consisted of 500 KTRs—is the SECRET (The Study on Evaluation of Candesartan Cilexetil after Renal Transplantation) study in which KTRs were compared while using candesartan or placebo [53••]. Philipp et al. have reported that proteinuria was lower and serum creatinine and potassium were higher in patients using candesartan [53••]. The authors have also noted that RAAS inhibitor allows better blood pressure control with less antihypertensive co-medication than the usual antihypertensive medication [53••]. In the second year, all-cause mortality, cardiovascular mortality, and graft survival were similar between candesartan and placebo group [53••]. Enrollment into this study was halted prematurely because of a lower than expected primary event rate (all-cause mortality, cardiovascular mortality, and graft survival) which would not cross the statistical boundary required to establish the superiority of one treatment over the other. The authors reported that none of the group comparisons for the different components of all-cause mortality, cardiovascular morbidity and all-cause graft failure was statistically significant [53••].
Hiremath et al. conducted a meta-analysis of 8 studies using RAAS blockers and placebo in 1500 KTRs [54••]. Their analysis showed RAAS blockers did not significantly change all-cause mortality (RR, 0.96; 95%CI, 0.62–1.51), graft survival (RR, 0.76; 95%CI, 0.49–1.18), and serum creatinine doubling (RR, 0.84, 95%CI, 0.51–1.39) compared with control group [54••]. The authors concluded that a sample wider than 10.000 patients would be needed to definitively answer whether RAAS blockade improves allograft survival in this patient population [54••].
Therefore, definitive evidence for the benefit of RAAS blockers in KTRs is still lacking. According to our opinion, RAAS blockers and/or RAAS blockers/CCBs could be a first choice of anti-hypertensive therapy in KTRs after stabilization of early transplantation period. We especially suggest RAAS blockers use in diabetic and/or proteinuric patients and also for post-transplant erythrocytosis; but its use could also be considered in non-proteinuric and non-diabetic KTRs. On the other hand, we agree with the general clinical nephrology practice that supports CCBs as a first choice of anti-hypertensive medication in the first 6 months of the post-transplantation period.
Beta-blockers could be selected in transplant recipients with coronary artery disease and/or arrhythmia. Alpha-blockers can also be used in patients with prostate hyperplasia. Although transplant nephrologists do not prefer in their clinical transplantation practice, it is also possible to use thiazide diuretics and mineralocorticoid receptor blockers in patients who can tolerate these antihypertensives.
Conclusion
In summary, we suggest that the blood pressure target could be considered as less than 140/90 mmHg to supply adequate organ perfusion in the first few months of transplantation. A target less than 130/80 mmHg to prevent target organ damage, cardiovascular morbidity, and prolong graft survival could be considered after stabilization. In KTRs with proteinuria, diabetes, or coronary artery disease, the RAAS blockers or RAAS blocker/CCB combination seems to be the best option. Further prospective, controlled trials are needed to define optimal blood pressure goals and therapies.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lentine KL, Costa SP, Weir MR, Robb JF, Fleisher LA, Kasiske BL, et al. American Heart Association Council on the Kidney in Cardiovascular Disease and Council on Peripheral Vascular Disease: American Heart Association; American College of Cardiology Foundation. Cardiac disease evaluation and management among kidney and liver transplantation candidates: a scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2012;126(5):617–63.
Collins AJ, Foley RN, Chavers B, Gilbertson D, Herzog C, Johansen K, et al. United States Renal Data System 2011 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States. Am J Kidney Dis. 2012;59(1 Suppl 1):A7 e1-420.
Rigatto C. Clinical epidemiology of cardiac disease in renal transplant recipients. Semin Dial. 2003;16(2):106–10.
Kasiske BL, Anium S, Shah R, Skogen J, Kandaswamy C, Danielson B, et al. Hypertension after kidney transplantation. Am J Kidney Dis. 2004;43(6):1071–81.
Glicklich D, Lamba R, Pawar R. Hypertension in the kidney transplant recipient: overview of pathogenesis, clinical assessment, and treatment. Cardiol Rev. 2017;25(3):102–9.
Mange KC, Cizman B, Joffe M, Feldman HI. Arterial hypertension and renal allograft survival. JAMA. 2000;283(5):633–8.
Opelz G, Dohler B. Collobrative Transplant Study. Improved long-term outcomes after renal transplantation associated with blood pressure control. Am J Transplant. 2005;5(11):2725–31.
Opelz G, Wujciak T, Ritz E. Association of chronic kidney graft failure with recipient blood pressure. Collaborative Transplant Study. Kidney Int. 1998;53(1):217–22.
Paoletti E, Gherzi M, Amidone M, Massarino F, Cannella G. Association of arterial hypertension with renal target organ damage in kidney transplant recipients: the predictive role of ambulatory blood pressure monitoring. Transplantation. 2009 Jun;87(12):1864–9.
Rigatto C, Foley R, Jeffery J, Negrijn C, Tribula C, Parfrey P. Electrocardiographic left ventricular hypertrophy in renal transplant recipients: prognostic value and impact of blood pressure and anemia. J Am Soc Nephrol. 2003;14(2):462–8.
Lafranca JA, IJermans JN, Betjes MG, Dor FJ. Body mass index and outcome in renal transplant recipients: a systematic review and meta-analysis. BMC Med. 2015;13:111.
Molnar MZ, Lazar AS, Lindner A, Fornadi K, Czira ME, Dunai A, et al. Sleep apnea is associated with cardiovascular risk factors among kidney transplant recipients. Clin J Am Soc Nephrol. 2010;5(1):125–32.
Weir MR, Burgess ED, Cooper JE, Fenves AZ, Goldsmith D, McKay D, et al. Assessment and management of hypertension in transplant patients. J Am Soc Nephrol. 2015;26(6):1248–60.
Hausberg M, Kosch M, Harmelink P, Barenbrock M, Hohage H, Kisters K, et al. Sympathetic nerve activity in end stage renal disease. Circulation. 2002;106(15):1974–9.
Curtis JJ, Luke RG, Diethelm AG, Whelchel JD, Jones P. Benefits of removal of native kidneys in hypertension after renal transplantation. Lancet. 1985;2(8458):739–42.
Huysmans FT, Hoitsma AJ, Koene RA. Factors determining the prevalence of hypertension after renal transplantation. Nephrol Dial Transplant. 1987;2(1):34–8.
Fricke L, Doehn C, Steinhoff J, Sack K, Jocham D, Fornara P. Treatment of posttransplant hypertension by laparoscopic bilateral nephrectomy? Transplantation. 1998;65(9):1182–7.
Fornara P, Doehn C, Fricke L, Durek C, Thyssen G, Jocham D. Laparoscopic bilateral nephrectomy: results in 11 renal transplant patients. J Urol. 1997;157(2):445–9.
Midvedt K, Hartmann A, Bentdal O, Brekke IB, Fauchald P. Bilateral nephrectomy simultaneously with renal allografting does not alleviate hypertension 3 months following living-donor transplantation. Nephrol Dial Transplant. 1996;11(10):2045–9.
Guidi E, Menghetti D, Milani S, Montagnino G, Palazzi P, Bianchi G. Hypertension may be transplanted with the kidney in humans: a long-term historical prospective follow-up of recipients grafted with kidneys coming from donors with or without hypertension in their families. J Am Soc Nephrol. 1996;7(8):1131–8.
Blanca L, Jimenes T, Cabello M, Sola E, Gutierrez C, Burgos D, et al. Cardiovascular risk in recipients with kidney transplants from expanded criteria donors. Transplant Proc. 2012;44(9):2579–81.
El-Agroudy AE, Hassan NA, Bakr MA, Foda MA, Shokeir AA, Shehab el-Dein AB. Effect of donor/recipient body weight mismatch on patient and graft outcome in living-donor kidney transplantation. Am J Nephrol. 2003;23(5):294–9.
Vella J, Brennan D [Internet]. US: Hypertension after kidney transplantation. Available from: www.uptodate.com Accessed 02/04/2021
Knight SR, Morris PJ. Steroid avoidance or withdrawal after renal transplantation increases the risk of acute rejection but decreases cardiovascular risk. Meta-anal Transplant. 2010;89(1):1–14.
Curtis JJ, Galla JH, Kotchen TA, Lucas B, McRoberts JW, Luke RG. Prevalance of hypertension in a renal transplant population on alternate-day steroid therapy. Clin Nephrol. 1976;5(3):123–7.
Textor SC, Canzanello VJ, Taler SJ, Wilson DJ, Schwartz LL, Augustine JE, et al. Cyclosporine-induced hypertension after transplantation. Mayo Clin Proc. 1994;69(12):1182–93.
Taler SJ, Textor SC, Canzanello VJ, Schwartz L. Cyclosporin-induced hypertension: incidence, pathogenesis and management. Drug Saf. 1999;20(5):437–49.
Margreiter R. European Tacrolimus vs Ciclosporin Microemulsion Renal Transplantation Study Group. Efficacy and safety of tacrolimus compared with ciclosporin microemulsion in renal transplantation: a randomized multicentre study. Lancet. 2002;359(9308):741–6.
Kramer BK, Del Castillo D, Margreiter R, Sperschneider H, Olbricht CJ, Ortuno J, et al. Efficacy and safety of tacrolimus compared with ciclosporin A in renal transplantation: three-year observational results. Nephrol Dial Transplant. 200;23(7):2386–92.
Arjang Djamali DB [Internet]. US: Prevention and treatment of antibody-mediated rejection of the renal allograft. Available from: www.uptodate.com Accessed 02/04/2021
Beecroft JR, Rajan DK, Clark TW, Robinette M, Stavropoulos SW. Transplant renal artery stenosis: outcome after percutaneous intervention. J Vasc Interv Radiol. 2004;15(12):1407–13.
Hurst FP, Abbott KC, Neff RT, Elster EA, Falta EM, Lentine KL, et al. Incidence, predictors and outcomes of transplant renal artery stenosis after kidney transplantation: analysis of USRDS. Am J Nephrol. 2009;30(5):459–67.
Chen LX, De Mattos A, Bang H, Vu CT, Gandhi M, Alnimri M, et al. Angioplasty vs stent in the treatment of transplant renal artery stenosis. Clin Transpl. 2018;32(4):e13217.
Aydoğdu S, Güler K, Bayram F, Altun B, Derici Ü, Abacı A. ve ark. 2019 Turkish Hypertension Consensus Report. Turk Kardiyol Dern Ars. 2019;47(6):535–46.
Unger T, Borghi C, Charchar F, Khan NA, Poulter NR, Prabhakaran D, et al. 2020 International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension. 2020;75(6):1334–57.
James PA, Oparil S, Carter BL, Cushman WC, Dennison-Himmelfarb C, Handler J, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA. 2014;311(5):507–20.
Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M, et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J. 2013;34(28):2159–219.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Associaton Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):e13–e115 This paper is the only guideline which outlines the blood pressure goal in kidney transplant recipients.
SPRINT Research Group, Wright JT Jr, Williamson JD, Whelton PK, Snyder JK, Sink KM, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103–16 The recommendation of blood pressure goal in kidney transplant recipients in 2017 ACC/AHA/AAPA/ABC/ACPM/AGS Hypertension Guideline was based on the results of this study. The blood pressure goal was extrapolated from chronic kidney disease patients' data, since no transplanted patient was included.
Taler SJ, Agarwal R, Bakris GL, Flynn JT, Nilsson PM, Rahman M, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for management of blood pressure in CKD. Am J Kidney Dis. 2013;62(2):201–13.
Lamb SA, Al Hamarneh YN, Houle SKD, Leung AA, Tsuyuki RT. Hypertension Canada’s 2017 guidelines for diagnosis, risk assessment, prevention and treatment of hypertension in adults for pharmacists: An update. Can Pharm J (Ott). 2017;151(1):33–42.
Xie X, Atkins E, Lv J, Bennett A, Neal B, Ninomiya T, et al. Effects of intensive blood pressure lowering on cardiovascular and renal outcomes: updated systemic review and meta-analysis. Lancet. 2016;387(10017):435–43.
Kuypers DR, Neumayer HH, Fritsche L, Budde K, Rodicio JL, Vanrenterghem Y, et al. Calcium channel blockade and presevation of renal graft function in cyclosporine-treated recipients: a prospective randomized placebo-controlled 2-year study. Transplantation. 2004;78(8):1204–11.
De-Mattos AM, Olyaei AJ, Benet WM. Pharmacology of immunosuppressive medications used in renal diseases and transplantation. Am J Kidney Dis. 1996;28(5):631–67.
Ladefoged SD, Anderson CB. Calcium channel blockers in kidney transplantation. Clin Transpl. 1994;8(2 Pt 1):128–33.
Cross NB, Webster AC, Masson P, O’Connell PJ, Craig JC. Antihypertensive treatment for kidney transplant recipients. Cochrane Database Syst Rev. 2009;(3):CD003598. This study demontrated that calcium channel blockers may be preferred as first-line antihypertensive treatment in kidney transplant recipients. The data showed that calcium channel blockers reduces graft loss, and improves glomerular filtration rate; however RAAS blockers have inconclusive results for glomerular filtration rate and graft loss in kidney transplant recipients.
Grassi G, Robles NR, Seravalle G, Fici F. Lercanidipine in the Management of Hypertension: An Update. J Pharmacol Pharmacother. 2017;8(4):155–65.
Rademacher J, Mengel M, Ellis S, Stuht S, Hiss M, Schwarz A, et al. The renal arterial resistance index and renal allograft survival. N Engl J Med. 2013;349(2):115–24.
Vlahakos DV, Canzanello VJ, Madaio MP, Madias NE. Enalapril-associated anemia in renal transplant recipients treated for hypertension. Am J Kidney Dis. 1991;17(2):199–205.
Shin JI, Palta M, Djamali A, Kaufman DB, Astor BC. The association between renin-angiotensin system blockade and long-term outcomes in renal transplant recipients: The Wisconsin Allograft Recipient Database (WisARD). Transplantation. 2016;100(7):1541–9.
Heinze G, Mitterbauer C, Regele H, Kramar R, Winkelmayer WC, Curhan GC, et al. Angiotensin-converting enzyme inhibitor or angiotensin II receptor antagonist therapy is associated with prolonged patient and graft survival after renal transplantation. J Am Soc Nephrol. 2006;17(3):889–99.
Knoll GA, Fergusson D, Chasse M, Hebert P, Wells G, Tibbles LA, et al. Ramipril versus placebo in kidney transplant patients with proteinuria: a multicentre, double-blind, randomised controlled trial. Lancet Diabetes Endocrinol. 2016;4(4):318–26.
Philipp T, Martinez F, Geiger H, Moulin B, Mourad G, Schmieder R, et al. Candesartan improves blood pressure control and reduces proteinuria in renal transplant recipients: results from SECRET. Nephrol Dial Transplant. 2010;25(3):967–76 This study demonstrated that candesartan reduces blood pressure and proteinuria, while increases serum creatinine and potassium in kidney transplant recipients. Although, all-cause mortality, cardiovascular mortality, and graft survival was reported as similar between candesartan and placebo; enrollment into this study was halted prematurely because of a lower than expected primary event rate in candesartan group.
Hiremath S, Fergusson DA, Fergusson N, Bennett A, Knoll GA. Renin-angiotensin system blockade and long-term clinical outcomes in kidney transplant recipients: a meta-analysis of randomized controlled trials. Am J Kidney Dis. 2017;69(1):78–86 This meta-analysis reported that RAAS blockers did not change all-cause mortality, graft survival, and serum creatinine doubling when compared to non-RAAS blockage group. It is not possible to answer definitively whether RAAS blockage improves allograft survival in kidney transplant recipients.
Funding
There are no funding sources to declare.
Author information
Authors and Affiliations
Contributions
All the 3 authors have contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
There is no conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Hypertension and the Kidney
Rights and permissions
About this article
Cite this article
Ari, E., Fici, F. & Robles, N.R. Hypertension in Kidney Transplant Recipients: Where Are We Today?. Curr Hypertens Rep 23, 21 (2021). https://doi.org/10.1007/s11906-021-01139-4
Accepted:
Published:
DOI: https://doi.org/10.1007/s11906-021-01139-4