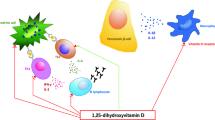
Abstract
Purpose of Review
There is a growing, largely inconsistent, literature on the role of vitamin D in association with type 2 diabetes, insulin resistance/insulin secretion, glycemic indices, and complications of type 2 diabetes. Pathophysiologic, bystander, preventive, and treatment roles of vitamin D have all been proposed. In this focused review, we attempt to organize and clarify our current information in this area.
Recent Findings
Clinical study interpretation is difficult because of variability in dosage, dosage form, study duration, and populations studied, as well as recently reported normal human polymorphisms in vitamin D synthesis and catabolism, vitamin D-binding protein, and vitamin D receptors in addition to a host of potential epigenetic confounders. Low vitamin D status appears to be associated with type 2 diabetes and most other insulin resistance disorders reported to date. The extraskeletal benefits of supplementation/repletion in these disorders in our species, with a few highlighted exceptions, remain to be established.
Summary
This focused review attempts to summarize our current knowledge in this burgeoning area through a review of key meta-analyses, observational studies, randomized control trials, and Mendelian randomization studies and will hopefully serve as a guide to indicate future research directions and current best practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There is considerable interest in vitamin D’s role in type 2 diabetes (T2DM) and insulin resistance beyond its role in bone/calcium metabolism. A recent review summarized reports showing significant positive correlation between serum 1,25-OH-vitamin D (the active form) and both insulin sensitivity and secretion, a negative association between vitamin D deficiency and glycemic control, and a positive association of vitamin D deficiency with T2DM [1••]. Randomized control trials (RCTs) of supplementation in healthy people, those with prediabetes and those with T2DM, yielded varied results. Small glycemic benefits from supplementation are reported mostly with baseline vitamin D deficiency. Most meta-analyses of interventional RCTs show inconsistent benefits on glycemic control in T2DM.
Vitamin D—Insulin Sensitivity/Insulin Secretion
Gulseth studied supplementation’s effects on insulin sensitivity and secretion in people with T2DM with replete baseline serum 25-OH-vitamin D (25OHD) (38.0 + 12.6 nmol/L) [2••]. Despite raising the mean serum 25OHD to 96.9 ± 18.3 at 4 weeks and 53.7 ± 9.2 after 6 months, there was no significant change in insulin sensitivity/secretion, or glycemic control, possibly because patients were replete at baseline.
One study reported that vitamin D3 10,000 IU/day × 8 weeks was associated with a significant decrease in acute insulin response to glucose (P = 0.011) and a significant increase in insulin sensitivity (P = 0.012) in 8 people with prediabetes [3]. From these studies, it appears that vitamin D supplementation may improve insulin sensitivity in patients at increased risk for T2DM with baseline low vitamin D status.
Vitamin D—Type 2 Diabetes Prevention
This subject was reviewed by Angellotti and Pittas [4•]. Despite evidence for prevention from mechanistic and observational studies, a definitive answer is impossible because the possibility of reverse causation cannot be eliminated. Serum 25OHD is not only a marker of vitamin D status, but of overall health, confounding interpretation. Small studies and post hoc analyses in people with normal glucose tolerance do not support a preventive role for vitamin D in normals; however, a preventive possibility exists in high-risk populations, e.g., those with prediabetes and pending adequately powered RCTs, such as the recently completed D2d trial [5]. The just released results of this pivotal trial will be discussed later in this article. It should be noted that there is no consensus on what treatment doses of vitamin D are appropriate in intervention trials, given the variety of preparations available, the baseline vitamin D status, and individual variability in vitamin D absorption [5]. The Institute of Medicine recommends most adults take 400 IU/day of vitamin D and 800 IU for those 71 years old and older. The D2d study used 4000 IU daily based on studies showing that for most adults, this will achieve a sustained serum 25OHD of 30–50 nmol/L There is consensus that serum 25-OH-vitamin D levels < 20 nmol/L are deficient, while those > 20 < 30 are insufficient [5]. The rationale for and limitations of these definitions are discussed later.
In Forouhi’s study, baseline serum 25OHD correlated inversely with serum glucose and insulin 10 years later [6]. This study is strengthened by its community, prospective design, and controlling for confounders (physical activity, obesity), but is limited by sample size (n = 524) and 50% dropout.
A study of 25 postmenopausal women taking 2000 IU vitamin D3/day reported no effect on insulin resistance or glycemia [7].
In post hoc analysis from a trial studying skeletal outcomes using 700 IU vitamin D3 + calcium/day, a significant decrease in homeostasis model assessment of insulin resistance (HOMA-IR) was noted in patients with prediabetes, but not in those with normal glucose tolerance [8]. The beneficial effect apparent in this study, as contrasted with the previous study, despite a considerably lower daily dose of vitamin D, may be related to the fact that the patients in this trial were at higher risk for T2DM. The foregoing studies suggest that vitamin D supplementation may reduce the risk of developing T2DM in those with low baseline vitamin D status and at high risk of developing T2DM. Results of intervention trials are summarized in Table 1. Mendelian randomization studies, discussed later in this article, also yield somewhat conflicting results on the role of vitamin D in prevention of T2DM. For now, the role of vitamin D in T2DM prevention remains unclear.
Vitamin D—Glycemic Control in Type 2 Diabetes
Santos systematically reviewed literature on vitamin D status and glycemic control [9••]. An inverse relationship between serum 25OHD and indices of glucose metabolism, e.g., HOMA-IR, was confirmed. Most studies showed no supplementation benefit on glycemic control or insulin sensitivity indicators such as A1c, HOMA-IR, or quantitative insulin sensitivity check index (QUICKI). While better glycemic control is associated with higher vitamin D status in people with T2DM, the reviewed studies did not show any clear glycemic benefit from vitamin D supplementation/repletion.
Vitamin D—Diabetic Peripheral Neuropathy
Qu reported an association between vitamin D deficiency/insufficiency and diabetic peripheral neuropathy in a meta-analysis [10••]. A significant difference in mean serum 25OHD between people with diabetes with/without neuropathy was seen in people of white and Asian background. The reviewed studies did not discuss preventive or therapeutic effects of vitamin D repletion/supplementation. Low vitamin D status is associated with diabetic peripheral neuropathy; causality is not confirmed.
Vitamin D—Diabetic Autonomic Neuropathy
Hansen reported a U-shaped association of both low and high serum 25OHD with cardiac neuropathy. Linear regression models demonstrated that a rise in serum 25OHD from 25 to 50 paralleled a 3.9% (95% CI 0.1–7.9) rise in heart rate response to deep breathing (E/I ratio) and 4.8% (95% CI 4.7–9.3) to standing (30/15 ratio). A rise from 125 to 150 in serum 25OHD was associated with decreases of 2.6% (95% CI − 5.8–0.1) and 4.1% (95% CI − 5.8 to − 0.5), respectively [11••]. Kedar studied relationships between vitamin D status and gastric emptying time in idiopathic gastroparesis (n = 42) and diabetic gastroparesis (n = 17) [12]. Increasing serum 25OHD was associated with reduced gastric emptying time throughout the study period in people with idiopathic gastroparesis. There was significant association at hour 2 (P = 0.069) and for total test period (P = 0.046). At hours 1 and 4, correlation was particularly strong (P = 0.034 and 0.076, respectively). No associations were observed between diabetic gastroparesis and vitamin D status.
High and low vitamin D levels were associated with cardiovascular autonomic neuropathy in people with diabetes. There is no association of gastric emptying time with vitamin D status in people with diabetic gastroparesis. No intervention studies have been reported as yet.
Vitamin D—Erectile Dysfunction in Type 2 Diabetes
Erectile dysfunction (ED) is concerning, not only with regard to quality of life but also because it is a “red flag” for generalized atherosclerosis and future cardiovascular events. Basat studied this in 98 people with T2DM, age 18–80, using the International Index of Erectile Function (IIEF-5) questionnaire. Patients were stratified into three groups of IIEF-5 scores: 5–10—severe ED (n = 32), 11–20—moderate ED (n = 45), and 21–25—without ED (n = 21). Serum 25OHD was lower in groups 5–10. There was a positive correlation between IIED% score and serum 25OHD (r = 0.21, P = 0.038) [13••]. Low vitamin D status is associated with ED; the effects of repletion/supplementation on prevention or treatment of ED in people with T2DM are not yet known.
Vitamin D—Diabetic Retinopathy
Currently, there is insufficient evidence showing whether vitamin D deficiency is related to diabetic retinopathy, and the determination of this relationship has rarely been studied. Luo conducted a meta-analysis on vitamin D status/diabetic retinopathy association using a systematic literature search, including 15 observational studies involving 17,664 people with T2DM [14••]. People with vitamin D deficiency had a significantly increased risk of diabetic retinopathy (OR = 2.03, 95% CI 1.07–3.86), and a decrease of 1.7 nmol/L (95% CI − 2.72–0.66) in serum 25OHD was found in diabetic retinopathy. Sensitivity analysis showed that exclusion of any single study did not materially change outcome, confirming the association between vitamin D deficiency and increased risk of diabetic retinopathy. In summary, low vitamin D status is associated with diabetic retinopathy. The effects of vitamin D repletion/supplementation on prevention and treatment of diabetic retinopathy are not currently known; considering the high prevalence of vitamin D deficiency and the burden of diabetic retinopathy, screening people with type 2 diabetes, who are at risk for vitamin D deficiency, should be considered.
Vitamin D—Diabetic Nephropathy
Derakhshanian’s meta-analysis of vitamin D status/diabetic nephropathy associations and repletion effects systematically searched articles published 2009–2014 [15]. The pooled OR from cross-sectional studies was 1.80 (95% CI 1.25–2.59; P = 0.002), demonstrating negative serum 25OHD/diabetic nephropathy risk association. Pooled data of urine albumin-to-creatinine ratios (UACR) in vitamin D supplementation trials demonstrated no significant change (17.98; 95% CI − 35.35–71.32; P = 0.51), supporting higher diabetic nephropathy risk in vitamin D deficiency. In summary, low vitamin D status is associated with higher risk for nephropathy in patients with T2DM. Pooling the results of available clinical trials after vitamin D supplementation did not support causality in this association.
Chokhandre performed a meta-analysis on supplementation of vitamin D or its analogs in chronic kidney disease [16]. Inflammation and oxidative stress participate in diabetic nephropathy pathogenesis. Both are believed to be regulated via nuclear receptors including vitamin D receptors. They summarized data on vitamin D’s possible role in diabetic nephropathy, searching databases for relevant trials. Trials assessing renal functions including UACR, albuminuria, and estimated glomerular filtration rate (eGFR) were reviewed. Quality and risk of bias assessments were conducted. Effects on 25OHD, calcium, and HbA1c were assessed. The mean or its percent change along with their standard deviation was used for result reporting. Six studies assessed effects of cholecalciferol, calcitriol, and paricalcitol, respectively, in diabetic nephropathy. Trial designs differed (3 randomized, 1 nonrandomized, 2 uncontrolled studies) with differing degrees of quality and bias risks. Analogs achieved significant renal function improvement in 2 RCTs (perhaps because they eliminate the need for a renally dependent 1-α-hydroxylation step). No study reported significant hypercalcemia incidence. Larger, longer RCTs, comparing efficacy/safety of vitamin D/analogs, are needed.
Li reviewed associations of vitamin D/diabetic nephropathy, reinforcing awareness that diabetic nephropathy is the leading diabetic renal complication and a major cause of renal failure [17]. The renin-angiotensin-aldosterone system (RAAS) is a principal cause of progressive kidney damage. RAAS antagonists are the major diabetic nephropathy pharmacotherapy. Limiting their efficacy is a compensatory increase in renin via interference with renin feedback inhibition. Vitamin D renoprotectively downregulates the RAAS through suppression of renin expression. Vitamin D receptor-null mice demonstrate worse diabetic nephropathy, likely due to impaired downregulation of RAAS activation. Combinations using RAAS inhibition + vitamin D analogs dramatically decreased renal injuries via analog inhibition of compensatory renin increase. Recent data suggest vitamin D and its analogs are renoprotective in diabetic nephropathy. In summary, low vitamin D status appears to be associated with worse renal function in people with T2DM-associated CKD. Supplementation with analogs of the active form of vitamin D, particularly when combined with RAAS inhibitors, appears to improve renal function in this demographic.
Vitamin D—Type 2 Diabetes Macrovascular/Microvascular Complications
The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) Study examined relationships between vitamin D status and macrovascular/microvascular complications [18]. Associations between baseline vitamin D status and macrovascular/microvascular disease incidences were analyzed using the Cox proportional hazards models and logistic regression in a 5-year observational study; 50% had vitamin D deficiency/insufficiency. Participants with serum 25OHD < 50 nmol/L had a greater cumulative incidence of macrovascular/microvascular occurrences than those with levels ≥ 50 nmol/L. Treatment-stratified multivariate analysis, adjusted for potential confounders, found that vitamin D status was an independent macrovascular outcome predictor. A 50 nmol/L difference in serum 25OHD was associated with a 23% (P = 0.007) change in macrovascular event risk. Relevant adjustments did not affect outcomes. Unadjusted risk for microvascular complications was 18% (P = 0.006) higher during the study, although excess risk fell to 11–14% and lost statistical significance after adjustments. In summary, lower vitamin D status was associated with greater macrovascular/microvascular occurrence risk; causality was not established.
Yu’s meta-analysis of 13 RCTs assessed vitamin D supplementation effects on macrovascular inflammatory risk factors [19••]. Supplementation significantly decreased hs-CRP level by 0.45 μg/mL, but did not affect TNF-α or IL-6. Subgroup analysis demonstrated that supplementation significantly lowered hs-CRP by 0.34 μg/mL in trials with daily dose ≤ 4000 IU and by 0.31 μg/mL among trials lasting > 12 weeks.
Norman and Powell summarized research on vitamin D and cardiovascular disease [20]. Vitamin D metabolites function extraskeletally, where 1-α-hydroxylation affects regulatory pathways via paracrine and autocrine actions. The active metabolite binds to vitamin D receptors, affecting gene expression in processes likely relevant to cardiovascular disease. Vitamin D receptors exist in all important cardiovascular cell types. Studies confirm a role for vitamin D metabolites in pathways integral to cardiovascular function and disease. Clinical studies mostly show independent associations between vitamin D deficiency and various cardiovascular disease expressions. Supplementation’s role in cardiovascular disease is uncertain.
Few studies have investigated the association between vitamin D and peripheral arterial disease (PAD) in patients with T2DM. Li studied associations of PAD with vitamin D status in 1028 people with T2DM [21••]. PAD was defined as an ankle-brachial index (ABI) < 0.9. Known surrogate PAD markers (blood pressure, HbA1c, lipids, comorbidities, carotid intima-media thickness, and vitamin D status) were measured. PAD prevalence was higher in participants with decreased (23.9%) than in those with normal (15.6%) 25OHD (≥ 30 nmol/L, P < 0.01). Low vitamin D status was associated with increased risk of PAD (OR = 1.69, 95% CI 1.17–2.44, P < 0.001), and PAD was significantly more likely to occur in participants ≥ 65 years of age (OR = 2.56, 95% CI 1.51–4.48 vs. 1.21, 95% CI 0.80–1.83). After adjusting for known cardiovascular risk factors and potential confounding variables, the association of decreased 25(OH)D and PAD remained significant in patients < 65 years of age (OR = 1.55; 95% CI 1.14–2.12, P = 0.006). In conclusion, low vitamin D levels were associated with a decreased ABI and increased prevalence of PAD in patients with T2DM. Vitamin D deficiency might be an independent risk factor for PAD in patients with T2DM < 65 years of age. Whether vitamin D supplementation could significantly improve vascular outcomes remains unclear.
Vitamin D—Type 2 Diabetes Mortality
Joergensen studied associations between vitamin D status and mortality in a longitudinal observational follow-up study of 289 people with T2DM (normoalbuminuria (n = 172), microalbuminuria (n = 73), macroalbuminuria (n = 44) at baseline) who they followed for a median (range) of 15.0 (0.2–23.0) years [22]. Median plasma 25OHD = 35.7 (5–136.7) nmol/L. Vitamin D status was unassociated with age, sex, eGFR, UACR, or HbA1C at baseline. Vitamin D deficiency was weakly associated with high systolic blood pressure (r = 0.13, P = 0.03). During the study, 196 (68%) patients expired. All-cause mortality was higher with severe vitamin D deficiency (HR = 1.96; 95% CI 1.29–2.98) and remained significant following adjustment for UACR, A1C, T2DM duration, and classical cardiovascular risk factors (OR = 2.03). Profound vitamin D deficiency was associated with excess cardiovascular mortality (OR = 1.95) and remained significant following adjustment (OR = 1.90). Profound baseline vitamin D deficiency failed to predict development of micro/macroalbuminuria but predicted increased rates of all-cause/cardiovascular mortality, independently of UACR and traditional risk factors. In summary, low vitamin D status is independently associated with increased cardiovascular and overall mortality risk in people with T2DM; it is unknown whether supplementation/repletion can reduce this risk.
Vitamin D—Nonalcoholic Fatty Liver Disease (NAFLD)
Barchetta studied associations between vitamin D deficiency and nonalcoholic fatty liver disease (NAFLD) and repletion trials [23••]. Epidemiological studies generally found associations between vitamin D deficiency and NAFLD/steatohepatitis (NASH), after adjustment for confounders. Several reported antifibrotic, antiinflammatory, and insulin-sensitizing vitamin D effects on the liver, yet results from interventional studies are inconsistent. The number of interventional RCTs in NAFLD/NASH is small. Promising data came from recent trials using newer dosing regimens. Further, well-designed studies are needed to robustly recommend optimizing Vitamin D status as a preventive and/or treatment strategy.
Vitamin D—Other Disorders Associated with Insulin Resistance
Polycystic ovarian syndrome, classic and nonclassic adrenal hyperplasia, and gout are all, like T2DM, associated with insulin resistance, which play a role in their pathophysiologies and are frequent comorbidities of T2DM. Insulin-sensitizing interventions, including vitamin D repletion/supplementation, have been reported to ameliorate these conditions; however, detailed discussion of these topics is beyond the scope and space allowance of this article. Interested readers may refer to references [24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81, 82••, 83,84,85, 86••].
Please see Table 2 summarizing vitamin D associations with T2DM complications.
Conclusions
While there is considerable preclinical evidence that low vitamin D status contributes to the pathogenesis of insulin resistance and T2DM and that repletion improves glycemic control in some species, vitamin D’s role in the pathogenesis, prevention, and treatment of insulin resistance T2DM in humans remains unclear. There is widespread agreement from epidemiologic studies that hypovitaminosis D is prevalent in people with these disorders [87, 88]. Most people with T2DM have low serum 25OHD, while high vitamin D status is associated with lower incident diabetes risk [89, 90]. Sipahi studied the relationship of VDS with parameters including glycemic control, serum uric acid, and microalbuminuria in T2DM and chronic kidney disease. They reported that higher HbA1c predicted lower vitamin D status S [82]. Despite consensus that serum 25OHD < 30 nmol/L indicates suboptimal vitamin D status, one must recognize that many factors contribute to whether a given serum concentration is adequate.
Inverse correlations between obesity indicators and vitamin D status have been frequently reported [24,25,26, 29, 34, 38]. Since vitamin D is fat soluble, a greater percentage of it is sequestered in fat in overweight and obese subjects, reducing serum concentrations. Overweight and obese people also have a greater volume of distribution, potentially diluting serum 25OHD concentrations.
Polymorphisms in vitamin D-binding protein (VDBP) affect serum 25OHD [86••, 91]. Binding affinity differences among these VDBP forms affect serum-free 25OHD and the amount convertible to 1,25-OH-vitamin D.
Selection of 30 nmol/L as the lower limit of sufficiency is based on the observation that most people will not have an elevated serum parathyroid hormone (PTH) with a serum 25OHD > 30 nmol/L, resulting in less net bone loss; however, this says nothing about what concentration(s) are sufficient to obtain purported nonskeletal benefits.
There are also vitamin D receptor polymorphisms determining whether a given serum 25OHD concentration is adequate and possibly predicting the metabolic response to supplementation in people with T2DM [92, 93••, 94, 95••].
As stated by Al-Dahgri et al., there is inconsistent evidence of benefit of supplementation on metabolic profile in T2DM, possibly due to vitamin D receptor variants [93••]. They investigated the metabolic effects of 1 year of supplementation in people with T2DM according to their vitamin D receptor polymorphisms. A total of 204 patients took 2000 IU vitamin D3 daily. Serum 25OHD and metabolic profiles were determined at entry and at 1 year. Vitamin D receptor polymorphisms (Taq-I, Bsm-I, Apa-I, and Fok-I) were identified using TaqMan genotyping assays. Supplementation was significantly associated with increased HOMA β-cell function (P = 0.003) and significantly decreased triglycerides and total and LDL cholesterol (P < 0.001). The lowest increment in 25OHD levels was found in Fok-I CC genotypes (P < 0.0001). With supplementation, Taq-I GG genotype carriers experienced significant improvements in triglycerides and LDL and total cholesterol, insulin, HbA1c, and HOMA-IR (P < 0.005, 0.01, < 0.001, < 0.005, 0.03, and 0.01, respectively). Bsm-I TT genotype carriers experienced significant improvements in triglycerides (P = 0.01), insulin, and HOMA-IR (P values < 0.05). Improvements in metabolic profile associated with supplementation are affected by VDR polymorphisms, specifically for Taq-I GG and Bsm-I TT genotypes.
Polymorphisms in the 1-α-hydroxylase gene as well as renal function variation may affect available serum 1,25-OH-vitamin D [94, 95••]. Such potentially confounding polymorphisms and epigenetic differences in vitamin D expression are the norm in humans and are less likely to be an issue in preclinical studies, which often have genetically identical (or nearly so) subjects. This may explain some discrepancies between human and animal studies, where vitamin D’s role in the pathogenesis, prevention, and treatment of IR and T2DM seems clearer.
Mendelian randomization studies have used single nucleotide polymorphisms (SNPs) associated with serum 25OHD concentrations, identified via genome wide association studies, to assess causality of vitamin D status in incident T2DM [96, 97]. When SNPs involved in vitamin D synthesis as well as its transport and catabolism were considered, causality could not be inferred in a European population. When only SNPs involved in vitamin D synthesis with no known pleiotropic effects were considered, excluding those involved in vitamin D transport and catabolism, a causally protective effect of higher vitamin D status in T2DM prevention could be inferred in both Chinese and European populations; however, the results of meta-analyses of ongoing trials in high-risk populations using higher vitamin D doses, such as the D2d trial, will be necessary to answer the question of causality more definitively [5]. Following the initial submission of this manuscript, the results of the D2d study were simultaneously presented at the annual scientific meeting of the American Diabetes Association and published online [98••]. A total of 2423 participants who met the previously described eligibility criteria, 80% of whom were vitamin D replete at baseline, were randomized to receive either placebo or 4000 IU cholecalciferol/day. At 2 years after randomization, mean serum 25-hydroxyvitamin D level in the vitamin D group was 54.3 ng/mL (from 27.7 ng per/mL at baseline) vs. 28.8 ng/mL in the placebo group (from 28.2 ng/mL at baseline). After a median follow-up of 2.5 years, the difference in incident T2DM between the 2 groups of people with 2 indicators for prediabetes at baseline was not statistically significant; the hazard ratio for vitamin D supplementation vs. placebo was 0.88 (95% CI 0.75–1.04; P = 0.12). In subgroup analysis, those individuals with hypovitaminosis D at baseline appeared to have a lower risk of developing T2DM with supplementation. While D2d is the largest intervention trial to date and shows that relatively high dose vitamin D supplementation in people at high risk for T2DM (having 2 indicators for pre-DM) was not protective against incident T2DM over a median follow-up period of 2.5 years, it leaves a number of important questions unanswered. If, for example, at study end, the highest and lowest quintiles of serum 25OHD had been compared for incident T2DM, would a significant difference have emerged? Had the eligibility criteria specified that subjects have vitamin D deficiency at baseline, might repletion/supplementation been proven to be effective in reducing incident T2DM? Could it be that interventions such as vitamin D supplementation are timing-dependent, e.g., would intervention earlier in the continuum from normal glucose tolerance to T2DM have resulted in a different outcome? Would significant benefit be demonstrated given a longer study duration? Might vitamin D reduce incident T2DM when combined with another lifestyle or pharmacologic intervention? Finally, might it be that any reduction in incident T2DM with vitamin D is dependent upon one or more polymorphisms in vitamin D-binding protein, vitamin D receptor, 1-α-hydroxylase, vitamin D synthesis, transport, or catabolism? To be effective in T2DM prevention should vitamin D treatment be as genetically targeted as biologic and chemotherapeutic cancer treatments are becoming?
The role of vitamin D status in the pathogenesis, prevention, and treatment of T2DM complications is unclear, though promising [45, 99–103]. As noted, there is a significant association between low vitamin D status and diabetic peripheral neuropathy. Both low and high vitamin D status are associated with diabetic cardiac neuropathy. There was no association between vitamin D status and gastric emptying time in people with diabetic gastroparesis, while in idiopathic gastroparesis, rising vitamin D concentrations were associated with shorter gastric emptying time.
No studies surfaced in our search concerning vitamin D and other diabetic autonomic neuropathies, mononeuropathy, or amyotrophy; however, Basat and colleagues found that the lowest vitamin D status was in men with T2DM with the most severe ED. Their findings are hard to interpret because ED results from a complex interplay of neurologic, vascular, hormonal, pharmacologic, toxicological, and psychologic factors.
The association of low vitamin D status with increased diabetic retinopathy risk in people with T2DM appears to be well established as does the association of low vitamin D status with increased diabetic nephropathy risk. Interventional studies suggest that vitamin D analogs potentiate the renoprotective effects of RAAS inhibitors.
Low vitamin D status is consistently associated with poor macrovascular outcomes in people with T2DM. Vitamin D deficiency appears to be independently associated with PAD risk in people with T2DM. It is unclear whether repletion reduces the risk of incident T2DM macrovascular complications or their severity.
Low vitamin D status is associated with systolic hypertension, and the HR for all-cause mortality in people with T2DM was almost doubled that in those with VDD, even after correction for confounders. There is a similar increase in excess cardiovascular mortality with severe VDD. The effect of repletion on overall and cardiovascular mortality in people with T2DM is unknown.
Most studies show an association between low vitamin D status and NAFLD in T2DM. There is scarce, inconsistent data on the effects of supplementation on prevention or treatment.
Remaining confusion concerning vitamin D’s association with T2DM arises because low vitamin D status is seen in many chronic and acute illnesses, and it is unknown whether it plays a causative role or is simply a bystander and general health indicator. Further confusion results because many apparently healthy people have vitamin D deficiency/insufficiency, and there is considerable heterogeneity in humans in vitamin D status due to polymorphisms in vitamin D synthesis, transport, 1-α-hydroxylation, transport/VDBP, catabolism, VDR, and in other epigenetic factors. It seems prudent to suggest that people at increased risk for T2DM, as well as those with T2DM, periodically have their vitamin D status monitored and maintain it in the mid-reference range.
Learning points
-
Low vitamin D status is highly associated with insulin resistance, impaired insulin secretion, and increased risk of incident T2DM, particularly in people already at high risk for incident T2DM.
-
In some other species, the association of low vitamin D status with risk of incident T2DM appears to be causal.
-
Causality of low vitamin D status in incident T2DM in humans is not yet established due to conflicting results of interventional studies and Mendelian randomization studies on both incidence and glycemic markers. Intervention studies have varied much in terms of dosages and forms of vitamin D used, duration of intervention, and study populations. The just completed D2d study does not support a preventive role for vitamin D in people with 2 markers for prediabetes, not selected for baseline vitamin D deficiency.
-
Low vitamin D status is highly associated with a number of microvascular and macrovascular complications of T2DM including peripheral neuropathy, erectile dysfunction, retinopathy, diabetic kidney disease, and overall mortality. It does not appear to be associated with diabetic gastroparesis. The relationship between vitamin D status and diabetic cardiac neuropathy is U-shaped.
-
Intervention studies in people with T2DM and chronic kidney disease have shown significant therapeutic benefit when vitamin D analogs have been combined with RAAS inhibitors. Causality of low vitamin D status in T2DM complications, with the exception of renal complications, is not established.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
•• Lips P, Eekhoff M, van Schoor N, Oosterwerff M, de Jongh R, Krul-Poel Y, et al. Vitamin D and type 2 diabetes. J Steroid Biochem Mol Biol. 2017;173:280–5. A recent review summarizing reports showing significant positive correlation between serum 1,25-OH-vitamin D (the active form) and both insulin sensitivity and secretion, a negative association between vitamin D deficiency and glycemic control, and a positive association of VDD with incident T2DM.
•• Gulseth HL, Wium C, Angel K, Eriksen EF, Birkeland KI. Effects of vitamin D supplementation on insulin sensitivity and insulin secretion in subjects with type 2 diabetes and vitamin D deficiency: a randomized controlled trial. Diabetes Care. 2017;40(7):872–8. A recent study of supplementation’s effects on insulin sensitivity and secretion and glycemic indicators in people with T2DM with replete baseline serum 25-OH-vitamin D.
Nazarian S, St Peter JV, Boston RC, Jones SA, Mariash CN. Vitamin D3 supplementation improves insulin sensitivity in subjects with impaired fasting glucose. Transl Res. 2011 Nov;158(5):276–81.
• Angellotti E, Pittas AG. The role of vitamin D in the prevention of type 2 diabetes: to D or not to D? Endocrinology. 2017;158(7):2013–21. In this recent focused review despite evidence for prevention from mechanistic and observational studies, a definitive answer regarding T2DM prevention with vitamin D is impossible because reverse causation cannot be excluded.
Pittas AG, Dawson-Hughes B, Sheehan PR, Rosen CJ, Ware JH, Knowler WC, et al. Rationale and design of the vitamin D and type 2 diabetes (D2d) study: a diabetes prevention trial. Diabetes Care. 2014;37(12):3227–34.
Forouhi NG, Luan J, Cooper A, Boucher BJ, Wareham N. Baseline serum 25-hydroxy vitamin D is predictive of future glycemic status and insulin resistance: the Medical Research Council Ely prospective study 1990–2000. Diabetes. 2008;57:2619–25.
Nilas L, Christiansen C. Treatment with vitamin D or its analogues does not change body weight or blood glucose level in postmenopausal women. Int J Obes. 1984;8:407–11.
Pittas AG, Harris SS, Stark PC, Dawson-Hughes B. The effects of calcium and vitamin D supplementation on blood glucose and markers of inflammation in nondiabetic adults. Diabetes Care. 2007;30:980–6.
•• Santos RKF, Brandão-Lima PN, Tete RMDD, Freire ARS, Pires LV. Vitamin D ratio and glycaemic control in individuals with type 2 diabetes mellitus: a systematic review. Diabetes Metab Res Rev. 2018;34(3). A recent focused review of studies assessing the association of vitamin D status with glycemic indices in people with T2DM.
•• Qu G, Wang L, Tang X, Wu W, Suna Y. The association between vitamin D level and diabetic peripheral neuropathy in patients with type 2 diabetes mellitus: an update systematic review and meta-analysis. J Clin Transl Endocrinol. 2017;9:25–31. A recent focused review and meta-analysis of studies exploring associations between vitamin D status and the common diabetic complication of peripheral neuropathy.
•• Hansen CS, Fleischer J, Vistisen D, Ridderstråle M, Jensen JS, Jørgensen ME. High and low vitamin D level is associated with cardiovascular autonomic neuropathy in people with type 1 and type 2 diabetes. Diabet Med. 2017;34(3):364–71. A recent study of the association of vitamin D status with cardiac autonomic neuropathy in people with either T2DM or T1DM.
Kedar A, Nikitina Y, Henry OR, Abell KB, Vedanarayanan V, Griswold ME, et al. Gastric dysmotility and low serum vitamin D levels in patients with gastroparesis. Horm Metab Res. 2013;45(1):47–53.
•• Basat S, Sivritepe R, Ortaboz D, Sevim Çalık E, Küçük EV, Şimşek B, et al. The relationship between vitamin D level and erectile dysfunction in patients with type 2 diabetes mellitus. Aging Male. 2018;21(2):111–5. An important recent cross-sectional study of the association of erectile dysfunction with vitamin D status.
•• Luo BA, Gao F, Qin LL. The association between vitamin D deficiency and diabetic retinopathy in type 2 diabetes: a meta-analysis of observational studies. Nutrients. 2017;9(3):307. An important recent study on the association of low vitamin D status and diabetic retinopathy.
Derakhshanian H, Shab-Bidar S, Speakman JR, Nadimi H, Djafarian K. Vitamin D and diabetic nephropathy: a systematic review and meta-analysis. Nutrition. 2015;31(10):1189–94.
Chokhandre MK, Mahmoud MI, Hakami T, Jafer M, Inamdar AS. Vitamin D & its analogues in type 2 diabetic nephropathy: a systematic review. J Diabetes Metab Disord. 2015;14:58.
Li YC. Vitamin D and diabetic nephropathy. Curr Diab Rep. 2008;8(6):464–9.
Herrmann M, Sullivan DR, Veillard AS, McCorquodale T, Straub IR, Scott R, et al. Serum 25-hydroxyvitamin D: a predictor of macrovascular and microvascular complications in patients with type 2 diabetes. Diabetes Care. 2015;38(3):521–8.
•• Yu Y, Tian L, Xiao Y, Huang G, Zhang M. Effect of vitamin D supplementation on some inflammatory biomarkers in type 2 diabetes mellitus subjects: a systematic review and meta-analysis of randomized controlled trials. Ann Nutr Metab. 2018;73(1):62–73. A recent focused review and meta-analysis of interventional RCTs studying impact of vitamin D supplementation on surrogate markers for macrovascular complications.
Norman PE, Powell JT. Vitamin D and cardiovascular disease. Circ Res. 2014;114:379–93.
•• Li DM, Zhang Y, Li Q, Xu XH, Ding B, Ma JH. Low 25-hydroxyvitamin D level is associated with peripheral arterial disease in type 2 diabetes patients. Arch Med Res. 2016;47(1):49–54. A recent cross-sectional study exploring the association of low vitamin D status with peripheral arterial disease.
Joergensen C, Gall MA, Schmedes A, Tarnow L, Parving HH, Rossing P. Vitamin D levels and mortality in type 2 diabetes. Diabetes Care. 2010;33(10):2238–43.
•• Barchetta I, Cimini FA, Cavallo MG. Vitamin D supplementation and non-alcoholic fatty liver disease: present and future. Nutrients. 2017;9(9):1015. Recent focused review of observational studies on associations between low vitamin D status and NAFLD and intervention studies.
Kim JJ, Choi YM, Chae SJ, Hwang KR, Yoon SH, Kim MJ, et al. Vitamin D deficiency in women with polycystic ovary syndrome. Clin Exp Reprod Med. 2014;41(2):80–5.
Panidis D, Balaris C, Farmakiotis D, Rousso D, Kourtis A, Balaris V, et al. Serum parathyroid hormone concentrations are increased in women with polycystic ovary syndrome. Clin Chem. 2005;51:1691–7.
Hahn S, Haselhorst U, Tan S, Quadbeck B, Schmidt M, Roesler S, et al. Low serum 25-hydroxyvitamin D concentrations are associated with insulin resistance and obesity in women with polycystic ovary syndrome. Exp Clin Endocrinol Diabetes. 2006;114:577–83.
Wehr E, Pilz S, Schweighofer N, Giuliani A, Kopera D, Pieber TR, et al. Association of hypovitaminosis D with metabolic disturbances in polycystic ovary syndrome. Eur J Endocrinol. 2009;161:575–82.
Mahmoudi T, Gourabi H, Ashrafi M, Yazdi RS, Ezabadi Z. Calciotropic hormones, insulin resistance, and the polycystic ovary syndrome. Fertil Steril. 2010;93:1208–14.
Selimoglu H, Duran C, Kiyici S, Ersoy C, Guclu M, Ozkaya G, et al. The effect of vitamin D replacement therapy on insulin resistance and androgen levels in women with polycystic ovary syndrome. J Endocrinol Investig. 2010;33:234–8.
Li HW, Brereton RE, Anderson RA, Wallace AM, Ho CK. Vitamin D deficiency is common and associated with metabolic risk factors in patients with polycystic ovary syndrome. Metabolism. 2011;60:1475–81.
Ngo DT, Chan WP, Rajendran S, Heresztyn T, Amarasekera A, Sverdlov AL, et al. Determinants of insulin responsiveness in young women: impact of polycystic ovarian syndrome, nitric oxide, and vitamin D. Nitric Oxide. 2011;25:326–30.
Ranjzad F, Mahban A, Shemirani AI, Mahmoudi T, Vahedi M, Nikzamir A, et al. Influence of gene variants related to calcium homeostasis on biochemical parameters of women with polycystic ovary syndrome. J Assist Reprod Genet. 2011;28:225–32.
Wehr E, Trummer O, Giuliani A, Gruber HJ, Pieber TR, Obermayer-Pietsch B. Vitamin D-associated polymorphisms are related to insulin resistance and vitamin D deficiency in polycystic ovary syndrome. Eur J Endocrinol. 2011;164:741–9.
Mazloomi S, Sharifi F, Hajihosseini R, Kalantari S, Mazloomzadeh S. Association between hypoadiponectinemia and low serum concentrations of calcium and vitamin D in women with polycystic ovary syndrome. ISRN Endocrinol. 2012;2012:949427.
Muscogiuri G, Policola C, Prioletta A, Sorice G, Mezza T, Lassandro A, et al. Low levels of 25(OH)D and insulin-resistance: 2 unrelated features or a cause-effect in PCOS? Clin Nutr. 2012;31:476–80.
El-Shal AS, Shalaby SM, Aly NM, Rashad NM, Abdelaziz AM. Genetic variation in the vitamin D receptor gene and vitamin D serum levels in Egyptian women with polycystic ovary syndrome. Mol Biol Rep. 2013;40:6063–73.
Krul-Poel YH, Snackey C, Louwers Y, Lips P, Lambalk CB, Laven JS, et al. The role of vitamin D in metabolic disturbances in polycystic ovary syndrome: a systematic review. Eur J Endocrinol. 2013;169:853–65.
Thomson RL, Spedding S, Buckley JD. Vitamin D in the aetiology and management of polycystic ovary syndrome. Clin Endocrinol. 2012;77:343–50.
Yildizhan R, Kurdoglu M, Adali E, Kolusari A, Yildizhan B, Sahin HG, et al. Serum 25-hydroxyvitamin D concentrations in obese and non-obese women with polycystic ovary syndrome. Arch Gynecol Obstet. 2009;280:559–63.
Thys-Jacobs S, Donovan D, Papadopoulos A, Sarrel P, Bilezikian JP. Vitamin D and calcium dysregulation in the polycystic ovarian syndrome. Steroids. 1999;64:430–5.
Kotsa K, Yavropoulou MP, Anastasiou O, Yovos JG. Role of vitamin D treatment in glucose metabolism in polycystic ovary syndrome. Fertil Steril. 2009;92:1053–8.
Wehr E, Pieber TR, Obermayer-Pietsch B. Effect of vitamin D3 treatment on glucose metabolism and menstrual frequency in polycystic ovary syndrome women: a pilot study. J Endocrinol Investig. 2011;34:757–63.
Prentice A. Vitamin D deficiency: a global perspective. Nutr Rev. 2008;66:S153–64.
Lips P. Worldwide status of vitamin D nutrition. J Steroid Biochem Mol Biol. 2010;121:297–300.
Adams JS, Hewison M. Update in vitamin D. J Clin Endocrinol Metab. 2010;95:471–8.
Butts SF, Seifer DB, Koelper N, et al. Vitamin D deficiency is associated with poor ovarian stimulation outcome in PCOS but not unexplained infertility. J Clin Endocrinol Metab. 2019;104(2):369–78.
Kuliczkowska-Plaksej J, Pasquali R, Milewicz A, et al. Serum vitamin D binding protein level associated with metabolic cardiovascular risk factors in women with the polycystic ovary syndrome. Horm Metab Res. 2019;51(1):54–61.
Fang F, Ni K, Cai Y, Shang J, Zhang X, Xiong C. Effect of vitamin D supplementation on polycystic ovary syndrome: a systematic review and meta-analysis of randomized controlled trials. Complement Ther Clin Pract. 2017;26:53–60.
Mogili KD, Karuppusami R, Thomas S, Chandy A, Kamath MS, TKA. Prevalence of vitamin D deficiency in infertile women with polycystic ovarian syndrome and its association with metabolic syndrome - a prospective observational study. Eur J Obstet Gynecol Reprod Biol. 2018;229:15–9.
Rashidi B, Haghollahi F, Shariat M, Zayerii F. The effects of calcium-vitamin D and metformin on polycystic ovary syndrome: a pilot study. Taiwan J Obstet Gynecol. 2009;48(2):142–7.
Speiser PW, Serrat J, New MI, Gertner JM. Insulin insensitivity in adrenal hyperplasia due to nonclassical steroid 21-hydroxylase deficiency. J Clin Endocrinol Metab. 1992;75:1421–4.
Saygili F, Oge A, Yilmaz C. Hyperinsulinemia and insulin insensitivity in women with nonclassical congenital adrenal hyperplasia due to 21-hydroxylase deficiency: the relationship between serum leptin levels and chronic hyperinsulinemia. Horm Res. 2005;63(6):270–4.
Charmandari E, Weise M, Bornstein SR, Eisenhofer G, Keil MF, Chrousos GP, et al. Children with classic congenital adrenal hyperplasia have elevated serum leptin concentrations and insulin resistance: potential clinical implications. J Clin Endocrinol Metab. 2002;87(5):2114–20.
Sacerdote A, Bahtiyar G “Treatment of congenital adrenal hyperplasia by reducing insulin resistance and cysticercosis induced polycystic ovarian syndrome,” in Darwish A editor Contemporary Gynecologic Practice, A. Darwish, Ed., InTech; 2015.
Bahtiyar G, Sacerdote A. “Management approaches to congenital adrenal hyperplasia in adolescents and adults; latest therapeutic developments,” in Chatterjee A editor Amenorrhea, InTech; 2011.
Thomas N, Kalani A, Vincent R, Luis Lam M, Bahtiyar G, Borensztein A, et al. Effect of vitamin D in a patient with classical adrenal hyperplasia due to 11-hydroxylase deficiency. J Med Cases. 2013;4(8):569–75.
Luis Lam M, Sacerdote AS, Bahtiyar G. Normalization of serum 11-deoxycortisol in a patient with non-classic adrenal hyperplasia due to 11-hydroxylase deficiency treated with vitamin D and a glucagon-like peptide(GLP)-1 agonist. Presented at The 96th Annual Meeting of The Endocrine Society, 6/21/2014, Chicago, IL. Presentation Number: SAT-0763.
Gröber U, Kisters K. Influence of drugs on vitamin D and calcium metabolism. Dermatoendocrinol. 2012;4(2):158–66.
Dono E, Inoue T, Ezeji G, Baby A, Bahtiyar G, Sacerdote AS. Amelioration of non-classic 11-hydroxylase deficiency in a vitamin D deficient patient with weight loss and ergocalciferol. press.endocrine.org/doi/abs/10.1210/endo-meetings.2015.AHPAA.7.THR-415.
Sacerdote, A.: Weight-dependent expression of non-classical aldosterone synthase deficiency. Program and Abstracts, Endo-2002, The Endocrine Society’s 84th Annual Meeting: 599, 2002.
Lundqvist J, Wikvall K, Norlin M. Vitamin D-mediated regulation of CYP21A2 transcription – a novel mechanism for vitamin D action. BiochimBiophysActa. 1820;2012:1553–9.
Fenteany G, Inoue T, Bahtiyar G, Sacerdote AS. Association of vitamin D repletion with normalization of elevated serum 17- OH-progesterone. Med Cas Rep. 2017;3:3.
Krysiak R, Kowalcze K, Bednarska-Czerwińska A, Okopień B. The effect of simvastatin treatment on plasma steroid levels in females with non-classic congenital adrenal hyperplasia. Exp Clin Endocrinol Diabetes. 2016;124(04):215–9.
Smith E, Hoy D, Cross M, Merriman TR, Vos T, Buchbinder R, et al. The global burden of gout: estimates from the global burden of disease 2010 study. Ann Rheum Dis. 2014;73:1470–6.
Roddy E, Doherty M. Gout. Epidemiology of gout. Arthritis Res Ther. 2010;12:223.
Kuo CF, Grainge MJ, Zhang W, Doherty M. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol. 2015;11:649–62.
Dalbeth N, Merriman TR, Stamp LK. Gout. Lancet. 2016;388:2039–52.
Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, et al. Diagnosis and management of the metabolic syndrome. An American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Curr Opin Cardiol. 2006;21(1):1–6.
International Diabetes Federation (IDF): The IDF consensus worldwide definition of the metabolic syndrome. https://www.idf.org/e-library/consensus-statements/60-idfconsensus-worldwide-definitionof-the-metabolic
Yu TY, Jee JH, Bae JC, Jin SM, Baek JH, Lee MK, et al. Serum uric acid: a strong and independent predictor of metabolic syndrome after adjusting for body composition. Metab Clin Exp. 2016;65:432–40.
Yuan H, Yu C, Li X, Sun L, Zhu X, Zhao C, et al. Serum uric acid levels and risk of metabolic syndrome: a dose-response meta-analysis of prospective studies. J Clin Endocrinol Metab. 2015;100:4198–207.
De Oliveira EP, Burini RC. High plasma uric acid concentration: causes and consequences. Diabetol Metab Syndr. 2012;4:2.
Perez-Ruiz F, Aniel-Quiroga MA, Herrero-Beites AM, Chinchilla SP, Erauskin GG, Merriman T. Renal clearance of uric acid is linked to insulin resistance and lower excretion of sodium in gout patients. Rheumatol Int. 2015;35:1519–24.
Jung JH, Song GG, Ji JD, Lee YH, Kim JH, Seo YH, et al. Metabolic syndrome: prevalence and risk factors in Korean gout patients. Korean J Intern Med. 2018;33(4):815–22.
Adachi S, Yoshizawa F, Yagasaki K. Hyperuricemia in type 2 diabetic model KK-Ay/Ta mice: a potent animal model with positive correlation between insulin resistance and plasma high uric acid levels. BMC Res Notes. 2017;10(1):577.
Pereira RM, De Carvalho JF, Bonfa E. Metabolic syndrome in rheumatological diseases. Autoimmun Rev. 2009;8:415–9.
Fraile JM, Puig JG, Torres RJ, de Miguel E, Martínez P, Vázquez JJ. Uric acid metabolism in patients with primary gout and the metabolic syndrome. Nucleosides Nucleotides Nucleic Acids. 2010;29:330–4.
Niu SW, Chang KT, Lin HY, Kuo IC, Chang YH, Chen YH, et al. Decreased incidence of gout in diabetic patients using pioglitazone. Rheumatology (Oxford). 2018;57(1):92–9.
Chen W, Roncal-Jimenez C, Lanaspa M, Gerard S, Chonchol M, Johnson RJ, et al. Uric acid suppresses 1 alpha hydroxylase in vitro and in vivo. Metabolism. 2014;63:150–60.
Vanholder R, Patel S, Hsu CH. Effect of uric acid on plasma levels of 1,25(OH)2D in renal failure. J Am Soc Nephrol. 1993;4:1035–8.
Takahashi S, Yamamoto T, Moriwaki Y, Tsutsumi Z, Yamakita JI, Higashino K. Decreased serum concentrations of 1,25(OH)2 vitamin D3 in patients with gout. Metabolism. 1998;47:336–8.
•• Sipahi S, Acikgoz SB, Genc AB, Yildirim M, Solak Y, Tamer A. The association of vitamin D status and vitamin D replacement therapy with glycemic control, serum uric acid levels, and microalbuminuria in patients with type 2 diabetes and chronic kidney disease. Med Princ Pract. 2017;26:146–51. A recent study of associations between vitamin D status and glycemic indicators and renal function in people with T2DM and CKD.
Hayashino Y, Okamura S, Tsujii S, Ishii H. Association of serum uric acid levels with the risk of development or progression of albuminuria among Japanese patients with type 2 diabetes: a prospective cohort study (Diabetes Distress and Care Registry at Tenri [DDCRT 10]). Acta Diabetol. 2016;53:599–607.
Peng H, Li H, Li C, Chao X, Zhang Q, Zhang Y. Association between vitamin D insufficiency and elevated serum uric acid among middle-aged and elderly Chinese Han women. PLoS One. 2013;8(4):e61159.
Yandı YE, Gencer E, Kılavuz B, Baki AE, Şahin H, Bilici M, et al. Cyanocobalamin and 25-hydroxy vitamin D levels in gout patients: an overlooked issue. Open Journal of Rheumatology and Autoimmune Diseases. 2016;6:96–101.
•• Langer-Gould A, Lucas RM, Xiang AH, Wu J, Chen LH, Gonzales E, Haraszti S, Smith JB, Quach H, Barcellos LF. Vitamin D-binding protein polymorphisms, 25-hydroxyvitamin D, sunshine and multiple sclerosis. Nutrients. 2018;10(2). An important recent discussion about how VDBP polymorphisms affect the biologic expression of vitamin D.
Mathieu C. Vitamin D and diabetes: where do we stand? Diabetes Res Clin Pract. 2015;108:201–9.
Al-Shoumer KA, Al-Essa TM. Is there a relationship between vitamin D with insulin resistance and diabetes mellitus? World J Diabetes. 2015;6:1057–64.
Pittas AG, Nelson J, Mitri J, Hillmann W, Garganta C, Nathan DM, et al. Plasma 25-hydroxyvitamin D and progression to diabetes in patients at risk for diabetes: an ancillary analysis in the Diabetes Prevention Program. Diabetes Care. 2012;35:565–373.
Thomas GN, ó Hartaigh B, Bosch JA, et al. Vitamin D levels predict all-cause and cardiovascular disease mortality in subjects with the metabolic syndrome: the Ludwigshafen Risk and Cardiovascular Health (LURIC) Study. Diabetes Care. 2012;35:1158–64.
Sinotte M, Diorio C, Bérubé S, Pollak M, Brisson J. Genetic polymorphisms of the vitamin D binding protein and plasma concentrations of 25-hydroxyvitamin D in premenopausal women. Am J Clin Nutr. 2009;89(2):634–40.
Valdivielso JM, Fernandez E. Vitamin D receptor polymorphisms and diseases. Clin Chim Acta. 2006;371(1–2):1–12.
•• Al-Daghri NM, Mohammed AK, Al-Attas OS, Ansari MGA, Wani K, Hussain SD, Sabico S, Tripathi G, Majed S, Alokail MS. Vitamin D receptor gene polymorphisms modify cardiometabolic response to vitamin D supplementation in T2DM patients. Sci Rep 2017, Article number: 8280. An important 1 year vitamin D intervention trial assessing cardiometabolic effects of vitamin D administration as a function of vitamin D receptor polymorphisms.
Jacobs ET, Van Pelt C, Forster RE, Zaidi W, Hibler EA, Galligan MA, et al. CYP24A1 and CYP27B1 polymorphisms modulate vitamin D metabolism in colon cancer cells. Cancer Res. 2013;73(8):2563–73.
•• Bahrami A, Sadeghnia HR, Tabatabaeizadeh SA, Bahrami-Taghanaki H, Behboodi N, Esmaeili H, et al. Genetic and epigenetic factors influencing vitamin D status. J Cell Physiol. 2018;233(5):4033–43. A good recent review of genetic and epigenetic influences on vitamin D status.
Ye Z, Sharpe SJ, Burgess S, Scott RA, Imamura F, Langenberg C, et al. Association between circulating 25-hydroxyvitamin D and incident type 2 diabetes: a mendelian randomisation study. Lancet Diabetes Endocrinol. 2015;3(1):P35–42.
Lu L, Bennett DA, Millwood IY, Parish S, McCarthy MI, Mahajan A, et al. Mendelian randomization study in European and Chinese adults. PLoS Med. 2018;15(5):e1002566. A recent Mendelian randomization study in which only 2 SNPs associated with vitamin D synthesis, for which possible pleiotropic effects on incident T2DM risk had been excluded, from which the investigators were able to infer a causative, genetically instrumented protective effect of vitamin D against incident T2DM.
•• Pittas AG, Dawson-Hughes B, Patricia Sheehan P et al. for the D2d Research Group. Vitamin D supplementation and prevention of type 2 diabetes. June 7, 2019. https://doi.org/10.1056/NEJMoa1900906. The largest randomized, placebo-controlled vitamin D intervention study to date in people at high risk for incident T2DM not preselected for vitamin D deficiency.
Matyjaszek-Matuszek B, Lenart-Lipińska M, Woźniakowska E. Clinical implications of vitamin D deficiency. Prz Menopauzalny. 2015;14:75–81.
Kajbaf F, Mentaverri R, Diouf M, Fournier A, Kamel S, Lalau JD. The association between 25-hydroxyvitamin D and hemoglobin A1c levels in patients with type 2 diabetes and stage 1-5 chronic kidney disease. Int J Endocrinol. 2014;2014:142468–6. https://doi.org/10.1155/2014/142468.
Thacher TD, Clarke BL. Vitamin D insufficiency. Mayo Clin Proc. 2011;86:50–60.
Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96:53–8.
Levin A, Bakris GL, Molitch M, Smulders M, Tian J, Williams LA, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 2007;71:31–8.
Author information
Authors and Affiliations
Contributions
AS is the corresponding author and reviewed about 65% of the articles cited. PD reviewed about 35% of the articles cited, chiefly those on gout, insulin resistance, and vitamin D, and she also prepared the 1st and 2nd drafts of the summary tables. GB, PD, VL, and AS all reviewed and edited the final presubmission draft.
Corresponding author
Ethics declarations
Conflict of Interest
Alan Sacerdote, Paulomi Dave, Vladimir Lokshin, and Gül Bahtiyar declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki declaration and its amendments, institutional/national research committee standards, and international/national institutional guidelines).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Pharmacologic Treatment of Type 2 Diabetes
Rights and permissions
About this article
Cite this article
Sacerdote, A., Dave, P., Lokshin, V. et al. Type 2 Diabetes Mellitus, Insulin Resistance, and Vitamin D. Curr Diab Rep 19, 101 (2019). https://doi.org/10.1007/s11892-019-1201-y
Published:
DOI: https://doi.org/10.1007/s11892-019-1201-y