Abstract
Background
Radiotherapy (RT) is a key treatment modality in the curative treatment of patients with non-small cell lung cancer (NSCLC). Incorrect definition of the gross, or clinical, target volume is a common source of error which can lead to a reduced probability of tumour control.
Objective
This was a pilot and a phase II study. The pilot evaluated the technical feasibility of integrating positron emission tomography–computed tomography (PET-CT) fusion. The primary outcome of the phase II study was to evaluate the safety of PET-CT scan–based RT by evaluating the rate of loco-regional recurrence outside the PET-CT planning target volume (PTV) but within conventional 3-D PTV.
Methods
Patients underwent standard post-treatment follow-up, including repeated three monthly CT scans of the thorax. In case of loco-regional recurrence, three categories were considered, with only extra-PET scan PTV and intra-CT scan PTV recurrences considered as a failure. Our hypothesis was that the rate of these events would be < 10%.
Results
Twelve patients were recruited; the study closed early due to poor recruitment. The primary endpoint of the pilot was met; it was feasible to deliver a PET-CT-based plan to ≥ 60% of patients. Two patients had intra-PET scan PTV recurrences, six had extra-PET scan PTV and extra-CT, and three patients had both. Another patient had extra-PET scan PTV and extra-CT as well as extra-PET scan PTV and intra-CT scan PTV recurrence.
Conclusion/advances in knowledge
PET-based planning has the potential to reduce radiation treatment volumes because of the avoidance of mediastinal lymph nodes that are PET negative.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Lung cancer is amongst the main causes of cancer-related death in Western countries. In the EU, the lung cancer–related mortality rate is estimated at 68.3/100,000 in men and at 14.3/100,000 in women, and this is increasing [1]. In the USA, 228,190 patients are expected to be diagnosed with lung cancer annually with an estimated 399,431 people currently living with this malignancy [2]. Patients with non-operable early-stage or locally advanced non-small cell lung cancer (NSCLC) have a poor prognosis, due both to local control failure and metastatic progression.
Radiotherapy (RT) is a key treatment modality for NSCLC patients being treated with curative intent. Careful staging and selection of patients are vital to achieve the maximal chance of prolonged survival with side effects that are acceptable. The accurate and precise delineation of target volumes is of utmost importance for preventing geographical misses. Incorrect definition of the gross tumour volume (GTV) (i.e., detectable tumour) or clinical target volume (CTV) (tumour plus a margin for microscopic extension) is a common source of error. Such errors can result in under-treatment and a reduced probability of local control. Advances in computer-assisted 3-D planning such as three-dimensional conformal radiotherapy (3-DCRT), intensity-modulated radiation therapy (IMRT) [3], and image-guided radiation therapy (IGRT) have facilitated the delivery of higher doses of radiation to the tumour and increased sparing of normal tissue [4].
Positron emission tomography (PET) scanning is a significant advance in cancer imaging [5], and when combined with structural imaging, such as computed tomography (CT), (fludeoxyglucose) 18FDG-PET provides the best available information on tumour extent for many common cancers [6]. PET imaging plays a role in diagnosis, staging, and patient selection for treatment of NSCLC. Indeed, 18FDG-based imaging represents one of the most significant advances in the pre-therapeutic management of a patient with NSCLC in the last decade, by more accurately diagnosing local and regional nodal and metastatic tumour extension [7, 8]. The role of PET imaging is formally established for patients undergoing surgery; several publications have shown promising results in patients treated by RT with or without chemotherapy [9,10,11]. At present, PET imaging is considered standard of care in those patients with seemingly localised NSCLC before the consideration of any radical local approach. Some contemporary studies have shown that the hybrid PET/CT examination has both higher sensitivity and specificity as a diagnostic tool in lung cancer [12]. The futile thoracotomy rate has been reduced by approximately 50% amongst non-operable patients with NSCLC as a result of pre-operative evaluation with PET imaging [13]. The utilisation of PET in the staging of patients treated with RT or chemoradiotherapy improves their apparent survival [14], by excluding incurable patients. In one large prospective trial, almost a third of potential candidates for high-dose RT on the basis of conventional staging received only palliation after PET, because of unexpected distant metastasis (20%) or very extensive intrathoracic disease (10%) [15].
The question of the impact of PET in RT planning is relevant and interesting; there is no phase III study confirming that PET planning is superior to CT planning, and such a study is not possible due to issues around clinical equipoise. This study had both a pilot and a phase II component. With the pilot, we aimed to evaluate the technical feasibility of integrating positron emission tomography–computed tomography (PET-CT) fusion in our institution. The primary outcome of the phase II study was to evaluate the safety of PET-CT scan–based RT by evaluating the rate of loco-regional recurrence outside the PET-CT planning target volume (PTV) but within conventional 3-D PTV.
Aims/objectives
Pilot study
The primary objective of the pilot study was to evaluate the technical feasibility of integrating PET-CT fusion in our institution. The secondary objective was a comparison of GTV, PTV (planning target volume), and OAR (organ at risk) DVHs (dose-volume histograms) between conventional 3-DCRT plan and PET-CT-based 3-DCRT plan. A rate of successful fusion and PET-CT scan–based radiotherapy of ≥ 60% will be considered acceptable.
Phase II study
The primary outcome of the phase II study was to evaluate the safety of PET-CT scan–based RT by evaluating the rate of loco-regional recurrence outside the PET-CT PTV but within conventional 3-D PTV. The secondary outcome was the same as that of the pilot study.
Methods
Patients received a total dose of 60–66 Gy in 30–33 fractions with 3-DCRT. The median dose received for the 12 patients was 66 Gy. Two separate plans using the CT-GTV and the PET-CT-GTV were generated, and patients were treated using the PET-CT-GTV plan. The pilot study investigated the technological feasibility of this method. The Phase II study was a two-stage phase II study to evaluate its safety.
Inclusion criteria required trial participants to be 18 years or older and have histologically proven NSCLC, TNM clinical non-operable stage I/II and non-resectable stage IIIa/b without pleural effusion, Karnofsky Performance Status (KPS) > 60, and no other malignancy (except non-melanomatous skin cancer) within 5 years prior to participation. Informed consent must have been provided. Patients needed to be suitable for radical 3-DCRT, they must not have had prior RT to the thorax, weight loss must not have exceeded 10% within the 3 months prior to diagnosis, and they must not have received chemotherapy prior to a pre-chemotherapy diagnostic PET-CT. Pre-chemotherapy PET-CTs and planning PET-CTs were directly compared, and any alteration in signal established. Patients with any significant change in FDG uptake were deemed unsuitable for treatment on a PET-CT plan as such treatment could result in a reduced volume that would not be reflective of the actual disease. This could result in active disease being omitted from the RT treatment volume. Specified exclusion criteria included a forced expiratory volume (FEV1) < 1, evidence of any other significant clinical disorder, or laboratory finding that made it undesirable for them to participate in the trial or felt by the research/medical team likely to make them unable be able to comply with protocol.
The planning CT was done with the patient in the standard supine position with lung board and kneefix immobilisation. Reference set-up marks were tattooed at CT. The planning PET/CT was the same position, with patient aligned to tattoos using lasers. Safety checks were essential from a radiation protection perspective and were performed to ensure that the PET/CT scan could be acquired prior to injecting the patient. The possibility of the patient colliding with the gantry was checked and eliminated, and the patient’s ability to tolerate the enclosed scanner was confirmed. The patient was then injected with FDG. The PET/CT scan (the hot session) was acquired 1 h after the injection. The radiation therapists were responsible for establishing the patient into the correct treatment position and for the acquisition of the CT scan. The diagnostic radiographers were responsible for the PET capture. All contouring was done on Advantage Sim Workstation.
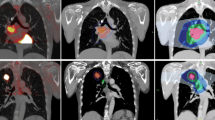
The GTV was delineated twice. Firstly, this was done with conventional planning CT scan alone (CT-GTV) without reference to the PET-CT. The GTV was restricted to the visible primary tumour defined on diagnostic CT scan and bronchoscopy, and to any mediastinal node enlarged (> 1 cm diameter) on diagnostic CT scan and/or proven histologically involved on mediastinoscopy. A second GTV (PET-GTV) was drawn using fused PET-CT and planning CT images. Similarly, GTV was defined as the visible primary tumour, defined on diagnostic CT scan and bronchoscopy which is visible on the fused PET-CT images, and nodes considered as involved on a standard PET-CT scan and/or proven histologically involved on mediastinoscopy. An uptake threshold of 35% was applied to avoid any possible exclusion of active disease.
The clinical target volume (CTV) was created by expanding the GTV by 0.6 cm and 0.8 cm isocentrically for squamous cell carcinomas and adenocarcinomas respectively. The CTV was then edited to take account of natural barriers to tumour spread like uninvolved bone or great vessels and organs like the heart and oesophagus.
The PTV was created by expanding the CTV by 1-cm margin isocentrically to take into account set-up error and respiratory motion. In the case of lower lobe tumours, 1.5-cm margins were used in a superior and inferior direction. Nodes were not irradiated electively.
Separate plans based on the CT and PET/CT volumes were generated. The minimum prescribed dose of radiation was 60 Gy at 2 Gy per fraction. Ninety-five percent of the dose covered 100% of the PTV for both CT and PET/CT plans. The patient was subsequently treated using the PET/CT plan.
Patients underwent standard post-treatment follow-up, including repeated three monthly CT scans of the thorax. In case of loco-regional recurrence, three categories were considered, with only the category 2 events considered as a failure:
-
1.
Intra-PET scan PTV recurrence
-
2.
Extra-PET scan PTV and intra-CT scan PTV recurrence
-
3.
Extra-PET scan PTV and extra-CT
Our hypothesis was that the rate of category 2 events would be < 10%.
Sample size and statistical analysis
Based on the advice from an external statistician, a sample size of 10 evaluable patients was considered sufficient to evaluate the technical feasibility of integrating PET-CT fusion into treatment planning in the pilot study.
In the phase II study, the primary objective was the rate of loco-regional recurrence outside the PET-CT PTV but within conventional 3-D PTV. Based on a recurrence rate of 10% in and out of field (suspected on CT but excluded on basis of PET), a total of 35 evaluable patients (to include the 10 from the pilot study) was required to provide 95% confidence that the true population proportion falls into the range from 0.06 to 19.94%. Evaluable patients were those who completed treatment and whose disease recurrence was confirmed by PET.
The dose delivery to the organ-at-risks was compared between the conventional 3-DCRT plan and the PET-CT-based 3-DCRT plan using the Wilcoxon signed rank test. All statistical tests were two-sided and assessed for significance at the 0.05 level. All available data were included in the analyses. Statistical analyses were carried out using IBM® SPSS® statistical software version 23.
Results
Pilot study
The primary endpoint of the pilot study was met in that it was feasible to deliver a PET-CT-based plan to ≥ 60% of patients (Fig. 1). The secondary endpoint of the pilot study was a comparison of GTV, PTV, and OAR DVHs between conventional 3-D plan and PET-CT-based 3D plans, and this was also achieved. It was feasible to analyse and to compare the differences in the DVCs to the OARs on the CT and PET-CT-based plans.
There were no serious adverse events (SAEs), and all toxicities and adverse events (AEs) were in keeping with those in common practice and reported literature.
Phase II study
Patient accrual and characteristics
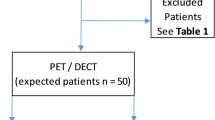
From August 2007 to February 2013, 49 eligible patients were enrolled. Three patients were found to be subsequently ineligible, 9 had disease progression, 1 patient died before they received RT, and 13 others did not receive RT for other reasons. Three patients died before the 3-month follow-up CT, 4 patients did not have their progression confirmed with a PET scan, and 4 others did not progress. Consequently, data from 12 patients were available for analysis of the primary endpoint. Figure 2 is a participant flow diagram that includes the numbers enrolled, received intended treatment, and analysed for the primary outcome.
Baseline demographic and clinical characteristics of the 12 evaluable patients are shown in Table 1. The vast majority of patients (67%) were stage IIIA patients. The mean age at enrolment was 67.5 years (range 47.4–85.0). Patients were followed until death or for a median of 20.9 months (range 6.3–69.3) from consent.
All 12 patients progressed, and all 12 died. The median overall survival was 20.4 months from the date of consent and 19.2 months from the start of RT. The median disease-free survival was 8.9 months from date of consent and was 7.4 months from the start of RT
Primary endpoint
The study closed early due to poor recruitment. The target for this study was 35 patients including those in the pilot study. The proportion of extra-PET scan PTV and intra-CT scan PTV recurrence was 0.8% and not 10% as hypothesised. Two patients had intra-PET scan PTV recurrences, six had extra-PET scan PTV and extra-CT, and three patients had both. Another patient had extra-PET scan PTV and extra-CT as well as extra-PET scan PTV and intra-CT scan PTV recurrence.
Secondary endpoints
GTV, PTV, and OAR DVHs
One of the 12 patients had a 2-phase plan, so for some variables data were analysed for 11 patients.
A Wilcoxon signed rank test revealed a statistically significantly higher maximum dose to the PTV and a higher oesophageal V45Gy with the conventional 3-DCRT plan compared with the PET-CT-based 3-DCRT plan. No other statistically significant differences in the PTV or the OAR DVHs were found between the conventional 3-DCRT plan and the PET-CT-based plan. The PET-CT and conventional 3-DCRT max dose to PTV and oesophageal V45Gy are shown in Table 2.
Discussion
Local failure continues to be a major cause of mortality in patients with inoperable NSCLC treated with RT. A systematic review by Kepka et al. [16] showed the failure of PET-CT to reduce elective nodal failures (defined as regional failures that occur without local recurrence irrespective of distant metastases status) in patients who underwent PET-CT for staging. However, many studies have shown elective nodal irradiation (ENI) to be ineffective and associated with increased toxicity and RT morbidity, which can limit the degree of dose escalation [16,17,18]. Due to the results of a phase I dose-escalation study in NSCLC [19], many experts terminated the inclusion of elective nodal regions in their treatment volumes to reduce toxicity [20]. The suggestion that ENI may not be necessary is strengthened by the acceptably low failure rate in uninvolved nodal regions that have resulted from the omission of ENI [20]. In a review of four Radiation Therapy Oncology Group trials (1705 NSCLC patients), Emami et al. [21] examined the effect of ENI and found that treatment of the ipsilateral hilar region was the only elective nodal region that influenced the outcome. Other trials have used ENI in the setting of early-stage disease with limited toxicity and had excellent clinical results [21, 22].
Absolute proof that PET-planning is superior to CT-planning would require randomised trials in which some patients were randomised to a less accurate (non-PET) staging workup, thereby presenting issues of clinical equipoise [23]. As such, there is no phase III trial evidence that supports the use of PET-CT in planning, although, PET-CT-based planning is recommended and is used by many. In 2006, an international panel reviewed the use of PET and PET-CT in this context. The International Atomic Energy Agency (IAEA) organised two synchronised and overlapping consultants’ meetings with international experts [23]. Nine experts and three IAEA staff evaluated the available data on the use of PET in RT planning and considered practical methods for making it a routine practice. They concluded that there is a strong case for the routine use of FDG-PET in RT planning for NSCLC. This was due to the remarkable accuracy of PET in staging and the demonstration of a powerful effect in all the published RT planning studies.
As mentioned, this study closed early due to poor recruitment despite our best efforts. Palliative studies, such as ours, can be notoriously difficult to recruit to, and this study represents an Irish real-world experience of this. Our study did, however, demonstrate the feasibility of delivering a PET-CT-based plan in an Irish centre. The secondary endpoint of the pilot study was a comparison of GTV, PTV, and OAR DVHs between conventional 3-D plan and PET-CT-based 3-D plans, and this was also achieved. It was feasible to analyse and to compare the differences in the DVCs to the OARs on the CT and PET-CT-based plans.
Staging PET scans should ideally be performed in the RT treatment position, facilitating dual use of imaging for staging and RT planning. Integrated PET/CT [24] is best, but PET/CT image co-registration, preferentially using fiducial markers, can be used [25]. In PET-CT fusion prior to RT, it is important that the fusion between the planning CT and the PET scan matches as closely as is possible to allow for the most accurate target volume delineation possible. Currently, the PET and planning CT scans have to be manually registered with a minimum of three common points in order to fuse images. Unfortunately, the PET scans are often performed with the patient in a different position to that of the planning CT scan, using a different tabletop and without immobilisation. Clearly, this results in sub-optimal co-registration of the two data sets, with poor anatomic correlation between the PET and CT scans.
The use of diagnostic PET-CT scans for RT treatment planning at first glance appears very attractive and has the potential to save on both time and resource. However, when diagnostic scans are used, disadvantages such as the influence of time delay between the diagnosis and initiation of RT on tumour volume, nodal extent [26], tracer uptake, and differences in positioning between data sets must be considered. Like other authors [27], we would recommend the use of dedicated PET-CT scans for both staging and RT planning in patients with NSCLC.
Despite its higher accuracy, the limitations of PET must be borne in mind. One study demonstrated the rate of false-negative lymph node station assessment (post-test probability) in NSCLC RT candidates to be 5–10% [28]. Some factors are particularly associated with false-negative findings: carcinoid tumours, low-grade adenocarcinomas including broncho-alveolar carcinomas, and very small lesions (especially in the context of raised blood sugar levels). Institutional standardisation is needed for image acquisition (breathing protocol, adjustment of dose calibrator, and many technical, physical, and biological factors influencing the SUV), data display and analysis, and the transfer of PET-CT data to the planning system [29,30,31]. PET acquisition takes several minutes, and this leads to tumour motion due to respiration or cardiac motion. This results in PET “GTVs” that incorporate the effects of this motion. As such, the breathing protocol used for PET image acquisition must match that of the CT used for RT planning [32]. Respiration-gated PET acquisition techniques have been developed [33] and are currently being evaluated in clinical trials. Delineation protocols influence the results very significantly and must be standardised [34].
Loco-regional failure remains a major challenge in treating NSCLC with RT. Dose escalation has been shown to reduce local failure rates but, in the setting of ENI, is often limited by pulmonary toxicity. Improvement of patient outcomes will come both from an improvement of treatment modalities and improvements in patient selection. The inadequacy of conventional standard RT to achieve local control has been clearly demonstrated. While dose escalation has the potential to improve local control, it can be achieved only by a significant reduction of the irradiated volume, by using conformal RT and optimising the tumour target volume. PET-based planning has the potential to reduce radiation treatment volumes because of the avoidance of mediastinal lymph nodes that are PET negative.
A small sample size, as mentioned, is a limitation of this study. Additionally, 3-D radiotherapy was used, rather than the more modern IMRT.
Conclusions
In patients with NSCLC, FDG-PET scans allow more accurate staging than conventional CT scans. This can avoid unnecessary, aggressive, and toxic treatments in patients who would not stand to benefit from their use. PET-based planning has the potential to reduce RT volumes due to the avoidance of mediastinal lymph nodes that are PET negative. This in turn has the potential to reduce toxicity associated with a particular radiation dose, or to enable radiation dose escalation with the same toxicity. The use of planning PET/CT in the RT planning of patients with NSCLC has a crucial impact on the precise determination of target volumes allowing for enhanced precision in the staging of disease, and thus also enables changes in treatment strategy.
References
Levi F, Lucchini F, La Vecchia C, Negri E (1999) Trends in mortality from cancer in the European Union, 1955-94. Lancet (London, England). 354(9180):742–743
Surveillance, Epidemiology and End Results (SEER) database, Feb 2014. Available from: https://seer.cancer.gov/data/
Dogan N, Leybovich LB, Sethi A, Emami B (2002) Improvement of dose distributions in abutment regions of intensity modulated radiation therapy and electron fields. Med Phys 29(1):38–44
Zelefsky MJ, Leibel SA, Kutcher GJ, Fuks Z (1998) Three-dimensional conformal radiotherapy and dose escalation: where do we stand? Semin Radiat Oncol 8(2):107–114
Hawkins DS, Conrad EU 3rd, Butrynski JE, Schuetze SM, Eary JF (2009) [F-18]-fluorodeoxy-D-glucose-positron emission tomography response is associated with outcome for extremity osteosarcoma in children and young adults. Cancer. 115(15):3519–3525
Ling CC, Humm J, Larson S, Amols H, Fuks Z, Leibel S, Koutcher JA (2000) Towards multidimensional radiotherapy (MD-CRT): biological imaging and biological conformality. Int J Radiat Oncol Biol Phys 47(3):551–560
Dwamena BA, Sonnad SS, Angobaldo JO, Wahl RL (1999) Metastases from non-small cell lung cancer: mediastinal staging in the 1990s--meta-analytic comparison of PET and CT. Radiology. 213(2):530–536
Gould MK, Maclean CC, Kuschner WG, Rydzak CE, Owens DK (2001) Accuracy of positron emission tomography for diagnosis of pulmonary nodules and mass lesions: a meta-analysis. Jama. 285(7):914–924
Cerfolio RJ, Ojha B, Mukherjee S, Pask AH, Bass CS, Katholi CR (2003) Positron emission tomography scanning with 2-fluoro-2-deoxy-d-glucose as a predictor of response of neoadjuvant treatment for non-small cell carcinoma. J Thorac Cardiovasc Surg 125(4):938–944
Hicks RJ, Mac Manus MP, Matthews JP, Hogg A, Binns D, Rischin D, Ball DL, Peters LJ (2004) Early FDG-PET imaging after radical radiotherapy for non-small-cell lung cancer: inflammatory changes in normal tissues correlate with tumor response and do not confound therapeutic response evaluation. Int J Radiat Oncol Biol Phys 60(2):412–418
Mac Manus MP, Hicks RJ, Matthews JP, McKenzie A, Rischin D, Salminen EK, Ball DL (2003) Positron emission tomography is superior to computed tomography scanning for response-assessment after radical radiotherapy or chemoradiotherapy in patients with non-small-cell lung cancer. J Clin Oncol 21(7):1285–1292
Lardinois D, Weder W, Hany TF, Kamel EM, Korom S, Seifert B, von Schulthess GK, Steinert HC (2003) Staging of non-small-cell lung cancer with integrated positron-emission tomography and computed tomography. N Engl J Med 348(25):2500–2507
van Tinteren H, Hoekstra OS, Smit EF, van den Bergh JH, Schreurs AJ, Stallaert RA et al (2002) Effectiveness of positron emission tomography in the preoperative assessment of patients with suspected non-small-cell lung cancer: the PLUS multicentre randomised trial. Lancet (London, England) 359(9315):1388–1393
Mac Manus MP, Wong K, Hicks RJ, Matthews JP, Wirth A, Ball DL (2002) Early mortality after radical radiotherapy for non-small-cell lung cancer: comparison of PET-staged and conventionally staged cohorts treated at a large tertiary referral center. Int J Radiat Oncol Biol Phys 52(2):351–361
Mac Manus MP, Hicks RJ, Ball DL, Kalff V, Matthews JP, Salminen E, Khaw P, Wirth A, Rischin D, McKenzie A (2001) F-18 fluorodeoxyglucose positron emission tomography staging in radical radiotherapy candidates with nonsmall cell lung carcinoma: powerful correlation with survival and high impact on treatment. Cancer. 92(4):886–895
Kepka L, Socha J (2015) PET-CT use and the occurrence of elective nodal failure in involved field radiotherapy for non-small cell lung cancer: a systematic review. Radiother Oncol 115(2):151–156
Pu AT, Harrison AS, Robertson JM, Hayman JA, Martel MK et al (1997) The toxicity of elective nodal irradiation in the definitive treatment of non-small cell carcinoma of the lung. Int J Radiat Oncol Biol Phys 39(abst 2):196
Marks LB, Prosnitz LR (1999) Assessing the impact of elective regional radiotherapy on survival. Cancer J Sci Am 5(2):92–100
Rosenzweig KE, Fox JL, Yorke E, Amols H, Jackson A, Rusch V, Kris MG, Ling CC, Leibel SA (2005) Results of a phase I dose-escalation study using three-dimensional conformal radiotherapy in the treatment of inoperable nonsmall cell lung carcinoma. Cancer. 103(10):2118–2127
Rosenzweig KE, Sura S, Jackson A, Yorke E (2007) Involved-field radiation therapy for inoperable non small-cell lung cancer. J Clin Oncol 25(35):5557–5561
Emami B, Mirkovic N, Scott C, Byhardt R, Graham MV, James Andras E, John M, Herskovic A, Urtasun R, Asbell SO, Perez CA, Cox J (2003) The impact of regional nodal radiotherapy (dose/volume) on regional progression and survival in unresectable non-small cell lung cancer: an analysis of RTOG data. Lung Cancer (Amsterdam, Netherlands) 41(2):207–214
Jeremic B, Shibamoto Y, Acimovic L, Milisavljevic S (1997) Hyperfractionated radiotherapy alone for clinical stage I nonsmall cell lung cancer. Int J Radiat Oncol Biol Phys 38(3):521–525
MacManus M, Nestle U, Rosenzweig KE, Carrio I, Messa C, Belohlavek O, Danna M, Inoue T, Deniaud-Alexandre E, Schipani S, Watanabe N, Dondi M, Jeremic B (2009) Use of PET and PET/CT for radiation therapy planning: IAEA expert report 2006-2007. Radiother Oncol 91(1):85–94
Messa C, Di Muzio N, Picchio M, Gilardi MC, Bettinardi V, Fazio F (2006) PET/CT and radiotherapy. Q J Nucl Med Mol Imaging 50(1):4–14
Deniaud-Alexandre E, Touboul E, Lerouge D, Grahek D, Foulquier JN, Petegnief Y, Grès B, el Balaa H, Keraudy K, Kerrou K, Montravers F, Milleron B, Lebeau B, Talbot JN (2005) Impact of computed tomography and 18F-deoxyglucose coincidence detection emission tomography image fusion for optimization of conformal radiotherapy in non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 63(5):1432–1441
Everitt S, Herschtal A, Callahan J, Plumridge N, Ball D, Kron T, Schneider-Kolsky M, Binns D, Hicks RJ, MacManus M (2010) High rates of tumor growth and disease progression detected on serial pretreatment fluorodeoxyglucose-positron emission tomography/computed tomography scans in radical radiotherapy candidates with nonsmall cell lung cancer. Cancer. 116(21):5030–5037
De Ruysscher D, Nestle U, Jeraj R, Macmanus M (2012) PET scans in radiotherapy planning of lung cancer. Lung Cancer (Amsterdam, Netherlands). 75(2):141–145
Graeter TP, Hellwig D, Hoffmann K, Ukena D, Kirsch CM, Schafers HJ (2003) Mediastinal lymph node staging in suspected lung cancer: comparison of positron emission tomography with F-18-fluorodeoxyglucose and mediastinoscopy. Ann Thorac Surg 75(1):231–235 discussion 5-6
Hellwig D, Ukena D, Paulsen F, Bamberg M, Kirsch CM (2001) Meta-analysis of the efficacy of positron emission tomography with F-18-fluorodeoxyglucose in lung tumors. Basis for discussion of the German Consensus Conference on PET in Oncology 2000. Pneumologie (Stuttgart, Germany) 55(8):367–377
Sattler B, Lee JA, Lonsdale M, Coche E (2010) PET/CT (and CT) instrumentation, image reconstruction and data transfer for radiotherapy planning. Radiother Oncol 96(3):288–297
Weber WA, Dietlein M, Hellwig D, Kirsch CM, Schicha H, Schwaiger M (2003) PET with (18)F-fluorodeoxyglucose for staging of non-small cell lung cancer. Nuklearmedizin 42(4):135–144
Grgic A, Nestle U, Schaefer-Schuler A, Kremp S, Kirsch CM, Hellwig D (2009) FDG-PET-based radiotherapy planning in lung cancer: optimum breathing protocol and patient positioning--an intraindividual comparison. Int J Radiat Oncol Biol Phys 73(1):103–111
Nehmeh SA, Erdi YE, Rosenzweig KE, Schoder H, Larson SM, Squire OD et al (2003) Reduction of respiratory motion artifacts in PET imaging of lung cancer by respiratory correlated dynamic PET: methodology and comparison with respiratory gated PET. J Nucl Med 44(10):1644–1648
Nestle U, Kremp S, Schaefer-Schuler A, Sebastian-Welsch C, Hellwig D, Rube C et al (2005) Comparison of different methods for delineation of 18F-FDG PET-positive tissue for target volume definition in radiotherapy of patients with non-small cell lung cancer. J Nucl Med 46(8):1342–1348
Funding
The Health Research Board
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, K.A., Rangaswamy, G., Lavan, N.A. et al. ICORG 06-35: a prospective evaluation of PET-CT scan in patients with non-operable or non-resectable non-small cell lung cancer treated by radical 3-dimensional conformal radiation therapy: a phase II study. Ir J Med Sci 188, 1155–1161 (2019). https://doi.org/10.1007/s11845-019-02019-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-019-02019-2