Abstract
Background
Previously, no large-scale literature reviews have focussed on the relationship between dietary protein and its impact on bone mineral density (BMD) and fracture risk—as measures of bone health—in older adults and its potential impact as a primary prevention tool.
Aims
The aim of this study was to assess the impact of varying dietary protein levels on bone health.
Methods
A literature review of trials concerning older adults’ (>50 years of age) and animals’ varying protein intake in the diet and its effect on BMD (human and animal) and fracture risk (human only) was carried out. Additionally, a review of dietary assessment tools used in these studies was also analysed.
Results
Ten out of fourteen trials assessing BMD and dietary protein quantity in humans and 3/4 in animal trials found a positive relationship between these two parameters. Four out of seven trials investigating the relationship between dietary protein quantity and fracture risk displayed a positive, protective effect of dietary protein levels on fracture risk. Sixty-two percent of studies used the Food-Frequency Questionnaire assessment method.
Discussion
Increased protein intake in the diet is beneficial to bone health and reduces morbidity and mortality. The importance of using dietary protein, along with calcium and vitamin D, as a primary preventative strategy should be stressed, given the health and cost benefits that this would deliver, with a possible need for a higher level of protein in the diet of an elderly person than what is currently recommended.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a systemic skeletal system disorder, characterised by diminishment of both bone architecture and bone mass, precipitating in an increase in bone fragility, risk of fracture [1, 2] and increased hospital admission and co-morbidities [3]. There is a significant social cost associated with hip fractures in Ireland, with the total in-patient cost alone being €58 million per annum for those over 65 years, which is set to increase [4]. Bone mineral density (BMD) is an indirect measurement tool for diagnosing osteoporosis, which is itself routinely measured using dual-energy x-ray absorptiometry (DXA), with osteoporosis defined as a T-score ≤ −2.5 (2 · 5 standard deviations below the young adult’s average) [5]. The fracture-risk assessment tool (FRAX) is used to judge an individual’s 10-year risk of developing a hip fracture and/or major osteoporotic fracture [6].
Weight bearing exercise, genetic factors, tobacco smoking, alcohol intake, age, hormone levels, gender, race, and nutrition all contribute to bone health [7,8,9,10,11,12,13]. Nutrients, including micronutrients—calcium, vitamin D, phosphorous and magnesium—are key components in the process of bone formation. However, macronutrients such as protein are an often neglected bone element and contributor to bone health through muscle mass and falls prevention, and the quantifying of its consumption in the population is important to physicians when considering treatment bone-related diseases [13,14,15,16,17,18,19,20]. Furthermore, dietary protein may also influence bone mineral content and density through its contribution to greater muscle contractions [21,22,23,24].
Protein accounts for approximately 30% of the mass of bone tissue and 50% of its volume [25]. The RDA of dietary protein is 0.6–0.8 g/kg per day for adult males and females, although studies have suggested that this figure could be increased to 1.0 g/kg or higher, especially for elderly populations [26, 27]. Certain mechanisms have been postulated while others have been established in relation to the association of protein with calcium absorption and urinary secretion. Protein has been observed to increase insulin-like growth factor (IGF-1) hormone production and secretion [13]. IGF-1 contributes variously to bone growth, including triggering chondrocyte proliferation and differentiation in the epiphyseal growth plate, involvement in osteoblast proliferation, differentiation and mineralization, trabecular and cortical bone formation and collagen synthesis [28,29,30]. It has also been observed that a decreased concentration of IGF-1, which can result from reduced dietary protein intakes [31], can indirectly lead to a decrease in calcium and phosphorous absorption in the intestine [32].
Dietary protein intake and its influence on the body’s calcium balance, and therefore bone growth, are complex. An increase in protein consumption has been associated with an increase in urinary calcium concentrations [33, 34]. It has been suggested that during the oxidation of sulphur-containing amino acids, protons are released, which increase renal acid excretion and acid production [35]. This translates to the oxidation process contributing to the body’s net acid load, causing increased bone dissolution, with the overall acid-base balance of the body being an important contributor to bone health [36]. The increased bone resorption of calcium was thought to act as a buffer to this increased acid load, with increased renal acid excretion of calcium as a result [37,38,39]. But more recent studies using a much larger dataset contradicted this, showing no significant difference between the biochemical indicators of bone turnover or whole body calcium retention in high protein or low protein diets [40]. This contradiction could be explained, among others, by the fact that foods which contain protein also contain hypocalciuric elements such as phosphorous [41,42,43]. Kerstetter et al. reported a strong association between dietary protein intake and intestinal calcium absorption [44] while conversely, both Surdykowski et al. and Fenton et al. concluded that increased protein intakes in the diet are not associated with significant blood pH changes and dispute the acid-base hypothesis [45, 46].
This confusion may have been due to the assumption of some studies that an increase in urinary calcium related to changes in bone turnover or bone health generally. Gaffney-Stomberg et al. reported a net gain of calcium with a high protein diet in rats, even when the resultant increase of urinary calcium was taken into account, with no changes in indicators of bone turnover [47]. In human isotopic studies, this trend has been repeated [40, 44, 48].
Advances have been made in understanding the underlying mechanism to which protein, or more accurately, the amino acids can increase calcium absorption. It is possible that calcium and protein act synergistically at the level of the cell, with one relying on the other to exist for the maintenance and growth of bone [30]. The calcium-sensing receptor (CaR), which is present in multiple organs, has been shown to be significant in maintaining calcium levels as well as contributing to skeletal growth and maintenance. Amino acids can increase the sensitivity of these receptors to calcium, therefore resulting in an increased uptake of calcium through these cells [13, 30].
There is considerable debate on whether protein quality, either vegetable or animal protein, is an important contributor to protecting and enhancing bone health. In some studies, vegetable protein was found to confer a greater benefit when compared with animal protein [49]. There is a degree of inconsistency in studies exploring the superiority of protein from vegetable rather than animal sources [13]. Furthermore, a potential disadvantage of consuming protein from vegetable sources is that vegetables may not contain all of the essential amino acids, unlike animal sources.
There is much controversy on the ideal quantity of protein intake and type among different population groups (especially older adults), as well as the most accurate and efficient means of evaluating dietary protein intake. The “one size fits all” model of adequate dietary protein intake among all age and population groups is not appropriate and may have unintended consequences for the elderly population, who require more dietary protein [27].
Therefore, our aims are to (1) review the evidence of the relationship between BMD and protein quantity in animal subjects as well as assessing this relationship among those ≥50 years of age and (2) assess the association between the concentration of protein consumed in the diet and bone fracture (hip fracture).
Materials and methods
We searched studies on various databases, including ScienceDirect, PubMed and EBSCO. The search strategy involved using specific terms: “Protein, bone, elderly, BMD” and “Protein, bone, elderly, fracture risk”. The reference list of each study and previous review articles was also examined. The search was not restricted regarding the date of the studies, with any articles meeting the inclusion criteria included in the review. The initial population was further narrowed, with any literature which included populations under 50 years of age being discounted. The majority of studies which assessed BMD utilised DXA scanning, with a small number of studies using single-photon absorptiometry or ultrasound technology [50,51,52]. One other study used dual photon absorptiometry for the baseline readings, with the 4-year follow-up using DXA [53]. Studies fit for qualification in the review process were also assessed by two reviewers with clinical and laboratory expertise. There was no journal article which posed a language restriction problem, and no authors had to be contacted for the literature review. All studies that were included in the main review had to be published, and reference was made to literature related to opinions in the discussion. A small proportion of unpublished literature was included only in the analysis of the conclusions of the studies, rather than the studies/trials themselves.
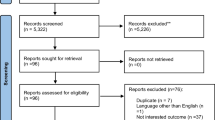
For assessing the relationship between protein quantity in the diet and BMD, we included studies which measured BMD using a DXA scan at sites such as the radius, femoral neck, total hip, lumbar spine, total hip and total body. For assessing the protein quantity and fracture risk/history association, publications which analysed hospital records were reviewed, depending on each individual study. Fractures caused by events such as road traffic accidents were discounted in many of the studies. The review of the assessment of dietary intake was carried out in the context of the studies assessing an individual’s protein consumption and BMD and fracture risk/history, respectively. We reviewed all publications on patients older than 50 years which related to protein quantity and fracture risk as well as BMD. There was a requirement for the subjects to be greater than 50 years, with studies discounted even if the average age was above 50 years but the range included those under 50 years, e.g. Sahni et al. [54]. Studies which included a maximum of two variables were included in the study, with the review of the studies taking this into account in the conclusions that were reached. This exclusion criteria and the process of selection is displayed in the flowchart below (Fig. 1 ).
To identify the volume of protein consumed in an individual’s diet, dietary assessment tools such as the Food Frequency Questionnaire (FFQ), dietary records and food diaries are used in the screening of large populations and to accurately identify those who are at risk due to their consumption patterns.
Results
BMD (Tables 1 and 2)
Of the 287 titles that we identified, 18 studies aimed to evaluate the relationship between protein quantity intake in the diet and BMD. Animal studies (Table 2) composed 4 of these, while 14 were on human subjects (Table 1). Of these studies, 13 found a positive correlation between protein and BMD, while 3 found either no relationship or possibly negative relationship. Two studies exhibited inconclusive results.
Regarding the animal studies, one of these displayed no association while the remaining three showed a positive correlation. All of the population in the animal studies were growing or adult rats, either male or female. None were ovariectomized. Of the positive results, the studies focussed on the low BMD effects of a low-protein diet, along with negative effects on cancellous bone mass and trabecular thickness in some instances also found [58]. A control was used in all the animal studies, which was 15% casein for 2 of the studies and 18% and 24% protein diet for the remaining studies, representing adequate levels of protein in the diet.
Of the 14 studies on human subjects >50 years, 10 found a positive association between protein quantity in the diet and BMD. Of these, eight had protein as an independent variable in their assessment. The two remaining studies had a variation in calcium or vitamin D intake. Aside from these, the remaining 8, some of which were wide-ranging epidemiological studies, do show a strong positive correlation, independent of other factors. There was a significant variation in the number of subjects in each of the studies. Chan et al. had the highest number of participants, with 1225 men and 992 women. Geinoz et al.’s study of 74 hospitalised patients had the lowest number of participants. In the studies which portrayed a positive correlation, a significantly higher BMD was found in the femoral neck and lumbar spine of the human subjects [50, 53, 57], as well as total hip [51, 55] and total body [27, 59] of those who were not protein malnourished.
Bone fracture
A total of seven studies were identified which aimed to evaluate the relationship between protein quantity and bone fracture risk in those over 50 years (Table 3). Of these, four studies found a positive correlation between dietary protein quantity and bone fracture risk. One study found no association, with two studies with mixed results, depending on the ratio of animal to vegetable protein consumed.
Wengreen et al. observed that those who consumed the highest quartile of protein (17.4–30.8% of total energy) had a 65% reduction in the risk of hip fracture. The study also reported that the association between protein consumption and its protective effects from fracture risk dissipated with increased age. The authors also observed that a higher protein intake conferred a protective effect from hip fracture in 50–69-year olds, with this protective effect disappearing in the 70–89-year age bracket [61].
Discussion
Protein quantity in the diet and BMD
The results from these studies convey the positive change in BMD with an increase in protein consumption, with 72% of the studies consistent with this finding. The majority of the studies in this review observed a positive relationship between protein consumption and BMD in older adults and the importance of the interaction between protein and calcium in bone formation and maintenance. This emphasises the need for adequate intake of all key nutrients in the prevention of bone-related diseases such as osteoporosis. However, not all studies were in agreement—2 of the 14 studies observed no association between consumption and BMD [21, 34].
For example, Kerstetter et al. in a combination of 13 younger and postmenopausal women observed no association between protein intake and femoral neck and lumbar spine BMD [44]. However, in this study, the relationship between a ratio of protein and calcium with bone was assessed, rather than protein on its own.
Importantly and as previous studies have suggested that although BMD is a good marker of bone strength, it is only one factor in the process, with bone mineral mass and architecture among other important contributors to bone strength [64]. Even with these, variance on the development of osteoporosis can occur. Therefore, assessing the role of intrinsic tissue quality may be a more accurate measure of the risk of osteoporosis developing [65]. Furthermore, the measurement and assessment of important structural proteins such as collagen and bone turnover (and the technical challenges that accompany this) in determining overall bone health should be considered for future studies.
Protein quantity in the diet and risk of fracture
The majority of studies reviewed displayed a positive relationship between protein consumption and fracture risk [59,60,61,62]. Many of the studies which displayed a positive correlation between protein quantity and fracture risk also emphasised the importance of correct calcium intake as well as protein intake in the diet [54]. However, this interdependent phenomenon between protein and calcium was not as evident among more elderly participants (aged >50 years [54]) which reinforces the hypothesis that the interaction of protein and calcium is complex in bone formation and stability [30].
The results of Wengreen et al. who investigated the different associations between protein intake and bone fracture risk between different age groups in the elderly population also requires further research. The renal tubular re-absorption and intestinal absorption of calcium are decreased among the elderly population, and as protein has been shown to potentially influence these mechanisms, the change in dietary protein would have to reflect this alteration in the elderly population [66]. This also emphasises the need for early intervention to maximise bone health before this life stage is reached, as previously stated by both Zhu et al. and Delmi et al. [56, 67].
Dargent-Molina et al. who observed a negative association between protein intake and bone health had calcium intakes which were designed to be low among the participants, while protein intakes were either moderate or high. The low calcium intake, rather than the moderate or high protein intake, could have contributed to the risk of the increased fractures. Interestingly, this study which looked at fracture risk of postmenopausal women (40–65 years) at baseline and 15 years later found that high animal protein intake actually conferred a greater fracture risk in the presence of reduced calcium intake whilst displaying a benefit in the presence of adequate calcium intake [36]. The complex interaction these two nutrients have is supported by Sahni et al. (which was not included in this review as the age range included subjects who were pre-menopausal), where there was a negative correlation between animal protein and risk of hip fracture in the presence of adequate calcium intake, as opposed to an increased risk of fracture associated with moderate to high intake of animal protein in the presence of low calcium intake in the diet [54].
As observed with BMD, the ratio of animal to vegetable protein may also play a role in the prevention of hip fractures. Sellmeyer et al. observed that elderly women with a high animal-plant ratio had a higher risk of hip fracture (relative risk = 3.7, P = 0.04) [63]. The acid-load hypothesis or a different, unknown mechanism may help explain this observed difference. However, this requires further exploration to elicit whether the issue is one of the animal protein vs plant protein or rather a question of macronutrients and micronutrients [13].
It has been observed that elderly fracture patients undergo deterioration in nutritional status during their hospital stay, with a negative association between low dietary protein intake and the risk of bone fracture and longer rehabilitation [61, 68, 69]. Furthermore, their functional outcome after bone fracture is well documented in literature [59, 60, 67, 70, 71]. Therefore, protein intake could also be a determinant of a patient’s morbidity and mortality and the length of their stay in hospital [70, 72, 73]. For example, protein-energy malnutrition has been found to be an outcome determinant for the patient, affecting immune function, among others [74, 75]. In three studies, protein energy malnutrition was identified in a significant percentage of admissions to long-term care settings, an orthopaedic surgery department and a community resident home, respectively [76,77,78].
Strengths and limitations of this review
From an initial literature review of 7941 papers, 25 were found to meet inclusion criteria. A total of 25 papers were assessed to examine the relationship between protein, BMD, and hip fracture, with reference to quantity rather than quality of protein. This review is one of the few studies that focussed predominantly on bone health in the over 50 age group. As there is a degree of variation within the FFQ format itself (with some FFQs going into more detail and using slightly different tools compared to others), the conclusions of this review had to reflect the degree of non-standardisation when deciding its effectiveness [79]. Furthermore, it was unknown if some of the studies included the impact of polypharmacy on absorption and utilisation of micronutrients which is a limitation of this paper.
Conclusions
The results of this review suggest that the quantity of protein consumed in the diet is associated with a greater BMD in the elderly. Furthermore, it was observed that protein bestowed a protective effect from the risk of fracture. These observations suggest that there is a need for a greater effort in ensuring that those >50 years meet their protein requirements as a means of bone protection [27]. Although there are weaknesses associated with the FFQ, the most common assessment tool in these studies, there are currently no equally alternative methods which have displayed greater utility in data attainment in this regard, in large studies. Ideally, the combination of biomarkers with BMD would yield the most accurate results by far, but there is limited cost and time, especially in the clinical environment. Improvements in the current FFQ model, such as creating a more detailed questionnaire, may prove to be more accurate in eliciting information [80]. As a result, the FFQ will continue to be the mainstay method of choice until further evidence suggests the need for a complete diet record of micro and macro nutrition.
References
(1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser 843:1–129
Strom O, Borgstrom F, Zethraeus N, Johnell O, Lidgren L, Ponzer S, Svensson O, Abdon P, Ornstein E, Ceder L, Thorngren KG, Sernbo I, Jonsson B (2008) Long-term cost and effect on quality of life of osteoporosis-related fractures in Sweden. Acta Orthop 79:269–280
Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA (1999) Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet 353:878–882
Gannon B, O'Shea E, Hudson E (2008) Economic consequences of falls and fractures among older people. Ir Med J 101:170–173
Poole KE, Compston JE (2006) Osteoporosis and its management. BMJ 333:1251–1256, England
Unnanuntana A, Gladnick BP, Donnelly E, Lane JM (2010) The assessment of fracture risk. J Bone Joint Surg Am 92:743–753
Grainge MJ, Coupland CA, Cliffe SJ, Chilvers CE, Hosking DJ (1998) Cigarette smoking, alcohol and caffeine consumption, and bone mineral density in postmenopausal women. The Nottingham EPIC study group. Osteoporos Int 8:355–363
Kanis JA, Johnell O, Oden A, Johansson H, De Laet C, Eisman JA, Fujiwara S, Kroger H, McCloskey EV, Mellstrom D, Melton LJ, Pols H, Reeve J, Silman A, Tenenhouse A (2005) Smoking and fracture risk: a meta-analysis. Osteoporos Int 16:155–162
Sinnesael M, Claessens F, Boonen S, Vanderschueren D (2013) Novel insights in the regulation and mechanism of androgen action on bone. Curr Opin Endocrinol Diabetes Obes 20:240–244
Melton LJ 3rd (2003) Epidemiology worldwide. Endocrinol Metab Clin N am 32:1–13, v
Marcus R (1996) Mechanisms of exercise effects on bone. Principles of Bone Biology 151:1135–1146
Bradney M, Pearce G, Naughton G, Sullivan C, Bass S, Beck T, Carlson J, Seeman E (1998) Moderate exercise during growth in prepubertal boys: changes in bone mass, size, volumetric density, and bone strength: a controlled prospective study. J Bone Miner Res 13:1814–1821
Holick MF, Nieves JW, Bonjour J-P, Chevalley T, Amman P, Rizzoli R (2015) Protein intake and bone health. Nutrition and bone health. Springer, New York, pp 301–317
Houston DK, Nicklas BJ, Ding J, Harris TB, Tylavsky FA, Newman AB, Lee JS, Sahyoun NR, Visser M, Kritchevsky SB (2008) Dietary protein intake is associated with lean mass change in older, community-dwelling adults: the health, Aging, and body composition (health ABC) study. Am J Clin Nutr 87:150–155
De Souza Genaro P, Martini LA (2010) Effect of protein intake on bone and muscle mass in the elderly. Nutr Rev 68:616–623
Murton AJ (2015) Muscle protein turnover in the elderly and its potential contribution to the development of sarcopenia. Proc Nutr Soc 74:387–396. doi: 10.1017/S0029665115000130.
Witard OC, Wardle SL, Macnaughton LS, Hodgson AB, Tipton KD (2016) Protein considerations for Optimising skeletal muscle mass in healthy young and older adults. Nutrients 8:181
Szulc P, Beck TJ, Marchand F, Delmas PD (2005) Low skeletal muscle mass is associated with poor structural parameters of bone and impaired balance in elderly men--the MINOS study. J Bone Miner Res 20:721–729
Landi F, Cruz-Jentoft AJ, Liperoti R, Russo A, Giovannini S, Tosato M, Capoluongo E, Bernabei R, Onder G (2013) Sarcopenia and mortality risk in frail older persons aged 80 years and older: results from ilSIRENTE study. Age Ageing 42:203–209
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K (2013) Frailty in elderly people. Lancet 381:752–762
Frost HM (1997) On our age-related bone loss: insights from a new paradigm. J Bone Miner Res 12:1539–1546
Ducher G, Jaffre C, Arlettaz A, Benhamou CL, Courteix D (2005) Effects of long-term tennis playing on the muscle-bone relationship in the dominant and nondominant forearms. Can J Appl Physiol 30:3–17
Pang MY, Eng JJ (2005) Muscle strength is a determinant of bone mineral content in the hemiparetic upper extremity: implications for stroke rehabilitation. Bone 37:103–111
Heaney RP (2009) Dairy and bone health. J Am Coll Nutr 28(Suppl 1):82s–90s
Heaney RP, Layman DK (2008) Amount and type of protein influences bone health. Am J Clin Nutr 87:1567S–1570S
Ilich JZ, Brownbill RA, Tamborini L (2003) Bone and nutrition in elderly women: protein, energy, and calcium as main determinants of bone mineral density. Eur J Clin Nutr 57:554–565
Bauer J, Biolo G, Cederholm T, Cesari M, Cruz-Jentoft AJ, Morley JE, Phillips S, Sieber C, Stehle P, Teta D, Visvanathan R, Volpi E, Boirie Y (2013) Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE study group. J Am Med Dir Assoc 14:542–559
Zhang W, Shen X, Wan C, Zhao Q, Zhang L, Zhou Q, Deng L (2012) Effects of insulin and insulin-like growth factor 1 on osteoblast proliferation and differentiation: differential signalling via Akt and ERK. Cell Biochem Funct 30:297–302
Babraj JA, Smith K, Cuthbertson DJ, Rickhuss P, Dorling JS, Rennie MJ (2005) Human bone collagen synthesis is a rapid, nutritionally modulated process. J Bone Miner Res 20:930–937
Conigrave AD, Brown EM, Rizzoli R (2008) Dietary protein and bone health: roles of amino acid-sensing receptors in the control of calcium metabolism and bone homeostasis. Annu Rev Nutr 28:131–155
Ammann P, Bourrin S, Bonjour JP, Meyer JM, Rizzoli R (2000) Protein undernutrition-induced bone loss is associated with decreased IGF-I levels and estrogen deficiency. J Bone Miner Res 15:683–690
Rizzoli R, Bianchi ML, Garabedian M, McKay HA, Moreno LA (2010) Maximizing bone mineral mass gain during growth for the prevention of fractures in the adolescents and the elderly. Bone 46:294–305
Margen S, Chu J-Y, Kaufmann N, Calloway D (1974) Studies in calcium metabolism. I. The calciuretic effect of dietary protein. Am J Clin Nutr 27:584–589
Johnson NE, Alcantara EN, Linkswiler H (1970) Effect of level of protein intake on urinary and fecal calcium and calcium retention of young adult males. J Nutr 100:1425–1430
Lemann J Jr (1999) Relationship between urinary calcium and net acid excretion as determined by dietary protein and potassium: a review. Nephron 81(Suppl 1):18–25
Dargent-Molina P, Sabia S, Touvier M, Kesse E, Breart G, Clavel-Chapelon F, Boutron-Ruault MC (2008) Proteins, dietary acid load, and calcium and risk of postmenopausal fractures in the E3N French women prospective study. J Bone Miner Res 23:1915–1922
Arnett T (2003) Regulation of bone cell function by acid-base balance. Proc Nutr Soc 62:511–520
Jehle S, Zanetti A, Muser J, Hulter HN, Krapf R (2006) Partial neutralization of the acidogenic western diet with potassium citrate increases bone mass in postmenopausal women with osteopenia. J Am Soc Nephrol 17:3213–3222
Sebastian A, Harris ST, Ottaway JH, Todd KM, Morris RC Jr (1994) Improved mineral balance and skeletal metabolism in postmenopausal women treated with potassium bicarbonate. N Engl J Med 330:1776–1781
Roughead ZKF, Johnson LK, Lykken GI, Hunt JR (2003) Controlled high meat diets do not affect calcium retention or indices of bone status in healthy postmenopausal women. J Nutr 133:1020–1026
Spencer H, Kramer L, Osis D, Norris C (1978) Effect of a high protein (meat) intake on calcium metabolism in man. Am J Clin Nutr 31:2167–2180
Kerstetter JE, O’Brien KO, Insogna KL (2003) Dietary protein, calcium metabolism, and skeletal homeostasis revisited. Am J Clin Nutr 78:584S–592S
Rizzoli R, Bonjour JP (2004) Dietary protein and bone health. J Bone Miner Res 19:527–531
Kerstetter JE, O'Brien KO, Caseria DM, Wall DE, Insogna KL (2005) The impact of dietary protein on calcium absorption and kinetic measures of bone turnover in women. J Clin Endocrinol Metab 90:26–31
Surdykowski AK, Kenny AM, Insogna KL, Kerstetter JE (2010) Optimizing bone health in older adults: the importance of dietary protein. Aging Health 6:345–357
Fenton TR, Lyon AW, Eliasziw M, Tough SC, Hanley DA (2009) Meta-analysis of the effect of the acid-ash hypothesis of osteoporosis on calcium balance. J Bone Miner Res 24:1835–1840
Gaffney-Stomberg E, Sun BH, Cucchi CE, Simpson CA, Gundberg C, Kerstetter JE, Insogna KL (2010) The effect of dietary protein on intestinal calcium absorption in rats. Endocrinology 151:1071–1078
Hunt JR, Johnson LK, Roughead ZF (2009) Dietary protein and calcium interact to influence calcium retention: a controlled feeding study. Am J Clin Nutr 89:1357–1365
Kim J, Lim SY, Kim JH (2008) Nutrient intake risk factors of osteoporosis in postmenopausal women. Asia Pac J Clin Nutr 17:270–275
Geinoz G, Rapin CH, Rizzoli R, Kraemer R, Buchs B, Slosman D, Michel JP, Bonjour JP (1993) Relationship between bone mineral density and dietary intakes in the elderly. Osteoporos Int 3:242–248
Devine A, Dick IM, Islam AF, Dhaliwal SS, Prince RL (2005) Protein consumption is an important predictor of lower limb bone mass in elderly women. Am J Clin Nutr 81:1423–1428
Metz JA, Anderson JJ, Gallagher PN Jr (1993) Intakes of calcium, phosphorus, and protein, and physical-activity level are related to radial bone mass in young adult women. Am J Clin Nutr 58:537–542
Hannan MT, Tucker KL, Dawson-Hughes B, Cupples LA, Felson DT, Kiel DP (2000) Effect of dietary protein on bone loss in elderly men and women: the Framingham osteoporosis study. J Bone Miner Res 15:2504–2512
Sahni S, Cupples LA, Mclean RR, Tucker KL, Broe KE, Kiel DP, Hannan MT (2010) Protective effect of high protein and calcium intake on the risk of hip fracture in the Framingham offspring cohort. J Bone Miner Res 25:2770–2776
Thorpe M, Mojtahedi MC, Chapman-Novakofski K, McAuley E, Evans EM (2008) A positive association of lumbar spine bone mineral density with dietary protein is suppressed by a negative association with protein sulfur. J Nutr 138:80–85
Zhu K, Meng X, Kerr DA, Devine A, Solah V, Binns CW, Prince RL (2011) The effects of a two-year randomized, controlled trial of whey protein supplementation on bone structure, IGF-1, and urinary calcium excretion in older postmenopausal women. J Bone Miner Res 26:2298–2306
Kerstetter JE, Looker AC, Insogna KL (2000) Low dietary protein and low bone density. Calcif Tissue Int 66:313
Bourrin S, Toromanoff A, Ammann P, Bonjour J, Rizzoli R (2000) Dietary protein deficiency induces osteoporosis in aged male rats. J Bone Miner Res 15:1555–1563
Misra D, Berry SD, Broe KE, McLean RR, Cupples LA, Tucker KL, Kiel DP, Hannan MT (2011) Does dietary protein reduce hip fracture risk in elders? The Framingham osteoporosis study. Osteoporos Int 22(1):345–349. doi:10.1007/s00198-010-1179-4.
Munger RG, Cerhan JR, Chiu BC (1999) Prospective study of dietary protein intake and risk of hip fracture in postmenopausal women. Am J Clin Nutr 69:147–152
Wengreen HJ, Munger RG, West NA, Cutler DR, Corcoran CD, Zhang J, Sassano NE (2004) Dietary protein intake and risk of osteoporotic hip fracture in elderly residents of Utah. J Bone Miner Res 19:537–545
Martínez-Ramírez MJ, Delgado-Martínez AD, Ruiz-Bailén M, de la Fuente C, Martínez-González MÁ, Delgado-Rodríguez M (2012) Protein intake and fracture risk in elderly people: a case-control study. Clin Nutr 31:391–395
Sellmeyer DE, Stone KL, Sebastian A, Cummings SR (2001) A high ratio of dietary animal to vegetable protein increases the rate of bone loss and the risk of fracture in postmenopausal women. Study of osteoporotic fractures research group. Am J Clin Nutr 73:118–122
Darling, A.L., Millward, D.J., Torgerson, D.J., Hewitt, C.E., Lanham-New, S.A. (2009) Dietary protein and bone health: a systematic review and meta-analysis. Am J Clin Nutr 27799
Hengsberger S, Ammann P, Legros B, Rizzoli R, Zysset P (2005) Intrinsic bone tissue properties in adult rat vertebrae: modulation by dietary protein. Bone 36:134–141
Rizzoli R, Boonen S, Brandi ML, Burlet N, Delmas P, Reginster JY (2008) The role of calcium and vitamin D in the management of osteoporosis. Bone 42:246–249
Delmi M, Rapin CH, Bengoa JM, Delmas PD, Vasey H, Bonjour JP (1990) Dietary supplementation in elderly patients with fractured neck of the femur. Lancet 335:1013–1016
Paillaud E, Bories PN, Le Parco JC, Campillo B (2000) Nutritional status and energy expenditure in elderly patients with recent hip fracture during a 2-month follow-up. Br J Nutr 83:97–103
Bonjour JP, Schurch MA, Rizzoli R (1996) Nutritional aspects of hip fractures. Bone 18:139s–144s
Tkatch L, Rapin C-H, Rizzoli R, Slosman D, Nydegger V, Vasey H, Bonjour J-P (1992) Benefits of oral protein supplementation in elderly patients with fracture of the proximal femur. J am Coll Nutr 11:519–525
Schurch MA, Rizzoli R, Slosman D, Vadas L, Vergnaud P, Bonjour JP (1998) Protein supplements increase serum insulin-like growth factor-I levels and attenuate proximal femur bone loss in patients with recent hip fracture. A randomized, double-blind, placebo-controlled trial. Ann Intern Med 128:801–809
Roche JJW, Wenn RT, Sahota O, Moran CG (2005) Effect of comorbidities and postoperative complications on mortality after hip fracture in elderly people: prospective observational cohort study. BMJ Br Med J 331(7529):1374. doi:10.1136/bmj.38643.663843.55.
Keene GS, Parker MJ, Pryor GA (1993) Mortality and morbidity after hip fractures. BMJ 307(6914):1248–1250
Chandra R, Joshi P, Au B, Woodford G, Chandra S (1982) Nutrition and immunocompetence of the elderly: effect of short-term nutritional supplementation on cell-mediated immunity and lymphocyte subsets. Nutr Res 2:223–232
Potter J, Klipstein K, Reilly J, Roberts M (1995) The Nutrititional status and clinical course of acute admissions to a geriatric unit. Age Ageing 24:131–136
Thomas DR, Verdery RB, Gardner L, Kant A, Lindsay J (1991) A prospective study of outcome from protein-energy malnutrition in nursing home residents. JPEN J Parenter Enteral Nutr 15:400–404
Christensson L, Unosson M, Ek AC (1999) Malnutrition in elderly people newly admitted to a community resident home. J Nutr Health Aging 3:133–139
Drevet S, Bioteau C, Mazière S, Couturier P, Merloz P, Tonetti J, Gavazzi G (2014) Prevalence of protein-energy malnutrition in hospital patients over 75years of age admitted for hip fracture. Orthop Traumatol Surg Res 100:669
Willett WC, Hu FB (2006) Not the time to abandon the food frequency questionnaire: point. Cancer Epidemiol Biomark Prev 15:1757–1758
Kanis JA, Kanis JA (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: Synopsis of a WHO report. Osteoporos Int 4(6):368–381
Dawson-Hughes B, Harris, SS (2002) Calcium intake influences the association of protein intake with rates of bone loss in elderly men and women. The American Journal of Clinical Nutrition 75, 773–779
Chan R, Woo J, Leung J (2011) Effects of food groups and dietary nutrients on bone loss in elderly Chinese population. J Nutr Health Aging 15, 287–294
Kerstetter JE, Bihuniak, JD, Brindisi J, Sullivan, RR, Mangano KM, Larocque S, Kotler BM, Simpson CA, Maria Cusano A, Gaffney-Stomberg E, Kleppinger A, Reynolds J, Dziura J, Kenny AM, Insogna K.L (2015) The effect of a whey protein supplement on bone mass in older Caucasian adults. J Clin Endocrinol Metab jc20143792
Rapuri PB, Gallagher JC, Haynatzka V (2003) Protein intake: effects on bone mineral density and the rate of bone loss in elderly women. Am J Clin Nutr 77, 1517–1525
Wang MC, Luz Villa M, Marcus R, Kelsey JL (1997) Associations of vitamin C, calcium and protein with bone mass in postmenopausal Mexican American women. Osteoporos Int 7, 533–538
Orwoll E, Ware M, Stribrska L, Bikle D, Sanchez T, Andon M, Li H (1992) Effects of dietary protein deficiency on mineral metabolism and bone mineral density. The American journal of clinical nutrition 56, 314–319
Demigné C, Bloch-Faure M, Picard N, Sabboh H, Besson C, Remesy C, Geoffroy V, Gaston AT, Nicoletti A, Hagege A, Menard J, Meneton P (2006) Mice chronically fed a westernized experimental diet as a model of obesity, metabolic syndrome and osteoporosis. Eur J Nutr 45, 298–306
Zhong Y, Okoro CA, Balluz LS (2009) Association of total calcium and dietary protein intakes with fracture risk in postmenopausal women: the 1999–2002 National Health and Nutrition Examination Survey (NHANES). Nutrition 25, 647–654
Acknowledgements
JC, EL and MC conceived and designed the literature review. JC collected and interpreted the data. MC and EL made corrections to the draft, which JC amended. All authors approved the abstract and review for publication. No funding was received from any institution.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Key points
Summary
-We found that a higher quantity of protein consumed in the diet of those >50 years resulted in a higher bone mineral density and therefore confers a protective benefit for the bone health of this population.
-It was also found that a higher quantity of protein in this cohort also reduced their fracture risk, and therefore, their morbidity and mortality.
-We recommend that a higher recommended daily amount of dietary protein should be instituted in those who are more at-risk of fractures due to declining bone health, including those >50 years of age.
Research questions
-Although protein quantity is well discussed in our paper, the importance of protein quality on the bone health of this population, either animal or vegetable, is also an important question to discuss in the future.
-Further studies to determine more accurately the exact benefit that increased protein quantity in the diet would have on specific cohorts (i.e. 70–80 years of age etc.) would be most beneficial.
-In our conclusions, we recommend a greater RDA of dietary protein in those >50 years than what is currently recommended. Greater discussion and debate on this topic would be most welcome in this regard.
Rights and permissions
About this article
Cite this article
Curneen, J.M.G., Casey, M. & Laird, E. The relationship between protein quantity, BMD and fractures in older adults. Ir J Med Sci 187, 111–121 (2018). https://doi.org/10.1007/s11845-017-1642-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-017-1642-8