Abstract
Crataegus species (Rosaceae) or hawthorns form dense thickets and displace native vegetation out of their native range. In South Africa, Crataegus monogyna and C. mexicana are under surveillance but there is lack of information on their pollination ecology. Therefore, this study sought to investigate the pollination mechanism and potential insect pollinators of these plants. Pollinator exclusion experiments were conducted on ten plants each of C. monogyna and C. mexicana near the towns of Hogsback and Seymour respectively, in the Eastern Cape Province. Flowers of each of these species were also observed to determine the diversity and distribution of insect visitors throughout the day (i.e. morning, mid day and afternoon). The results showed that C. monogyna and C. mexicana are capable of self and cross-pollination. Fruit set from open inflorescences of C. monogyna was 40.88 ± 0.08%, significantly higher than bagged inflorences. Fruit set from open inflorescences of C. mexicana was 48.18 ± 0.03%, significantly higher than those of bagged inflorescences. Hymenopterans were the most frequent visitors on C. monogyna flowers while dipterans were the most frequent visitors on C. mexicana flowers. The distribution of insects did not vary with the time of day for each plant species. Insect pollination, therefore, plays a significant role in the fruiting of C. monogyna and C. mexicana in the Eastern Cape. The results of this study contribute to the understanding of the ecology and phenology of Crataegus species in South Africa and thus the evaluation of their invasive status.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Globalisation has led to an increase in the movement and establishment of species into new natural habitats far away from their native range (Banks et al. 2015). Some of these alien species have the potential to become invasive and cause damage to biodiversity and thus the economy of their new geographical environment (Cassey et al. 2015; Blackburn et al. 2019). These species have been found to extirpate native species and replace them (Simberloff et al. 2013). They also decrease the survival or reproduction of native species. In addition, alien plants can transform ecosystems, when functional traits that drive ecosystem processes differ between alien and native plants (Wardle et al. 2011). Due to the fact that alien plants add physical structures in their new environments, erosion regimes are altered and thus change habitat suitability for other species (Simberloff 2011; Simberloff et al. 2013).
Many alien plant species in a new environment require insect pollinators and dispersal vectors to become successful invaders (Blackburn et al. 2011; Menz et al. 2011; Saunders 2018). The contribution facilitated by recruited insect pollinators is crucial in assisting further spread and invasion in a new area. While recruiting new pollinators in a new area may be beneficial to invasive alien plants, the altered behaviour of recruited pollinators might have negative ecological consequences (Stout and Tiedeken 2017). These may include native species being neglected by native insect pollinators and a decrease in the production of fruits and seeds for future generations of native species (Brown et al. 2002; New 2016). With native species being neglected by pollinators, more areas would be invaded by alien plants, changing the entire species network and threatening biodiversity (Padron et al. 2009).
Crataegus species (Rosaceae), commonly known as hawthorns, comprise about 280 species worldwide, distributed naturally throughout North Africa, North America, Central Asia, East Asia, and Europe, with minor occurrence in Peru and Ecuador (Nieto-Ángel et al. 2009; Bechkri et al. 2017; Naghipour et al. 2021). Members of the genus are deciduous spiny hard wood shrubs or little trees that can grow to about 10 m in sandy, stony, shallow and dry soils (Kumar et al. 2012; Naghipour et al. 2021). Habitat suitability of these plants is determined by elevation, precipitation and temperature (Jafari et al. 2019; Rafiee mo et al. 2020). Many species from this genus are used as ornamentals and for medicinal purposes, with some regarded as the oldest pharmaceutical plants used in western medicine (Bahorun et al. 2003; Naghipour et al. 2021). The leaves, flowers, and fruits have been used for various cardiovascular and respiratory problems or their anti-inflammatory, antioxidant and anti-carcinogenic properties (Edwards et al. 2012; Bechkri et al. 2017). In addition, the majority of species of these plants are used globally as firewood by locals (Eberly 1989; Singh et al. 2018).
In countries such as Australia, New Zealand, USA and Canada where Crataegus species have become invasive, they form dense thickets which alter the structure of understory plants in a forest, displacing native vegetation (Alverson and Sigg 2008; Pasiecznik 2018; Tropical Plants Database 2023). Their fruits may be preferred by birds over those from native plants, resulting in the reduction of native species being dispersed (Pasiecznik 2018; Tropical Plants Database 2023). In Oregon, United States of America for example, the Willamette Valley Nature Preserve had to be abandoned due to invasion by C. monogyna and a lack of adequate resources to control the species (Alverson and Sigg 2008; Pasiecznik 2018). Similarly, there have been some reports on the threat to Garry Oak ecosystem in British Columbia, Canada by C. monogyna species (Pasiecznik 2018). Due to the fact that Crataegus species are closely related and share similar habitats with Pyracantha species and Cotoneaster species, they are likely to cause similar adverse effects in South Africa. These include the formation of dense thickets that exclude other native plants, block access by humans and livestock to valuable grazing and riverine habitats, protected areas, road ways, fences and power lines (Chari et al. 2020).
In South Africa, Crataegus monogyna Jacq and Crataegus mexicana DC (Rosaceae) are not listed in any of the four categories of the National Environmental Management: Biodiversity Act (NEMBA) of 2004 (CABI 2021). This is because these species are present in the country but little is known about them. Such species are listed as species under surveillance (category no. 4) decreed under the Conservation of Agricultural Resources Act (CARA) regulations, which is a draft categorisation of the Invasive Alien Plants in South Africa (CARA 2006). These species are of great concern in South Africa as they have the potential of becoming invasive in the future due to the history of the invasion of these plants in other countries. In South Africa, the pollination ecology and pollinators of the hawthorn plants is currently unknown. Understanding the pollination and pollinators of invasive or alien species in a new environment is crucial to making effective decisions for managing or eradicating the species (O’Connor et al. 2019). Similarly, their interaction with insect pollinators has never been studied in the country. Therefore, this study focused on the reproductive ecology of these species, with emphasis on the pollination mechanism and potential pollinators of C. monogyna and C. mexicana, to inform effective decision making and management of these alien species in the country. To complete this, we performed a pollination exclusion experiment and assessed fruit set to determine the main pollination mechanism and discover the potential pollinators of C. monogyna and C. mexicana.
Materials and methods
Study area and plants
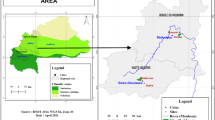
This study was carried out near the town of Seymour at 800 m asl (− 32.455756, 26.845530) and Hogsback Arboretum at 1200 m asl (− 32.590871, 26.934574) in the Eastern Cape Province of South Africa (Fig. 1). Crataegus monogyna was dominant in Hogsback while C. mexicana was dominant in Seymour. Hogsback Arboretum site is found in the Afromontane forest biome while Seymour site is located in the grassland biome surrounded by a livestock farm in the province. The grasslands of the province are mostly semi-arid Karoo characterised by dryness and sparse rain of about 625 mm per year with frosty winters and hot summers (Mandleni 2011), while the higher lying areas of Afromontane forest is wetter with mean annual rainfall of more than 725 mm and snowfall in some years.
Crataegus monogyna and C. mexicana have hermaphroditic flowers with single-seeded fruits. In their native ranges they are reported to be self-compatible with variable autogamy even though cross pollination may increase fruit set (Guitián and Fuentes 1992; Chacoff et al. 2008). The flowers are actinomorphic, perfect and possess a hypanthium (Evan and Dickinson 2005). The corollas comprise five free white petals that when open are perpendicular to the long axis of the flower at anthesis (Fig. 2e, g). Flowers grow in small clusters and are entomophilous (Chacoff et al. 2008; Tropical Plants Database 2023). The fruits of these plants are single-seeded drupes, red in C. mongyna and yellow in C. mexicana when ripe (Fig. 2f, h). Timing of flowering for these species were noted in this study.
Open and bagged inflorence, flowers, insect flower visitor and fruits of Crataegus monogyna and C. mexicana. a Tagged open inflorescence of C. mexicana and b labelled bagged inflorence of C. monogyna. c Lucilia sericata on C. mexicana. d Eristalinus taeniops on C. monogyna. e Flowers of C. monogyna. f Fruits of C. monogyna. g Flowers of C. mexicana. h Fruits of C. mexicana
Insect exclusion experiment
Insect exclusion experiments were set up at each study site to determine the role and significance of self pollination and insect or cross pollination in Crataegus monogyna and C. mexicana. Within each study site, ten mature trees were randomly selected before anthesis (Fig. 2a) for insect exclusion experiments. They were at least 3 m from each other. On each tree, three branches were randomly selected and tagged with labelled white flags as control, group bagging and single bagging treatments (Fig. 2b) according to Jacobs et al. (2009). Each of the treatments was approximately 1–2.5 m above the ground to easily access floral buds. On the control branch, ten randomly selected inflorescences were tagged and marked with weather proof enamel paint from 1 to 10 and left open for insects to visit. On the group bagging branch, ten inflorescences were equally randomly selected, marked with weather proof enamel paint on the bags from 1 to 10. Each of the ten inflorescences was then bagged separately. Lastly, on the single bagging branch another ten randomly selected inflorescences were marked with weather proof enamel paint from 1 to 10 on the bags with only one flower bud from each inflorescence bagged. The bagging of one flower bud was done to exclude it from pollen of other flowers on the same inflorescence. Bagging (either single or group) was done at the “balloon” stage (i.e. before anthesis) during which it is assumed no pollination has occurred (Fig. 2a). The bags were handmade muslin bags (Jacobs et al. 2009) covered with a wire frame. The mesh was placed over the frame and secured with a labelled twist tie. The end of the bag was sealed on to the branch using insulating tape to prevent insects from crawling inside. The wire frame avoided the likelihood of contact between the bag and the reproductive organs of the flowers and prevented stigmas from protruding through the bag. Muslin bags are resilient to thorns and have mesh size of 0.5–0.7 mm capable of excluding insects and pollen.
On the ten tagged Crataegus monogyna plants, a total of 1086 flower buds were left open as control treatments while 1020 buds were group bagged and 100 buds singly bagged. On the tagged C. mexicana plants, 390 flower buds were left opened as control treatments while 365 buds were bagged in groups and 100 buds singly bagged.
The bagging of C. monogyna flower buds took place in early September, while bagging for C. mexicana was done in early October, coinciding with the time each species produces flower buds. In fruit-producing plants, abscission of unfertilized immature fruits (which may be due to inadequate pollination) occurs soon after flowering (Jacobs et al. 2009). Therefore, only fruits that have been successfully pollinated, fertilized and retained by the plant were counted for each treatment. Counting of the fruits from each treatment was done 30 days after experimental set up.
Observations of insects’ flower visitors
To determine which insects visited Crataegus monogyna and C. mexicana flowers during the flowering season, ten trees from each study site were randomly selected. This was done in spring, mid-September for C. monogyna and mid-October for C. mexicana. All observations were made when it was sunny; this is when the flower visitors are more active, and flowers are fully open to attract pollinators (Fig. 2c, d). The time of day for the observations was categorised as morning (07h00–10h00), mid day (10h00–13h00) and afternoon (13h00–16h00) (Larue et al. 2016; Howlett et al. 2018). A 2 m × 2 m (4 m2) quadrat was selected for examination on the tree side where flowers were present (O’Connor et al. 2019). Observation of flower visitors was done with the naked eyes and using binoculars. Each tree was observed for 15 min taking note of any insect that spent more than 10 s on the flowers of each species (Larue et al. 2016). To aid the identification of insects observed visiting flowers, pictures of the visiting insects were taken from a distance of at least 1 m to minimise the disturbance of the visitors. A tally counter was used to count each visitor landing on the flower of the plants. Each tree was sampled five times over the flowering season.
Samples of insects that could not be identified in situ were collected using sweep nets, pooters (aspirator), and forceps in the two study sites. All collections were preserved and taken to the laboratory for identification with the help of a power stereo microscope and identification guides by Picker et al. (2004). Furthermore, the identification of all insects was verified using insect databases such as African Insect Atlas Database, Ispotnature Database, iNaturalist and Insect Identification Key Database. Lastly, these identification were further verified with the help of experts. All insects collected are preserved in insect boxes in the Entomology Laboratory in the Universiversity of Fort Hare, Alice campus, South Africa.
Data analysis
The proportion of flowers that set fruits for each of the treatments (single, group and open pollination) were calculated for each plant species. Due to some of the treatments having zeros, the data were square root transformed and the means compared using analysis of variance and least significant difference post hoc test. Means were back-transformed before reporting.
The total number and means of all insects visiting the flowers of each of the plant species per 4 m2 quadrat were calculated and the means compared using ANOVA of square root transformed data. The diversity indices for the different times of visitation were also calculated and compared for each of the plant species. Species abundance and species numbers are not dimensionless quantities and so tend to be less informative. Hence, the following diversity indices were calculated for each of the plant species across time of the day: Margalef’s species richness (d), Shannon–Wiener diversity index (H′) and Pielou’s evenness index (J′) (Clarke and Gorley 2006). These indices were chosen because each expresses either species richness and/or equitability with the number of individuals belonging to each species as a common unit used to calculate the different indices as follows:
Unlike the total number of species (S) per time of the day, Margalef’s index (d) incorporates the total number of individuals (N) and is a measure of the number of species present for a given number of individuals i.e.
Shannon–Wiener diversity index (H′) measures the degree of diversity across the different times of the day i.e.
where \(Pi\) is the proportion of the total count arising from the \(i\mathrm{th}\) species that visited the flowers.
Pielou’s evenness index (J′) measures whether the species were equitably distributed across the different times of the day i.e.
where \({H}{\prime}\mathrm{max}\) is the maximum possible value of Shannon–Wiener diversity i.e. that which would be achieved if all species were equally abundant (\(\mathrm{log}S)\).
The p level for all statistical analyses was set at P ≤ 0.05. Microsoft Excel softwares was used in organising the data while IBM Statistical Package 25 (SPSS) and Primer V.6 were used in performing statistical analysis.
This study was conducted following the South African and international biodiversity rights and in accordance with the internationally accepted principles for use of animals.
Results
Pollination in Crataegus monogyna and C. mexicana
All treatments of Crataegus monogyna produced fruits following exclusion experiments with the control treatment producing the highest number of fruits. This suggests that C. monogyna is capable of both self (i.e. autogamy and geitonogamy) and cross pollination but relies more on cross pollination (Table 1). Only the control and group bagged treatments of C. mexicana produced fruits following exclusion experiments and like C. monogyna are capable of both cross and self pollination. However, self pollination in the flowers of C. mexicana is geitonogamous and not autogamous (Table 1).
Insect exclusion experiments on Crataegus monogyna and C. mexicana plants
There was a significant difference (P < 0.001) in the percentage of fruits produced by the control (open) treatment of inflorescences compared to either group or single-bagged inflorescences of C. monogyna plants. The control treatment produced the highest percentage of fruits followed by group bagged where as single-bagged treatment plants produced the least percentage fruits (Table 2). Similarly, there was significant difference (P < 0.001) in the percentage of fruits produced by control (open), group and single-bagged treatment inflorescences of C. mexicana. Control treatment produced the highest percentage of fruits followed by the group bagged treatment, while the single-bagged treatment producing no fruits (Table 2).
Flower visitors of Crataegus mongyna and C. mexicana
A total of 5086 insects visited the flowers of Crategus monogyna within the 4 m2 quadrat over the study period. These insects were classified into 15 species belonging to the orders; Coleoptera, Diptera, Hymenoptera and Lepidoptera. The most floral visitors were Apis mellifera L., Episyrphus balteatus De Geer, Acrae horta L., and Megachile species where as the least visitors were Exkorynetes analis Klug, Cladophorus marshalli Marshall and Papilio echeriodes Trimen. The means (± SE) of most of these insects were significantly (P ≤ 0.05) different from each other (Table 3).
On Crataegus mexicana flowers, a total of 5014 insect visitors were counted within the 4 m2 quadrat over the study period. These insects were identified to 14 species in the orders; Coleoptera, Diptera, Hymenoptera and Lepidoptera. The most floral visitors were Apis mellifera L., Lucilia sericata Meigen, Scathophaga stercoraria L., Episyrphus balteatus De Geer and Megachile species while the least visitors were Belenois zochalia Boisduval and Stomoxys calcitrans L. The means (± SE) of most of these insects were significantly (P ≤ 0.05) different from each other (Table 4).
Insect floral visitors’ distribution across different times of the day on Crataegus monogyna and C. mexicana
Within the 4 m2 quadrat, the number of insects visiting the flowers of Crataegus monogyna was significantly higher at mid-day than either in the morning or afternoon. However, the number of species, species richness, species diversity and evenness was not significantly (P ≥ 0.05) different across the different times of the day (Table 5). On C. mexicana the total number of insects, number of species and species diversity was significant (P ≤ 0.05) higher in the morning than either at mid-day or during the afternoon. However, species richness and evenness was not significantly different for either morming, mid day or afternoon (Table 6).
Discussion
Less than 50% of flowers produced fruit for both Crataegus monogyna and C. mexicana in the Eastern Cape Province of South Africa. This may be as a result of pollen limitation, limited resources for maturation of fruits, or a self incompatibility mechanism (Chacoff et al. 2008; Rodriguez-Garcia et al. 2019; Gao et al. 2021). Crataegus monogyna and C. mexicana are capable of both self-pollination and cross pollination by insects (entomophily) to produce fruits. There are different species of insects in the orders Coleoptera, Diptera, Hymenoptera and Lepidoptera capable of pollinating the flowers of these plants in South Africa. Insect visitation varies significantly between plants species. The distribution of these insects in terms of species richness, diversity and evenness do not vary significantly throughout the day on C. monogyna. On C. mexicana, the diversity of insects was higher in the morning than the rest of the day. However, species richness and evenness were statistically the same throughout the day.
Insect pollination is an important ecosystem service that plays a crucial role in the conservation of different plant communities through reproduction (Ashman et al. 2004). Understanding the relationship between pollination, insect pollinators, and fruiting is very important in determining the reproduction system of an entrant species in a new environment, such as alien plants. This is because the most important providers of pollination services are insects (O’Connor et al. 2019). In addition, there is a global concern about the declines in the diversity of insect pollinators and consequences for pollination services (Potts et al. 2016). Indeed, studies continue to demonstrate that invasive non-native species, land-use change, climate change, pesticides, pests and diseases may act to cause decline in pollinating insects (Vanbergen et al. 2013).
In the Eastern Cape Province of South Africa C. monogyna start flowering early September i.e. the beginning of Spring while C. mexicana starts flowering early October i.e. mid-spring season. The flowering of both species starts to increase towards the peak within 2 weeks just after some flowers start opening fully. Both species bloom for about four to 6 weeks, and then fruits start forming. This is similar to what happens in the native ranges of these plants (Barros et al. 2010; Spellenberg and Gil 2014).
Crataegus monogyna fruit ripens between March and April which is early autumn, while C. mexicana ripens from late April to June which is late autumn to early winter in the Eastern Cape Province of South Africa. In Europe, the fruits of C. monogyna can last through autumn while the fruits of C. mexicana last up to late winter in the mountains of Mexico (USDA-NRCS 2018; CAL-IPC 2018; CABI 2021; Tropical Plants Database 2023). Crataegus monogyna produced 40.88% while C. mexicana produced 48.18% of fruits in this study. This is less than 50% of number of floral inflorescences produced. Even though Crataegus monogyna can have between five to seven flowers on each inflorescence while C. mexicana have between three to six flowers in each inflorescence, pollen limitation, limited resources for maturation of fruits or self incompatibility mechanism (Chacoff et al. 2008) continue to play an equal part in both plants.
Insect pollinators contribute to the fruiting of both species in South Africa during the flowering season. When insect pollinators are excluded from visiting flowers of both species, a reduction in fruit set was observed with the majority of flowers being aborted. This confirms that insect pollination plays a critical role in the fruiting of the two species in South Africa, as they do for most plant species globally (Fijen et al. 2018). Self pollination is common in the plant family Rosaceae but flower morphology does not allow for complete self pollination resulting in fewer number of fruits (Hill and Hulley 2000; Andrikopoulos and Cane 2018) as found in this study. Insect visitation therefore facilitate and maximize the reproduction of C. monogyna and C. mexicana plants. Insect pollination is one of the barriers that an alien plant needs to overcome to become a successful invader in a new environment (Blackburn et al. 2011). Similar results have also been reported in the United Kingdom, where Jacobs et al. (2009) showed evidence that C. monogyna and other fruit-bearing hedges require insect pollinators to set more fruits.
This study showed that a reduction of fruit set only happens when insects are excluded from visiting flowers or inflorescences. When a single flower is excluded from other flowers and insect visitation, self-pollination by autogamy occurs though to a lesser extent on C. monogyna but does not occur on flowers of C. mexicana. However, when only insects are excluded and flowers are bagged together or as an inflorescence, self-pollination by geitonogamy occurs in both species. This showed that Crataegus monogyna and C. mexicana could self-pollinate when insect pollinators are excluded from visiting the flowers. There is, however, a decrease in the fruit set observed in the absence of insect pollinators. This is because insects are crucial for the transfer of pollen from one flower to another. Similar results were also observed for C. monogyna in studies conducted in the United Kingdom (Jacobs et al. 2009). Therefore, insect pollinators have a positive influence on the rate of reproduction of these plants in South Africa given that more fruits are set in their presence for both species. According to Love et al. (2016), self-pollination is one of the characteristics that plant species employ to secure the next generation.
Generally, insect pollinators are vital agents in flowering plants contributing to the production of fruits and then seeds. Hence, these insects are important in maintaining plant diversity in natural habitats (Ojija et al. 2019). In South Africa, C. monogyna is visited by various species from order Lepidoptera, Hymenoptera, Coleoptera and Diptera with the Orders Hymenoptera and Diptera contributing more vistors. This is similar in north-western Spain where C. monogyna is native (Guitián and Fuentes 1992). The majority of these frequent visitors on C. monogyna flowers are generalist pollinators such as Apis mellifera, Episyrphus balteatus, Eristalinus taeniops and Acrae horta (Picker and Griffiths 1989; Gaigher et al. 2019; Bonoan et al. 2020; Rossi-Rotondi et al. 2020). However, Cassionympha cassius is the only frequent forest specialist pollinator that visits C. monogyna flowers in this study (Gaigher et al. 2019).
The main insect orders that visit C. mexicana flowers were also from the insect Orders Hymenoptera, Diptera, Lepidoptera and Coleoptera with insects in the Orders Diptera and Hymenoptera contributing most visitors. This corroborates results of Lo et al. (2007) and Ken (2019) who reported that C. mexicana in its native range is primarily pollinated by insects in the Orders Diptera and Hymenoptera. According to Guitián and Fuentes (1992), Blanckenhorn et al. (2010) and Niederegger et al. (2013), Scathophaga species and Calliphora species are the main pollinators of Crataegus mexicana. Indeed, Calliphora vomitoria and Scathophaga stercoraria equally visited the flowers of this plant in this study. Other frequent floral visitors were Lucilia sericata, A. mellifera, E. balteatus and Megachile species. These insects are some of the most vital insect pollinators that increase productivity and fruit-set of many plant species in the wild (Shuttleworth and Johnson 2009). They are therefore referred to as generalist pollinators of C. mexicana flowers (Williams et al. 2014; Bonoan et al. 2020; Rossi-Rotondi et al. 2020).
The distribution of insect visitors on the flowers of C. monogyna and C. mexicana did not generally vary with time of the day. Nevertheless, there were differences in the insect community visiting either Crataegus monogyna or C. mexicana flowers in this study. Indeed, insect species diversity depends on the area or type of habitat the species are found in or surrounded with (Humphrey et al. 1999; Emmanuel and Anuoluwa 2019).The distribution of Crataegus spp. are generally determined by elevation, precipitation and temperature (Jafari et al. 2019; Rafiee mo et al. 2020). Indeed, Crataegus monogyna populations are mostly found in high altitude forests habitat of Hogsback while C. mexicana populations are found in low altitude grassland surrounded by farms in Seymour in the Eastern Cape Province of South Africa. The number of individuals and insect species were higher for C. monogyna than C. mexicana in this study. This may be due to the alteration of the habitat in farmlands providing less suitable habitat for insect pollinators. The majority of the insect visitors found in areas surrounded by farms were flies (Order Diptera). These flies could be attracted by the waste produced by livestock but end up also visiting the flowers of this plant.
Flower traits may influence the visitation rate by certain insect species; this is achieved by creating a communication channel between the flower and pollinator (Bergamo et al. 2016; Fornoff et al. 2017; Reeth et al. 2019). The flower colour acts as a decisive cue for insect pollinators, directly influencing the visitation rate (Bergamo et al. 2016). Our findings showed that the insect visitors for C. monogyna and C. mexicana flowers in South Africa are also the pollinators for these plant species in their native range. These insects are mostly generalist pollinators (Guitián and Fuentes 1992; Lo et al. 2007; Tropical Plants Database 2023). Therefore, these insects may contribute significantly to an increase in the reproduction of these plant species in South Africa in the future.
The results from this study provide important information that will be useful in understanding the ecology and phenology of C. monogyna and C. mexicana in South Africa. This is the first step in understanding the risk posed by the Crataegus species, and in working towards management plans for the invaders. It is therefore, recommended that future studies be conducted for other regions of the country and investigate if pollinators prefer these non-native plants over native species in their natural environment.
Data availability
The dataset generated and analysed during this study are available on https://doi.org/10.6084/m9.figshare.23500998.v1.
References
Alverson E, Sigg J (2008) Crataegus monogyna. California invasive plant council (Cal-IPC). http://ucce.ucdavis.edu/datastore/detailreport.cfm?Username=37&surveynumber=182
Andrikopoulos CJ, Cane JH (2018) Comparative pollination efficacies of five bee species on raspberry. J Econ Entomol 111(6):2513–2519
Ashman T, Knight T, Steets J, Amarasekare P, Burd M, Campbell D, Dudash M, Johnston MO, Mazer S, Mitchell R, Morgan M, Wilson W (2004) Pollen limitation of plant reproduction: ecological and evolutionary causes and consequences. Ecology 85:2408–2421
Bahorun T, Aumjaud E, Ramphul H, Rycha M, Luximon-Ramma A, Trotin F, Aruoma OI (2003) Phenolic constituents and antioxidant capacities of Crataegus monogyna (Hawthorn) callus extracts. Nahrung 47:191–198
Banks NC, Paini DR, Louise BK, Hodda M (2015) The role of global trade and transport network topology in the human-mediated dispersal of alien species. Ecol Lett 18:188–199
Barros L, Carvalho AM, Ferreira ICFR (2010) Comparing the composition and bioactivity of Crataegus Monogyna flowers and fruits used in folk medicine. Phytochem Anal 22:181–188
Bechkri S, Berrehal D, Semra Z, Bachari K, Kabouche A, Kabouche Z (2017) Composition and biological activities of seeds oils of two Crataegus species growing in Algeria. J Mater Environ Sci 8:1023–1028
Bergamo PJ, Rech AR, Brito V, Icius LG, Sazima M (2016) Flower colour and visitation rates of Costus arabicus support the ‘bee avoidance’ hypothesis for red-reflecting hummingbird-pollinated flowers. Funct Ecol 30:710–720
Blackburn T, Pyšek P, Bacher S, Carlton TJ, Duncan R, Jarošík V, Wilson JR, Richardson DM (2011) A proposed unified framework for biological invasions. Trends Ecol Evol 26:333–339
Blackburn TM, Bellard C, Ricciardi A (2019) Alien versus native species as drivers of recent extinctions. Front Ecol Environ 17:203–207
Blanckenhorn WU, Pemberton AJ, Bussière LF, Roembke J, Floate KD (2010) A review of the natural history and laboratory culture methods for the yellow dung fly, Scathophaga stercoraria. J Insect Sci 10:11. https://doi.org/10.1673/031.010.1101
Bonoan RE, Gonzalez J, Starks PT (2020) The perilsof forcing a generalist to be a specialist: lack of dietary essential amino acids impacts honeybee pollen foraging and colony growth. J Apic Res 59:95–103
Brown BJ, Mitchell RJ, Graham SA (2002) Competition for pollination between an invasive species (purple loosestrife) and a native congener. Ecology 83:2328–2336
CABI (2021) Invasive species compendium. https://www.cabi.org/isc/datasheet/16496
CAL-IPC (2018) Crataegus monogyna. https://www.cal-ipc.org/plants/impact/
CARA (2006) Conservation of Agricultural Act (Act No. 43 of 1983). Weed control scheme amendment, governement gazette No. 28847, Vol 491
Cassey P, Blackburn TM, Blackburn TM (2015) Alien species as a driver of recent extinctions. Biol Lett 12:20150623
Chacoff NP, García D, Obeso JR (2008) Effects of pollen quality and quantity on pollen limitation in Crataegus monogyna (Rosaceae) in NW Spain. Flora 203:499–507
Chari LD, Martin GD, Steenhuisen SL, Adams LD, Clark VR (2020) Biology of invasive plants 1. Pyracantha angustifolia (Franch.) C.K. Schneid. Invasive Plant Sci Manag 13(3):120–142
Clarke KR, Gorley RN (2006) PRIMER V6: user manual/tutorial. PRIMER-E Ltd, Plymouth, pp 1–190
Eberly SS (1989) A thorn among the lilies: the hawthorn in medieval love allegory. Folklore 100:41–52
Edwards JE, Brown PN, Talent N, Dickinson TA, Shipley PR (2012) Phytochemistry: a review of the chemistry of the genus Crataegus. Phytochemistry 79:5–26
Emmanuel O, Anuoluwa YO (2019) A study on the diversity and relative abundance of insect fauna in Wukari, Taraba State, Nigeria. Int J Adv Biol Biomed Res 7:129–141
Evans R, Dickinson T (2005) Floral ontogeny and morphology in Gillenia (Spiraeoidea) and subfamily Maloideae C. Weber (Rosaceae). Int J Plant Sci 166:427–447
Fijen TPM, Scheper JA, Boom TM, Janssen N, Raemakers I, Kleijn D (2018) Insect pollination is at least as important for marketable crop yield as plant quality in a seed crop. Ecol Lett 21:1704–1713
Fornoff F, Hartig AKF, Benadi G, Venjakob C, Martin HS, Ebeling A (2017) Functional flower traits and their diversity drive pollinator visitation. Oikos 000:001–011
Gaigher R, Pryke JS, Samways MJ (2019) Divergent fire management leads to multiple beneficial outcomes for butterfly conservation in a production mosaic. J Appl Ecol 56:1322–1332
Gao L, Yu G, Hu F, Li Z, Li W, Peng C (2021) The patterns of male and female flowers in flowering stage may not be optimal resource allocation for fruit and seed growth. Plants 10:2819
Guitián J, Fuentes M (1992) Reproductive biology of Crataegus monogyna in northwestern Spain. Acta Ecol 13:3–11
Hill MP, Hulley PE (2000) Aspects of phenology and ecology of the Southern American weed, Solanum sisymbriifolium, in the Eastern Cape Province of South Africa. Afr Plant Prot 6:53–59
Howlett BG, Evans LJ, Kendall LK, Rader R, Mcbrydie HM, Read SFJ, Cutting BT, Pattemore DE, Willcox BK (2018) Surveying insect flower visitors to crops in New Zealand and Australia. bioRxiv. https://doi.org/10.1101/373126
Humphrey JW, Hawes C, Peace AJ, Ferris-Kaan R, Jukes MR (1999) Relationships between insect diversity and habitat characteristics in plantation forests. For Ecol Manag 113:11–21
Jacobs JH, Clark SJ, Denholm I, Goulson D, Stoate C, Osborne JL, Annals S, December N (2009) Pollination biology of fruit-bearing hedgerow plants and the role of flower-visiting insects in fruit-set. Ann Bot 104:1397–1404
Jafari A, Alipour M, Abbasi M, Soltani A (2019) Distribution modeling of hawthorn (Crataegus azarolus L.) in Chaharmahal and Bakhtiari Province using the maximum entropy method. J Environ Stud 45(2):223–235
Kumar D, Arya V, Bhat A, Ahmad N (2012) The genus Crataegus: chemical and pharmacological perspectives. Braz J Pharmacogn 22:1187–1200
Larue C, Raguso RA, Junker RR (2016) Experimental manipulation of floral scent bouquets restructures flower—visitor interactions in the field. J Anim Ecol 85:396–408
Lo EYY, Stefanović S, Dickinson TA (2007) Molecular reappraisal of relationships between Crataegus and Mespilus (Rosaceae, Pyreae): two genera or one? Syst Bot 32:596–616
Love J, Graham SW, Irwin JA, Ashton PA, Abbott RJ (2016) Self-pollination, style length development and seed set in self-compatible Asteraceae: evidence from Senecio vulgaris L. Plant Ecol Divers 9:371–379
Mandleni B (2011) Impact of climate change and adaptation on cattle and sheep farming in the Eastern Cape Province of South Africa. PhD Thesis, University of South Africa, South Africa, pp 1–154
Menz MHM, Phillips RD, Winfree R, Kremen C, Aizen MA, Johnson SD, Dixon KW (2011) Reconnecting plants and pollinators: challenges in the restoration of pollination mutualisms. Trends Plant Sci 16:4–12
Naghipour AA, Asl ST, Ashrafzadeh MR, Haidarian M (2021) Predicting the potential distribution of Crataegus azarolus L. under climate change in central Zagros, Iran. J Wildl Biodivers 5(4):28–43
New TR (2016) Alien species and insect conservation: alien plants and insect conservation. Springer International Publishing, Cham. https://doi.org/10.1007/978-3-319-38774-1
Niederegger S, Wartenberg N, Spiess R, Mall G (2013) Influence of food substrates on the development of the blowflies Calliphora vicina and Calliphora vomitoria (Diptera, Calliphoridae). Parasitol Res 112:2847–2853
Nieto-Ángel R, Pérez-Ortega SA, Núñez-Colín CA, Martínez-Solís J, González-Andrés F (2009) Seed and endocarp traits as markers of the biodiversity of regional sources of germplasm of tejocote (Crataegus spp.) from Central and Southern Mexico. Sci Hortic 121:166–170
O’Connor R, Kunin WE, Garratt MPD, Potts SG, Roy HE, Andrews C, Jones CM, Peyton JM, Savage J, Harvey MC, Morris RKA, Roberts SPM, Wright I, Vanbergen AJ, Carvell C (2019) Monitoring insect pollinators and flower visitation: the effectiveness and feasility of different survey methods. Methods Ecol Evol 10:2129–2140
Ojija F, Arnold SEJ, Treydte AC (2019) Impacts of alien invasive Parthenium hysterophorus on flower visitation by insects to co-flowering plants. Arthropod Plant Interact 13:719–734
Padron B, Traveset A, Biedenweg T, Di D, Nogales M, Olesen JM (2009) Impact of alien plant invaders on pollination networks in two Archipelagos. PLoS ONE 4:e6275
Pasiecznik N (2018) Crataegus monogyna (hawthorn). https://www.cabi.org/isc/datasheet/16496. Accessed 28 Mar 2018
Ken Fern. (2019). Temperate plant database. https://www.thefern.info.temperate.thefern.info/plant/Crataegus+mexicana
Picker M, Griffiths C (1989) Insects within insects. Sagittarius 4:12–15
Picker M, Griffiths C, Weaving A (2004) Field guide to insects of South Africa, New edn. New Holland publishing, Rivonia
Potts SG, Imperatriz-Fonseca V, Ngo HT, Aizen MA, Biesmeijer JC, Breeze TD, Vanbergen AJ (2016) Safeguarding pollinators and their values to human well-being. Nature 540(7632):220–229
Rafiee mo G, Jafari R, Matinkhah SH, Tarkesh isfahani M, Karimzadeh HR, Jafari Z (2020) Predicting the potential habitat distribution of Crataegus pontica C. Koch, using a combined modeling approach in Lorestan Province. Iran J Appl Ecol 9(2):45–59
Reeth CV, Michel N, Bockstaller C, Caro G (2019) Influences of oilseed rape area and aggregation on pollinator abundance and reproductive success of a co-flowering wild plant. Agr Ecosyst Environ 280:35–42
Rodriguez-Garcia E, Olano JM, Leroux O, Mezquida ET (2019) Deciphering the role of reproductive investment, pollination success and predispersal seed predation on reproductive output in Juniperus thurifera. Plant Ecol Divers 12:37–49
Rossi-Rotondi BA, Videla M, Beccacece HM, Fenoglio MS (2020) New records of the exotic Band-eyed Drone Fly, Eristalinus taeniops (Wiedemann, 1818) (Diptera, Syrphidae), in Argentina. CheckList 16:1523–1529
Saunders ME (2018) Insect pollinators collect pollen from wind-pollinated plants: implications for pollination ecology and sustainable agriculture. Insect Conserv Divers 11:13–31
Shuttleworth A, Johnson SD (2009) New records of insect pollinators for South African asclepiads (Apocynaceae: Asclepiadoideae). S Afr J Bot 75:689–698
Simberloff D (2011) How common are invasion-induced ecosystem impacts? Biol Invasions 13:1255–1268
Simberloff D, Martin J-L, Genovesi P, Maris V, Wardle DA, Aronson J, Courchamp F, Galil B, García-Berthou E, Pascal M, Pyšek P, Sousa R, Tabacchi E, Vilà M (2013) Impacts of biological invasions: what’s what and the way forward. Trends Ecol Evol 28:58–66
Singh R, Negi PS, Dwivedi SK (2018) Indian hawthorn (Pyracantha crenulata). New age herbals. Springer, Singapore, pp 135–149
Spellenberg RCJE, Gil N (2014) Trees of western North America. Princeton University Press, Princeton
Stout JC, Tiedeken EJ (2017) Direct interactions between invasive plants and native pollinators: evidence, impacts and approaches. Funct Ecol 31:38–46
Tropical Plants Database (2023) Ken Fern. Tropical.theferns.info/viewtropical.php?id Crataegus+mexicana
USDA-NRCS (2018) The PLANTS database. USA National Plant Data Team, Greensboro. https://plants.sc.egov.usda.gov
Vanbergen AJ, Baude M, Biesmeijer JC, Britton NF, Brown M, Wright GA (2013) Threats to an ecosystem service: pressures on pollinators. Front Ecol Environ 11(5):251–259
Wardle DA, Bardgett RD, Callaway RM, Van Der Putten WH (2011) Terrestrial ecosystem responses to species gains and losses. Science 332:1273-m1277
Williams KA, Richards CS, Villet MH (2014) Predicting the geographic distribution of Lucilia sericata and Lucilia cuprina (Diptera: Calliphoridae) in South Africa. Afr Invertebr 55:157–170
Acknowledgements
We thank the South African National Biodiversity Institute (SANBI), the National Research Foundation (NRF) and the Govan Mbeki Research and Development Centre (GMRDC) University of Fort Hare for supporting this study financially. Chris of the Waterloo Farm and Hogsback Arboretum club are greatly acknowledged for giving us permissions to conduct this study on their properties. We are also grateful to the editor and two anonymous reviewers whose comments and editorial work greatly improved the manuscript.
Funding
Open access funding provided by University of Fort Hare.
Author information
Authors and Affiliations
Contributions
All the authors contributed to the conception and design of the study. The authors equally contributed to the material preparation, data collection, analysis, drafting, editing and commenting on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Financial support was received from South African National Biodiversity Institute (SANBI), the National Research Foundation (NRF) and the Govan Mbeki Research and Development Centre (GMRDC) University of Fort Hare. The authors have no conflicting interest to declare.
Additional information
Handling Editor: Avery L. Russell.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tshilingalinga, S., Heshula, L.UN.P., Forbanka, D.N. et al. Pollination and floral insect visitors of non-native Crataegus monogyna Jacq and Crataegus mexicana DC (Rosaceae) in the Eastern Cape Province, South Africa. Arthropod-Plant Interactions 17, 581–591 (2023). https://doi.org/10.1007/s11829-023-09991-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-023-09991-8