Abstract
Background
Apnea-related hypoxia, hypercapnia, and blood pressure fluctuations cause production of various proinflammatory cytokines and trigger a vicious cycle that results in vascular endothelial damage and systemic inflammation in obstructive sleep apnea (OSA). Endothelial function is frequently impaired in OSA even in the absence of significant cardiac or vascular disorders.
Aims
This study aimed to investigate the serum endocan and serglycin levels in OSA patients.
Methods
This prospective study included 78 consecutive patients who admitted to the sleep laboratory of a tertiary referral center with the complaints of daytime sleepiness, witnessed sleep apnea, and/or snoring and who underwent all-night polysomnography (PSG). After PSG, the patients were divided into four groups in relation with their apnea–hypopnea indexes. The groups were compared for endocan and serglycin levels and their correlations with OSA severity. The correlations with demographic data and PSG findings were also investigated.
Results
The OSA and the control groups had significantly different endocan and serglycin levels (\(p = 0.001\), for both). On univariate logistic regression analysis, it was found that serglycin and endocan levels and BMI were predictors of OSA. Multiple logistic regression analysis showed that endocan and serglycin levels were independent predictors for OSA (\(p = 0.027\) and \(p = 0.003\), respectively).
Conclusions
We have demonstrated that elevated endocan and serglycin levels are predictors for OSA. Furthermore, we have showed for the first time in literature that serglycin is correlated with OSA and is an independent predictor for OSA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea (OSA) is characterized by recurrent upper airway obstructions during sleep, and it is a frequently encountered disorder affecting approximately 15–30% of the adults in the United States [1, 2]. Apnea-related hypoxia, hypercapnia, and blood pressure fluctuations cause production of various proinflammatory cytokines and trigger a vicious cycle that results in vascular endothelial damage and systemic inflammation in OSA, which forms the basis for a number of comorbidities including cardiovascular, neurodegenerative, and cognitive disorders [3, 4]. Endothelial function is frequently impaired in OSA even in the absence of significant cardiac or vascular disorders [5].
Endocan is a 50 kDa proteoglycan containing a single dermatan sulfate chain called endothelial specific molecule-1 (ESM-1) [6]. Significantly high endocan levels have been reported in disorders going together with vascular endothelial damage including type 2 diabetes mellitus (T2DM), OSA, and septic shock, and endocan has been suggested as a significant endothelial damage marker [7-10].
Serglycin is a proteoglycan and has an essential role in the modulation of activated immune system cells and inflammatory reactions [11]. Inflammatory cells synthesize and store serglycin in granules to react with mediators such as growth factors, cytokines, proteases, and chemokines and when needed [12, 13]. Serglycin may be involved in the development of vascular disorders, including atherosclerosis [14].
In the light of aforementioned information, we have hypothesized that endocan and serglycin may be used as predictive markers in OSA, and we aimed to investigate the serum endocan and serglycin levels in OSA patients. This study is the first one that has investigated serglycin levels in OSA patients.
Material and methods
This prospective study included 171 consecutive patients aged 18–65 years who were admitted to the sleep laboratory of a tertiary referral center with the complaints of daytime sleepiness, witnessed sleep apnea, and/or snoring and who underwent all-night polysomnography (PSG). A total of 93 patients were excluded due to history of hypertension, diabetes mellitus, hematological disorders, chronic inflammatory disorders, hyperlipidemia, stroke, liver conditions, malignancy, myocardial infarction, and coronary artery surgery or refusal to participate in the study. At the end, 78 patients were included in the study.
Blood sampling
Blood samples for endocan and serglycin levels and other standard laboratory tests were obtained on the day of PSG. An automated complete blood analyzer device was used to analyze complete blood count (CBC) parameters. Enzyme-linked immunosorbent assay method was employed to determine plasma serglycin and endocan levels, as described previously [8, 15].
A sleep technician-supervised full-night PSG was recorded with Alice 5 PSG device (Philips Respironics, The Netherlands) during spontaneous sleep, as described previously [4]. A PSG and sleep disorder-certified ENT physician scored all PSG data manually [16]. After PSG, the patients were divided into four groups in relation with their apnea–hypopnea indexes (AHI) as follows: simple snoring (control group) (AHI < 5), mild OSA (5 ≤ AHI < 15), moderate OSA (15 ≤ AHI < 30), and severe OSA (AHI ≥ 30).
The groups were compared for endocan and serglycin levels and their correlations with OSA severity. The correlations with demographic data and PSG findings were also investigated.
Local ethics committee approved the study protocol (Project No. E-16–933). The study was conducted in agreement with the ethical principles of Declaration of Helsinki. All participants provided their informed consents before participating in the study.
For the statistical analyses, IBM-SPSS for Windows v.21.0 software (IBM Corporation, Armonk, NY, USA) program was used. Continuous variables were presented as mean ± standard deviation, and categorical variables were presented as percentages. Data with normal distribution were compared with an independent sample t-test. Two groups with abnormal distributions were compared with the Mann–Whitney U test. Three or more groups without normal distributions were compared with the Kruskal–Wallis test. Pearson’s correlation analysis was employed for correlation analysis. The independent predictors of OSA were determined with multiple logistic regression analysis. Statistical significance was considered as \(p < 0.05\).
Results
Table 1 shows endocan and serglycin levels, CBC parameters, PSG findings, and demographic features of the control and OSA groups. There were 24 (38.7%) female, 38 (61.3%) male, and a total of 62 patients in the OSA group. The control group consisted of 7 (43.75%) male, 9 (56.25%) female, and a total of 16 patients. The participants’ mean age was 47.09 ± 8.44 years in the OSA group and 44.81 ± 4.60 years in control group. Age and gender were similar in two groups (\(p = 0.205\) and \(p = 0.302\), respectively). There was no significant difference between the two groups for CBC parameters (\(p > 0.05\)). BMI value was significantly higher in the OSA group compared to the control group (\(p = 0.021\)). The OSA and control groups had significantly different endocan and serglycin levels (\(p = 0.001\), for both) (Table 1).
Serglycin and endocan levels in relation with OSA severity
Table 2 shows the comparisons of endocan and serglycin levels and BMI and PSG findings in the control group and the OSA subgroups.
The mean endocan levels were 214.43 ± 43.08, 242.57 ± 42.14, 282.07 ± 36.98, and 292.06 ± 61.04 pg/ml, in control, mild, moderate, and severe OSA subgroups, respectively. Endocan levels differed significantly among the groups (\(p = 0.001\), Fig. 1). Except for control–mild OSA and moderate OSA–severe OSA groups, binary comparisons showed significant differences between other subgroups (Table 2).
The mean serglycin levels were 12.91 ± 2.79, 17.75 ± 4.18, 17.93 ± 4.07, and 20.75 ± 4.29 ng/ml, in control, mild, moderate, and severe OSA subgroups, respectively. Serglycin levels were significantly different in the groups (\(p = 0.001\), Fig. 2). The serglycin level was significantly different in the control group compared to the other OSA subgroups in binary comparisons. On the other hand, binary comparisons of the other OSA subgroups did not reveal any significantly difference for serglycin levels (Table 2).
The correlations of serglycin and endocan levels with BMI and PSG findings
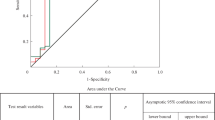
The serglycin and endocan levels had positive correlations with AHI, ST90, and BMI (\(p =0.001\) for AHI and ST90, \(p = 0.015\) and \(p = 0.019\) for BMI, respectively) and negative correlations with Min O2 (\(p = 0.001\)) (Table 3 and Figs. 3 and 4).
Multivariate logistic regression analysis for existence of OSA
On univariate logistic regression analysis, it was found that serglycin and endocan levels and BMI were predictors of OSA. Multiple logistic regression analysis showed that endocan and serglycin levels were independent predictors for OSA (\(p = 0.027\) and \(p = 0.003\), respectively) (Table 4).
Discussion
In the present study, we have demonstrated that elevated endocan and serglycin levels are predictors for OSA. Furthermore, we have showed for the first time in literature that serglycin is correlated with OSA and is an independent predictor for OSA.
OSA results in systemic inflammation and may be accompanied by various comorbid disorders including cardiovascular conditions, stroke, and cognitive disorders. The systemic inflammation in OSA has been supposed to be triggered by recurrent sleep-related upper airway obstructions, and resulting vascular endothelial injury and the comorbid disorders create a vicious cycle that aggravates OSA [17]. Hypoxia, sympathetic nervous system overactivity, and inflammation due to repetitive upper respiratory tract obstructions trigger excrete of vasoactive substances and cause endothelial dysfunction in OSA [18-20].
Inflammation, endothelial dysfunction, and OSA are strongly linked [21]. Repetitive obstructive sleep apnea events result in endothelial cell activation, signifying onset of an inflammatory response [22], and it has been reported that endothelial dysfunction may occur even without any clinical evidence of cardiovascular disorders in OSA patients [5].
It has been proposed that endocan promotes adhesion of monocytes to endothelial cells and it plays a significant role in endothelial dysfunction in inflammatory disorders [23]. High endocan levels have been reported in various disorders including chronic renal failure, sepsis, transplant rejection, hypertension, and T2DM [8, 24-27]. Balamir et al. investigated endothelial dysfunction indicators in T2DM patients and reported endocan as an endothelial dysfunction indicator positive correlated with the other indicators in T2DM [8].
The relevance of endocan has been investigated in patients with OSA-related comorbidities. However, the researchers excluded the patients with OSA alone, which is a condition that may increase the endocan levels by itself.
Altıntas et al. reported high endocan levels in the patients with OSA and revealed that continuous positive airway pressure (CPAP) therapy caused improvement in the serum endocan levels after 3 months. They also indicated endocan as a useful marker for monitoring treatment response in OSA [10]. Another study reported endocan as an independent predictor for severe OSA [9]. Sun et al. have reported that endocan may be employed as a biomarker to monitor development and progression of coronary artery disease (CAD) in OSA [23]. Kanbay et al. found significantly higher endocan levels and lower flow-mediated dilatation (FMD) values in OSA patients when compared to healthy controls and suggested that serum endocan level and FMD were correlated. They also showed that BMI was positively correlated with endocan levels [21]. In our study, the endocan levels were higher in OSA patients compared to the controls, and endocan levels showed a positive correlation with OSA severity. Our results indicated endocan as an independent predictor for presence of OSA.
Obesity is a significant predisposing issue for OSA [28], and AHI increases as the weight of the patient increases [29]. In our study, we found that BMI was correlated with presence and severity of OSA. Univariate analysis showed correlation of BMI with presence of OSA; however, multivariate analysis did not show any correlation.
Recently, Yazan et al. reported significant correlation of serum endocan levels with AHI in hypertensive OSA patients although this correlation was not evident in normotensive ones [27]. Although we excluded hypertensive patients, we found higher endocan levels in OSA patients in our study. This result offers evidence that endothelial dysfunction in OSA is multifactorial and cannot be attributed to hypertension alone. Furthermore, we may conclude that endothelial dysfunction may occur in OSA even in the absence of clinically evident cardiovascular disease, and cardiovascular disorders may be diagnosed at an early stage by determining the endocan levels in OSA patients.
Serglycin is mainly a hematopoietic cell proteoglycan and is released by hematopoietic cells such as mast cells, natural killer cells (NK), platelets, and macrophages as well as non-hematopoietic endothelial cells. Serglycin release is triggered by cytokines in endothelial cells and monocytes, and this plays a significant role in atheroma formation and other inflammatory processes [12, 30-34].
OSA patients have higher inflammatory mediator levels including intracellular adhesion molecule-1 (ICAM-1), IL-6, IL-8, and tumor necrosis factor-alpha (TNF-α) compared to the normal population [35, 36]. Liposaccharide, TNF-α, and interleukin 1-β are critical inflammatory mediators that upregulate synthesis of serglycin [32]. This signifies that serglycin plays a role in the augmentation of inflammatory response. Serglycin plays an important part in endothelial functions. Higher serglycin synthesis and secretion was reported in activated endothelial cells compared to the inactive endothelial cells [37]. In fact, serglycin plays various functions and physiological roles in reproduction, apoptosis, cell growth, immunity, and hemostasis and is also involved in several inflammatory conditions.
Considering that serglycin may play a role in vascular disorders and coronary artery disease, Bolayir et al. performed a study and reported that plasma serglycin level was correlated with the existence of coronary artery disease and the severity of coronary stenosis in patients with stable angina pectoris [38]. Ilgın et al. found elevated serglycin levels in ST-segment elevation myocardial infarction (STEMI) patients and reported a positive relationship with troponin and CRP levels, and they suggested serglycin as a useful marker in patients with STEMI [34]. Kundi et al. reported significantly and independently higher serglycin levels in patients with coronary artery ectasia (CAE), and they suggested that inflammation might play a role in the development of CAE. In a recent study, Doncheva et al. investigated the effect of serglycin expression on diet-induced adipose tissue inflammation and obesity. They found that serglycin played a role in the occurrence of obesity-induced adipose tissue inflammation [39]. In our study, we found higher serglycin levels in patients with OSA compared to the control group. In addition, we showed serglycin as an independent predictor for presence of OSA. In our study, high BMIs of the OSA patients might have affected the results concerning serglycin levels. Therefore, apart from multifactorial endothelial damage and inflammation caused by OSA, high BMI values might have caused high serglycin levels in our study.
Our study has some limitations. First, serglycin and endocan levels were not measured after CPAP therapy. Second, this is a single-center study. Third, we did not evaluate the smoking status or CRP and TNF-α levels. However, we excluded the patients with inflammatory conditions since those conditions could affect serum serglycin and endocan levels, and this strengthens our results. Further multicenter studies are required on larger and more homogeneous study populations.
In conclusion, to the best of our knowledge, this is the first study that investigated the correlation of serglycin with OSA. We found that high serglycin and endocan levels were correlated with the presence and severity of OSA. Serglycin and endocan might be useful early markers for cardiovascular comorbidities in OSA, before the onset of clinically apparent disease. More comprehensive studies are needed to further bring underlying mechanisms and the predictive value of serglycin and endocan to light in patients with OSA.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Young T, Palta M, Dempsey J et al (2009) Burden of sleep apnea: rationale, design, and major findings of the Wisconsin Sleep Cohort study. WMJ 108(5):246–249
Peppard PE, Young T, Barnet JH et al (2013) Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol 177(9):1006–1014. https://doi.org/10.1093/aje/kws342
Lee S, Thomas RJ, Kim H et al (2014) Association between high nocturnal blood pressure and white matter change and its interaction by obstructive sleep apnoea among normotensive adults. J Hypertens 32(10):2005–2012; discussion 2012. https://doi.org/10.1097/hjh.0000000000000290
Kum RO, Baklaci D, Ozcan M et al (2017) Increased risk of cerebral white matter lesions in obstructive sleep apnea syndrome. Sleep Biol Rhythms 15(1):49–55. https://doi.org/10.1007/s41105-016-0079-1
Schulz R, Schmidt D, Blum A et al (2000) Decreased plasma levels of nitric oxide derivatives in obstructive sleep apnoea: response to CPAP therapy. Thorax 55(12):1046–1051. https://doi.org/10.1136/thorax.55.12.1046
Sarrazin S, Adam E, Lyon M, Depontieu F (2006) Endocan or endothelial cell specific molecule-1 (ESM-1): a potential novel endothelial cell marker and a new target for cancer therapy. Biochim Biophys Acta (BBA) Rev Cancer 1765(1):25–37
Zhang SM, Zuo L, Zhou Q et al (2012) Expression and distribution of endocan in human tissues. Biotech Histochem 87(3):172–178. https://doi.org/10.3109/10520295.2011.577754
Balamir I, Ates I, Topcuoglu C, Turhan T (2018) Association of endocan, ischemia-modified albumin, and hsCRP levels with endothelial dysfunction in type 2 diabetes mellitus. Angiology 69(7):609–616. https://doi.org/10.1177/0003319717740781
Pusuroglu H, Somuncu U, Bolat I et al (2020) Assessment of the relationship between endocan and obstructive sleep apnea severity. Arch Med Sci 16(1)
Altintas N, Mutlu LC, Akkoyun DC et al (2016) Effect of CPAP on new endothelial dysfunction marker, endocan, in people with obstructive sleep apnea. Angiology 67(4):364–374. https://doi.org/10.1177/0003319715590558
Kolset SO, Tveit H (2008) Serglycin – structure and biology. Cell Mol Life Sci 65:1073–1085
Kolset SO, Pejler G (2011) Serglycin: a structural and functional chameleon with wide impact on immune cells. J Immunol 187(10):4927–4933. https://doi.org/10.4049/jimmunol.1100806
Niemann CU, Abrink M, Pejler G et al (2007) Neutrophil elastase depends on serglycin proteoglycan for localization in granules. Blood 109(10):4478–4486. https://doi.org/10.1182/blood-2006-02-001719
Kundi H, Gok M, Topcuoglu C, Ornek E (2017) Association of serglycin levels with isolated coronary artery ectasia. Kardiol Pol 75(10):990–996. https://doi.org/10.5603/KP.a2017.0119
Reine TM, Vuong TT, Jenssen TG, Kolset SO (2014) Serglycin secretion is part of the inflammatory response in activated primary human endothelial cells in vitro. Biochim Biophys Acta Gen Subj 1840(8):2498–2505
Berry RB, Brooks R, Gamaldo CE et al (2012) The AASM manual for the scoring of sleep and associated events. Rules, Terminology and Technical Specifications, Darien, Illinois, American Academy of Sleep Medicine
Hatipoglu U, Rubinstein I (2003) Inflammation and obstructive sleep apnea syndrome pathogenesis: a working hypothesis. Respiration 70(6):665–671. https://doi.org/10.1159/000075218
Torelli F, Moscufo N, Garreffa G et al (2011) Cognitive profile and brain morphological changes in obstructive sleep apnea. Neuroimage 54(2):787–793. https://doi.org/10.1016/j.neuroimage.2010.09.065
Bauters F, Rietzschel ER, Hertegonne KB, Chirinos JA (2016) The link between obstructive sleep apnea and cardiovascular disease. Curr Atheroscler Rep 18(1):1. https://doi.org/10.1007/s11883-015-0556-z
Ifergane G, Ovanyan A, Toledano R et al (2016) Obstructive sleep apnea in acute stroke: a role for systemic inflammation. Stroke 47(5):1207–1212. https://doi.org/10.1161/STROKEAHA.115.011749
Kanbay A, Kaya E, Büyükoğlan H et al (2015) Correlation between pentraxin-3 and endothelial dysfunction in obstructive sleep apnea syndrome. Ann Thorac Med 10(3):199
Nacher M, Serrano-Mollar A, Farre R et al (2007) Recurrent obstructive apneas trigger early systemic inflammation in a rat model of sleep apnea. Respir Physiol Neurobiol 155(1):93–96. https://doi.org/10.1016/j.resp.2006.06.004
Sun H, Zhang H, Li K et al (2019) ESM-1 promotes adhesion between monocytes and endothelial cells under intermittent hypoxia. J Cell Physiol 234(2):1512–1521
Scherpereel A, Depontieu F, Grigoriu B et al (2006) Endocan, a new endothelial marker in human sepsis. Crit Care Med 34(2):532–537. https://doi.org/10.1097/01.ccm.0000198525.82124.74
Malyszko J, Koc-Zorawska E, Malyszko JS (2018) Endocan concentration in kidney transplant recipients. Transplant Proc 50(6):1798–1801. https://doi.org/10.1016/j.transproceed.2018.02.123
El-Senosy FM, Abd El Aziz REM, Kasim SA et al (2022) Serum endocan levels and subclinical atherosclerosis in patients with chronic kidney and end-stage renal diseases. Int J Clin Pract 2022:4524637. https://doi.org/10.1155/2022/4524637
Yazan S, Karakurt H, Pusuroglu H (2023) Relationship between obstructive sleep apnea severity and serum endocan levels in patients with hypertension. Tex Heart Inst J 50(1). https://doi.org/10.14503/THIJ-21-7664
Kum RO, Kundi FCS, Baklaci D et al (2018) Predicting severe sleep apnea in patients with complaints: pulse oximetry and body mass index. Turk Arch Otorhinolaryngol 56(3):149–154. https://doi.org/10.5152/tao.2018.2928
Kritikou I, Basta M, Tappouni R et al (2013) Sleep apnoea and visceral adiposity in middle-aged male and female subjects. Eur Respir J 41(3):601–609. https://doi.org/10.1183/09031936.00183411
Woulfe DS, Lilliendahl JK, August S et al (2008) Serglycin proteoglycan deletion induces defects in platelet aggregation and thrombus formation in mice. Blood 111(7):3458–3467
Schmidt RE, MacDermott RP, Bartley G et al (1985) Specific release of proteoglycans from human natural killer cells during target lysis. Nature 318(6043):289
Korpetinou A, Skandalis SS, Labropoulou VT et al (2014) Serglycin: at the crossroad of inflammation and malignancy. Front Oncol 3:327. https://doi.org/10.3389/fonc.2013.00327
Meen AJ, Øynebråten I, Reine TM et al (2011) Serglycin is a major proteoglycan in polarized human endothelial cells and is implicated in the secretion of the chemokine GROα/CXCL1. J Biol Chem 286(4):2636–2647
Ilgın BU, Kızıltunç E, Gök M et al (2021) Association between serum serglycin levels and St-segment elevation myocardial infarction. Arq Bras Cardiol 116:756–762
Ohga E, Nagase T, Tomita T et al (1999) Increased levels of circulating ICAM-1, VCAM-1, and L-selectin in obstructive sleep apnea syndrome. J Appl Physiol 87(1):10–14
Ohga ETT, Wada H, Yamamoto H et al (2003) Effects of obstructive sleep apnea on circulating ICAM-1, IL-8, and MCP-1. J Appl Physiol 94(1):179–184
Woulfe DS, Lilliendahl JK, August S et al (2008) Serglycin proteoglycan deletion induces defects in platelet aggregation and thrombus formation in mice. Blood 111(7):3458–3467. https://doi.org/10.1182/blood-2007-07-104703
Bolayir HA, Kivrak T, Gunes H et al (2018) The association between serum serglycin level and coronary artery disease severity in patients with stable angina pectoris. Kardiol Pol 76(4):783–790. https://doi.org/10.5603/KP.2018.0007
Doncheva AI, Norheim FA, Hjorth M et al (2022) Serglycin is involved in adipose tissue inflammation in obesity. J Immunol 208(1):121–132
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material and method preparation were performed by R.O.K., F.C.S.K., and C.T. Data collection and analysis were performed by R.O.K. and C.T. Statistical analysis and interpretation were performed by R.O.K. All the authors contributed to writing and read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Our study was approved by the local ethics committee and conducted in accordance with the ethical principles described by the Declaration of Helsinki (Project No. E16-933).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kum, R.O., Sazak Kundi, F.C., Topcuoglu, C. et al. Investigation of serum endocan and serglycin levels in obstructive sleep apnea. Ir J Med Sci 192, 2909–2915 (2023). https://doi.org/10.1007/s11845-023-03360-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-023-03360-3