Abstract
The PROMISE (Prospective multicenter imaging study for evaluation of chest pain) trial compared the effectiveness of coronary CT angiography and functional testing as initial diagnostic test for patients with suspicion for stable coronary artery disease (CAD). With 10,003 patients randomized at 193 sites, the PROMISE trial provides a snapshot of real-world care for this very common presentation. Over a median follow-up of 25 months, PROMISE did not find significant differences in major clinical events (composite endpoint 164 vs. 151, HR 1.04 (0.83–1.29); p = 0.75) between the two strategies. Other major findings were the large discrepancy between estimates of pre-test likelihood and observed prevalence for obstructive CAD (≥50 %) and the proportion of noninvasive tests positive for ischemia or obstructive CAD (53 vs. 11 %; respectively) and the better efficiency of coronary computed tomography angiography (CTA) to select patients for invasive coronary angiography (ICA) who had obstructive CAD (72 vs. 48 % for coronary CTA and functional testing, respectively). Radiation exposure was higher in the CT arm compared to all functional testing but lower than for nuclear perfusion stress testing. Improvement of patient selection for diagnostic testing and risk stratification will be keys to increase efficacy and efficiency of management of patients with suspicion for stable CAD.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction: Background and Rationale for the PROMISE Trial
Symptoms suggestive of stable coronary artery disease (CAD) are one of the most common presentations in the USA [1]. Many of the 4 million who present with de novo angina pectoris [2] undergo functional diagnostic testing including exercise ECG, echocardiography, or stress nuclear imaging during exercise or pharmacological stress for the evaluation of inducible myocardial ischemia. Functional testing has a class Ia recommendation from the American Heart Association and American College of Cardiology guidelines for this indication [3]. Large registries suggest a limited diagnostic yield of functional testing to identify patients with obstructive CAD on subsequent invasive coronary angiography (ICA) (40–50 %) [4].
Coronary computed tomography angiography (CTA) is a newer technique that permits noninvasive visualization of the coronary arteries including the evaluation of coronary artery plaque and luminal narrowing. Studies have demonstrated the excellent diagnostic accuracy of coronary CTA to detect the presence and extent of obstructive CAD (defined as either 50 or 70 % stenosis) as compared to ICA with a sensitivity of 95 % (range 91–99 %) and a specificity of 83 % (range 76 to 91 %) [2, 5–7]. In addition, coronary CTA due its ability to provide a more granular assessment of presence and extent of CAD, including non-calcified plaque, provides important prognostic information to risk stratify patients according to their risk of future major adverse cardiac events (MACE) [8–10]. A metaanalysis of 32 studies including 41,960 patients with suspected CAD and a mean follow-up of 2 years demonstrated that the risk for cardiac death or myocardial infarction (MI) was increased six to fifteen times in patients with non-obstructive and obstructive CAD (OR 6.41; 95%CI (2.44–16.84) and OR 14.92; 95%CI (6.78–32.85)), respectively [11]. At the same, the absence of CAD is associated with a very low event rate over the next 5 years (<0.3 % annually) [12, 13].
However, according to the 2008 Medicare Part B database, clinical adoption of CCTA has been slow, for example the utilization rate of myocardial perfusion imaging (MPI) was 15–44 times that of CCTA [14, 15].
Together, these data provided the motivation for the PROMISE (prospective multicenter imaging study for evaluation of chest pain) [16•] trial with the goal to determine whether one of the two available strategies would render benefits on health and economical outcomes in patients with suspicion of stable obstructive CAD in a generalizable real-world care setting.
Design and Population of the PROMISE Trial
The PROMISE trial was a randomized comparative effectiveness trial in stable outpatient chest pain patients who required noninvasive cardiac testing to determine the presence or absence of obstructive CAD or myocardial ischemia. Eligible patients were randomly assigned to either coronary CTA or functional testing (exercise electrocardiography, stress echocardiography, or nuclear stress testing). Major inclusion criteria for the PROMISE trial were men ≥45 and women ≥50 years of age with symptoms suspicious for obstructive CAD but without known CAD. Major exclusion criteria were acute or unstable presentation or any class I indication for urgent invasive catheterization, LVEF <40 %, and contraindications for CTA. The study was performed at 193 community and academic medical centers in the USA and Canada. Participating sites had established expertise in cardiology, radiology, primary care, urgent care, and anesthesiology. The sites were certified and had experienced readers interpreting cardiac test results. The trial provided recommendations for patient management but in keeping with the principles of an effectiveness trial, care was decided by local physicians.
The population of the PROMISE trial included 53 % women and 17 % ethnic minorities. Patients were middle-aged (mean age 60.8 ± 8.3 years), had a high burden of cardiovascular risk factors (mean 2.4 ± 1.1 of the following 5 risk factors; 21.4 % had diabetes, 65.0 % had hypertension, 51.1 % were past or current tobacco users, 67.7 % had dyslipidemia, and 32.1 % had family history of premature CAD); the majority had atypical chest pain (77.7 %) resulting in a mean pre-test likelihood for obstructive CAD of 53 % based on the combined Diamond and Forrester (DF) and Coronary Artery Surgery Study risk score. In 67.6 % of all patients, the 10-year risk for ASCVD events was ≥7.5 %. Overall, patients enrolled in the PROMISE trial had demographics, cardiovascular risk profile, and pre-test likelihood consistent with low to intermediate risk of CAD and in whom a noninvasive test was reasonable and indicated per guidelines [3].
Results of the PROMISE Trial
The primary endpoint of the PROMISE trial was a composite of death, myocardial infarction, hospitalization for unstable angina, or major procedural complication. After a median follow-up of 25 months (interquartile range 18 to 34 months), there was no significant difference in time to the primary composite endpoint in the CTA-based strategy as compared to functional testing (164 vs. 151; HR 1.04; 95%CI (0.83–1.29); p = 0.75) or any of its components (death or nonfatal myocardial infarction 104 vs. 112; HR 0.88; 95%CI (0.67–1.15); p = 0.35; death or nonfatal myocardial infarction or hospitalization for unstable angina 162 vs. 148; HR 1.04; 95%CI (0.84–1.31); p = 0.70), and in the combination of the primary endpoint plus catheterization showing no obstructive CAD (332 vs. 353; HR 0.91; 95%CI (0.78–1.06); p = 0.22) between two study groups. There was a significantly lower number of ICAs showing no obstructive CAD in the CTA arm as compared to the functional arm (170/4996 (3.4 %) vs. 213/5007 (4.3 %); p = 0.02) (see Table 1).
Across the trial, we observed a low rate of both ICA and revascularization (1015/10,003 (10.1 %) and 469/10,003 (4.7 %), respectively). However, compared to functional testing, more patients in the CT arm underwent ICA and revascularization (609/4996 (12.2 %) vs. 406/5007 (8.1 %) and 311/4996 (6.2 %) vs. 158/5007 (3.2 %), respectively; both p < 0.001).
Prevalence of Obstructive CAD and Myocardial Ischemia
The mean pre-test likelihood of obstructive CAD in the PROMISE trial based on a combined Diamond and Forrester and Coronary Artery Surgery Study risk score was 53.3 ± 21.4 %. This was much higher than the observed prevalence of obstructive CAD after coronary CTA (12.6 %) or myocardial ischemia after functional testing (11.7 %) [17]. Similar findings were published recently by the CONFIRM registry in 14,048 patients reporting a substantially lower prevalence of obstructive CAD in coronary CTA than that predicted by Diamond and Forrester criteria (18 % vs. 51 % and 10 % vs. 42 % for ≥50 and ≥70 % stenosis thresholds, respectively) [18].
The primary reason for this discrepancy is that the patient population assessed for obstructive CAD has significantly changed since George Diamond and James Forrester reported their findings in 1979 [19]. For instance, lifestyle changes have led to a marked decrease in cigarette smoking from 42 % in 1965 to 30 % in 1985, and 18 % in 2014 [20, 21]. In addition, Diamond and Forrester based their observations on ICA. Several studies suggest that modifying the Diamond Forrester model by inclusion of risk factors, the agreement between the projected, and observed prevalence of obstructive CAD can be improved in contemporary populations using coronary CTA as the gold standard for obstructive CAD [22–24].
Health Outcomes
The primary composite endpoint (death, myocardial infarction, hospitalization for unstable angina, or major procedural complication) occurred in 3.1 % of patients during a median of 25 months of follow-up in the PROMISE trial, which is lower than the anticipated 8 % over 2.5 years in the functional arm based on historical and national claims data [10, 17, 25, 26]. Contributing factors include a lower than expected disease burden, changes in lifestyle and increase in preventive medical therapy such as aspirin and statins as compared to historical populations (45 % of patients were on antiplatelet therapy and 46 % on statin therapy), and perhaps in a minor way a small decrease in the minimum follow-up from 2 to 1 years. However, the observed annual event rate in PROMISE (1.5 %) is consistent with other recent trials such as SCOT-HEART [27] (annual event rate 0.9 %), suggesting that contemporary populations of patients with suspicion of stable CAD are at intermediate Framingham Risk, making medical therapy the preferred choice of treatment.
Coronary Revascularization and Health Outcomes
Another observation of the PROMISE trial was that the higher rate of revascularization after coronary CTA as compared to functional testing CT arm (311/4996 (6.2 %) vs. 158/5007 (3.2 %); p < 0.001) did not translate into a lower MACE rate. Similar results were previously reported from randomized comparative effectiveness trials in the acute chest pain setting (ROMICAT-II, ACRIN-PA, and CT-STAT) [28–30]. Whether the effectiveness of coronary revascularization in stable chest pain can be improved by limiting this procedure to lesions with proven hemodynamic significance is the motivation and primary hypothesis of an ongoing NHLBI funded trial—International Study of Comparative Health Effectiveness with Medical and Invasive Approaches (ISCHEMIA).
In addition, the fact that caregivers were aware of the results of noninvasive diagnostic testing could have lead to differences in the outcome of unstable angina requiring hospitalization (ascertainment bias). For example, a patient in the CT arm, diagnosed with a 50 % stenosis who was subsequently medically managed and who presents with recurring symptoms may have been more likely hospitalized for work-up as compared to a patient who also has 50 % stenosis but, as most of these patients do, had a normal functional test and who is more likely to undergo outpatient work-up given similar presentation. Indeed, 50 % more patients were diagnosed with “hospitalization for unstable angina” after coronary CTA as compared to functional testing (61 vs. 41, respectively), while this trend was reversed for myocardial infarction (30 vs. 40, respectively).
Opportunities to Improve Risk Stratification and Selection for PCI Candidates in Stable Chest Pain Patients CAD
Improved Risk Stratification—High-Risk Coronary Plaque
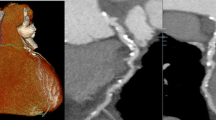
While coronary artery stenosis has been the hallmark for diagnostic and prognostic assessment, data suggest that up to two thirds of acute myocardial infarctions (MI) occur at locations in the coronary artery tree where there previously was no obstructive CAD [31, 32]. The PROSPECT trial for instance, in patients undergoing coronary revascularization after MI using intravascular ultrasound (IVUS), found that 50 % of recurrent cardiovascular events originated from non-stenotic plaque characterized by a thin fibrous cap, plaque burden >70 % (plaque occupying >70 % of cross-sectional vessel area), and luminal diameter <4 mm [2]. These features were associated with a three- to fivefold increased risk for MACE [33•].
Technical progress in coronary CTA has enabled a more granular noninvasive assessment of coronary plaque morphology and composition. For example, low CT attenuation (<30 HU) and Napkin Ring Sign (NRS) in CT accurately represent plaque with a large lipid-rich/necrotic core and a thin fibrous cap in intravascular imaging and histology [34–45], and remodeling index and increased plaque burden can be accurately detected and measured by coronary CTA as compared to IVUS [46–48]. Several studies in populations similar to the PROMISE trial suggest that presence of high-risk plaque confers a six- and twelvefold excess risk for MACE, independent of traditional CV risk factors and obstructive CAD [49–51, 52•]. Hence, high-risk plaque may improve risk stratification, especially in those with non-obstructive disease.
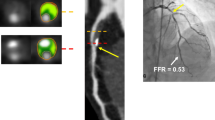
Improved Selection for Candidates for PCI–FFR-CT
In addition, recent studies suggest that noninvasive estimation of fractional coronary flow reserve (FFR) is possible using modeled computational fluid dynamics based on regular contrast enhanced coronary CTA data sets [53]. Using this methodology, FFR-CT can be calculated in good correlation with invasive FFR measurements (r = 0.63–0.82) [54–56] and may have favorably increased the specificity of coronary CTA. For example, the NXT trial in 252 patients reported an increase in specificity compared to invasive FFR per patient from 34 % based on anatomic CTA assessment to 79 % based on FFR-CT [56]. This may have a favorable effect on resource use as compared to a strategy of referring patients directly to ICA with more than a 50 % reduction of initially planned ICA procedures in the FFR-CT group [57].
Radiation Exposure
In the PROMISE trial, the overall mean radiation exposure of the CTA arm was higher compared to the functional arm (12.0 ± 8.5 vs. 10.1 ± 9.0 mSv; p < 0.001). However, functional testing with no radiation exposure (exercise ECG and stress echocardiography, see Table 1) were included in these calculations. These tests were performed in 10.2 % and 22.5 % of the patients undergoing a functional testing strategy, respectively. The majority of patients underwent nuclear myocardial perfusion imaging (67.3 %). Among the group of patients who were intended to undergo nuclear myocardial perfusion imaging before randomization (n = 6781), the 90-day cumulative radiation exposure was significantly higher as compared to the CTA group (12.0 ± 8.4 vs. 14.1 ± 7.6; p < 0.001).
As both nuclear and CT technology improve over time, a decrease in radiation dose can be achieved using advanced technology. For instance, a rapid decrease in radiation exposure during a 6-year period between 2005 and 2010 has been observed with CTA with median doses decreasing by nearly 75 % from 13.1 to 3.3 mSv [58].
Conclusion
The PROMISE trial compared the effectiveness of coronary CT angiography and functional testing as initial diagnostic test for patients with suspicion for stable CAD and provided a snapshot of real-world care for this very common presentation. PROMISE did not find significant differences in major clinical events between the two strategies. However, lessons from PROMISE include the need to improve patient selection for diagnostic testing and coronary revascularization as well as to focus on improvement of risk stratification and medical therapy.
Abbreviations
- 95%CI:
-
95 % confidence interval
- CAD:
-
Coronary artery disease
- CTA:
-
CT angiography
- CVD:
-
Cardiovascular disease
- FFR:
-
Fractional flow reserve
- HR:
-
Hazard ratio
- ICA:
-
Invasive coronary angiography
- MACE:
-
Major adverse cardiac event
- OMT:
-
Optimal medical therapy
- PCI:
-
Percutaneous coronary intervention
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cherry DK, Woodwell DA, Rechtsteiner EA. National ambulatory medical care survey: 2005 summary. Adv Data. 2007;2007(387):1–39.
Miller JM, Rochitte CE, Dewey M, et al. Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med. 2008;359(22):2324–36.
Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2012;126(25):e354–471.
Patel MR, Peterson ED, Dai D, et al. Low diagnostic yield of elective coronary angiography. N Engl J Med. 2010;362(10):886–95.
Budoff MJ, Dowe D, Jollis JG, et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) trial. J Am Coll Cardiol. 2008;52(21):1724–32.
Meijboom WB, Meijs MF, Schuijf JD, et al. Diagnostic accuracy of 64-slice computed tomography coronary angiography: a prospective, multicenter, multivendor study. J Am Coll Cardiol. 2008;52(25):2135–44.
Janne d’Othée B, Siebert U, Cury R, Jadvar H, Dunn EJ, Hoffmann U. A systematic review on diagnostic accuracy of CT-based detection of significant coronary artery disease. Eur J Radiol. 2008;65(3):449–61.
Ostrom MP, Gopal A, Ahmadi N, et al. Mortality incidence and the severity of coronary atherosclerosis assessed by computed tomography angiography. J Am Coll Cardiol. 2008;52(16):1335–43.
Hadamitzky M, Taubert S, Deseive S, et al. Prognostic value of coronary computed tomography angiography during 5 years of follow-up in patients with suspected coronary artery disease. Eur Heart J. 2013;34(42):3277–85.
Bamberg F, Sommer WH, Hoffmann V, et al. Meta-analysis and systematic review of the long-term predictive value of assessment of coronary atherosclerosis by contrast-enhanced coronary computed tomography angiography. J Am Coll Cardiol. 2011;57(24):2426–36.
Habib PJ, Green J, Butterfield RC, et al. Association of cardiac events with coronary artery disease detected by 64-slice or greater coronary CT angiography: a systematic review and meta-analysis. Int J Cardiol. 2013;169(2):112–20.
Min JK, Shaw LJ, Devereux RB, et al. Prognostic value of multidetector coronary computed tomographic angiography for prediction of all-cause mortality. J Am Coll Cardiol. 2007;50(12):1161–70.
Min JK, Dunning A, Lin FY, et al. Age- and sex-related differences in all-cause mortality risk based on coronary computed tomography angiography findings results from the International Multicenter CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter Registry) of 23,854 patients without known coronary artery disease. J Am Coll Cardiol. 2011;58(8):849–60.
Levin DC, Parker L, Halpern EJ, Julsrud PR, Rao VM. The lack of growth in use of coronary CT angiography: is it being appropriately used? AJR Am J Roentgenol.196(4):862–867
Shreibati JB, Baker LC, Hlatky MA. Association of coronary CT angiography or stress testing with subsequent utilization and spending among Medicare beneficiaries. JAMA. 2011;306:2128–36.
Douglas PS, Hoffmann U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med. 2015;372(14):1291–300. Randomized comparative effectiveness trial providing a snapshot of contemporary cardiology practice of noninvasive imaging in stable chest pain in the US.
Douglas PS, Hoffmann U, Lee KL, et al. PROspective Multicenter Imaging Study for Evaluation of chest pain: rationale and design of the PROMISE trial. Am Heart J. 2014;167(6):796–803.e791.
Cheng VY, Berman DS, Rozanski A, et al. Performance of the traditional age, sex, and angina typicality-based approach for estimating pretest probability of angiographically significant coronary artery disease in patients undergoing coronary computed tomographic angiography: results from the multinational coronary CT angiography evaluation for clinical outcomes: an international multicenter registry (CONFIRM). Circulation. 2011;124(22):2423–32. 2421–2428.
Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med. 1979;300(24):1350–8.
National Center for Health Statistics. Health, United States, 2012: with special feature on emergency care. Hyattsville (MD): National Center for Health Statistics (US); 2013 May. Report No.: 2013–1232.2013
Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics–2014 update: a report from the American Heart Association. Circulation. 2014;129(3):e28–292.
Genders TS, Steyerberg EW, Hunink MG, et al. Prediction model to estimate presence of coronary artery disease: retrospective pooled analysis of existing cohorts. BMJ. 2012;344, e3485.
Min JK, Dunning A, Gransar H, et al. Medical history for prognostic risk assessment and diagnosis of stable patients with suspected coronary artery disease. Am J Med. 2015
Genders TS, Steyerberg EW, Alkadhi H, et al. A clinical prediction rule for the diagnosis of coronary artery disease: validation, updating, and extension. Eur Heart J. 2011;32(11):1316–30.
Health Quality O. Non-invasive cardiac imaging technologies for the diagnosis of coronary artery disease: a summary of evidence-based analyses. Ont Health Technol Assess Ser. 2010;10(7):1–40.
Mudrick DW, Cowper PA, Shah BR, et al. Downstream procedures and outcomes after stress testing for chest pain without known coronary artery disease in the United States. Am Heart J. 2012;163(3):454–61.
S-H investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet. 2015;385(9985):2383–91.
Hoffmann U, Truong QA, Schoenfeld DA, et al. Coronary CT angiography versus standard evaluation in acute chest pain. N Engl J Med. 2012;367(4):299–308.
Litt HI, Gatsonis C, Snyder B, et al. CT angiography for safe discharge of patients with possible acute coronary syndromes. N Engl J Med. 2012;366(15):1393–403.
Goldstein JA, Chinnaiyan KM, Abidov A, et al. The CT-STAT (Coronary Computed Tomographic Angiography for Systematic Triage of Acute Chest Pain Patients to Treatment) trial. J Am Coll Cardiol. 2011;58(14):1414–22.
Little WC, Constantinescu M, Applegate RJ, et al. Can coronary angiography predict the site of a subsequent myocardial infarction in patients with mild-to-moderate coronary artery disease? Circulation. 1988;78(5 Pt 1):1157–66.
Ambrose JA, Tannenbaum MA, Alexopoulos D, et al. Angiographic progression of coronary artery disease and the development of myocardial infarction. J Am Coll Cardiol. 1988;12(1):56–62.
Stone GW, Maehara A, Lansky AJ, et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med.364(3):226–235. Landmark study supporting the concept of high risk plaque originating from non-culprit lesions using IVUS.
Papadopoulou S-L, Garcia-Garcia HM, Rossi A, et al. Reproducibility of computed tomography angiography data analysis using semiautomated plaque quantification software: implications for the design of longitudinal studies. Int J Cardiovasc Imaging. 2013;29:1095–104.
Kashiwagi M, Tanaka A, Kitabata H, et al. Feasibility of noninvasive assessment of thin-cap fibroatheroma by multidetector computed tomography. JACC Cardiovasc Imaging. 2009;2:1412–9.
Leber AW, Knez A, Becker A, et al. Accuracy of multidetector spiral computed tomography in identifying and differentiating the composition of coronary atherosclerotic plaques: a comparative study with intracoronary ultrasound. J Am Coll Cardiol. 2004;43:1241–7.
Marwan M, Taher MA, El Meniawy K, et al. In vivo CT detection of lipid-rich coronary artery atherosclerotic plaques using quantitative histogram analysis: a head to head comparison with IVUS. Atherosclerosis. 2011;215:110–5.
Otsuka M, Bruining N, Van Pelt NC, et al. Quantification of coronary plaque by 64-slice computed tomography: a comparison with quantitative intracoronary ultrasound. Investig Radiol. 2008;43(5):314–21.
Pundziute G, Schuijf JD, Jukema JW, et al. Head-to-head comparison of coronary plaque evaluation between multislice computed tomography and intravascular ultrasound radiofrequency data analysis. JACC Cardiovasc Interv. 2008;1(2):176–82.
Voros S, Rinehart S, Qian Z, et al. Prospective validation of standardized, 3-dimensional, quantitative coronary computed tomographic plaque measurements using radiofrequency backscatter intravascular ultrasound as reference standard in intermediate coronary arterial lesions: results from the ATLANTA (assessment of tissue characteristics, lesion morphology, and hemodynamics by angiography with fractional flow reserve, intravascular ultrasound and virtual histology, and noninvasive computed tomography in atherosclerotic plaques) I study. JACC Cardiovasc Interv. 2011;4:198–208.
Achenbach S, Moselewski F, Ropers D, et al. Detection of calcified and noncalcified coronary atherosclerotic plaque by contrast-enhanced, submillimeter multidetector spiral computed tomography: a segment-based comparison with intravascular ultrasound. Circulation. 2004;109(1):14–7.
Brodoefel H, Burgstahler C, Sabir A, et al. Coronary plaque quantification by voxel analysis: dual-source MDCT angiography versus intravascular sonography. AJR Am J Roentgenol. 2009;192(3):W84–9.
Leber AW, Knez A, von Ziegler F, et al. Quantification of obstructive and nonobstructive coronary lesions by 64-slice computed tomography: a comparative study with quantitative coronary angiography and intravascular ultrasound. J Am Coll Cardiol. 2005;46:147–54.
Pohle K, Achenbach S, Macneill B, et al. Characterization of non-calcified coronary atherosclerotic plaque by multi-detector row CT: comparison to IVUS. Atherosclerosis. 2007;190:174–80.
Schroeder S, Kopp AF, Baumbach A, et al. Noninvasive detection and evaluation of atherosclerotic coronary plaques with multislice computed tomography1. J Am Coll Cardiol. 2001;37:1430–5.
Achenbach S, Ropers D, Hoffmann U, et al. Assessment of coronary remodeling in stenotic and nonstenotic coronary atherosclerotic lesions by multidetector spiral computed tomography. J Am Coll Cardiol. 2004;43(5):842–7.
Gauss S, Achenbach S, Pflederer T, Schuhbäck A, Daniel WG, Marwan M. Assessment of coronary artery remodelling by dual-source CT: a head-to-head comparison with intravascular ultrasound. Heart. 2011;97:991–7.
Voros S, Rinehart S, Qian Z, et al. Coronary atherosclerosis imaging by coronary CT angiography: current status, correlation with intravascular interrogation and meta-analysis. JACC Cardiovasc Imaging. 2011;4:537–48.
Motoyama S, Sarai M, Harigaya H, et al. Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J Am Coll Cardiol. 2009;54(1):49–57.
Otsuka K, Fukuda S, Tanaka A, et al. Napkin-ring sign on coronary CT angiography for the prediction of acute coronary syndrome. JACC Cardiovasc Imaging. 2013;6(4):448–57.
Yamamoto H, Kitagawa T, Ohashi N, et al. Noncalcified atherosclerotic lesions with vulnerable characteristics detected by coronary CT angiography and future coronary events. J Cardiovasc Comput Tomogr. 2013;7(3):192–9.
Motoyama S, Ito H, Sarai M, et al. Plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up. J Am Coll Cardiol. 2015;66(4):337–46. Landmark study confirming the validity of the concept of high risk plaque as determined by coronary CTA.
Taylor CA, Fonte TA, Min JK. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J Am Coll Cardiol. 2013;61(22):2233–41.
Koo BK, Erglis A, Doh JH, et al. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms. Results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional Flow Reserve) study. J Am Coll Cardiol. 2011;58(19):1989–97.
Min JK, Leipsic J, Pencina MJ, et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA. 2012;308(12):1237–45.
Norgaard BL, Leipsic J, Gaur S, et al. Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: the NXT trial (Analysis of Coronary Blood Flow Using CT Angiography: Next Steps). J Am Coll Cardiol. 2014;63(12):1145–55.
Douglas PS, Pontone G, Hlatky MA, et al. Clinical outcomes of fractional flow reserve by computed tomographic angiography-guided diagnostic strategies vs. usual care in patients with suspected coronary artery disease: the prospective longitudinal trial of FFRct: outcome and resource impacts study. Eur Heart J. 2015.
Ghoshhajra BB, Engel LC, Major GP, et al. Evolution of coronary computed tomography radiation dose reduction at a tertiary referral center. Am J Med. 2012;125(8):764–72.
Acknowledgments
Maros Ferencik reports receiving the following award (American Heart Association Fellow to Faculty Award 13FTF16450001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Daniel O. Bittner and Maros Ferencik declare that they have no conflict of interest.
Udo Hoffmann reports grants from HeartFlow, Siemens HealthCare, and ACRIN, as well as fees from the AHA.
Pamela S. Douglas reports grants from HeartFlow, and grants and DSMB membership for GE HealthCare.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Cardiac PET, CT, and MRI
Rights and permissions
About this article
Cite this article
Bittner, D.O., Ferencik, M., Douglas, P.S. et al. Coronary CT Angiography as a Diagnostic and Prognostic Tool: Perspective from a Multicenter Randomized Controlled Trial: PROMISE. Curr Cardiol Rep 18, 40 (2016). https://doi.org/10.1007/s11886-016-0718-9
Published:
DOI: https://doi.org/10.1007/s11886-016-0718-9