Abstract
Purpose of Review
Team-based care has been proposed as a tool to improve health care delivery, especially for the treatment of complex medical conditions. Chronic limb-threatening ischemia (CLTI) is a complex disease associated with significant morbidity and mortality which often involves the care of multiple specialty providers. Coordination of efforts across the multiple physician specialists, nurses, wound care specialists, and administrators is essential to providing high-quality and efficient care. The aim of this review is to discuss the multiple facets of care of the CLTI patient and to describe components important for a team-based care approach.
Recent Findings
Observational studies have reported improved outcomes when using a team-based care approach in the care of the patients with CLTI, including reduction in mean wound healing times, decreasing rate of amputations, and readmissions.
Summary
Team-based care can streamline care of CLTI patients by raising awareness, facilitating early recognition, and providing prompt vascular assessment, revascularization, and surveillance. This approach has the potential to improve patient outcomes and reduce downstream health care costs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chronic limb-threatening ischemia (CLTI), the most severe form of peripheral artery disease (PAD), may present as ischemic rest pain, gangrene, or non-healing ulceration due to arterial insufficiency [1]. In the USA, approximately 8.5 million adults have PAD and it is estimated that about 11% present with CLTI [2, 3]. Associated with significant morbidity and mortality, patients with CLTI face amputation rates of up to 40% at 1 year and mortality rates of up to 50% at 5 years [4, 5]. Furthermore, CLTI represents a large economic burden for individual patients and society, with health care costs estimated to be greater than $4 billion annually [6].
The care of patients with CLTI requires a multidisciplinary team approach that addresses risk factor management, revascularization, and optimal wound care. Multiple physicians are often involved in addressing different aspects of care necessary to reduce cardiovascular and limb-related morbidity in CLTI patients [7]. Given the presence of multiple specialty providers, collaboration and consensus in standards of clinical care are needed to avoid fallouts in the management of this complex patient population. One of the many challenges CLTI providers face is how to best deliver the highest quality of care in the most efficient way particularly under the limitations of current clinical evidence [8].
As bundled payment structures are increasingly adopted over fee-for-service financial models, multidisciplinary teams are needed to collaborate in providing highest quality and cost-effective care. Team-based care has been defined as “the provision of health services to individual, families and/or their communities by at least 2 health providers who work collaboratively with patients and their caregivers to accomplish shared goals within and across settings to achieve coordinated, high-quality care” [9]. The goal of team-based care goes above the usual collaboration that occurs across individual providers in health care. Team-based care is the deliberate, intentional integration, coordination, and collaboration of health professionals to deliver care [10]. The goal of this review is to discuss the multiple facets of care of the CLTI patient, defining components relevant for a team-based care approach, and its implementation in current day practice.
Evidence for Team-Based Care
The team-based care approach has increasingly been adopted across different health care areas, particularly in primary care as well as in cardiovascular and surgical settings. In primary care, the team-based care approach has allowed different practices to create partnerships between providers, medical assistants, registered nurses, pharmacists, and behavioral specialists to engage in patient care to the maximum extent. This enables providers to focus on tasks that only they can perform, such as pharmacists and medical assistants managing titration of chronic medications including antihypertensives or insulin regimens [11, 12]. In the care of patients with chronic heart failure, team-based care involving a multidisciplinary team of heart failure cardiologists, nurses, pharmacists, social workers, and dieticians has proven to be essential in reducing readmissions, improving clinical outcomes, and managing patient symptoms [13, 14]. Similarly, in cardiothoracic surgery a multidisciplinary team approach in the care of high-risk complex patients has demonstrated to improve outcomes and increase patient, family, and staff satisfaction [15]. The medical literature now has greater evidence of improved coordination of care and clinical outcomes from intentional and standardized multidisciplinary care, including heart teams, pulmonary embolism response teams, and shock teams [16,17,18,19].
Revascularization
The cornerstone of CLTI care is revascularization with the goal to restore inline flow to the affected limb and prevention of major limb loss. Multiple professional society guidelines recommend endovascular and/or surgical revascularization in patients with CLTI [8, 20]. Despite these recommendations, it is estimated that less than 50% of patients with CLTI receive prompt revascularization before amputation [21, 22]. With significant variability in amputation rates and use of revascularization procedures in the USA, the regions with more intensive vascular care have been noted to have the lowest amputation rates [21, 23]. This highlights the importance of raising patient awareness and physician identification of CLTI and increasing access to care nationwide.
Revascularization strategies for patients with CLTI include surgical, endovascular, or a hybrid combination of both. Infrainguinal lower-extremity bypass is commonly used in patients with CLTI with significant infrainguinal occlusive disease, displaying excellent clinical durability and limb salvage rates [24]. However, lower extremity bypass durability is significantly limited by the type and quality of the conduit used, with venous conduits having superior patency rates than prosthetic grafts [25]. Moreover, surgical bypass is associated with perioperative complications, including graft thrombosis and surgical site infection, as well as 30-day major adverse cardiovascular events and death of 6.2% and 2.7%, respectively [26, 27]. With a wide array of approaches and development of new techniques and devices, the use of endovascular revascularization procedures has increased steadily over the last decade [28•]. This has been associated with a concomitant decrease in in-hospital mortality, major amputations, and length of stay for patients admitted with CLTI [28•, 29]. Given its associated lower morbidity and mortality rates, many have argued for an endovascular-first approach in patients with CLTI and infrainguinal disease [30]. In some cases, hybrid procedures that include a combination of bypass techniques with endovascular therapies represent a reasonable option to achieve inline flow to the foot. One example is the use of iliac stenting combined with femoral endarterectomy and patch angioplasty in the management of patients with significant inflow disease, which has been reported to have good long-term results [31]. Overall, a multitude of factors can influence the choice of revascularization strategy, including patient comorbidities, lesion characteristics, availability of autogenous conduit, presence of local expertise, and referral patterns [32].
The persistent clinical equipoise regarding the optimal revascularization modality for CLTI is partly due to the paucity of high-quality evidence. Two ongoing trials, the BEST-CLI (Best Endovascular vs. Best Surgical Therapy in Patients With Critical Limb Ischemia) trial in North America and BASIL-2 (Bypass vs. Angioplasty in Severe Ischemia of the Leg 2) trial in the UK, comparing endovascular vs surgical strategies for CLTI, will provide the evidence needed to develop a standard of care to best guide the treatment of patients with CLTI [33, 34]. The 2016 AHA/ACC guidelines on management of lower extremity PAD recommend an evaluation for revascularization options by an interdisciplinary care team before amputation in patients with CLTI [8]. In some cases, patients may benefit from a primary amputation rather than revascularization. These may include patients with extensive non-reconstructable foot necrosis, severe concomitant life-threatening infection, unfavorable arterial anatomy, and patients with terminal illness [7, 20]. A multidisciplinary vascular team is essential in managing complex scenarios and driving the decision for the most optimal revascularization strategy in each patient [35•].
Revascularization procedures whether surgical or endovascular can be complex and technically challenging, requiring skilled providers with the resources to restore flow to the limb. When CLTI patients are undergoing evaluation for revascularization, particularly in complex cases, multidisciplinary discussions with a team consisting of a noninvasive vascular medicine specialist, endovascular interventionalist, and vascular surgeon as a “CLTI team” with overview of the noninvasive and angiographic findings can help determine a consensus for the best mode of revascularization [35•]. This approach is similar to the heart-team meetings held between noninvasive cardiologists, interventional cardiologists, and cardiothoracic surgeons to achieve a consensus on a best management strategy for patients presenting with multivessel coronary artery disease or valvular heart disease [18].
Wound Care
The management of ischemic ulcers and/or non-healing wounds is another pillar in the care of patients with CLTI and requires a team approach. Active wound care encompasses debridement of necrotic tissue, offloading, dressing changes, edema and infection control, and topical regenerative regimens [36]. These strategies require prompt and frequent evaluation by a podiatrist, vascular specialist, and wound care nurses which count with the special expertise and resources. In some instances, primary urgent debridement or amputation may be needed, such as in cases of gas gangrene or necrotizing fasciitis, to prevent systemic spread of possible life-threatening infection. Some wounds may require further adjunctive and specialized therapies such as negative pressure wound therapy (wound VAC), skin grafting, and hyperbaric oxygen therapy [37]. Complex wound closure may occasionally require a close collaboration between different specialties including podiatry, vascular surgery, plastic surgery, and dermatology [38].
Wound care centers play an important role in providing the outpatient wound care for CLTI patients. They are staffed with physicians, wound care nurses, and medical assistants, and since many wounds are of the lower extremity, both podiatrists and vascular surgeons tend to lead the management of these patients. Wound care centers provide space for regular, algorithm-based multidisciplinary care of lower extremity wounds, including debridement, offloading, infection control, and other advanced wound care therapies such as hyperbaric oxygen therapy. They also allow for coordination of the plan of care with other specialists. Aggressive wound care should also include an infectious disease specialist for appropriate management of soft tissue and bone infections [35•].
Observational studies have reported improved outcomes when using a team-based care approach in the care of the patients with CLTI. A single center study in 85 CLTI patients reported greater than two-fold amputation-free survival in those who received multidisciplinary care, involving a vascular, plastic, and podiatric surgeon who jointly managed wound care versus standard wound care [39]. Another center reported that implementation of an aggressive wound care strategy consisting of bedside surgical debridement and skin grafting improved 1-year wound healing rates from 80 to 92% and shortened mean wound healing time from 82 to 48 days [40].
Follow-up
Patients who have been successfully treated for CLTI are at relatively high risk of recurrence. Regular serial evaluation of the wound in the early stages after revascularization is needed until complete wound healing is achieved, to facilitate early identification of wound worsening and timely clinically driven reintervention and debridement. This is accompanied by further prevention of wounds, by using protective footwear and prosthetics, pressure reduction, and preventative education in patients at risk [41]. Orthotics and prosthetics specialists and occupational therapists are essential in patients that require minor or major amputation.
Follow-up clinical assessments also consist of evaluation for any return or progression of symptoms, vascular physical exam, and physiologic testing. A surveillance ultrasound is typically obtained following surgical or endovascular revascularization, and on a regular surveillance schedule for bypass grafts [8]. Although there is no official consensus on ultrasound surveillance for asymptomatic patients after endovascular intervention, some high-risk patients, such as those with multiple reinterventions or poor runoff, may benefit more than others from regular ultrasound surveillance.
Risk Factor Modification
The primary goal of risk factor modification is to reduce the incidence rate of major adverse cardiovascular events and death but also aid in improving limb outcomes. This approach includes medical therapy to reduce risk of atherothrombotic events and to achieve control of underlying hypertension, hyperlipidemia, and diabetes as well as lifestyle changes.
The AHA/ACC lower extremity PAD guidelines give a Class I recommendation for the use of antiplatelet therapy to reduce risk of myocardial infarction, stroke, and vascular death in patients with CLTI, as well as statins, since their use has shown to reduce cardiovascular events, mortality, and amputation rates in patients with PAD. Use of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers is a Class II recommendation, as they have been shown to reduce cardiovascular mortality in patients with CLTI [8, 42,43,44]. Furthermore, recent trials have shown a reduction in major adverse limb events and cardiovascular events with the addition of low-dose rivaroxaban in patients undergoing surgical or endovascular revascularization for PAD [45•]. Cardiologists and vascular medicine specialists serve an important role in the CLTI team, with their understanding of the risk of benefits of these medical therapies and how to implement them. Additionally, the care of CLTI patients with diabetes requires the integration of endocrinologists and nutritionists to ensure adequate glycemic control and improve nutrition and diet.
In terms of lifestyle changes, smoking is a major risk factor for the development of PAD and CLTI, and significantly increases the risk of lower extremity bypass failure and amputation in CLTI. Smoking cessation has been shown to reduce the progression to CLTI, need for amputation, and mortality in patients with CLTI [46].
Limb Preservation Programs
Understanding the need for coordination along the multiple facets of care of CLTI patients, many institutions have developed limb preservation programs, which incorporate the team-based care approach. These programs provide integration and access to all the physician team members needed to provide optimal care to the patient with CLTI (Fig. 1). Limb preservation programs also count with dedicated administrative support to provide clinical coordination to oversee the transition of care from the inpatient to outpatient setting, as well as arrangement of consultations among providers, and scheduling of surveillance ultrasound studies [47]. By using a multidisciplinary approach, a limb preservation program can help standardize the care of patients with CLTI by fostering protocol-driven management. These programs can streamline care of CLTI patients by providing prompt vascular assessment and revascularization, offering concomitant wound and podiatric care, and ensuring increase surveillance after revascularization. This approach has been reported by multiple institutions to improve outcomes in patients with CLTI, including better amputation-free survival and decreased 30-day readmissions [48, 49].
Another critical task these programs have is to raise awareness in both the public and providers of PAD and CLTI, as well as the treatments available for limb salvage within the institution. Educational and screening efforts directed to the local community can help raise awareness with the goal of increasing early evaluation and treatment. Within health care systems, physicians at different areas of entry, such as emergency room physicians, primary care doctors, hospitalists, and podiatrists, should maintain a high index of suspicion for arterial insufficiency when patients present with non-healing foot wounds o rest pain [7]. These physicians can order initial noninvasive vascular testing and/or refer to the vascular specialist for evaluation.
The team in a limb preservation program must include individuals from multiple areas of training, working collaboratively while sharing dedication and passion for limb salvage. In addition to a multidisciplinary team of physicians, the team should include physician assistants or nurse practitioners, registered nurses, and medical assistants. Advanced practice providers serve a crucial role in the success of CLTI practices; by working in teams with physicians, they allow programs to adjust to both the growing number of patients and the increasing intensity of care these patients require. Additionally, administrative support and clinical coordination support is key in guiding the program on its strategy goals and maintaining integration and communication across providers, as well as with their patients and families. Streamlined access to diagnostic vascular ultrasound and other diagnostic modalities will help facilitate prompt diagnosis and surveillance. A limb preservation program should also include social workers and case managers that can provide resources for education as well as provide support to patients living with CLTI and those adapting to amputations.
Conclusion
Team-based care can streamline care of CLTI patients by raising awareness and allowing early recognition, providing prompt vascular assessment and revascularization, as well as close surveillance. Establishment of an effective multidisciplinary team requires communication and commitment among all team members and has the potential to improve patient outcomes and to reduce downstream health care costs. The CLTI team faces many challenges to continue to optimize CLTI care including greater implementation of guideline-directed medical therapy, continuing research to identify strategies to improve limb-related outcomes, and promotion of strategies that drive cost-effective care.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Patel MR, Conte MS, Cutlip DE, Dib N, Geraghty P, Gray W, et al. Evaluation and treatment of patients with lower extremity peripheral artery disease: consensus definitions from Peripheral Academic Research Consortium (PARC). J Am Coll Cardiol. 2015;65(9):931–41. https://doi.org/10.1016/j.jacc.2014.12.036.
Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, Callaway CW, et al. Heart Disease and Stroke Statistics-2021 Update: a report from the American Heart Association. Circulation. 2021;143(8):e254–743. https://doi.org/10.1161/CIR.0000000000000950.
Nehler MR, Duval S, Diao L, Annex BH, Hiatt WR, Rogers K, et al. Epidemiology of peripheral arterial disease and critical limb ischemia in an insured national population. J Vasc Surg. 2014;60(3):686–95 e2. https://doi.org/10.1016/j.jvs.2014.03.290.
Abu Dabrh AM, Steffen MW, Undavalli C, Asi N, Wang Z, Elamin MB, et al. The natural history of untreated severe or critical limb ischemia. J Vasc Surg. 2015;62(6):1642–51 e3. https://doi.org/10.1016/j.jvs.2015.07.065.
Shishehbor MH, White CJ, Gray BH, Menard MT, Lookstein R, Rosenfield K, et al. Critical limb ischemia: an expert statement. J Am Coll Cardiol. 2016;68(18):2002–15. https://doi.org/10.1016/j.jacc.2016.04.071.
Sachs T, Pomposelli F, Hamdan A, Wyers M, Schermerhorn M. Trends in the national outcomes and costs for claudication and limb threatening ischemia: angioplasty vs bypass graft. J Vasc Surg. 2011;54(4):1021–31 e1. https://doi.org/10.1016/j.jvs.2011.03.281.
Armstrong EJ, Alam S, Henao S, Lee AC, DeRubertis BG, Montero-Baker M, et al. Multidisciplinary care for critical limb ischemia: current gaps and opportunities for improvement. J Endovasc Ther. 2019;26(2):199–212. https://doi.org/10.1177/1526602819826593.
Gerhard-Herman MD, Gornik HL, Barrett C, Barshes NR, Corriere MA, Drachman DE, et al. 2016 AHA/ACC Guideline on the management of patients with lower extremity peripheral artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2017;69(11):e71–126. https://doi.org/10.1016/j.jacc.2016.11.007.
Mitchell P, Wynia M, Golden R, McNellis B, Okun S, Webb CE, Rohrbach V, Von Kohorn I. Core principles & values of effective team-based health care. NAM Perspectives. 2012. https://nam.edu/wp-content/uploads/2015/06/VSRT-Team-Based-Care-Principles-Values.pdf.
Schottenfeld L PD, Peikes D, Ricciardi R, Burak H, McNellis R, Genevro J. Creating patient-centered team-based primary care. Rockville, MDMarch 2016.
Wagner EH, Flinter M, Hsu C, Cromp D, Austin BT, Etz R, et al. Effective team-based primary care: observations from innovative practices. BMC Fam Pract. 2017;18(1):13. https://doi.org/10.1186/s12875-017-0590-8.
Kennelty KA, Polgreen LA, Carter BL. Team-based care with pharmacists to improve blood pressure: a review of recent literature. Curr Hypertens Rep. 2018;20(1):1. https://doi.org/10.1007/s11906-018-0803-0.
Cooper LB, Hernandez AF. Assessing the quality and comparative effectiveness of team-based care for heart failure: who, what, where, when, and how. Heart Fail Clin. 2015;11(3):499–506. https://doi.org/10.1016/j.hfc.2015.03.011.
Brush JE Jr, Handberg EM, Biga C, Birtcher KK, Bove AA, Casale PN, et al. 2015 ACC health policy statement on cardiovascular team-based care and the role of advanced practice providers. J Am Coll Cardiol. 2015;65(19):2118–36. https://doi.org/10.1016/j.jacc.2015.03.550.
Crawford TC, Conte JV, Sanchez JA. Team-based care: the changing face of cardiothoracic surgery. Surg Clin North Am. 2017;97(4):801–10. https://doi.org/10.1016/j.suc.2017.03.003.
Tehrani BN, Truesdell AG, Sherwood MW, Desai S, Tran HA, Epps KC, et al. Standardized team-based care for cardiogenic shock. J Am Coll Cardiol. 2019;73(13):1659–69. https://doi.org/10.1016/j.jacc.2018.12.084.
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Fleisher LA, et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2017;70(2):252–89. https://doi.org/10.1016/j.jacc.2017.03.011.
Nallamothu BK, Cohen DJ. No “i” in Heart Team: incentivizing multidisciplinary care in cardiovascular medicine. Circ Cardiovasc Qual Outcomes. 2012;5(3):410–3. https://doi.org/10.1161/CIRCOUTCOMES.112.966101.
Dudzinski DM, Piazza G. Multidisciplinary pulmonary embolism response teams. Circulation. 2016;133(1):98–103. https://doi.org/10.1161/CIRCULATIONAHA.115.015086.
Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG, et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg. 2007;45 Suppl S:S5–67. https://doi.org/10.1016/j.jvs.2006.12.037.
Goodney PP, Travis LL, Nallamothu BK, Holman K, Suckow B, Henke PK, et al. Variation in the use of lower extremity vascular procedures for critical limb ischemia. Circ Cardiovasc Qual Outcomes. 2012;5(1):94–102. https://doi.org/10.1161/CIRCOUTCOMES.111.962233.
Stella J, Engelbertz C, Gebauer K, Hassu J, Meyborg M, Freisinger E, et al. Outcome of patients with chronic limb-threatening ischemia with and without revascularization. Vasa. 2020;49(2):121–7. https://doi.org/10.1024/0301-1526/a000831.
Goodney PP, Holman K, Henke PK, Travis LL, Dimick JB, Stukel TA, et al. Regional intensity of vascular care and lower extremity amputation rates. J Vasc Surg. 2013;57(6):1471–79, 80 e1–3; discussion 9–80. https://doi.org/10.1016/j.jvs.2012.11.068.
Farber A, Eberhardt RT. The current state of critical limb ischemia: a systematic review. JAMA Surg. 2016;151(11):1070–7. https://doi.org/10.1001/jamasurg.2016.2018.
Schanzer A, Hevelone N, Owens CD, Belkin M, Bandyk DF, Clowes AW, et al. Technical factors affecting autogenous vein graft failure: observations from a large multicenter trial. J Vasc Surg. 2007;46(6):1180–90; discussion 90. https://doi.org/10.1016/j.jvs.2007.08.033.
Conte MS, Geraghty PJ, Bradbury AW, Hevelone ND, Lipsitz SR, Moneta GL, et al. Suggested objective performance goals and clinical trial design for evaluating catheter-based treatment of critical limb ischemia. J Vasc Surg. 2009;50(6):1462–73 e1–3. https://doi.org/10.1016/j.jvs.2009.09.044.
Greenblatt DY, Rajamanickam V, Mell MW. Predictors of surgical site infection after open lower extremity revascularization. J Vasc Surg. 2011;54(2):433–9. https://doi.org/10.1016/j.jvs.2011.01.034.
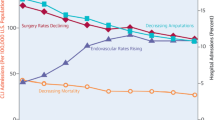
• Anantha-Narayanan M, Doshi RP, Patel K, Sheikh AB, Llanos-Chea F, Abbott JD, et al. Contemporary trends in hospital admissions and outcomes in patients with critical limb ischemia: an analysis from the National Inpatient Sample Database. Circ Cardiovasc Qual Outcomes. 2021;14(2):e007539. https://doi.org/10.1161/CIRCOUTCOMES.120.007539. This study highlights contemporary trends in CLTI admissions including increasing number of endovascular interventions, decreasing rates of amputation and mortality.
Dosluoglu HH, O’Brien-Irr MS, Lukan J, Harris LM, Dryjski ML, Cherr GS. Does preferential use of endovascular interventions by vascular surgeons improve limb salvage, control of symptoms, and survival of patients with critical limb ischemia? Am J Surg. 2006;192(5):572–6. https://doi.org/10.1016/j.amjsurg.2006.08.002.
Jaff MR, White CJ, Hiatt WR, Fowkes GR, Dormandy J, Razavi M, et al. An update on methods for revascularization and expansion of the TASC lesion classification to include below-the-knee arteries: a supplement to the inter-society consensus for the management of peripheral arterial disease (TASC II): The TASC steering committee. Catheter Cardiovasc Interv. 2015;86(4):611–25. https://doi.org/10.1002/ccd.26122.
Chang RW, Goodney PP, Baek JH, Nolan BW, Rzucidlo EM, Powell RJ. Long-term results of combined common femoral endarterectomy and iliac stenting/stent grafting for occlusive disease. J Vasc Surg. 2008;48(2):362–7. https://doi.org/10.1016/j.jvs.2008.03.042.
Bradbury A, Wilmink T, Lee AJ, Bell J, Prescott R, Gillespie I, et al. Bypass versus angioplasty to treat severe limb ischemia: factors that affect treatment preferences of UK surgeons and interventional radiologists. J Vasc Surg. 2004;39(5):1026–32. https://doi.org/10.1016/j.jvs.2004.01.031.
Menard MT, Farber A, Assmann SF, Choudhry NK, Conte MS, Creager MA, et al. Design and rationale of the best endovascular versus Best Surgical Therapy for Patients With Critical Limb Ischemia (BEST-CLI) Trial. J Am Heart Assoc. 2016;5(7). https://doi.org/10.1161/JAHA.116.003219.
Popplewell MA, Davies H, Jarrett H, Bate G, Grant M, Patel S, et al. Bypass versus Angioplasty in Severe Ischaemia of the Leg - 2 (BASIL-2) trial: study protocol for a randomised controlled trial. Trials. 2016;17:11. https://doi.org/10.1186/s13063-015-1114-2.
• Kolte D, Parikh SA, Piazza G, Shishehbor MH, Beckman JA, White CJ, et al. Vascular teams in peripheral vascular disease. J Am Coll Cardiol. 2019;73(19):2477–86. https://doi.org/10.1016/j.jacc.2019.03.463. This perspective paper by the ACC Peripheral Vascular Disease Council highlights the success of team-based care approach for in cardiac care and how it can be applied for the management of peripheral vascular disease.
Frykberg RG. Team approach toward lower extremity amputation prevention in diabetes. J Am Podiatr Med Assoc. 1997;87(7):305–12. https://doi.org/10.7547/87507315-87-7-305.
Kranke P, Bennett MH, Martyn-St James M, Schnabel A, Debus SE, Weibel S. Hyperbaric oxygen therapy for chronic wounds. Cochrane Database Syst Rev. 2015(6):CD004123. https://doi.org/10.1002/14651858.CD004123.pub4.
Sumpio BE, Armstrong DG, Lavery LA, Andros G, Society for Vascular S, American Podiatric Medical A. The role of interdisciplinary team approach in the management of the diabetic foot: a joint statement from the Society for Vascular Surgery and the American Podiatric Medical Association. J Am Podiatr Med Assoc. 2010;100(4):309–11. https://doi.org/10.7547/1000309.
Chung J, Modrall JG, Ahn C, Lavery LA, Valentine RJ. Multidisciplinary care improves amputation-free survival in patients with chronic critical limb ischemia. J Vasc Surg. 2015;61(1):162–9. https://doi.org/10.1016/j.jvs.2014.05.101.
Mii S, Tanaka K, Kyuragi R, Ishimura H, Yasukawa S, Guntani A, et al. Aggressive wound care by a multidisciplinary team improves wound healing after infrainguinal bypass in patients with critical limb ischemia. Ann Vasc Surg. 2017;41:196–204. https://doi.org/10.1016/j.avsg.2016.09.024.
Kim PJ, Attinger CE, Evans KK, Steinberg JS. Role of the podiatrist in diabetic limb salvage. J Vasc Surg. 2012;56(4):1168–72. https://doi.org/10.1016/j.jvs.2012.06.091.
Bodewes TCF, Darling JD, O'Donnell TFX, Deery SE, Shean KE, Mittleman MA, et al. Long-term mortality benefit of renin-angiotensin system inhibitors in patients with chronic limb-threatening ischemia undergoing vascular intervention. J Vasc Surg. 2018;67(3):800–8 e1. https://doi.org/10.1016/j.jvs.2017.07.130.
Arya S, Khakharia A, Binney ZO, DeMartino RR, Brewster LP, Goodney PP, et al. Association of statin dose with amputation and survival in patients with peripheral artery disease. Circulation. 2018;137(14):1435–46. https://doi.org/10.1161/CIRCULATIONAHA.117.032361.
Critical Leg Ischaemia Prevention Study G, Catalano M, Born G, Peto R. Prevention of serious vascular events by aspirin amongst patients with peripheral arterial disease: randomized, double-blind trial. J Intern Med. 2007;261(3):276–84. https://doi.org/10.1111/j.1365-2796.2006.01763.x.
• Bonaca MP, Bauersachs RM, Anand SS, Debus ES, Nehler MR, Patel MR et al. Rivaroxaban in peripheral artery disease after revascularization. N Engl J Med. 2020;382(21):1994–2004. https://doi.org/10.1056/NEJMoa2000052. This randomized controlled trial showed that the addition of low dose rivaroxaban after lower extremity revascularization significantly reduced major adverse limb events and major adverse cardiovascular events.
Armstrong EJ, Wu J, Singh GD, Dawson DL, Pevec WC, Amsterdam EA, et al. Smoking cessation is associated with decreased mortality and improved amputation-free survival among patients with symptomatic peripheral artery disease. J Vasc Surg. 2014;60(6):1565–71. https://doi.org/10.1016/j.jvs.2014.08.064.
Galmer AM, Selim SM, Giri J, Lau JF, Weinberg MD. Building a critical limb ischemia program. Curr Treat Options Cardiovasc Med. 2016;18(8):50. https://doi.org/10.1007/s11936-016-0476-4.
Hioki H, Miyashita Y, Miura T, Ebisawa S, Motoki H, Izawa A, et al. Prognostic improvement by multidisciplinary therapy in patients with critical limb ischemia. Angiology. 2015;66(2):187–94. https://doi.org/10.1177/0003319714523113.
Vartanian SM, Robinson KD, Ofili K, Eichler CM, Hiramoto JS, Reyzelman AM, et al. Outcomes of neuroischemic wounds treated by a multidisciplinary amputation prevention service. Ann Vasc Surg. 2015;29(3):534–42. https://doi.org/10.1016/j.avsg.2014.10.030.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Yulanka Castro-Dominguez declares no conflict of interest. Mehdi H. Shishehbor reports Advisory Board and consulting fees from Medtronic, Abbott Vascular, Boston Scientific, Phillips, and Terumo. He also reports leadership or fiduciary role in other board, society, committee, or advocacy group, paid or unpaid, for SCAI, ACC, and AHA.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Peripheral Vascular Disease
Rights and permissions
About this article
Cite this article
Castro-Dominguez, Y., Shishehbor, M.H. Team-Based Care in Patients with Chronic Limb-Threatening Ischemia. Curr Cardiol Rep 24, 217–223 (2022). https://doi.org/10.1007/s11886-022-01643-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-022-01643-2