Abstract
Background
Anaemia among haemodialysis patients is treated with iron and erythropoietin-stimulating agents (ESAs). ESAs reduce requirements for blood transfusions but are also expensive and overzealous use may be associated with adverse outcomes. Recent international trends have been characterised by reduced ESA doses and a greater reliance on intravenous (IV) iron. We determined trends in prescribing patterns of ESAs and IV iron for the treatment of anaemia in two representative Irish dialysis centres and correlated with current guidelines and international trends.
Methods
Patient data was accessed from the Kidney Disease Clinical Patient Management System (KDCPMS) for the period 2012 to 2014. We generated reports on ESA and iron doses, lab data (haemoglobin (Hb), transferrin saturation (TSAT) and ferritin) and patient population characteristics. We mapped the trends in ESA, iron dosing and lab parameters achieved. A linear mixed model determined the significance of these trends over time.
Results
ESA dosing became lower in the second, third and fourth quarters of 2014. Dosing of iron increased throughout but a large increase was seen in the third and fourth quarters of 2014. Ferritin levels decreased and TSAT and haemoglobin levels increased. Changes in iron dosing were significant with p value of < 0.05.
Conclusions
Our findings are consistent with recent global trends toward increasing iron use. Such trends may have economic implications given the high cost of ESAs and the relative affordability of iron. In addition, the potential harm of excessive iron dosing may need to be considered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The majority of chronic kidney disease (CKD) patients on dialysis suffer from anaemia, and anaemia contributes greatly to symptoms including fatigue, dyspnoea, reduced exercise tolerance and depression [1]. End stage kidney disease (ESKD)-associated anaemia is treated with a combination of erythropoiesis-stimulating agents (ESAs) and iron (occasionally oral but usually intravenous (IV)). Prescribing patterns for CKD-associated anaemia have changed over the years as guidelines have evolved, and continue to change, in the wake of notable clinical trials [2,3,4,5].
Following the approval of recombinant erythropoietin (EPO) by the food and drug administration (FDA) in 1989 (followed by other ESAs), these agents became the treatment of choice for anaemia of ESKD. Epoetin alfa and epoetin beta were the first ESAs available. Biosimilar drugs such as epoetin theta subsequently appeared on the market, as did ESAs with a longer half-life, such as darbepoetin alfa and methoxy polyethylene glycol-epoetin beta. These longer-acting agents may be more convenient than their predecessors but carry a significant expense [6].
ESA use has been associated with a reduction in the need for blood transfusions [3]. However, recent studies [4, 5] revealed that higher haemoglobin (Hb) targets and consequently higher doses of ESAs resulted in increased adverse outcomes including stroke and myocardial infarction. Following such trials, the FDA released a warning regarding high Hb targets for patients on ESAs. This has led to a trend of reducing ESA doses with a greater reliance on IV iron as an alternative.
Administration of IV iron is recommended for all CKD patients on dialysis who have an absolute deficiency of iron (transferrin saturation (TSAT) ≤ 30% and ferritin ≤ 500 ng/mL) [7]. In most cases, approximately 1000 mg is required to raise Hb from approximately 8 to 11–12 g/dL [8]. Once target Hb levels are achieved, approximately 250–500 mg of iron may be required every 3 months to maintain adequate iron stores. Kidney Disease: Improving Global Outcomes (KDIGO) recommends IV iron over oral iron in dialysis patients [7]. Treatment with IV iron is substantially less expensive when compared with ESAs but higher doses have also been linked with adverse events [9]. There is a suggestion of increased mortality that is not supported by all trials. One study suggested an increased rate of infection-related mortality with high IV iron dosing but these results were not statistically significant [10]. Thus, the safety profile of iron is far from clear at this time.
Guidelines and prescribing patterns for these agents have varied widely between countries. In the past, ESA doses in the United States (US) were noted to be significantly higher than those in Europe [7, 11]. Because of the significant cost involved, the choice of treatment has financial implications for the health system providing care.
Methods
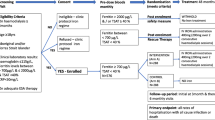
The time period studied was inclusive from January 2012 to December 2014. This period of 3 years was divided into 12 quarters, each of 3 months and these were used as the time points throughout.
Data source
All CKD patients in the Saolta group have all of their clinical data recorded on the Kidney Disease Clinical Patient Management System (KDCPMS). This system functions as a national electronic health record for CKD patients. Galway University Hospital and its affiliated dialysis units were selected as the initial pilot group for this system, and data has been captured on the system since 2012. Missing data from our study is low. In no instances was a Hb value not available for a patient in a given quarter. TSAT values were missing in < 2% of cases. Ferritin values were missing in < 3% of cases. The system keeps a detailed record of all dialysis treatments and drugs that were prescribed during dialysis. All lab parameters are entered into the system automatically from the patient administration system (PAS) via a dynamic interface that provides seamless access to patient data in the form of an electronic patient record. We used the KDCPMS to generate quarterly reports for the 3 years in question.
Patient population
In this study, we focused on two dialysis centres in the west of Ireland: Merlin Park Dialysis Unit and Mayo General Dialysis Unit. All patients included in the study were on chronic dialysis. In most cases, chronic dialysis is defined as dialysis for a minimum of 3 months. For this study, we included any patient who had had a minimum of 20 dialysis sessions in the 3 months prior to the quarter in question. On average, 105.6 patients per quarter met the inclusion criteria. The same patients were included in multiple quarters if they continued to meet the criteria. Information on age, sex, ethnicity, cause of CKD, previous transplantation and history of diabetes was gathered. A summary of patient population demographics by quarter is shown in Table 1. In total, data was collected on 215 different patients over the 3-year period. The prevalence of diabetes was 30.2%. Renal transplantation was carried out for 24.2% of patients. A summary of the cause of CKD, according to ICD subtypes, is shown in Table 2.
Measured parameters
Reports generated from the KDCPMS include information on ESA and iron doses prescribed and Hb, TSAT and ferritin levels achieved during each period.
The ESAs prescribed to the patients in this study were darbepoetin alfa (Aranesp), epoetin theta (Eporatio), epoetin beta (Neorecormon), epoetin zeta (Retacrit) and methoxy polyethylene glycol-epoetin beta (Mircera). Darbopoetin dosing is in micrograms whereas the dosing for all other ESAs mentioned is in units. To adjust for this discrepancy, we converted the ESA doses of all other agents to a darbopoetin equivalent dose in micrograms using a conversion factor of 200:1. This conversion factor has been used in the majority of recent publications in the field and thus should serve well when comparing similar studies [12]. However, it must be noted that this conversion is not ideal as ESA dosing is not normally distributed. Iron was administered intravenously in the form of iron sucrose.
Hb, TSAT and ferritin levels are surrogate markers of anaemia and iron storage and have typically been used as markers of treatment efficacy.
Statistical analysis
To plot the trends on a graph, we calculated average figures for each quarter. To calculate the average dose of ESA and iron for the patient group, we selected the dose prescribed for each patient as of the first day of each month. We then calculated the average dose for the patient group. We repeated the same for months 2 and 3 before calculating the average of these three figures for the quarterly average. A similar procedure was followed to calculate the average Hb, TSAT and ferritin values. The results were plotted on line graphs to display the trends.
To assess if there was a significant difference in the weekly dose of ESA over time, we ran a linear mixed model. As the residuals are not normally distributed, we ran the model with the log transformation on the response variable (i.e. weekly dose), which satisfies the model assumption. A similar analysis was used for analysis of iron dosing. To see where the differences exist, we performed a post hoc analysis.
Results
ESA dosing is represented in Fig. 1. The result of the model showed that there is no significant difference in the weekly dose of EPO over time (p value was found to be 0.1473). Although a decrease can be observed in the second, third and fourth quarters of 2014, the overall decrease spanning the 3-year period was not significant.
The trend in IV iron dosing is represented in Fig. 2. Dosing of IV iron increased throughout the period but a large increase was seen in the third and fourth quarters of 2014. There was a significant difference in the monthly dose of iron over time (p value was found to be < 0.05).
The trends in the markers of treatment efficacy are represented in Figs. 3, 4 and 5. A rise in Hb levels, a decreasing trend of ferritin and a rise in TSAT can be observed.
Discussion
In two Irish dialysis centres, there was a trend toward decreasing ESA use and increasing IV iron use for the treatment of ESKD-associated anaemia from January 2012 to December 2014. At the time, there was no specific local guideline on ESA/iron dosing or Hb/ferritin/TSAT targets but the practice in these units was to follow the KDIGO and National Institute of Clinical Excellence (NICE in the UK) guidelines with an associated awareness of European Best Practice (EBPG) [13].
In particular, ESA dosing decreased in 2014. In the second half of 2014, we saw a significant upward trend in IV iron dosing. There were no specific changes in local policy at this time but it is likely that there was increased awareness among staff of recently published data. The timing of these changes is consistent with changes in practice around the world.
In general, there is a policy among staff of avoiding significant iron dosing in patients with high ferritin levels. This may have minimised the expected rise in ferritin with the significant increase in IV iron dosing that was observed in the third and fourth quarters of 2014.
Anaemia of CKD is associated with significant morbidity and mortality [14] and can have a significant impact on quality of life. Currently available treatment options for ESKD patients include IV iron, oral iron and ESAs. Blood transfusions and androgens are now rarely used.
In 2014, 70% of patients in the US on dialysis were treated with IV iron each month [15]. In our study, 33.9% of the patient group were receiving IV iron at the beginning of 2012. By the end of 2014, this number had increased to 41.1%. In January 2012, 86.8% of patients were receiving ESA treatment. This figure had decreased to 82.7% by December 2014.
Target Hb levels remain controversial and recommendations and practice patterns differ across jurisdictions [7, 8, 13, 16,17,18]. Continuous administration of iron without a target in mind is not an ideal strategy as use of IV iron may have potential risks. Excess iron has been shown to cause oxidative stress and may result in increased cellular apoptosis in dialysis patients. Overtreatment with IV iron may increase the risk of cardiovascular events and infection-related mortality. A higher mortality rate has been demonstrated with increased IV iron dose in large analyses of dialysis patient data [9].
A number of large ESA studies have demonstrated a significant risk associated with overzealous use of ESAs [4, 5].
Guidance on the treatment of ESKD-associated anaemia varies from region to region. Disparities exist regarding when to initiate iron and ESA treatments as well as target Hb levels and maximum desired ferritin levels [19]. Thus, treatment strategies differ accordingly. The KDIGO international guidelines recommend that a 1–3-month trial of IV iron should be administered if TSAT is < 30% and ferritin is < 500 ng/ml. This can be used to avoid ESA therapy altogether or when aiming to decrease the ESA dose [7]. NICE specifies that most adult patients on dialysis will require 600–1000 mg of iron in a single or divided dose [18].
With more evidence available, guidelines on ESA use are generally more consistent in their treatment recommendations. In all cases, balancing potential risks with benefits of treatment is advised.
KDIGO recommends that iron stores must first be replenished before starting on an ESA, to gain maximum benefit from ESA therapy [19].
KDIGO suggests initiating treatment with an ESA when Hb falls to between 9 and 10 g/dl. Individualisation of therapy is stressed with dosing determined on Hb concentration, body weight and clinical circumstances. KDIGO currently recommends a Hb target of ≤ 11.5 g/dl [7].
NICE guidelines similarly recommend replenishing iron stores prior to commencing ESA therapy. A target of 10–12 g/dl, with a rate of increase between 10 and 20 g/l per month is advised [18]. The majority of patients throughout our study had Hb values within this range, as demonstrated in Fig. 6.
More caution needs to be taken in high-risk cases (those with cancer, diabetes and cardiovascular comorbidities) [20].
Trends in the treatment of CKD-associated anaemia have evolved following clinical trials and changes in reimbursement and regulatory policies in different countries [21]. When ESAs first came on the market (in the 1980s), there was a reduction in the use of IV iron in the management of anaemia. ESA use increases Hb but causes a shift of iron from storage tissues, and so iron deficiency became an issue [15]. This led to an increase in the use of IV iron to replenish iron stores. In 2011, a change in reimbursement for IV drugs used in the treatment of ESKD in the US may have resulted in a further increase in iron use [15].
For years, ESA use in the US was much higher than that in Europe or Asia [21]. From 2010 to 2013, both ESA doses and Hb levels decreased dramatically in the US. Fuller et al. attribute these observations to changes in national reimbursement policy and regulatory guidance [21]. In 2014, Thamer et al. reported that use of ESAs in the US following the TREAT study [5] declined significantly [22]. ESA use in stage 3 and 4 CKD patients decreased by 38 and 22% respectively.
Elsewhere in the world, patterns vary. A 2016 Swedish study showed that mean ESA dose decreased significantly in dialysis patients, by 10% over the time period studied. Mean Hb levels decreased slightly and iron use increased [23].
These patterns in ESA and iron use are similar to our results in the Irish population. The data we collected shows a decreasing trend in ESA prescribing in our units. This trend was not statistically significant for the 3-year period but a decrease can be observed in the year 2014. We did not see a major change in Hb levels, which may be explained by the shorter time period studied but there was a marked drop in ESA use at the end of our observation period.
Selecting an ideal treatment regimen for ESKD patients is highly complex. It involves balancing of risks and benefits. Although effective at increasing Hb levels (and reducing the need for blood transfusions), there are certainly adverse effects associated with ESA use. Increasing IV iron treatment results in lower ESA requirements (and additionally lower costs) but higher doses of iron may present its own problems.
Looking to the future, a greater consensus on treatment strategies for ESKD-associated anaemia is needed. The CKD Outcomes and Practice Patterns Study team is working to develop an evidence base on treatment strategies for advanced CKD. The goal of this team is to improve outcomes for CKD patients by exploring trends and comparisons in practices between different countries [20].
New markers of iron deficiency are required to better inform treatment strategies. Reticulocyte Hb has been shown to be a more sensitive marker of iron deficiency than both TSAT and ferritin [24]. Newer treatment options currently undergoing clinical trials include hypoxia-inducible factor (HIF) stabilisers and hepcidin (a regulator of iron metabolism). These drugs may complement or offer an alternative to existing therapies.
Our study has some limitations. In the first instance, we only included patients from two dialysis centres in the west of Ireland. Our results may not be representative of the entire national dialysis population. The sample size of 215 different patients over the 3-year period is also a limiting factor, as is the relatively short duration of the study. Missing data from the KDCPMS must also be taken into account when interpreting the results. If no ESA/iron dose was prescribed on the system, it was assumed that no dose was administered during that period. Ferritin and TSAT values were measured less consistently than Hb. We have not collected data on rates of blood transfusions and this can influence TSAT, ferritin and Hb values. In addition, ferritin is an acute phase reactant and thus may be elevated in patients who had an infectious/inflammatory illness at the time in question.
In our study of Irish dialysis patients, we observed a decreasing trend in ESA dosing and an increase in IV iron use. These findings were consistent with recent international trends in prescribing patterns. The patterns we observed may have economic implications given the high cost of ESAs and the relative affordability of iron.
Increasing iron doses result in decreased ESA administration and thus potentially less adverse effects attributable to ESAs. However, care must be taken when administering increasing doses of iron as this could also be linked to potential harm.
References
Revicki DA, Brown RE, Feeny DH, Henry D, Teehan BP, Rudnick MR, Benz RL (1995) Health-related quality of life associated with recombinant human erythropoietin therapy for predialysis chronic renal disease patients. Am J Kidney Dis 25(4):548–554
Besarab A, Bolton WK, Browne JK, Egrie JC, Nissenson AR, Okamoto DM, Schwab SJ, Goodkin DA (1998) The effects of normal as compared with low hematocrit values in patients with cardiac disease who are receiving hemodialysis and epoetin. N Engl J Med 339(9):584–590
Drüeke TB, Locatelli F, Clyne N, Eckardt KU, Macdougall IC, Tsakiris D, Burger HU, Scherhag A, CREATE Investigators (2006) Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med 355(20):2071–2084
Singh AK, Szczech L, Tang KL, Barnhart H, Sapp S, Wolfson M, Reddan D, CHOIR Investigators (2006) Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med 355(20):2085–2098
Pfeffer MA, Burdmann EA, Chen C-Y, Cooper ME, de Zeeuw D, Eckardt KU, Feyzi JM, Ivanovich P, Kewalramani R, Levey AS, Lewis EF, McGill J, McMurray J, Parfrey P, Parving HH, Remuzzi G, Singh AK, Solomon SD, Toto R, TREAT Investigators (2009) A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med 361(21):2019–2032
Del Vecchio L, Locatelli F (2014) New treatment approaches in chronic kidney disease-associated anaemia. Expert Opin Biol Ther 14(5):687–696
McMurray JJV, Parfrey PS, Adamson JW et al (2012) Kidney Disease: Improving Global Outcomes (KDIGO) anemia work group. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl 2(4):279–335
Madore F, White CT, Foley RN, Barrett BJ, Moist LM, Klarenbach SW, Culleton BF, Tonelli M, Manns BJ (2008) Clinical practice guidelines for assessment and management of iron deficiency. Kidney Int 74:S7–S11
Bailie GR, Larkina M, Goodkin DA, Li Y, Pisoni RL, Bieber B, Mason N, Tong L, Locatelli F, Marshall MR, Inaba M, Robinson BM (2015) Data from the Dialysis Outcomes and Practice Patterns Study validate an association between high intravenous iron doses and mortality. Kidney Int 87(1):162–168
Miskulin DC, Tangri N, Bandeen-Roche K, Zhou J, McDermott A, Meyer KB, Ephraim PL, Michels WM, Jaar BG, Crews DC, Scialla JJ, Sozio SM, Shafi T, Wu AW, Cook C, Boulware LE, for The Developing Evidence to Inform Decisions about Effectiveness (DEcIDE) Network Patient Outcomes in End Stage Renal Disease Study Investigators (2014) Intravenous iron exposure and mortality in patients on hemodialysis. Clin J Am Soc Nephrol 9:1930–1939. https://doi.org/10.2215/CJN.03370414
Locatelli F, Aljama P, Bárány P et al (2004) Revised European best practice guidelines for the management of anaemia in patients with chronic renal failure. Nephrol Dial Transplant 19(Suppl 2)
Bonafont X, Bock A, Carter D, et al (2009) A meta-analysis of the relative doses of erythropoiesis-stimulating agents in patients undergoing dialysis. Oxford University Press
Locatelli F, Aljama P, Bárány P et al (2004) Revised European best practice guidelines for the management of anaemia in patients with chronic renal failure. Nephrol Dial Transplant 19:ii1–i47
Schmidt RJ, Dalton CL (2007) Treating anemia of chronic kidney disease in the primary care setting: cardiovascular outcomes and management recommendations. Osteopath Med Prim Care 1(1):14
Fishbane S, Mathew AT, Wanchoo R (2014) Intravenous iron exposure and outcomes in patients on hemodialysis. Clin J Am Soc Nephrol 9(11):1837–1839
National KF (2006) KDOQI clinical practice guidelines and clinical practice recommendations for anemia in chronic kidney disease. Am J Kidney Dis 47(5 Suppl 3):S11
Moist LM, Foley RN, Barrett BJ, Madore F, White CT, Klarenbach SW, Culleton BF, Tonelli M, Manns BJ (2008) Clinical practice guidelines for evidence-based use of erythropoietic-stimulating agents. Kidney Int 74:S12–SS8
Excellence NIfHaC (2015) Chronic kidney disease: managing anaemia. [Available from: https://www.nice.org.uk/guidance/ng8
Locatelli F, Mazzaferro S, Yee J (2016) Iron therapy challenges for the treatment of nondialysis CKD patients. Clin J Am Soc Nephrol 11(7):1269–1280
Del Vecchio L, Locatelli F (2016) An overview on safety issues related to erythropoiesis-stimulating agents for the treatment of anaemia in patients with chronic kidney disease. Expert Opin Drug Saf 15(8):1021–1030
Fuller DS, Bieber BA, Pisoni RL, et al (2015) International comparisons to assess effects of payment and regulatory changes in the United States on anemia practice in patients on hemodialysis: the dialysis outcomes and practice patterns study. J Am Soc Nephrol. ASN. 2015060673
Thamer M, Zhang Y, Kshirsagar O, Cotter DJ, Kaufman JS (2014) Erythropoiesis-stimulating agent use among non–dialysis-dependent CKD patients before and after the Trial to Reduce Cardiovascular Events With Aranesp Therapy (TREAT) using a large US health plan database. Am J Kidney Dis 64(5):706–713
Evans M, Suttorp MM, Bellocco R et al (2015) Trends in haemoglobin, erythropoietin-stimulating agents and iron use in Swedish chronic kidney disease patients between 2008 and 2013. Nephrol Dial Transplant 31(4):628–635
Fishbane S, Shapiro W, Dutka P, Valenzuela OF, Faubert J (2001) A randomized trial of iron deficiency testing strategies in hemodialysis patients1. Kidney Int 60(6):2406–2411
Funding
This study was funded by the Health Research Board (grant number SS-2015-1431).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Roisin Gardiner has received a research grant from the Health Research Board.
Donal Reddan is a consultant with Akebia and is on the data safety board of phase 3 anaemia studies.
Davood Roshan declares that he has no conflict of interest.
Denise Connolly declares that she has no conflict of interest.
Anne Brennan declares that she has no conflict of interest.
Susan Murray declares that she has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Gardiner, R., Roshan, D., Brennan, A. et al. Trends in the treatment of chronic kidney disease-associated anaemia in a cohort of haemodialysis patients: the Irish experience. Ir J Med Sci 188, 223–230 (2019). https://doi.org/10.1007/s11845-018-1823-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-018-1823-0