Abstract
Characteristics of preferred or alternative host plants can impact the population dynamics of polyphagous insects. Chrysodeixis includens has become an important pest in economically important crops such as sunflower, cotton, soybean, and beans. In the present study, we evaluated the effects of basil cultivars Ocimum basilicum (alternative host) on C. includens. For this, larvae were fed daily with bean leaves (preferred host-control) and five basil cultivars (Maria Bonita, Mr. Burns, Italian Large Leaf, Vermelho Rubi, and Cinnamon). Subsequently, development, survival, pupae body mass, reproductive parameters, and life table parameters of insects were evaluated. The populations of C. includens fed with leaves of the Italian Large Leaf cultivar showed similar performance to the preferential host (beans). The other host plants prolonged the duration of the larval stage and negatively affected the reproductive and demographic parameters of C. includens. Mr. Burns negatively affected the development and reduced the reproductive potential of the insect. The cultivars Maria Bonita, Vermelho Rubi, and Cinnamon had a negative effect on the development and reproductive potential of this insect. The results indicate that basil cultivars have a negative effect on the development, longevity, and reproduction of C. includens. The use of basil host plants may be a safe alternative to be used concomitantly with other strategies for the management of C. includens.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plants have a great diversity of direct and indirect defense mechanisms that can affect the performance and maintenance of their consumer populations (Fürstenberg-Hägg et al. 2013; Schuman and Baldwin 2016; War et al. 2012). On the Other hand, herbivorous insects have developed different life histories in relation to the use of their hosts, which can range from monophagous feeding habits, which include the consumption of phylogenetically related plants (e.g., specialists), to the polyphagous habit, which consists of consuming of plants from different families (e.g., generalists) (Ali and Agrawal 2012; Forister et al. 2012). The nutritional quality of alternative hosts can be a major factor for the growth of populations of polyphagous insects (Tikkanen et al. 2000).
Polyphagous herbivores tend to tolerate a wider range of different defensive compounds when at low levels (Zhang et al. 2014). Thus, polyphagy allows herbivores to occur simultaneously in several plants, persisting throughout the growth period of their hosts, even at low population densities (Specht et al. 2015). However, since defenses vary greatly between individuals, cultivars, and plant species; the availability of hosts can interfere in the population dynamics of generalist herbivores. The quality of alternative host plants can change the selection for use by insects, either as food and/or shelter. When alternative hosts are chosen by insects, these can to some degree affect their fecundity, the hatching rate of their eggs, the development of the immature forms (larvae or nymphs), and the emergence and longevity of adults (Andrade et al. 2016; Schuman and Baldwin 2016).
Integrated Pest Management (IPM) systems have considered host plant characteristics as one of the insect control strategies (Fathipour et al. 2019; Jha et al. 2014; Moonga and Davis 2016). Several studies suggest that plants and their compounds act efficiently in the control of arthropod pests (Fathipour et al. 2019; Golikhajeh et al. 2016; Lima et al. 2020; Melo et al. 2018; Pereira et al. 2021). Between the wide range of pests that require control strategies, Chrysodeixis includens (Lepidoptera: Noctuidae)—a polyphagous insect that attacks several crops, with wide distribution in America (Alford and Hammond Junior 1982)—has become a serious phytosanitary problem in areas agricultural in Brazil (EMBRAPA 2013). The increase in attacks by this pest is mainly due to the expansion of planting areas, climatic modifications, indiscriminate use of insecticides and the use of pesticide mixtures for its control (Sosa-Gómez et al. 2010).
Basil plants have been recorded as one of the alternative hosts used by C. includens (Specht et al. 2015). The basil (Ocimum basilicum) is an herbaceous plant, cultivated worldwide, which has a great diversity of cultivars, with distinct physical, morphological, and chemical characteristics (Blank et al. 2004; Costa et al. 2015; Sousa et al. 2021). This plant is recognized for its great bioactive potential, either as an antimicrobial agent (Avetisyan et al. 2017; Oxenham et al. 2005), anthelmintic (Sousa et al. 2021), antioxidant (Avetisyan et al. 2017), and for its insecticidal activity (Chang et al. 2009; Murugan et al. 2007). Given these characteristics, there is no information on the effects of this alternative host plant (O. basilicum) on the biology and reproductive parameters of phytophagous insects such as C. includens.
Thus, the hypothesis of this research was that cultivars of basil can be used by C. includens as alternative hosts, can these cause negative effects on the performance and biology of this species. Thus, the objective of this study was to evaluate the biological development, reproductive and demographic parameters of C. includens fed with O. basilicum cultivars, aiming to understand the effect of alternative host plants on the population dynamics of this pest.
Materials and methods
Cultivation of host plants
To evaluate the development and reproductive and demographic parameters of C. includens, five basil cultivars and a variety of one of its preferred hosts—common bean (Phaseolus vulgaris) were used. Basil cultivars were selected based on visual variations in color and morphology, including: Maria Bonita, Mr. Burns, Italian Large Leaf, Vermelho Rubi, and Cinnamon. In addition, they are cultivars commonly grown in the region.
The cultivar Maria Bonita comes from accession PI 197442, from the North Central Regional PI Station Germoplasm bank, Iowa State University, USA. Currently, this cultivar has been maintained in the Germoplasm bank located at the Fazenda Experimental Campus Rural of the UFS, in the Municipality of São Cristóvão, SE. Initially, seeds of this cultivar were obtained, which were subsequently sown to obtain the plants to be used in the experiments. The other varieties composed by Mr. Burns, Italian Large Leaf, Cinnamon (Richters Herbs™), and Vermelho Rubi (Isla sementes LTDA) were commercially obtained.
The sowing of basil cultivars was carried out in 128-cell seedbeds with substrate in the proportion of 2:1 (coconut powder and humus) and maintained for 15 days. Subsequently, the seedlings were transplanted into 300 ml pots with substrate in the proportion of 2:1 (sterilized soil in oven at 100 °C) and humus (worm humus—3 kg GNÚMUS™). The beans were sown directly in 300 ml pots, following the same substrate proportion. The plants were sprinkled daily twice a day (5 min/irrigation), in the morning and afternoon and were kept in a greenhouse for 45 days until use in the experiments.
Rearing of Chrysodeixis includens
The insects used in the experiments were collected in the municipality of Fatima in Bahia—Brazil (10° 36′ 00″ S and 38° 13′ 00″ W). To make the sample representative, more than 1000 insects including eggs, larvae, pupae, and adults of C. includens were collected from five different cultivation areas in the municipality. Then all this material was packed in polyethylene plastic bags and submitted to the laboratory. Upon arrival from the field, the insects were kept in air-conditioned rooms (25 ± 1 °C, RH 70 ± 5%, 12 h photoperiod) in the Integrated Pest Management laboratory, at the Universidade Federal de Sergipe (UFS), São Cristóvão-SE (10° 54’ S, 37° 04’ W, 7 m altitude), Brazil. Before the insects were used in the experiments, they were quarantined to verify that they were not parasitized or contaminated with any type of pathogen.
Adults of C. includens were kept in 2 L plastic pots fully lined with sulfite paper (oviposition substrate). The bottom of the pot consisted of a Petri dish (14 × 1.5 cm) and the top sealed with organza-like fabric. 10% honey solution was provided ad libitum for adult feeding. Eggs were removed daily and placed in fully sealed 2 L plastic containers until the larvae hatched. When hatching the larvae were individualized in 50 ml plastic pots where they were fed with an artificial diet (Greene et al. 1976). When they reached the pupa stage were sexed after 24 h and placed in 2 L plastic pots lined with paper towels, staying in these until the emergence of adults, to start a new cycle began.
Experiments
Development, survival, pupae body mass, reproductive parameters, and life table parameters were evaluated in cohorts of C. includens fed with different cultivars of basil (Maria Bonita, Mr. Burns, Italian Large Leaf, Vermelho Rubi, and Cinnamon) and the preferred host (bean). The experimental design was completely randomized with 10 repetition, with each repetition composed of four individuals, each experiment was repeated 3 times in space and time, totaling 120 individuals per host plant (720 individuals in total).
Development and reproduction
The eggs and later the larvae were placed individually in a Petri dish (9 × 1.5 cm) containing filter paper moistened with distilled water (300 μl) and leaf discs (587.9 mm2) of the host plants. The discs were provided ad libitum for larval feeding. The plates were kept in an acclimatized room (25 ± 1 °C, RH 70 ± 5%, and 12 h photoperiod). The period of development and longevity were evaluated daily until the death of all tested insects.
After 24 h reaching the pupal stage, the individuals of C. includens were sexed and weighed. Adult females and males were kept isolated for two days until sexual maturity. Subsequently, ten couples from each of the treatments were separated into 2 L pots and fed ad libitum wih a 10% honey solution. Chrysodeixis includens adults were monitored daily until all individuals died. The following were recorded: i) the pre-oviposition, oviposition, and pos-oviposition periods, ii) the longevity of females and males, iii) the numbers of eggs and neonatal larvae per female, iv) the incubation period of the eggs, and v) the viability of eggs.
Life table parameters
From the survival and reproduction data of C. includens fed with the different host plants, the parameters were calculated: x = average age of insects (from egg stage), lx = survival rate (from age zero to initial age x), mx = specific fertility (number of females produced per surviving female in the age range x); lx.mx = total number of females born at age x.
For the fertility life table were calculated: the net reproductive rate (R0) (number of females added per female during her lifetime), the intrinsic rate of increase (rm) (rate of population increase per unit time), the finite rate of increase (λ), and the mean generation time (T) (time between the birth of parents and children) (Krebs 2009). The parameters were calculated by the following equations:
Statistical analysis
For all analyses, the Kolmogorov–Smirnov test was initially applied to verify the normality of the data and the Levene test to verify the homogeneity of the variance. Data on the duration of larval stages, instars (1st to 9th), pre-pupa, pupa, and adults; and the mass of females and males of C. includens were submitted to analysis of variance (ANOVA) followed by Dunnett’s test (p < 0.05) to compare basil cultivars with the preferred host (beans), using the PROC GLM procedure, with Dunnett (SAS Institute 2008). To verify differences between the mass of female and male pupae, for the same host plant, the paired t test was used (p < 0.05). The effects of host plants on reproductive and demographic parameters (life table) were compared using the 95% confidence interval (95%CI). Means were compared using the non-overlapping CI95 criterion with the origin of the interval. The life table parameters were estimated using the Jackknife technique in the SAS statistical program (Maia et al. 2000).
Results
Development and longevity
The duration of the larval stages (Fig. 1a) (F5,136 = 45.95; p < 0.001), pre-pupa (Fig. 1b) (F5,136 = 14.09; p < 0.001), pupa (Fig. 1c) (F5,136 = 7.38; p < 0.001), and adult (Fig. 1d) (F5,136 = 7.01; p < 0.001) of C. includens varied between host plants.
Duration of larval (a), pre-pupa (b), pupa (c) stages, longevity of adults (d) and larvae from 1st to 9th instars of Chrysodeixis includens (e) fed with bean leaves (CB) (control) and five basil cultivars (MBo Maria Bonita, MBu Mr. Burns, ILL Italian Large Leaf, VR Vermelho Rubi, and Ci Cinnamon). The error lines represent the 10th and 90th percentiles and the ends of the box the 25th and 75th percentiles. The solid and dashed line inside the box represent the median and mean, respectively. Means followed by an asterisk differ from the control (beans) by Dunnett’s test at p < 0.05
All basil cultivars increased the duration of the larval stage of C. includens, except Italian Large Leaf, which did not differ from the control (bean). On average, the Maria Bonita, Mr. Burns, Vermelho Rubi, and Cinnamon increased by 8.9; 3.4; 1.8; and 5.4 days the duration of the larval stage (Fig. 1a).
Chrysodeixis includens larvae fed on bean plants originated pre-pupa with 1.75 ± 0.054 days of duration. This phase was increased by 20.5% and reduced by 24.3%, in the cultivars Maria Bonita and Vermelho Rubi, respectively, when compared to the control (Fig. 1b). Differences in the duration of the pupal stage were observed only for the cultivar Italian Large Leaf, with an increase of about 2 days (Fig. 1c). The longevity of adults originating from larvae that fed on Mr. Burns, Vermelho Rubi, and Cinnamon was reduced by 2.2; 4.1; and 3.9 days, respectively (Fig. 1d).
Overall, C. includens larvae passed through seven instars, regardless of the host plant. However, only 5.5 and 0.8% of the larvae reached the 8th and 9th instars, respectively (Fig. 1e). The duration of the 2nd instar (F5,144 = 9.04; p < 0.001), 3rd instar (F5,135 = 17.64; p < 0.001), 5th instar (F5,143 = 3.33; p = 0.007), 6th instar (F5,144 = 9.15; p < 0.001), and 7th instar (F5,110 = 4.25; p = 0.001) of C. includens varied between the host plants. The increase in the larval stage was largely explained by the increase in the duration of the 7th instar (Fig. 1e). 7th instar larvae of C. includens fed on bean plants had a duration of 3.73 ± 0.3 days. On average, the Maria Bonita, Mr. Burns, Vermelho Rubi, and Cinnamon increased by 51; 37; 45; and 63% the duration of the 7th larval instar (Fig. 1e).
Mass of the insects
The mass of female (F5,97 = 34.43; p < 0.001) and males (F5,123 = 29.41; p < 0.001) of C. includens varied between host plants. The basil cultivars Italian Large Leaf and Cinnamon increased the mass of females (37 and 36 mg) and males (41 and 27 mg) (Fig. 2). Males from larvae that fed on the basil cultivar Mr. Burns had their mass reduced by 38 mg (Fig. 2). The mass of males that fed on beans (t = − 2.367; p = 0.021) and the Italian Large Leaf (t = − 2.070; p = 0.043) cultivar was about 10% greater than the mass of females (Fig. 2).
Mass of female and male pupae of Chrysodeixis includens fed with bean (CB) leaves (control) and five basil cultivars (MBo Maria Bonita, MBu Mr. Burns, ILL Italian Large Leaf, VR Vermelho Rubi, and Ci Cinnamon). The error lines represent the 10th and 90th percentiles and the ends of the box the 25th and 75th percentiles. The solid and dashed line inside the box represent the median and mean, respectively. Means followed by an asterisk differ from the control by Dunnett’s test at p < 0.05 and means followed by different letters indicate between sex differences by the paired t test at p < 0.05
Reproductive parameters
Chrysodeixis includens larvae that fed on Maria Bonita (n = 2), Mr. Burns (n = 2), Vermelho Rubi (n = 2), and Cinnamon (n = 5) basil cultivars resulted in fewer couples formed. The females formed from larvae that fed on the cultivars Vermelho Rubi and Cinnamon did not lay eggs (Fig. 3a, e, b). The period of pre-oviposition and incubation of eggs of females from larvae that fed on beans and Italian Large Leaf did not differ, as well as the period of oviposition between beans and the cultivars Maria Bonita, Mr. Burns, and Italian Large Leaf (Fig. 3a). The eggs laid by the females formed by larvae that fed on the cultivars Maria Bonita and Mr. Burns did not hatch (Fig. 3b). The numbers of eggs (324 and 204 eggs/female) and larvae per female did not differ between the common bean and the Italian Large Leaf basil cultivar. These eggs had a viability of 68 and 60%, respectively (Fig. 3b).
Duration of pre-oviposition, oviposition, and egg incubation periods (a) and average number of eggs and larvae per female (b) of Chrysodeixis includens fed on bean leaves (CB) (control) and five basil cultivars, Maria Bonita (MBo), Mr. Burns (MBu), Italian Large Leaf (ILL), Vermelho Rubi (VR), and Cinnamon (Ci). n = number of couples formed. Error bars represent the confidence interval at 95% probability
Demographic parameters
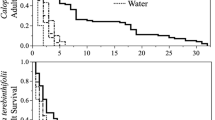
The survival rate (lx) of C. includens was higher for insects that fed on bean leaves (Fig. 4a) and the basil cultivar Italian Large Leaf (Fig. 4d). All demographic parameters studied do not vary between individuals of C. includens that fed on the leaves of these host plants (Fig. 4a, d). Individuals of C. includens fed on Vermelho Rubi leaves showed lower longevity (9 days) (Fig. 4e) and the cohort that presented the highest longevity was that of insects that fed on Cinnamon plants (21st day) (Fig. 4f).
Survival rate (lx) and specific fecundity (mx) of Chrysodeixis includens fed with bean leaves (control) (a) and five basil cultivars Maria Bonita (b), Mr. Burns (c), Italian Large Leaf (d), Vermelho Rubi (e) and Cinnamon (f), where n = number of pairs formed. R0 = net reproductive rate, rm = intrinsic rate of increase, λ = finite rate of increase, and T = mean generation time; ± the confidence interval at 95% probability
The specific fertility (mx) of C. includens fed on bean leaves (Fig. 4a) was higher when compared to the other host plants (Fig. 4b–f). Basil cultivars Vermelho Rubi and Cinnamon caused loss of reproductive capacity in C. includens females, and therefore life table parameters were not estimated (Fig. 4e, f). The other cultivars reduced the net reproductive rate (R0), the intrinsic rate of increase (rm), and the finite rate of increase (λ). The R0, rm, and λ also did not differ between Mr. Burns and Italian Large Leaf (Fig. 4a–c). The mean generation time (T) was the same for the cultivars Maria Bonita and Mr. Burns (Fig. 4d). The evaluation of demographic parameters is important to know the possible negative effects on arthropods population dynamics.
Discussion
The availability (e.g., nutritional quality and defenses) of alternative host plants can determine the growth potential of pests of agricultural importance. This is the first study to analyze the performance of C. includens in alternative hosts of basil cultivars. In general, our results show that the use of basil cultivars by C. includens negatively affected the development and reproductive and demographic parameters of this polyphagous herbivore.
The greater success of C. includens in common bean plants (P. vulgaris) can be explained by the adaptability of this herbivore to the defenses of its preferentially used plants. Domesticated plants may have a trade-off between defense and reproduction (Andrade et al. 2016; Hernández-Cumplido et al. 2021; Schuman and Baldwin 2016). As traits with greater palatability and productivity are selected in the field, there is a parallel loss of desirable traits to stop herbivory, such as morphological attributes and secondary metabolites (Tikkanen et al. 2000). Comparatively, P. vulgaris plants may have gone through more improvement processes when compared to O. basilicum, which explains the better performance of C. includens in its preferred host.
In turn, basil cultivars present aromatic volatiles, represented by the wide variety of terpenoid compounds present in their essential oils. Such compounds can trigger repellency, toxicity, attraction, or influence the feeding rates of herbivorous insect (Baldin et al. 2015; Lu et al. 2021; Moura et al. 2021; Papanastasiou et al. 2020; Tak and Isman 2016). Despite the basil cultivars being consumed by C. includens, these alternative plants increased the duration of the insect larval stage, except for Italian Large Leaf, which did not differ from the control (bean). This delay in development time may be due to nutritional factors and secondary substances present in plants that interfere with metabolism and, consequently, digestion and insect development (Baldin et al. 2019). Terpenoid compounds present in basil such as geraniol, linalool, eugenol, and citral (Guedes et al. 2020; Moore et al. 2014; Praveena and Sanjayan 2011; Tak et al. 2017; Vargas-Méndez et al. 2019; Melo et al. 2020) have proven insecticidal activity. High concentrations of linalool, for example—a compound present in all basil cultivars analyzed—is a parameter recognized for negatively affecting the survival and growth of Lepidoptera (Cruz et al. 2017).
However, although the larval parameters are related to the nutritional quality and defenses of the host plant, they may not be good predictors of insect fitness (Chen et al. 2017). On the other hand, in general, the increase in pupal mass is related to a greater fitness of the insect and may indicate an adequacy of the host plant (Nguyen et al. 2019). Here we verified that the mass of female and male pupae varied between the host plants, being higher in the Italian Large Leaf and Cinnamon cultivars. Nguyen et al. (2019), Reported that diet composition is an important factor that influences pupal mass, with protein-rich diets generating heavier pupae, while high-sugar diets generate lighter pupae. Insect populations that grew on the Italian Large Leaf cultivar also showed reproductive success similar to that observed for populations maintained on beans plants (Figs. 3, 4). Different hosts can positively affect oviposition and fecundity of females due to the high protein content absorbed by the insect as well as the low amount of active compounds that could inhibit the hatching of eggs (Guedes et al. 2020).
Life table studies are an important parameter that evaluates the growth, development, and survival of insects (Chen et al. 2017). Positive values of R0 > 1 and rm > 0, indicate host in which the herbivore may have potential for population growth (Chen et al. 2017). Our results show that populations of C. includens have potential for growth in Italian Large Leaf basil cultivars (R0 = 37.4 and rm = 0.129) and Mr. Burns (R0 = 10.3 and rm = 0.082). The values of these parameters found for Mr. Burns, although lower than that observed for populations that grew with beans, indicate susceptibility of this alternative host to the attack of C. includens (Farahani et al. 2012). Thus, despite C. includens presenting a reduction in reproductive parameters in relation to beans, the Mr. Burns and Italian Large Leaf, may allow the maintenance of populations of this herbivore in the absence of bean plants.
In general, polyphagous insects can perceive the signals emitted by different hosts and present a longer time to choose than monophagous insects (Salazar et al. 2016). Thus, agroecosystems that maintain a greater diversity of alternative plants, with different physical, morphological, and chemical characteristics, could result in a reduction of herbivore attack on crops (Salazar et al. 2016). The use of alternative hosts that reduce population growth rates could be a strategy used concomitantly with other tactics in Integrated Pest Management.
Our results showed that between the basil cultivars evaluated, the populations of C. includens fed with leaves of the Italian Large leaf cultivar showed similar performance to the preferential host (beans). The cultivars Mr. Burns, Maria Bonita, Vermelho Rubi, and Cinnamon may play a key role in breaking the cycle of C. includens, since they had a negative effect on the reproductive potential of this insect. Thus, those cultivars that gave low performance to C. includens could be used in intercropping with beans. Concomitantly with the use of other strategies, the offer of basil plants as alternative hosts to C. includens could contribute to a more efficient and safe management of this pest.
The intercropping of plants is a strategy that can be used to improve the growth of both species, in this case, beans and basil. Companion plants can directly repel pest insect species and/or indirectly control them by attracting natural enemies that kill them (Parker et al. 2013).
In addition, within a context related to pest management, it appears that these plants can increase their negative effects on herbivorous insects through attracting natural enemies, thus strengthening biological control services, when these plants are used in consortium with cultivated plants (Laffon et al. 2022).
Future studies will be able to assess whether the specific compounds present in basil leaves that directly interfere with the biology and demographic parameters of C. includens can promote the attraction of natural enemies.
Conclusion
Our results show that the vast majority of cultivars of alternative hosts caused an increase in the larval period, with the except of the Italian Large Leaf. Populations of C. includens that fed on this cultivar showed very similar performance to the larvae that fed on beans. The other cultivars, in addition to prolonging the larval period of C. includens, ended up negatively affecting their reproductive and demographic parameters. Thus, the use of basil plants can be a safe alternative to be used in association with other control measures to be used in the management of C. includens. This study is extremely important, as it is the first to demonstrate the effects of alternative hosts (basil cultivars) on the reproductive and demographic parameters of a pest (C. includens), and little information is available in the literature in this regard.
Data availability
The data and materials used to support the findings of this study are available from the corresponding author upon request.
References
Alford AR, Hammond Junior AM (1982) Plusiinae (Lepidoptera: Noctuidae) populations in Louisiana soybean ecosystems as determined with looplure-baited traps. J Econ Entomol 75:647–650
Ali JG, Agrawal AA (2012) Specialist versus generalist insect herbivores and plant defense. Trends Plant Sci 17:293–302. https://doi.org/10.1016/j.tplants.2012.02.006
Andrade K, Bueno AF, Silva DM, Stecca CS, Pasini A, Oliveira MCN (2016) Bioecological characteristics of Chrysodeixis includens (Lepidoptera: Noctuidae) fed on different hosts. Austral Entomol 55:449–454. https://doi.org/10.1111/aen.12208
Avetisyan A, Markosian A, Petrosyan M, Sahakyan N, Babayan A, Aloyan S, Trchounian A (2017) Chemical composition and some biological activities of the essential oils from basil Ocimum different cultivars. BMC Complement Altern Med 17:1–8. https://doi.org/10.1186/s12906-017-1587-5
Baldin ELL, Aguiar GP, Fanela TLM, Soares MCE, Groppo M, Crotti AEM (2015) Bioactivity of Pelargonium graveolens essential oil and related monoterpenoids against sweet potato whitefly, Bemisia tabaci biotype B. J Pest Sci 88:191–199. https://doi.org/10.1007/s10340-014-0580-8
Baldin ELL, Vendramim JD, Lorenção AL (2019) Resistência de plantas a insetos: Fundamentos e Aplicações. Fundação Estud. Agrários Luiz Queiroz-FEALQ 1–493.
Blank AF, Carvalho Filho JLS, Santos Neto AL, Alves PB, Arrigoni-Blank MF, Silva-Mann R, Mendonça MC (2004) Caracterização morfológica e agronômica de acessos de manjericão e alfavaca. Hortic Bras 22:113–116. https://doi.org/10.1590/s0102-05362004000100024
Chang CL, Cho IIK, Li QX (2009) Insecticidal activity of basil oil, trans-anethole, estragole, and linalool to adult fruit flies of Ceratitis capitata, Bactrocera dorsalis, and Bactrocera cucurbitae. J Econ Entomol 102:203–209. https://doi.org/10.1603/029.102.0129
Chen Q, Li N, Wang X, Ma L, Huang JB, Huang GH (2017) Age-stage, two-sex life table of Parapoynx crisonalis (Lepidoptera: Pyralidae) at different temperatures. PLoS ONE. https://doi.org/10.1371/journal.pone.0173380
Costa AS, Arrigoni-Blank MDF, Carvalho Filho JLS, Santana ADD, Santos DDA, Alves PB, Blank AF (2015) Chemical diversity in basil (Ocimum sp.) Germplasm. Sci World J 1–9. https://doi.org/10.1155/2015/352638
Cruz GS, Wanderley-Teixeira V, Silva LM, Dutra KA, Guedes CA, Oliveira JV, Navarro DMAF, Araújo BC, Teixeira ÁAC (2017) Chemical composition and insecticidal activity of the essential oils of Foeniculum vulgare Mill., Ocimum basilicum L., Eucalyptus staigeriana F. Muell. ex Bailey, Eucalyptus citriodora Hook and Ocimum gratissimum L. and. J Essent Oil Bear Plants 20:1360–1369. https://doi.org/10.1080/0972060X.2017.1383192
EMBRAPA (2013) Tecnologias de Produção de Soja - Região Central do Brasil 2005, Sistemas de Produção 16: Tecnologias de produção de soja - Região Central do Brasil 2014.
Farahani S, Talebi AA, Fathipour Y (2012) Life table of Spodoptera exigua (Lepidoptera: Noctuidae) on five soybean cultivars. Psyche: A Journal of Entomology 1–7. https://doi.org/10.1155/2012/513824
Fathipour Y, Kianpour R, Bagheri A, Karimzadeh J, Hosseininaveh V (2019) Bottom-up effects of Brassica genotypes on performance of diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae). Crop Prot 115:135–141. https://doi.org/10.1016/j.cropro.2018.09.020
Forister ML, Dyer LA, Singer MS, Stireman JO, Lill JT (2012) Revisiting the evolution of ecological specialization, with emphasis on insect-plant interactions. Ecology 93:981–991. https://doi.org/10.1890/11-0650.1
Fürstenberg-Hägg J, Zagrobelny M, Bak S (2013) Plant defense against insect herbivores. Int J Mol Sci 14:10242–10297. https://doi.org/10.3390/ijms140510242
Golikhajeh N, Naseri B, Razmjou J (2016) Effect of geographic population and host cultivar on demographic parameters of Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae). J Asia-Pac Entomol 19:743–751. https://doi.org/10.1016/j.aspen.2016.07.002
Greene GL, Leppla NC, Dickerson WA (1976) Velvetbean Caterpillar: a rearing procedure and artificial medium. J Econ Entomol 69:487–488. https://doi.org/10.1093/jee/69.4.487
Guedes CA, Teixeira VW, Dutra KA, Navarro DMAF, Cruz GS, Lapa Neto CJC, Correia AA, Sandes JM, Brayner FA, Alves LC, Teixeira ÁAC (2020) Evaluation of Piper marginatum (Piperales: Piperaceae) oil and geraniol on the embryonic development of Spodoptera frugiperda (Lepidoptera: Noctuidae) in comparison to formulated products. J Econ Entomol 113:239–248. https://doi.org/10.1093/jee/toz300
Hernández-Cumplido J, Rodriguez-Saona C, Ruíz-Rodríguez CE, Guevara-Fefer P, Aguirre-Paleo S, Miranda Trejo S, Callejas-Chavero A (2021) Genotypic variation in plant traits, chemical defenses, and resistance against insect herbivores in avocado (Persea americana) across a domestication gradient. Front Agron 2:1–12. https://doi.org/10.3389/fagro.2020.616553
Jha RK, Tuan S-J, Chi H, Tang L-C (2014) Life table and consumption capacity of corn earworm, Helicoverpa armigera, fed asparagus, Asparagus officinalis. J Insect Sci 14:1–17. https://doi.org/10.1673/031.014.34
Krebs CJ (2009) Ecology: the experimental analysis of distribution and abundance. Harper Collins College Publishers, New York
Laffon L, Bischoff A, Gautier H, Gilles F, Gomez L, Lescourret F, Franck P (2022) Conservation biological control of codling moth (Cydia pomonella): effects of two aromatic plants, basil (Ocimum basilicum) and french marigolds (Tagetes patula). InSects 13:908. https://doi.org/10.3390/insects13100908
Lima APS, Santana EDR, Santos ACC, Silva JE, Ribeiro GT, Pinheiro AM, Santos ITBF, Blank AF, Araújo APA, Bacci L (2020) Insecticide activity of botanical compounds against Spodoptera frugiperda and selectivity to the predatory bug Podisus nigrispinus. J. Crop Protect 136:105230. https://doi.org/10.1016/j.cropro.2020.105230
Lu XX, Hu NN, Du YS, Almaz B, Zhang X, Du SS (2021) Chemical compositions and repellent activity of Clerodendrum bungei Steud. essential oil against three stored product insects. DARU J Pharm Sci 29:469–475. https://doi.org/10.1007/s40199-021-00398-5
Maia AHN, Luiz AJB, Campanhola C (2000) Statistical inference on associated fertility life table parameters using jackknife technique: computational aspects. J Econ Entomol 93:511–518. https://doi.org/10.1603/0022-0493-93.2.511
Melo CR, Picanço MC, Santos AA, Santos IB, Pimentel MF, Santos ACC, Blank AF, Araújo APA, Cristaldo PF, Bacci L (2018) Toxicity of essential oils of Lippia gracilis chemotypes and their major compounds on Diaphania hyalinata and non-target species. Crop Prot 104:47–51. https://doi.org/10.1016/j.cropro.2017.10.013
Melo CR, Oliveira BMS, Santos ACC, Silva JE, Ribeiro GT, Blank AF, Araújo APA, Bacci L (2020) Synergistic effect of aromatic plant essential oils on the ant Acromyrmex balzani (Hymenoptera: Formicidae) and antifungal activity on its symbiotic fungus Leucoagaricus gongylophorus (Agaricales: Agaricaceae). Environ Sci Pollut Res 27:17303–17313. https://doi.org/10.1007/s11356-020-08170-z
Moonga MN, Davis JA (2016) Partial life history of Chrysodeixis includens (Lepidoptera: Noctuidae) on summer hosts. J Econ Entomol 109:1713–1719. https://doi.org/10.1093/jee/tow156
Moore BD, Andrew RL, Külheim C, Foley WJ (2014) Explaining intraspecific diversity in plant secondary metabolites in an ecological context. New Phytol 201:733–750. https://doi.org/10.1111/nph.12526
Moura ES, Faroni LRD, Heleno FF, Rodrigues AAZ (2021) Toxicological stability of Ocimum basilicum essential oil and its major components in the control of Sitophilus zeamais. Molecules 26:1–11. https://doi.org/10.3390/molecules26216483
Murugan K, Murugan P, Noortheen A (2007) Larvicidal and repellent potential of Albizzia amara Boivin and Ocimum basilicum Linn against dengue vector, Aedes aegypti (Insecta:Diptera:Culicidae). Biores Technol 98:198–201. https://doi.org/10.1016/j.biortech.2005.12.009
Nguyen B, Ponton F, Than A, Taylor PW, Chapman T, Morimoto J (2019) Interactions between ecological factors in the developmental environment modulate pupal and adult traits in a polyphagous fly. Ecol Evol 9:6342–6352. https://doi.org/10.1002/ece3.5206
Oxenham SK, Svoboda KP, Walters DR (2005) Antifungal activity of the essential oil of basil (Ocimum basilicum). J Phytopathol 153:174–180
Papanastasiou SA, Loannou CS, Papadopoulos NT (2020) Oviposition-deterrent effect of linalool—a compound of citrus essential oils—on female Mediterranean fruit flies, Ceratitis capitata (Diptera: Tephritidae). Pest Manag Sci 76:3066–3077. https://doi.org/10.1002/ps.5858
Parker JE, Snyder WE, Hamilton GC, Rodriguez-Saona C (2013) Companion planting and insect pest control. Weed Pest Control. https://doi.org/10.5772/55044.
Pereira JM, Santos TTM, Soliman EP, Dias TKR, Baldin ELL, Wilcken CF (2021) Survival and performance of Sarsina violascens (Lepidoptera:Lymantriidae) larvae on Eucalyptus species and hybrids. Phytoparasitica 50:13–20. https://doi.org/10.1007/s12600-021-00933-9
Praveena A, Sanjayan KP (2011) Inhibition of acetylcholinesterase in three insects of economic importance by linalool, a monoterpene phytochemical. Entomol Res 340–345.
Salazar D, Jaramillo A, Marquis RJ (2016) The impact of plant chemical diversity on plant–herbivore interactions at the community level. Oecologia 181:1199–1208. https://doi.org/10.1007/s00442-016-3629-y
SAS Institute (2008). Introduction to statistical modeling with SAS/STAT software (Book Excerpt). SAS/STAT 9.3 User’s Guid.
Schuman MC, Baldwin IT (2016) The layers of plant responses to insect herbivores. Annu Rev Entomol 61:373–394. https://doi.org/10.1146/annurev-ento-010715-023851
Sosa-Gómez DR, Lastra CCL, Humber RA (2010) An overview of arthropod-associated fungi from Argentina and Brazil. Mycopathologia 170:61–76. https://doi.org/10.1007/s11046-010-9288-3
Sousa AIP, Silva CR, Costa-Júnior HN, Silva NCS, Pinto JAO, Blank AF, Soares AMS, Costa-Júnior LM (2021) Essential oils from Ocimum basilicum cultivars: analysis of their composition and determination of the effect of the major compounds on Haemonchus contortus eggs. J Helminthol 95:1–5. https://doi.org/10.1017/S0022149X21000080
Specht A, Moraes SVP, Sosa-Gómez DR (2015) Host plants of Chrysodeixis includens (Walker) (Lepidoptera, Noctuidae, Plusiinae). Revista Brasileira De Entomologia 59:343–345. https://doi.org/10.1016/j.rbe.2015.09.002
Tak JH, Isman MB (2016) Metabolism of citral, the major constituent of lemongrass oil, in the cabbage looper, Trichoplusia ni, and effects of enzyme inhibitors on toxicity and metabolism. Pestic Biochem Physiol 133:20–25. https://doi.org/10.1016/j.pestbp.2016.03.009
Tak JH, Jovel E, Isman MB (2017) Effects of rosemary, thyme and lemongrass oils and their major constituents on detoxifying enzyme activity and insecticidal activity in Trichoplusia ni. Pestic Biochem Physiol 140:9–16. https://doi.org/10.1016/j.pestbp.2017.01.012
Tikkanen AO, Niemelä P, Keränen J (2000) International association for ecology growth and development of a generalist insect herbivore, Operophtera brumata, on original and alternative host plants. Oecologia 122:529–536
Vargas-Méndez LY, Sanabria-Flórez PL, Saavedra-Reyes LM, Merchan-Arenas DR, Kouznetsov VV (2019) Bioactivity of semisynthetic eugenol derivatives against Spodoptera frugiperda (Lepidoptera: Noctuidae) larvae infesting maize in Colombia. Saudi J Biol Sci 26:1613–1620. https://doi.org/10.1016/j.sjbs.2018.09.010
War AR, Paulraj MG, Ahmad T, Buhroo AA, Hussain B, Ignacimuthu S, Sharma HC (2012) Mechanisms of plant defense against insect herbivores. Plant Signal Behav 7:1306–1320
Zhang K, Di N, Ridsdill-Smith J, Zhang BW, Tan XL, Cao HH, Liu YH, Liu TX (2014) Does a multi-plant diet benefit a polyphagous herbivore? A case study with Bemisia tabaci. Entomol Exp Appl 152:148–156. https://doi.org/10.1111/eea.12210
Funding
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico – Brazil (CNPq), the Fundação de Apoio à Pesquisa e a Inovação Tecnológica do Estado de Sergipe (FAPITEC/SE) – Brazil, the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) – Finance Code 001, and the Financiadora de Estudos e Projetos – Brazil (FINEP).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. ACCS was responsible for data curation, investigation, writing—original draft. APAA was responsible for writing-review & editing, visualization. JdOD was responsible for data curation and investigation. JEdS was responsible for investigation, writing—review and editing. AMP was responsible for data curation and investigation. VSA was responsible for data curation and investigation. NCS was responsible for data curation and investigation. SRSAT was responsible for data curation and investigation. AFB was responsible for resource and investigation. LB was responsible for conceptualization, methodology, validation, formal analysis, writing-review and editing, supervision. The final version of the manuscript was reviewed and approved by all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to participate
Not applicable.
Consent to publish
All authors have given their consent to publish this research article.
Ethical approval
Not applicable.
Additional information
Handling Editor: Xoaquín Moreira.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Santos, A.C.C., Araújo, A.P.A., Dantas, J.O. et al. Alternative hosts can affect the development, reproduction, and demographic parameters of Chrysodeixis includens. Arthropod-Plant Interactions 17, 351–361 (2023). https://doi.org/10.1007/s11829-023-09969-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-023-09969-6