Abstract
Background
This study aimed to investigate the occurrence and predictive factors of restenosis in coronary heart disease (CHD) patients underwent percutaneous coronary intervention (PCI) with sirolimus-eluting stent (SES).
Methods
Demographic data, clinical features, and laboratory tests of 398 CHD patients underwent PCI with SES were retrospectively reviewed. Coronary angiography was performed to evaluate coronary stenosis before PCI and in-stent restenosis at 1-year follow-up.
Results
There were 37 (9.3%) patients suffered restenosis, but 361 (90.7%) patients did not develop restenosis at 1-year follow-up. Demographic characteristic (age), cardiovascular risk factors (hypertension and hyperuricemia), biochemical indexes (fasting blood-glucose, total cholesterol, low density lipoprotein cholesterol (LDL-C) and high-sensitivity C-reactive protein (HsCRP)), cardiac function index (cardiac troponin I), lesion features (multivessel artery lesions, target lesion at left circumflex artery (LCX), two target lesions and length of target lesion), and operation procedure (length of stent) were correlated with higher restenosis risk. Moreover, age, hypertension, diabetes mellitus, LDL-C, HsCRP, and target lesion at LCX were independent predictive factors for raised restenosis risk. Based on these independent predictive factors, we established a restenosis risk prediction model, and receiver-operating characteristic curves displayed that this model exhibited an excellent predictive value for higher restenosis risk (areas under the curve 0.953 (95% CI 0.926–0.981)).
Conclusion
Our findings provide a new insight into the prediction for restenosis in CHD patients underwent PCI with SES.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coronary heart disease (CHD), as the most common type of ischemic cardiovascular disorders, is related to serious syndromes such as stable angina and asymptomatic or silent ischemia [1, 2]. According to the 2016 World Health Organization global disease assessment report, CHD has become the leading cause of death in the past 10 years, and it is estimated that the CHD related death will reach 25 million worldwide by 2020 [3,4,5]. From pathological perspective, it is characterized by coronary atherosclerosis and results from the accumulation of fatty deposits on the arterial vessel walls, which eventually gives rise to stenosis of the arteries [6]. Regarding the treatments to remove stenosis, vascular stent represents the gold standard treatment in CHD during percutaneous coronary intervention (PCI) for most lesions, and drug-eluting stent (DES) is currently the primary choice among the vascular stents [7,8,9]. As to the drugs applied in DES, sirolimus-eluting stent (SES), which belongs to the first-generation DES, is more frequently applied in clinical practices compared with other DESs (such as paclitaxel-eluting stent, everolimus-eluting stent, and zotarolimus-eluting stent) since it is the first commercially available DES in China; moreover, it has shown superiority in terms of reducing the need for target vessel revascularization compared with the traditional bare-metal stents [10, 11]. Although SES is known as an efficacy and well-tolerated PCI strategy, in-stent restenosis still occurs in around 5–15% cases, which is a major problem in the clinical application of PCI with SES [12, 13]. Hence, it is urgently needed to explore factors that predict restenosis risk in CHD patients underwent PCI with SES implantation, which may help establish individual treatment and further improve prognosis in these patients.
According to a few studies, the occurrence of restenosis may be more frequently in patients with multiple conditions such as chronic disease histories (diabetes mellitus, hypertension and congestive heart failure), complex lesions (bypass grafts, bifurcations and longer lesion length), and abnormal biochemical indexes (low density lipoprotein (LDL), white cell count and serum uric acid), and some of these conditions have been identified to be predictive factors for restenosis risk in CHD patients underwent PCI with DES [14,15,16,17,18]. Whereas, most of the previous studies are conducted with small sample size, which may result in weak statistical power to make the data not convincing enough; moreover, comprehensive analysis of the predictive factors for restenosis risk in CHD patients underwent PCI with SES is limited; thus, further study with large sample size to comprehensively analyze the potentially predictive factors for restenosis risk is necessary. In this study, we enrolled a large population to investigate the occurrence, and predictive factors of restenosis in CHD patients underwent PCI with SES.
Methods
Patients
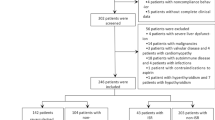
We retrospectively reviewed 398 CHD patients who underwent PCI with SES between January 2014 and June 2018. The screening criteria included the following: (i) diagnosed as CHD; (ii) age ≥ 18 years; (iii) underwent PCI with sirolimus-eluting stents; (iv) 1-year restenosis information were complete and available; (v) medical records and follow-up records were complete (at least included baseline characteristics, operation procedures and post-procedure managements); (vi) no history of PCI, coronary artery bypass grafting or other cardiovascular major surgery; (vii) no history of malignancies. This study was approved by the Institutional Review Board of The Second Hospital of Hebei Medical University, and the written informed consents were provided by patients or their guardians.
Data collection
The clinical data of patients were collected from medical records, which included (1) demographic characteristics (such as age, gender, and body mass index (BMI)); (2) cardiovascular risk factors (such as current smoke status, hypertension, diabetes mellitus, hypercholesteremia, hyperuricemia, and family history of coronary artery disease (CAD)); (3) blood pressure index (mean arterial pressure (MAP)); (4) biochemical index (such as fasting blood-glucose (FBG), glycated hemoglobin, triglyceride (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), high-sensitivity C-reactive protein (Hs-CRP), erythrocyte sedimentation rate (ESR), white blood cell (WBC), neutrophil, serum creatinine (Scr), and serum uric acid (SUA)); (6) cardiac function index (such as left ventricular ejection fraction (LVEF), cardiac troponin I (cTnl), and N-terminal probrain natriuretic peptide (NT-proBNP)); (6) angiographic information (such as multivessel artery lesions, location of target lesion, two target lesions, stenosis degree of target lesion, and length of target lesion); (7) operation procedures (such as length of stent, diameter of stent, time of stent dilation, and balloon dilation pre-stent); (8) medication used after surgery (such as aspirin, nitrates, statins, β receptor blockers, angiotensin converting enzymes inhibitors (ACEIs), angiotensin receptor blockers (ARBs), and calcium channel blockers).
In-stent restenosis assessment
For all patients, the PCI procedure and the implantation of sirolimus-eluting stent (Lepu (Beijing) Medical Devices Co., Ltd. Beijing, China) were performed according to PCI guideline [19]. The information of coronary angiography follow-up was collected from the follow-up records. Before PCI, all patients received coronary angiography to assess the clinical status. Immediately post-PCI, coronary angiography was conducted for the patients to evaluate the degree of stenosis after PCI. After discharge, if the patients with clinical indication, coronary angiography was performed when clinical visit. And for the patients without clinical indication, coronary angiography was carried out at 1-year follow-up. Based on the coronary angiograms, in-stent restenosis was assessed by the quantitative coronary angiography (QCA) analysis as previous studies described [20, 21], and the percentage diameter stenosis (PDS) was automatically calculated by computer-based system cardiovascular angiographic analysis system (CAAS) II (Pie Medical Imaging, Maastricht, the Netherlands). The in-stent restenosis was defined as the PDS of stent-implanted segment at 1-year follow-up exceeded 50% compared with lumen assessed immediately after PCI [16]. According to the 1-year restenosis, patients were further classified as restenosis group and non-restenosis group.
Statistical analysis
The normality of continuous variables was analyzed by Kolmogorov-Smirnov test. The normally distributed continuous variables were displayed as mean ± standard deviation (SD), and the non-normal distributed continuous variables were presented as median and interquartile range (IQR). The categorical variables were expressed as count (percentage). Comparison of continuous variables between two groups was determined by Student’s t test or Wilcoxon rank sum test, and comparison of categorical variables between two groups was determined by chi-square test, Yates’ corrected chi-square test, or Fisher’s exact test. Factors predicting restenosis were analyzed by univariate logistic regression, and the variables with P value < 0.1 were further screened by forward stepwise multivariate logistic regression. Based on the independent predicting factors screened from multivariate logistic regression, the restenosis risk prediction model was generated as follows: P = [exp(−23.423 + 0.062 (age) + 3.109 (hypertension) + 3.240 (diabetes mellitus) + 3.040 (LDL-C) + 0.473 (HsCRP) + 1.208 (target lesion at LCX))]/ [1 + exp(− 23.423 + 0.062 (age) + 3.109 (hypertension) + 3.240 (diabetes mellitus) + 3.040 (LDL-C) + 0.473 (HsCRP) + 1.208 (target lesion at LCX))], − 2ln (likelihood ratio) = 107.321. The predicting performance of restenosis risk prediction model and independent predicting factors were analyzed using receiver operating characteristic (ROC) curves and the areas under the curve (AUC) with 95% confidence intervals (CI). SPSS 22.0 statistical software (SPSS Inc., Chicago, USA) was used for all statistical analyses, and GraphPad Prism 7.00 software (GraphPad Software Inc., San Diego, USA) was used to plot figures. P value < 0.05 was considered significant.
Results
Clinical characteristics of CHD patients
A total of 398 CHD patients underwent PCI with SES (including 71 (17.8%) females and 327 (82.2%) males) were enrolled in this study, with mean age of 64.0 ± 9.3 years and mean BMI value of 24.5 ± 3.7 kg/m2 (Table 1). Other information of clinical characteristics including cardiovascular risk factors, blood pressure indexes, biochemical indexes, cardiac function indexes, angiographic data, operation procedures, and medications used after surgery was exhibited in Table 1.
Incidence of 1-year restenosis
Among total patients, there were 37 (9.3%) patients suffered restenosis, and 361 (90.7%) patients did not develop restenosis at 1-year follow-up (Fig. 1), and patients were further classified as restenosis group as well as non-restenosis group respectively.
Comparison of clinical characteristics between restenosis group and non-restenosis group
The age in restenosis group (68.7 ± 10.3 years) was higher compared with non-restenosis group (63.5 ± 9.0 years) (P = 0.001) (Table 2). The proportions of patients with hypertension (P = 0.009) and hyperuricemia (P = 0.031) were both increased in restenosis group compared with non-restenosis group. For biochemical indexes, elevated levels of FBG (P = 0.023), TC (P = 0.016), LDL-C (P < 0.001), and HsCRP (P < 0.001) were observed in restenosis group compared with non-restenosis group. As to cardiac function indexes, restenosis group showed higher cTnl level compared with non-restenosis group (P = 0.039). For angiographic information (lesion features), multivessel artery lesions (P = 0.019), target lesion at LCX (P = 0.048), two target lesions (P = 0.016), and length of target lesion (P = 0.005) were increased in restenosis group compared with non-restenosis group. Regarding operation procedures, restenosis group had longer length of stent compared with non-restenosis group (P = 0.007). Besides, no difference of other characteristics was found between restenosis group and non-restenosis group (Table 2).
Analysis of factors affecting restenosis risk
Univariate logistic regression analysis showed that higher age (P = 0.001, OR = 1.068), hypertension (P = 0.014, OR = 3.759), hyperuricemia (P = 0.034, OR = 2.090), higher TC (P = 0.018, OR = 1.523), higher LDL-C (P < 0.001, OR = 2.737), higher HsCRP (P < 0.001, OR = 1.268), higher SUA (P = 0.035, OR = 1.005), multivessel artery lesions (P = 0.025, OR = 3.030), patients with two target lesions (P = 0.018, OR = 2.279), larger length of target lesion (P = 0.005, OR = 1.059), and larger length of stent (P = 0.007, OR = 1.055) were associated with elevated restenosis risk (Table 3). Moreover, multivariate logistic regression analysis displayed that higher age (P = 0.033, OR = 1.064), hypertension (P = 0.001, OR = 22.397), diabetes mellitus (P < 0.001, OR = 25.534), higher LDL-C (P < 0.001, OR = 20.911), higher HsCRP (P < 0.001, OR = 1.604), and target lesion at LCX (P = 0.027, OR = 3.348) independently predicted raised restenosis risk (Table 4).
Predictive values of candidate factors for restenosis risk
Since several factors were identified as independent predictive factors for restenosis risk by multivariate logistic regression, we performed ROC curve analysis to further evaluate the predictive values of these independent predictive factors. ROC curves showed that age (AUC 0.654 (95% CI 0.548–0.761)), hypertension (AUC 0.602 (95% CI 0.517–0.688)), LDL-C (AUC 0.676 (95% CI 0.591–0.761)), and HsCRP (AUC 0.876 (95% CI 0.830–0.921)) could predict elevated restenosis risk, while diabetes mellitus (AUC 0.566 (95% CI 0.465–0.667)) and target lesion at LCX (AUC 0.582 (95% CI 0.484–0.681)) presented with poor values in predicting restenosis risk (Fig. 2). Based on these independent predictive factors, we established a restenosis risk prediction model (P = [exp(− 23.423 + 0.062 (age) + 3.109 (hypertension) + 3.240 (diabetes mellitus) + 3.040 (LDL-C) + 0.473 (HsCRP) + 1.208 (target lesion at LCX))]/ [1 + exp(− 23.423 + 0.062 (age) + 3.109 (hypertension) + 3.240 (diabetes mellitus) + 3.040 (LDL-C) + 0.473 (HsCRP) + 1.208 (target lesion at LCX))], − 2ln (likelihood ratio) = 107.321). Compared with the predictive value of separately independent factor for restenosis risk, the restenosis risk prediction model exhibited a much higher predictive value for increased restenosis risk, with the AUC of 0.953 (95% CI 0.926–0.981).
Predictive values of various factors for restenosis risk by ROC curve analysis. Predictive values of age, hypertension, diabetes mellitus, LDL-C, HsCRP, target lesion at LCX, and restenosis risk prediction model for restenosis risk, which were evaluated by ROC curves. ROC curves, receiver operating characteristic curves; LDL-C, low density lipoprotein cholesterol; HsCRP, high-sensitivity C-reactive protein; LCX, left circumflex artery; AUC, area under the curve; CI, confidence interval
Discussion
In this study, we reviewed the comprehensive characteristics of 398 CHD patients underwent PCI with SES, and explored the 1-year restenosis occurrence as well as the predictors for restenosis risk in these patients. Firstly, we found that the incidence of 1-year restenosis was 9.3%. Secondly, age, hypertension, diabetes mellitus, LDL-C, and HsCRP as well as target lesion at LCX were independent predictive factors for increased restenosis risk. Thirdly, the restenosis risk prediction model involving these independent factors had an excellent value for predicting restenosis.
In clinical practices for treating CAD, PCI with DES is regarded as revolutionized technology in interventional cardiology, which has exhibited great performance in reducing restenosis and decreasing the need for repeated revascularization, especially PCI with SES, which is the first commercially available and most commonly used DES [7, 8]. A study enrolling 115 patients with ischemic heart disease has shown that the patients received SES presents with much lower risk of restenosis compare with patients received bare metal stent [18]. And a study reviewed 19 trials displays that SES results in a great reduction in the risk of target lesion revascularization compared with other PCI strategies including paclitaxel-eluting stent, drug-coated balloons, and bare-metal stents [22]. Thus, these data reveal that SES is advantageous in prevention of restenosis and target lesion revascularization, and the reason for the superiority of SES might due to the advantage of sirolimus in promoting aggressive neointimal suppression [23, 24]. Furthermore, regarding the restenosis rate in patients underwent PCI with SES, a study displays a 9-month restenosis rate of 13.7% in 73 chronic total occlusion patients underwent PCI with SES [25]. And another study displays a 6-month restenosis rate of 9.2% in 122 chronic total occlusion patients underwent PCI with SES [26]. In line with these data, our study showed that the 1-year restenosis rate was 9.3% in CHD patients underwent PCI with SES.
Regarding the predictive factors for restenosis, a few studies with small sample size disclose some potential factors that may forecast restenosis risk. For instance, a study displays that hypertension independently predicts elevated restenosis risk in 50 patients with ischemic heart diseases underwent PCI with SES [27]. Additionally, a study shows that higher CRP is an important predictor for raised restenosis occurrence in 167 hemodialysis patients with coronary lesions who underwent PCI with SES [28]. Besides, a study discloses that higher age is an independent predictive factor for higher restenosis occurrence in 49 patients with underwent PCI with SES [29]. Partially consistent with these previous studies, our study chose some factors that might potentially affect the restenosis in CHD patients, and investigated the predictive factors for restenosis in 398 CHD patients underwent PCI with SES, which sample size was larger than the previous studies, and we observed that age, hypertension, diabetes mellitus, LDL-C, HsCRP, and target lesion at LCX were independent predictive factors for increased restenosis risk in these patients. The following reasons might explain our results: (1) elder patients had thicker arterial wall and decreased anticoagulant ability, thus they were more liable to develop atherosclerosis, which led to restenosis [16, 17]; (2) hypertension resulted in increased blood flow impact on the vessels and caused injury on the vascular endothelium, which facilitated the formation of atherosclerotic plaque and further contributed to atherosclerosis, thus it predicted raised restenosis risk [30]; (3) the patients with diabetes mellitus might have elevated advanced glycation end products (AGE) level, which increased reactive oxygen species, accelerated vein graft arterialization as well as atherosclerosis, and eventually resulted in restenosis, thus diabetes mellitus predicted higher restenosis risk [17, 31, 32]; (4) excess LCL-C level caused more cholesterol accumulation on the arterial wall, thereby contributed to atherosclerosis and further led to restenosis [33]; (5) elevated HsCRP indicated the enhanced local inflammatory response, which activated platelets and fibrinogen to recruit to the stent-induced direct arterial wall injury and further facilitated atherosclerotic plaque to results in restenosis, thereby increased restenosis risk [17, 34, 35]; (6) stent implantation on the target lesion at LCX was subjected to flexion, torsion, as well as rotational forces, and it might induce shear stress due to the acute angle as well as hinge motion; thus, it promoted the vascular endothelium injury and enhanced the occurrence of restenosis in CHD patients underwent PCI with SES [36].
Based on the independent predictive factors screened from multivariate logistic regression analysis, we generated these factors into the restenosis risk prediction model (P = [exp(− 23.423 + 0.062 (age) + 3.109 (hypertension) + 3.240 (diabetes mellitus) + 3.040 (LDL-C) + 0.473 (HsCRP) + 1.208 (target lesion at LCX))]/[1 + exp(− 23.423 + 0.062 (age) + 3.109 (hypertension) + 3.240 (diabetes mellitus) + 3.040 (LDL-C) + 0.473 (HsCRP) + 1.208 (target lesion at LCX))], − 2ln (likelihood ratio) = 107.321) and speculated whether this model was able to predict restenosis risk. We observed that the restenosis risk prediction model presented with a great predictive value for raised restenosis risk (AUC 0.953 (95% CI 0.926–0.981)) in CHD patients underwent PCI with SES, and the predictive value was stronger compared with any other factor alone. These data indicated that our restenosis risk prediction model would contribute to the prevention of restenosis in CHD patients underwent PCI with SES.
Several limitations of our study needed to be highlighted. One was that, the follow-up duration (1 year) was relatively short, and restenosis rate assessment with longer follow-up duration was not investigated. Secondly, this was a retrospective study, and a prospective study was needed to validate our results. Thirdly, as a single-center study, selective bias might exist in our study; Finally, the effect of the common indexes on predicting the restenosis risk in recurrence/restenosis patients who initiate PCI with DES statement was not investigated in this present study. Further multi-center study enrolling recurrence/restenosis patients is needed to validate our findings.
In conclusion, the restenosis risk prediction model, which involves age, hypertension, diabetes mellitus, LDL-C, HsCRP, and target lesion at LCX, may serve as an excellent predictor for increased restenosis risk in CHD patients underwent PCI with SES implantation. These findings provide a new insight into the prediction for restenosis in CHD patients underwent PCI with SES.
References
Piccolo R, Giustino G, Mehran R, Windecker S (2015) Stable coronary artery disease: revascularisation and invasive strategies. Lancet 386(9994):702–713
Sanchis-Gomar F, Perez-Quilis C, Leischik R, Lucia A (2016) Epidemiology of coronary heart disease and acute coronary syndrome. Ann Transl Med 4(13):256
Zheng X, Zheng Y, Ma J, Zhang M, Zhang Y, Liu X, Chen L, Yang Q, Sun Y, Wu J, Yu B (2019) Effect of exercise-based cardiac rehabilitation on anxiety and depression in patients with myocardial infarction: a systematic review and meta-analysis. Heart Lung 48(1):1–7
Beatty AL, Doll JA, Schopfer DW, Maynard C, Plomondon ME, Shen H, Whooley MA (2018) Cardiac rehabilitation participation and mortality after percutaneous coronary intervention: insights from the veterans affairs clinical assessment, reporting, and tracking program. J Am Heart Assoc 7(19):e010010
Tian Y, Deng P, Li B, Wang J, Li J, Huang Y, Zheng Y (2019) Treatment models of cardiac rehabilitation in patients with coronary heart disease and related factors affecting patient compliance. Rev Cardiovasc Med 20(1):27–33
Nazneen F, Herzog G, Arrigan DW, Caplice N, Benvenuto P, Galvin P, Thompson M (2012) Surface chemical and physical modification in stent technology for the treatment of coronary artery disease. J Biomed Mater Res B Appl Biomater 100(7):1989–2014
Arafat M, Fouladian P, Blencowe A, Albrecht H, Song Y, Garg S (2019) Drug-eluting non-vascular stents for localised drug targeting in obstructive gastrointestinal cancers. J Control Release 308:209–231
Zahn R, Hamm CW, Schneider S, Zeymer U, Richardt G, Kelm M, Levenson B, Bonzel T, Tebbe U, Sabin G, Nienaber CA, Pfannebecker T, Senges J, German Cypher Stent R (2007) The Sirolimus-eluting coronary stent in daily routine practice in Germany: trends in indications over the years. Results from the prospective multi-centre German Cypher Stent registry. Clin Res Cardiol 96(8):548–556
Mazin I, Paul G, Asher E (2019) Neoatherosclerosis - from basic concept to clinical implication. Thromb Res 178:12–16
Ray GM, Nawarskas JJ, Frishman WH (2006) The paclitaxel-eluting stent in percutaneous coronary intervention: part II. Comparison with the sirolimus-eluting stent, economics, and unanswered questions. Cardiol Rev 14(3):143–150
Yates DJ, Savage ML, Walters DL, Raffel OC (2016) Retrospective study of first-generation drug-eluting stents, second-generation drug-eluting stents and non-drug eluting stent methods in the treatment of native vessel in-stent restenosis in real-world clinical practice. Heart Lung Circ 25(4):342–351
Farooq V, Gogas BD, Serruys PW (2011) Restenosis: delineating the numerous causes of drug-eluting stent restenosis. Circ Cardiovasc Interv 4(2):195–205
Xiu WJ, Yang HT, Zheng YY, Ma YT, Xie X (2018) Drug-eluting balloons versus second-generation drug-eluting stents for treating in-stent restenosis in coronary heart disease after PCI: a meta-analysis. Cardiol Res Pract 2018:7658145
Kastrati A, Dibra A, Mehilli J, Mayer S, Pinieck S, Pache J, Dirschinger J, Schomig A (2006) Predictive factors of restenosis after coronary implantation of sirolimus- or paclitaxel-eluting stents. Circulation 113(19):2293–2300
Zhao LP, Xu WT, Wang L, Li H, Shao CL, Gu HB, Chan SP, Xu HF, Yang XJ (2015) Influence of insulin resistance on in-stent restenosis in patients undergoing coronary drug-eluting stent implantation after long-term angiographic follow-up. Coron Artery Dis 26(1):5–10
Wu Y, Fu X (2019) Comprehensive analysis of predictive factors for rapid angiographic stenotic progression and restenosis risk in coronary artery disease patients underwent percutaneous coronary intervention with drug-eluting stents implantation. J Clin Lab Anal 33(2):e22666
Qian H, Luo Z, Xiao C, Chen J, Li D, Xu H, He P, Zhou X, Zhang T, Min X (2018) Red cell distribution width in coronary heart disease: prediction of restenosis and its relationship with inflammatory markers and lipids. Postgrad Med J 94(1115):489–494
Obata JE, Nakamura T, Kitta Y, Saito Y, Sano K, Fujioka D, Kawabata K, Kugiyama K (2013) In-stent restenosis is inhibited in a bare metal stent implanted distal to a sirolimus-eluting stent to treat a long de novo coronary lesion with small distal vessel diameter. Catheter Cardiovasc Interv 82(6):E777–E787
Levine GN, Bates ER, Blankenship JC, Bailey SR, Bittl JA, Cercek B, Chambers CE, Ellis SG, Guyton RA, Hollenberg SM, Khot UN, Lange RA, Mauri L, Mehran R, Moussa ID, Mukherjee D, Nallamothu BK, Ting HH, American College of Cardiology F, American Heart Association Task Force on Practice G, Society for Cardiovascular A, Interventions (2013) 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Catheter Cardiovasc Interv 82(4):E266–E355
Bruining N, Sabate M, de Feyter PJ, Kay IP, Ligthart J, Disco C, Kutryk MJ, Roelandt JR, Serruys PW (1999) Quantitative measurements of in-stent restenosis: a comparison between quantitative coronary ultrasound and quantitative coronary angiography. Catheter Cardiovasc Interv 48(2):133–142
Haase J, Escaned J, van Swijndregt EM, Ozaki Y, Gronenschild E, Slager CJ, Serruys PW (1993) Experimental validation of geometric and densitometric coronary measurements on the new generation Cardiovascular Angiography Analysis System (CAAS II). Catheter Cardiovasc Diagn 30(2):104–114
Siontis GC, Piccolo R, Praz F, Valgimigli M, Raber L, Mavridis D, Juni P, Windecker S (2016) Percutaneous coronary interventions for the treatment of stenoses in small coronary arteries: a network meta-analysis. JACC Cardiovasc Interv 9(13):1324–1334
Finn AV, Nakazawa G, Joner M, Kolodgie FD, Mont EK, Gold HK, Virmani R (2007) Vascular responses to drug eluting stents: importance of delayed healing. Arterioscler Thromb Vasc Biol 27(7):1500–1510
Nakazawa G, Nakano M, Otsuka F, Wilcox JN, Melder R, Pruitt S, Kolodgie FD, Virmani R (2011) Evaluation of polymer-based comparator drug-eluting stents using a rabbit model of iliac artery atherosclerosis. Circ Cardiovasc Interv 4(1):38–46
Park HJ, Kim HY, Lee JM, Choi YS, Park CS, Kim DB, Her SH, Koh YS, Park MW, Kwon BJ, Kim PJ, Chang K, Chung WS, Seung KB (2012) Randomized comparison of the efficacy and safety of zotarolimus-eluting stents vs. sirolimus-eluting stents for percutaneous coronary intervention in chronic total occlusion--CAtholic Total Occlusion Study (CATOS) trial. Circ J 76(4):868–875
Ge L, Iakovou I, Cosgrave J, Chieffo A, Montorfano M, Michev I, Airoldi F, Carlino M, Melzi G, Sangiorgi GM, Corvaja N, Colombo A (2005) Immediate and mid-term outcomes of sirolimus-eluting stent implantation for chronic total occlusions. Eur Heart J 26(11):1056–1062
Chatani K, Muramatsu T, Tsukahara R, Ito Y, Ishimori H, Hirano K, Nakano M, Yamawaki M, Araki M, Sakurai M, Iuchi K, Nozawa T, Inoue H (2009) Predictive factors of re-restenosis after repeated sirolimus-eluting stent implantation for SES restenosis and clinical outcomes after percutaneous coronary intervention for SES restenosis. J Interv Cardiol 22(4):354–361
Ishii H, Toriyama T, Aoyama T, Takahashi H, Amano T, Hayashi M, Tanaka M, Kawamura Y, Yasuda Y, Yuzawa Y, Maruyama S, Matsuo S, Matsubara T, Murohara T (2009) Prognostic values of C-reactive protein levels on clinical outcome after implantation of sirolimus-eluting stents in patients on hemodialysis. Circ Cardiovasc Interv 2(6):513–518
Asamasu-Sato S, Mita M, Ogawa R, Isaka M, Saito M (2007) Analysis of clinical factors that influence re-stenosis after percutaneous coronary stenting. Yakugaku Zasshi 127(8):1309–1315
Mohan S, Dhall A (2010) A comparative study of restenosis rates in bare metal and drug-eluting stents. Int J Angiol 19(2):e66–e72
Aronson D (2002) Potential role of advanced glycosylation end products in promoting restenosis in diabetes and renal failure. Med Hypotheses 59(3):297–301
Ping S, Liu S, Zhou Y, Li Z, Li Y, Liu K, Bardeesi AS, Wang L, Chen J, Deng L, Wang J, Wang H, Chen D, Zhang Z, Sheng P, Li C (2017) Protein disulfide isomerase-mediated apoptosis and proliferation of vascular smooth muscle cells induced by mechanical stress and advanced glycosylation end products result in diabetic mouse vein graft atherosclerosis. Cell Death Dis 8(5):e2818
Li C, Shen Y, Xu R, Dai Y, Chang S, Lu H, Dong Z, Deng J, Qian J, Ge J (2018) Evaluation of Preprocedural laboratory parameters as predictors of drug-eluting stent restenosis in coronary chronic total occlusion lesions. Angiology:3319717752245
Li JJ, Yan HB, Xiang XP, Qin XW, Zhang CY (2009) Comparison of changes in early inflammatory markers between sirolimus- and paclitaxel-eluting stent implantation. Cardiovasc Drugs Ther 23(2):137–143
Nakazawa KR, Wengerter SP, Power JR, Lookstein RA, Tadros RO, Ting W, Faries PL, Vouyouka AG (2017) Preoperative inflammatory status as a predictor of primary patency after femoropopliteal stent implantation. J Vasc Surg 66(1):151–159
Naganuma T, Chieffo A, Basavarajaiah S, Takagi K, Costopoulos C, Latib A, Carlino M, Montorfano M, Bernelli C, Nakamura S, Colombo A (2013) Single-stent crossover technique from distal unprotected left main coronary artery to the left circumflex artery. Catheter Cardiovasc Interv 82(5):757–764
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the Institutional Review Board of The Second Hospital of Hebei Medical University, and the written informed consents were provided by patients or their guardians.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, J., Wang, X., Wang, H. et al. Occurrence and predictive factors of restenosis in coronary heart disease patients underwent sirolimus-eluting stent implantation. Ir J Med Sci 189, 907–915 (2020). https://doi.org/10.1007/s11845-020-02176-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-020-02176-9