Abstract
Climatic parameters are able to influence the timing of phenological events affecting the degree of synchrony among plant species, their interactions, and reproductive success. Shrubs of Malpighiaceae family in the Brazilian Tropical Savanna present sequential flowering phenology. We verified variations in climatic factors (temperature and precipitation) over a period of 10 years (2005–2014) and correlated them with the onset of flowering of four of these Malpighiaceae species. Furthermore, we tested whether the phenological synchronization among species has changed over time affecting the herbivory and fruit set. Herbivory and fruit production were recorded during three reproductive seasons (2008/2009, 2011/2012, 2013/2014). We developed a mathematical model to estimate the flower and fruit production in response to phenological changes for the next 5 years. Results show that climatic factors changed, influencing the onset of species flowering. The degree of overlap among species also changed and the effects on species interactions were species specific. The mathematical model successfully presented a tendency on flower and fruit production contributing to the predictions of the outcomes in response to phenological changes. We confirm the effects of climate changes on plant phenological events and the importance of feature plasticity for better performance of species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Flowering phenology is a critical life-history trait in angiosperms and has potential significance for plant ecology and evolution (e.g., Amasino 2010; Torres and Galetto 2011). Due to its influence on a wide range of ecological processes, including plant interactions, and reproductive success (Pilson 2000; Kawagoe and Kudoh 2010; Del-Claro et al. 2016), the pattern of flowering is one of the most investigated aspects of plant phenology (Hegland et al. 2009; Vilela et al. 2014), especially nowadays with the occurrence of noticeable climate changes (Franks et al. 2013; CaraDonna et al. 2014). Shifts in the timing of flowering have great consequences on plant developmental traits and interactions with mutualistic (pollinators and seed dispersers) and antagonistic (herbivores) partners which may affect plant success and survival rates of species (Herrera et al. 2002; Memmott et al. 2007; Torezan-Silingardi 2011; Encinas-Viso et al. 2012; Velasque and Del-Claro 2016).
The flowering pattern of individual species varies widely (Forrest et al. 2010; Forrest and Thomson 2011). In the Cerrado habitat, for some species it is staggered over a long period while others present a more pronounced peak with relatively few individuals occurring in the beginning or final of the flowering period (Ferreira and Torezan-Silingardi 2013). These variations are likely the result of a combination of abiotic, biotic, and phylogenetic factors (Mitchell et al. 2009; Rusquist and Staton 2013; Del-Claro and Marquis 2015) and the action of these factors may have led to either the minimization or the maximization of flowering within and among plants species, inducing synchronous and asynchronous phenologies at population and community level (Van Schaik et al. 1993; Vilela et al. 2014).
Sympatric species can exhibit clumped flowering phenologies that overlap extensively, possibly reducing herbivory according to the Herbivore Satiation Hypothesis (Beattie et al. 1973), i.e., a plant-plant association in which one species could decrease the likelihood of detection or vulnerability of other species by insect herbivores (see Barbosa et al. 2009; Lamarre et al. 2014). Alternatively, less synchrony between herbivore and flowering plants (e.g., Forrest and Thomson 2011) with a mismatch in the timing of interacting organisms can also reduce the herbivore pressure (Hoye and Forchhammer 2008). Nevertheless, during periods of overlap, different plant species may attract the same pollinators which can lead to competition for these cross-pollination vectors (e.g., Tanakura et al. 2009; Muchhala and Thomson 2012). The Pollinator Competition Hypothesis introduced by Robertson (1895) assumes that pollinators are a limiting resource and flowering events should be evenly spread through time (staggered phenology) to reduce the competition for pollinators and increase the effectiveness of pollination. Thus, a balance in plant life-history strategies based on a trade-off between a staggered phenology and a more overlapped flowering is expected (see Herrera et al. 2002, Forrest et al. 2010).
Many plant species have presented phenological shifts in the timing of their life-history events (Rafferty and Ives 2011), affecting the strength of mutualists and antagonistic interactions (Burkle et al. 2013; Rafferty et al. 2013, 2015; Velasque and Del-Claro 2016) and consequently their fitness (see English-Loeb and Karban 1992; Thomson 2010; Lange and Del-Claro 2014). Recent studies attempt to answer why and how the phenological overlap of interacting species has been changing. Many have concluded that climate change is the main disturbance responsible for alteration of timing of life-history events in a wide array of communities (Forrest et al. 2010; Rafferty and Ive 2012; Diez et al. 2012), due to its potential to desynchronize the phenology of interdependent species (Rafferty et al. 2015) and vary the degree of overlap among related plant species (Forrest et al. 2010).
In a recent meta-analysis, Chambers et al. (2013) related phenological drivers and trends among Southern Hemisphere species and discussed how phenological information contributes to our knowledge of the adaptive capacity of species, their resilience and constraints. However, the authors concluded that our general understanding and capacity to predict phenological responses remains patchy and constrained. Furthermore, few studies have been able to directly address what are the consequences of phenological shifts for species interactions (but see van Asch et al. 2007; Fabina et al. 2010; Liu et al. 2011) and for plant fitness (but see Dominguez and Dirzo 1995; Miller-Rushing et al. 2010). Therefore, there is an urgency to translate a basic understanding of phenology into forecasts about continued phenological changes and to predict the ecological consequences of these changes (Diez et al. 2012), especially for South American species (Staggemeier and Morellato 2011; Morellato et al. 2013).
Integrating life-history theory and mathematical models into ecological studies should help in forecasting changes and in understanding what may be the consequences for plant interaction and reproductive success (Fabina et al. 2010). Mathematical models could be used to generate testable predictions that address how phenological shifts can be generated to investigate consequences for temporal overlap of interacting species (Memmott et al. 2007; Gilman et al. 2012) and how it could affect plant fitness.
Some empirical models have historically been proposed (Porter et al. 1993) but, being based only on experimental data, these are unable to predict plant responses to new scenarios. Only recently some mechanistic (methodologies that take into account cause–effect relations based on plant phenology and biology) models have been introduced (Hoogenboom 2015; Jones et al. 2003), having success in predicting tendencies of growth.
Cerrado (the Brazilian Tropical Savanna) is the second largest South American ecosystem and presents great seasonal variation as a remarkable characteristic (Oliveira-Filho and Ratter 2002). Initially, we (a) tested whether the climatic parameters (temperature and precipitation) have changed over 10 years (2005–2014) and then we used empirical data of four Malpighiaceae species of Cerrado, which present previously described sequential flowering (Torezan-Silingardi 2007; Vilela et al. 2014), to (b) answer whether the onset of flowering is correlated with those climatic parameters, and (c) whether the phenological synchrony among species has therefore changed over time. We also aimed to (d) detect the effects of a sequential flowering and a higher overlap phenology for herbivory and fruit set of species. We developed a mathematical model, CSVM program (Claro 2015), to (e) estimate the flower and fruit production of each analyzed plant species for the next 5 years in a scenario with continued phenological changes. We hypothesized that temperature and precipitation rates have presented variation over the years analyzed and the onsets of flowering have also changed affecting the degree of overlap among Malpighiaceae species. Furthermore, we expected that the degree of plant phenological overlap (staggered or synchronized) influences the herbivory and fruit set of species.
Materials and methods
Study site
Fieldwork was carried out from May 2008 to March of 2014, spanning three reproductive seasons for each species studied (2008/2009; 2012/2013; 2013/2014), at the Reserva Ecológica do Clube Caça e Pesca Itororó de Uberlândia (CCPIU) (18°59′S, 48°18′W), Uberlândia, Minas Gerais State, Brazil. We used a 400-ha Cerrado site consisting of a dense scrub of shrubs and trees, known as Cerrado sensu stricto (Oliveira-Filho and Ratter 2002).
Plant species
In the Cerrado of central Brazil, shrubs of Malpighiaceae family are diverse and abundant (Gates 1982; Anderson 1990; Ferreira and Torezan-Silingardi 2013). Several species exhibit a staggered phenological development, in which individuals of different species resprout, bloom, and set fruit sequentially over time (Munhoz and Felfili 2005; Torezan-Silingardi 2007; Mendes et al. 2011). Four of these Malpighiaceae species were selected for the study because they present a sequential flowering phenology previously described by Torezan-Silingardi (2007) and Vilela et al. (2014): Peixotoa tomentosa A. Juss., Banisteriopsis laevifolia (A. Juss) B. Gates, Banisteriopsis campestris (A. Juss.) Little and Banisteriopsis malifolia (Ness and Mart) B. Gates. These species are deciduous, hermaphroditic, bee-pollinated, self-compatible, and share both pollinators and herbivores. The sequential resprouting and flowering of these species in the Cerrado also provides an uninterrupted food supply to a diverse herbivore guild. The first to blossom is P. tomentosa in June, followed by B. laevifolia, B. campestris, and B. malifolia. The greatest similarity in herbivore composition occurs between P. tomentosa and B. laevifolia (70%). Between these two species and B. campestris the similarity is almost 65%. B. malifolia shared almost 50% of its herbivores with the other plant species (Vilela et al. 2014). The immature structures (e.g., young leaves, buds, and flowers) have low structural resistance to physical damage, making the shoots, and inflorescences especially attractive to chewing and sucking insects (see Torezan-Silingardi 2007; Vilela et al. 2014).
Climatic analysis
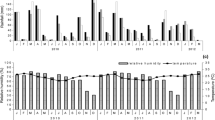
The climate at the study site is markedly seasonal, with dry/cold season from April to September (rainfall 22 ± 20 mm; temperature 19° ± 3 °C) and a wet/warm season occurring from October to March (rainfall, 270 ± 50 mm; temperature, 23° ± 5 °C) (additional details in Réu and Del-Claro 2005). The annual mean of temperature and total precipitation were provided by the Climate Station of the Geography Institute of Federal University of Uberlandia (Fig. 1). Initially, we performed the Repeated Measures ANOVA to compare the monthly means of temperature and monthly precipitations of each year from 2005 to 2014. We considered climatic factors since 2005 based on the sequential phenological results provided by Torezan-Silingardi (2007) for the same Malpighiaceae species at same place during 2005/2006. Further, we conducted Spearman’s correlations between climatic factors (monthly means of temperature and monthly precipitations) and phenological parameters (the number of individuals which begin their flowering in each month) (e.g., Morellato et al. 2010; Staggemeier and Morellato 2011).
Data collection
Individual plants (N = 30) of each plant species were monitored every 2 weeks during their reproductive period (see Vilela et al. 2014). Plant phenology (intensity and number of buds, flowers, and fruits) was recorded according to Torezan-Silingardi and Oliveira (2004) and Vilela et al. (2014) and fruit production was expressed as the proportion of fruit produced per buds (e.g., Del-Claro et al. 2013). At the end of the breeding season, we also quantified herbivory (leaf area loss). We recorded data from nine leaves per plant (three from the most apical stem, three from a middle stem, and three from the most basal stem) in order to determine the mean herbivory rate per species. This procedure was done without leaf removal. Measurements of herbivory rates on leaves were assessed by placing leaves on a transparent grid (divided into millimeters). An index of herbivory from each leaf was calculated as the proportion of points in the grid falling within damaged and undamaged areas of the leaf blade (see Moreira and Del-Claro 2005).
Statistical analysis
Species flowering variation
Circular statistical analyses were performed using the date of first flowering of each species separately for each year studied. To calculate the circular statistic parameters, months were converted to angles from 0 = January to 345 = December at intervals of 15 because the measurements were made every 2 weeks (e.g., Cardoso et al. 2012). The frequency of individuals at the onset of flowering for each species was considered for calculating the parameters: the mean vector (µ), length of mean vector (r), median, circular standard deviation, Rayleigh test (z). The mean date for each phenophase is determined by converting the mean angular directions to corresponding mean dates (see Morellato et al. 2000, 2010; Staggemeier et al. 2010). When the mean angle was significant, we performed the two-sample Watson-Williams tests (F) to determine differences among the mean data of the onset flowering of each species over time (Zar 1996).
Community flowering variation
Circular statistical analyses were also performed considering all species studied to analyze the degree of overlap in each reproductive period to verify for possible community flowering variation. In this case, we used more phenological variables: date of first bud, first flowering and first fruiting, date of peak of bud, flowering and fruiting. The phenological data were analyzed with the statistical software Oriana 4.0.
Intending to confirm a segregated phenology, a null model was performed to indicate if the temporal overlap among species was less than expected by chance. Overlap was quantified via Pianka (Pianka 1973) and Czechanowski (Feinsinger et al. 1981) indices and the values were generated using a randomization algorithm (Rosario). The algorithm Rosario was designed specifically for use with interval data important (Castro-Arellano et al. 2010). Rosario maintains the shape of the empirical activity distributions for each species in the randomly generated matrices by shifting entire activity patterns a random number of intervals. For each analysis, overlap indices were calculated for 10,000 randomly generated matrices of temporal flowering phenology, creating a null distribution of overlap values. Then a one-tailed test was conducted and the p value was calculated as the proportion of randomizations that resulted in overlap that is equal to or less than the empirical overlap value (observed) (e.g., Brito et al. 2012). Simulations were conducted with the TimeOverlap program (Castro-Arellano et al. 2010).
Herbivory and productivity analyses
We compared the rates of herbivory and fruit production between the periods with minimal (2008/2009) and higher overlap (2013/2014) using Paired test t after arcsine of the square root transformation of the percentage values for herbivory data and Wilcoxon test for fruit production, used for paired nonparametric data. The data were analyzed using the software Systat 12.
Mathematical modeling
The model we developed is part of a software (Programa CSVM ®—INPI—BR 51 2015 000482-4) to present a tendency of plant production in face of phenological shifts associated with climate variability in agricultural systems (Claro 2015). The software encompasses a mechanistic fuzzy logic approach, to describe the behavior of a population of plants. Considering that temporal and spatial variations on the abundance of specific partners could represent local variation on the results of flower and fruit production, the program intends to ensure a qualitative validity, by demonstrating a tendency in different scenarios. We adjusted and validated the model parameters by using Differential Evolution Optimization methods (Lobato and Steffen 2008; Vanderplaats 2005) to define some of the model’s constants, by comparing the real case with the program results via optimization. This procedure was made by estimating the interval in which the variables considered may vary, normalizing the variables, and then applying an optimization procedure to achieve the best approximation of the real parameters/variables in a specific scenario (Lobato and Steffen 2007). Initially, we considered the field data obtained by Torezan-Silingardi (2007) in 2005/2006 aiming to ensure a greater robustness for the model. In order to standardize the sample method, we employed the same methodology applied to Torezan-Silingardi (2007).
The inputs considered were rainfall, leaf sprouting and production of floral buds, flowers and fruits. Other parameters estimated by the researchers and adjusted by the optimization procedure, were the weekly rates of herbivory, pollination and autonomous self-pollination. The preliminary data as input parameters were: area of 2500 m2, with 100 × 100 square elements of 0.5 × 0.5 m each, simulating for 5 years of 48 weekly time steps each (1 time step equals 1.0863 week). Containing 50 B. malifolia individuals (3 × 3 elements in size), 50 P. tomentosa individuals (2 × 1 elements in size), 30 B. laevifolia individuals (2 × 2 elements in size), and 40 B. campestris individuals (1 × 1 elements in size) based on their real size and presence measured on the field. The herbivory and pollination were set to occur in 90% of the elements per repetition. The randomness of weekly herbivory considered was its species-specific base value with a random of 3%. The weekly randomness of insect pollination and autonomous self-pollination considered was also its species-specific base value with a random of 3%. We considered 13% of randomness for the baselines (rainfall, leaves sprouting, and flowering), applied both in time and geographical location. All variation parameters were estimated based on real field data.
An advance in the phenology of B. laevifolia was simulated, given that this phenomenon was already observed in 2012 and 2013, to investigate the consequences of a continuous shift in its phenology for the degree of overlap among species and the outcomes for plant reproduction, i.e., production of flowers and fruit set, for the next 5 years. We simulated three phenological scenarios with advancement of phenology in 2, 4, and 6 weeks on the B. laevifolia flowering, keeping all other parameters set as before.
Results
Climatic analysis
The monthly means of temperature and monthly precipitations varied substantially from 2005 to 2014 (Repeated-Measure One-Way ANOVA F = 3.785; df = 9; p < 0.001 for temperature and F = 2.21; df = 9; p = 0.027 for precipitation) (Fig. 1). The Spearman’s correlations between the onset of flowering of each species and the monthly temperature and precipitation data for each year studied (2008/2009, 2012/2013, 2013/2014) showed that the species were differently correlated with climatic factors over the years with P. tomentosa presenting the highest correlation results and B. malifolia as the species suffering less interference from climatic variations (see Table 1).
Species flowering variation
For all species, the mean angles of onset of flowering were significantly seasonal for whole analyzed period (Rayleigh test p < 0.05) and the lengths of mean vectors (r) were nearly 1 (Table 2). The vector r varies from 0 (when phenological activity is distributed uniformly throughout the year) to 1 (when phenological activity is concentrated around one single date or mean angle) (Zar 1996). Comparisons of the mean data among the years showed that the onset of species flowering varied over time (F = 59.176, p < 0.001 for P. tomentosa; F = 48.445, p < 0.001 for B. laevifolia; F = 33.087, p < 0.001 for ; F = 60.729, p < 0.001 for B. campestris B. malifolia, Table 2).
Community flowering variation
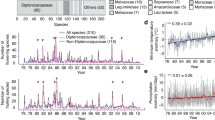
For the first reproductive season (2008/2009) the mean angles of onset and peak for all phenophases were not significantly seasonal (Rayleigh test p > 0.05) and the lengths of mean vectors (r) were nearly 0 (Table 3). These results show that phenological activity of the Malpighiaceae was distributed uniformly throughout the year displaying a segregated phenology. The null model simulations for this reproductive period indicated that the overlap of distributions was significantly less than expected by chance, which confirms the sequential flowering (Pianka = 0.202, p < 0.001; Czechanowski = 0.968, p < 0.001). For the second reproductive season (2012/2013) the mean angle of peak for buds and flowers and onset of fruit were not significantly seasonal (Rayleigh test p > 0.05) and the lengths of mean vectors (r) were nearly 0 (Table 3). These results show that phenological activity exhibited a degree of overlap among some phenophases. In the third year of study, the mean angles were significantly seasonal (Rayleigh test p < 0.05) and the lengths of mean vectors (r) were nearer 1 (Table 3). Linear graphics with the intensity of flowering of Malpighiaceae species showing the degree of overlap among the years were performed (Fig. 2).
Linear graphics with the intensity of flowering of Peixotoa tomentosa, Banisteriopsis laevifolia, Banisteriopsis campestris, and Banisteriopsis malifolia during 2008/2009, 2012/2013, and 2013/2014 in a Brazilian Tropical Savanna (axis y means intensity of flowering phenology being 1 = 25% and 2 = 75%; n = 30)
Herbivory and productivity analyses
The periods with higher overlap of flowering accounted for lower rates of herbivory for B. laevifolia (t = 2.382; df = 29; p = 0.024) and P. tomentosa (t = 2.883, df = 29, p = 0.007). However, there were no significant difference for B. campestris (t = 1.821; df = 29; p = 0.079) and B. malifolia (t = 0.7957, df = 29, p = 0.4327) (Fig. 3a). In relation to fruit set, the higher overlap period accounted for lower rates of productivity only for B.laevifolia (W = 168; n = 30; p = 0.044). There was no significant difference for either B. malifolia (W = 147; n = 30; p = 0.114) and P. tomentosa (W = 23; n = 30; p = 0.721). B. campestris presented an increase in productivity during the period with higher overlap, however, there was no significant difference (W = −62; n = 30; p = 0.219) (Fig. 3b).
Comparative analysis of herbivory (leaf area loss) a and fruit production, b of Peixotoa tomentosa, Banisteriopsis laevifolia, Banisteriopsis campestris and Banisteriopsis malifolia in a Brazilian Tropical Savanna between the period with lower (2008/2009) and greater overlap of flowering (2013/2014). (Test Paired T in (a) and Wilcoxon test in (b) (*) means statistical difference; p < 0.05, Means ± 1SE are presented)
Mathematical modeling
The CSVM program obtained a mean squared error of 3.087% when compared to the mean field data response, achieving a close fit between the model and the real-case scenario. All mean percentage squared error achieved for each species after the optimization procedure are exhibited in Table 4. The results of advance in the phenology of B. laevifolia demonstrated a tendency in increasing the production of flowers, however, a proportionally lower production of fruits (Fig. 4a). For P. tomentosa, the model indicated a tendency in increasing amounts of both flowers and fruits (Fig. 4b). There were no tendencies of variation for B. malifolia and B. campestris (Fig. 4c, d, respectively).
Results of flower and fruit production for Banisteriopsis laevifolia (a) Peixotoa tomentosa (b), Banisteriopsis malifolia (c), and Banisteriopsis campestris (d) in four different phenological simulations, considering an anticipated phenology of B. laevifolia in 2, 4, and 6 weeks. Default means the standard setting
Discussion
Our results present climatic variation in annual mean of temperature and precipitation from 2005 to 2014 and detected correlations with the onset of plant flowering during the three reproductive seasons studied (2008/2009, 2011/2012, 2013/2014). Shifts on the onset of flowering resulted in a greater overlap among Malpighiaceae species during the third reproductivity period analyzed, corroborating our first hypothesis. We also demonstrated that degree of plant phenological overlap (staggered or synchronized) influenced the herbivory and fruit set of species, corroborating our second hypothesis. The mathematical model indicated a tendency in flower and fruit production for each plant species contributing to a better understanding of how the phenology may vary on a continuing climate change (see a movie produced in CSVM program as supplementary material).
The correlations between onset of plant flowering, temperature, and precipitation suggest an influence of these climatic parameters on the phenology of plant species. Plants may advance or delay their reproductive period in a seasonal climate by avoiding the sprouting of new leaves before or during unfavorable periods (e.g., water stress) or by producing new leaves to coincide with the onset of periods of favorable conditions (e.g., high radiation) (Van Shaik et al. 1993; Del-Claro and Torezan-Silingardi 2012). The impacts of climate change on phenological shifts are among the most documented process occurring during the last years, as demonstrated by several studies (e.g., Forrest et al. 2010; Forrest and Thomson 2011; Rafferty and Ive 2012; Diez et al. 2012; Liuth et al.2013), especially the effects of rainfall and temperature in tropical climates (e.g., Morellato 2013; Bock et al. 2014). These environmental factors can affect the growth and development of the plants under different forms during the phenological phases, whereas solar radiation, and relative humidity influence many physiological processes related to flowering patterns (see Camargo 2010).
Changes in flowering patterns are likely to affect plant reproduction if competitive or facilitative interactions between plant species affect their associations with interacting animal partners (see Gross et al. 2000; Lazaro et al. 2009; Forrest et al. 2010). Annual variation on plant phenologies in subalpine meadows in Colorado, USA, produced a higher overlap among plant species leading to increased sharing of pollinators by some species (e.g., Forrest et al. 2010) and decreased fruit set for other species due to their decreased synchrony with pollinators (Thomson 2010). Rafferty and Ives (2011) showed that species experimentally induced to flower earlier in a wildflower community received more pollinator visits and Rafferty and Ives (2012) simulated changes in phenology of two perennial wildflowers and demonstrated that the seed set can vary as a result of changes in pollinator taxonomic composition and effectiveness. Therewith, flowering phenology is commonly thought to affect plant reproduction through the influence of pollinators, as well as the degree of plant dependence on this interaction (see Johnson et al. 2011).
In our system, all species are self-compatible, they can require animal pollinators to move pollen or reproduce via spontaneous autogamy, however, the rate of autonomous self-pollination varies greatly among species (Torezan-Silingardi et al. in preparation). Fruit set via spontaneous self-pollination in Banisteriopsis laevifolia and B. campestris is about only 2%, while in P. tomentosa this value can range up to 43%, and in B. malifolia accounts for about 30% of the fruit set. In this sense, the species of Malpighiaceae were affected differently by the greater overlap of flowering period with B. laevifolia, the only significantly affected in a negative way. Based on these results, we can suggest that P. tomentosa and B. malifolia presented a more robust response to a greater overlap of phenology due to their relative independence of pollinators. Banisteriopsis campestris cannot be considered in this line of thought because its flowering separated from the core of other species. Therefore, the staggered strategy is more essential for B. laevifolia due to its greater dependence in relation to pollinators. Bishop and Schemske (1998) and Moeller (2006) have demonstrated and discussed the importance of self-compatibility by increasing fruit and seed production as an important driver of plant mating system evolution.
Our results also showed that the herbivory rate was significantly reduced for P. tomentosa and B. laevifolia and not significant for B. malifolia and B.campestris in a scenario with greater overlap. Therefore, the effects of overlap of plants in reducing herbivory were corroborated and it is likely that the effect on B. malifolia and B.campestris was lower due to its less overlapping than P. tomentosa and B. laevifolia. In addition, these species flourish in the rainy season, the period with the greatest abundance and activity of herbivore insects (e.g., Alves-Silva et al. 2013, 2014; Alves-Silva and Del-Claro 2013; Vilela et al. 2014). Thus, we suggest that greater synchrony between P. tomentosa and B. laevifolia may have allowed the occurrence of dilution of herbivory (Herbivore Satiation Hypothesis) (e.g., Clark and Clark 1991; English-Loeb and Karban 1992). A classic study of Aide (1991) investigated this effect for leaves growing synchronously within the population of the sub-canopy tree Gustavia superba (Lecythidaceae) and found out that individuals out of synchrony with their conspecifics suffered significantly more herbivore damage. It is also possible that herbivory rate diminished due to less synchrony between herbivores and flowering time. The importance of synchrony for effective herbivore consumption was already evidenced for larva of Lepidoptera feeding on leaf flush from experimental studies (e.g., Hunter and Elkinton 2000; Van Asch et al. 2007).
The mathematical model we developed was able to demonstrate with great precision (mean squared error ≈ 3%) any given scenario with variations in the rainfall or herbivory for the species considered. In scenario extrapolations with input not directly linked to the species themselves (i.e., rainfall, herbivory, etc.) the model is also able to make very precise predictions, which has been optimized and validated by field measured data. In the opposite way, when the variations imposed as entries are directly related to the species (i.e., flowering), the results should be interpreted as a tendency with an unknown error, due to unavailability of real field data for comparisons and model adjustment. We considered variations in the onset of plant flowering and, therefore, we have to interpret the results as a valid tendency. The simulations showed a species-specific response, also demonstrated by Diez et al. (2012) to three North American plant communities in response to climate change.
In our study, the most pollinator-dependent species, B. laevifolia, presented a tendency in increasing the production of flowers possibly as a result of the dilution in herbivore pressure (e.g., Bishop and Schemske 1998) or lowering of herbivore synchrony with plant flowering (e.g., Forrest and Thomson 2011). However, the greater synchronization with P. tomentosa and possible sharing of pollinators associated with its dependence on these vectors resulted in a tendency of reduced production of fruits. On the other hand, plants possessing the combination of simultaneously autonomous self-pollination and escape from herbivores, as demonstrated by P. tomentosa, were able to keep a disproportionate fitness advantage over plants possessing any of the other possibilities (see Herrera et al. 2002). Thus, within communities, variation in species responses may determine community-level patterns of phenology and resilience of species (Memmott et al. 2007; Kaiser-Bunbury et al. 2010).
We demonstrated variations in climate parameters over the recent years and the manifestation of these changes on plant phenology and biotic interactions of different plant species from the Southern Hemisphere. There is a concern that climate change is altering the timing of life-history events in a wide array of species, especially for species from Northern Hemisphere. However, few papers have related the effects of climatic change on plant phenology and interactions of South American species (Morellato et al. 2013). Our findings of significant variability in species responses to their interacting animal partners suggest that plasticity in these traits will be important for the survival and reproduction of these plant species in the future. Thus, integrating phenological investigations in the field with the existing theories and mathematical modeling allow us to interpret different phenological synchronization degrees and enable us to describe the possible maintenance and evolution of life histories in response to climate change.
References
Aide TM (1991) Synchronous leaf production and herbivory in juveniles of Gustavia superba. Oecologia 88:511–514
Alves-Silva E, Del-Claro K (2013) Effect of post-fire resprouting on leaf fluctuating asymmetry, extrafloral nectar quality, and ant–plant–herbivore interactions. Naturwissenschaften 100:525–532
Alves-Silva E, Barônio GJ, Torezan-Silingardi HM, Del-Claro K (2013) Foraging behavior of Brachygastra lecheguana (Hymenoptera: Vespidae) on Banisteriopsis malifolia (Malpighiaceae): Extrafloral nectar consumption and herbivore predation in a tending ant system. Entomol Sci 16:162–169
Alves-Silva E, Bachtold A, Barônio GJ, Torezan-Silingardi HM, Del-Claro K (2014) Ant–herbivore interactions in an extrafloral nectaried plant: are ants good plant guards against curculionid beetles? J Nat Hist 49:841–851
Amasino R (2010) Seasonal and developmental timing of flowering. Plant J 61:1001–1013
Anderson WR (1990) The origin of the Malpighiaceae - the evidence from morphology. Mem N Y Bot Gard 64:210–224
Barbosa P, Hines J, Kaplan I, Martinson H, Szczepaniec A, Szendrei Z (2009) Associational resistance and associational susceptibility: having right or wrong neighbors. Annu Rev Ecol Evol Syst 40:1–20
Beattie AJ, Breedlove DE, Ehrlich PR (1973) The ecology of the pollination and predation of Frasera speciose. Ecology 54:81–91
Bishop JG, Schemske DW (1998) Variation in flowering phenology and its consequences for lupines colonizing Mount St Helens. Ecology 79:534–546
Bock A, Sparks TH, Estrella N, Jee N, Casebow A, Schunk C, Leuchner M, Menzel A (2014) Changes in first flowering dates and flowering duration of 232 plant species on the island of Guernsey. Glob Change Biol 20:3508–3519
Brito AF, Presley SJ, Santos GMM (2012) Temporal and trophic niche overlap in a guild of flower-visiting ants in a seasonal semi-arid tropical environment. J Arid Environ 87:161–167
Burkle LA, Marlin JC, Knight TM (2013) Plant-pollinator interactions over 120 years: loss of species, co-occurrence, and function. Science 339:1611–1615
Camargo MBP (2010) The impact of climatic variability and climate change on arabica coffee crop in Brazil. Brag Camp 69:239–247
CaraDonna PJ, Iler AM, Inouye DW (2014) Shifts in flowering phenology reshape a subalpine plant community. Proc Natl Acad Sci USA 111:4916–4921
Cardoso FCG, Marques R, Botosso PC, Marques MCM (2012) Stem growth and phenology of two tropical trees in contrasting soil conditions. Plant Soil 354:269–281
Castro-Arellano I, Lacher TE Jr, Willig MR, Rangel TF (2010) Assessment of assemblage-wide temporal-niche segregation using null models. Methods Ecol Evol 1:311–318
Chambers LE, Altwegg R, Barbraud C, Barnard P, Beaumont LJ, Crawford RJM et al (2013) Phenological changes in the southern hemisphere. PLoS ONE 8(10):e75514
Clark DB, Clark DA (1991) Herbivores, herbivory, and plant phenology: patterns and consequences in a tropical rain-forest cycad. In: Price PW, Lewinsohn TM, Fernandes GW, Benson WW (eds) Plant-animal interactions: evolutionary ecology in tropical and temperate regions. Wiley, New York, p 639
Claro VTS (2015) Programa CSVM®—INPI—BR 51 2015 000482-4 Revista de Propriedade Industrial, Patentes RPI—DOU, 179
Del-Claro K, Marquis RJ (2015) Ant species identity has a greater effect than fire on the outcome of an ant protection system in Brazilian Cerrado. Biotropica 47(4):1–9
Del-Claro K, Torezan-Silingardi HM (2012) Ecologia das interações plantas-animais: Uma abordagem ecológico-evolutiva. Technical Books, Rio de Janeiro
Del-Claro K, Stefani V, Lange D, Vilela AA, Nahas L, Velasques M, Torezan-Silingardi HM (2013) The importance of natural history studies for a better comprehension of animal-plant interactions networks. Biosci J 29:439–448
Del-Claro K, Rico-Gray V, Torezan-Silingardi HM, Aalves-Silva E, Fagundes R, Lange D, Dátillo W, Vilela AA, Aguirre A, Rodriguez-Morales D (2016) Loss and gains in ant–plant interactions mediated by extrafloral nectar: fidelity, cheats, and lies. Insect Soc 63:207–221
Diez JM, Ibanez I, Miller-Rushing AJ, Mazer SJ, Crimmins TM, Crimmins MA, Bertelsen CD, Inouye DW (2012) Forecasting phenology: from species variability to community patterns. Ecol Lett 15:545–553
Domınguez CA, Dirzo R (1995) Rainfall and flowering synchrony in a tropical shrub: variable selection on the flowering time of Erythroxylum havanense. Evol Ecol 9:204–216
Encinas-Viso F, Revilla TA, Etienne RS (2012) Phenology drives mutualistic network structure and diversity. Ecol Lett 15:198–208
English-Loeb GM, Karban R (1992) Consequences of variation in flowering phenology for seed head herbivory and reproductive success in Erigeron glaucus (Compositae). Oecologia 89:588–595
Fabina NS, Abbot KC, Gilman RT (2010) Sensitivity of plant - pollinator-herbivore communities to changes in phenology. Ecol Modell 221:453–458
Feinsinger P, Spears EE, Poole RW (1981) A simple measure of niche breadth. Ecology 62:27–32
Ferreira CA, Torezan Silingardi HM (2013) Implications of the floral herbivory on Malpighiaceae plant fitness: visual aspect of the flower affects the attractiveness to pollinators. Sociobiology 60:323–328
Forrest JRK, Thomson JD (2011) An examination of synchrony between insect emergence and flowering in Rocky Mountain meadows. Ecol Monogr 81:469–491
Forrest JRK, Inouye DW, Thomson JD (2010) Flowering phenology in subalpine meadows: does climate variation influence community co-flowering patterns? Ecology 91:431–440
Franks PJ, Adams MA, Amthor JS, Barbour MM, Berry JA, Ellsworth DS, Farquhar GD, Ghannoum O, Lloyd J, McDowell N, Norby RJ (2013) Sensitivity of plants to changing atmospheric CO2 concentration: from the geological past to the next century. New Phytol 197:1077–1094
Gates B (1982) Banisteriopsis, Diplopterys (Malpighiaceae). Fl Neotrop Monogr 30:1–236
Gilman RT, Fabina NS, Abbott KC, Rafferty NE (2012) Evolution of plant-pollinator mutualisms in response to climate change. Evol Appl 5:2–16
Gross CL, Mackay DA, Whalen MA (2000) Aggregated flowering phenologies among three sympatric legumes: the degree of non-randomness and the effect of overlap on fruit set. Plant Ecol 148:13–21
Hegland SJ, Nielsen A, Lazaro A, Bjerknes AL, Totland O (2009) How does climate warming affect plant–pollinator interactions? Ecol Lett 12:184–195
Herrera CM, Medrano M, Rey PJ, Sanchez-Lafuente M, Garcia MB, Guitian J, Manzaneda AJ (2002) Interaction of pollinators and herbivores on plant fitness suggests a pathway for correlated evolution of mutualism- and antagonism-related traits. Proc Natl Acad Sci USA 99:16823–16828
Hoogenboom, G, JW Jones, PW Wilkens, CH Porter, KJ Boote, LA Hunt, U Singh, JI Lizaso, JW White, O Uryasev, R Ogoshi, J Koo, V Shelia, GY Tsuji (2015) Decision Support System for Agrotechnology Transfer (DSSAT) Version 46 (http://dssat.net) DSSAT Foundation, Prosser, Washington
Hoye TT, Forchhammer MC (2008) Phenology of high-Arctic arthropods: effects of climate on spatial, seasonal and inter-annual variation. Adv Ecol Res 40:299–324
Hunter AF, Elkinton JS (2000) Effects of synchrony with host plant on populations of a spring-feeding Lepidopteran. Ecology 81:1248–1261
Johnson SD, Burgoyne PM, Harder LD, Dötterl S (2011) Mammal pollinators lured by the scent of a parasitic plant. Proc R Soc Lond B 278:2303–2310
Jones JW, Hoogenboom G, Porter CH, Boote KJ, Batchelor WD, Hunt LA, Wilkens PW, Singh U, Gijsman AJ, Ritchie JT (2003) DSSAT Cropping System. Model Eur J Agron 18:235–265
Kaiser-Bunbury CN, Traveset A, Hansen DM (2010) Conservation and restoration of plant-animal mutualisms on oceanic islands Perspect. Plant Ecol 12:131–143
Kawagoe T, Kudoh H (2010) Escape from floral herbivory by early flowering in Arabidopsis halleri subsp gemmifera. Oecologia 164:713–720
Lamarre GPA, Mendoza I, Fine PVA, Baraloto C (2014) Leaf synchrony and insect herbivory among tropical tree habitat specialists. Plant Ecol 215:209–220
Lange D, Del-Claro K (2014) Ant-plant interaction in a tropical savanna: may the network structure vary over time and influence on the outcomes of associations? PLoS ONE 9(8):e105574
Lazaro A, Lundgren R, Totland O (2009) Co-flowering neighbors influence the diversity and identity of pollinator groups visiting plant species. Oikos 118:691–702
Liu Y, Reich PB, Li G, Sun S (2011) Shifting phenology and abundance under experimental warming alters trophic relationships and plant reproductive capacity. Ecology 92:1201–1207
Liuth HS, Talora DC, Amorim AM (2013) Phenological synchrony and seasonality of understory Rubiaceae in the Atlantic Forest, Bahia, Brazil. Acta Bot Bras 27:195–204
Lobato FS, Steffen V (2007) Engineering system design with multi-objective differential evolution. Proc COBEM 19:1–10
Lobato FS, Steffen V (2008) Otimização multi-objetivo para o projeto de sistemas de engenharia Doc Thesis Universidade Federal de Uberlândia
Memmott J, Craze PG, Waser NM, Price MV (2007) Global warming and the disruption of plant-pollinator interactions. Ecol Lett 10:710–717
Mendes FN, Rêgo MMC, Albuquerque PMC (2011) Phenology and reproductive biology of two species of Byrsonima Rich. Biota Neotrop 11:104–115
Miller-Rushing AJ, Hoye TT, Inouye DW, Post ES (2010) The effects of phenological mismatches on demography. Philos Trans 365:3177–3186
Mitchell RJ, Irwin RE, Flanagan RJ, Karron JD (2009) Ecology and evolution of plant-pollinator interactions. Ann Bot 103:1355–1363
Moeller DA (2006) Geographic structure of pollinator communities, reproductive assurance, and the evolution of self-pollination. Ecology 87(6):1510–1522
Moreira VSS, Del-Claro K (2005) The outcomes of an ant–treehopper association on Solanum lycocarpum St Hill: increased membracid fecundity and reduced damage by chewing herbivores. Neotrop Entomol 34:881–887
Morellato LPC, Talora DC, Takahasi A, Bencke CSC, Romera EC, Zipparro V (2000) Phenology of atlantic rain forest trees: a comparative study. Biotropica 32:811–823
Morellato LPC, Alberti LF, Hudson IL (2010) Applications of circular statistics in plant phenology: a case studies approach. In: Keatley M, Hudson IL (eds) Phenological research: methods for environmental and climate change analysis. Springer, New York, pp 357–371
Morellato LPC, Camargo MGG, Gressler E (2013) A review of plant phenology in South and Central America. In: Schwartz MD (ed) Phenology: an integrative environmental science. Springer, Heidelberg, pp 91–113
Muchhala N, Thomson JD (2012) Interspecific competition in pollination systems: costs to male fitness via pollen misplacement. Funct Ecol 26:476–482
Munhoz CBR, Felfili JM (2005) Fenologia do estrato herbáceo-subarbustivo de uma comunidade de campo sujo na Fazenda Água Limpa no Distrito Federal, Brasil. Acta Bot Bras 19:981–990
Oliveira-Filho AT, Ratter JA (2002) Vegetation physiognomies and woody flora of the Cerrado Biome. In: Marquis RJ, Oliveira PS (eds) The Cerrados of Brazil PS. Ecology and Natural History of a Neotropical Savanna Columbia University, New York, pp 91–120
Pianka ER (1973) The structure of lizard communities. Annu Rev Ecol Syst 4:53–74
Pilson D (2000) Herbivory and natural selection on flowering phenology in wild sunflower, Helianthus annuus. Oecologia 122:72–82
Porter JR, Jamieson PD, Wilson DR (1993) A comparison of the wheat simulation models AFRCWHEAT2, CERES-wheat and SWHEAT for nonlimiting conditions of growth. Field Crop Res 33:131–157
Rafferty NE, Ives AR (2011) Effects of experimental shifts in flowering phenology on plant -pollinator interactions. Ecol Lett 14:69–74
Rafferty NE, Ives AR (2012) Pollinator effectiveness varies with experimental shifts in flowering time. Ecology 93:803–814
Rafferty NE, CaraDonna PJ, Burkle LA, Iler AM, Bronstein JL (2013) Phenological overlap of interacting species in a changing climate: an assessment of available approaches. Ecol Evol 3:3183–3193
Rafferty NE, CaraDonna PJ, Bronstein JL (2015) Phenological shifts and the fate of mutualisms. Oikos 124:14–21
Réu WF, Del-Claro K (2005) Natural history and biology of Chlamisus minax Lacordaire (Chrysomelidae: Chlamisinae). Neotrop Entomol 34:357–362
Robertson C (1895) The philosophy of flower seasons and the phenological relations of the entomophilous flora and the anthophilous insect fauna. Am Nat 29:97–117
Runquist RB, Stanton ML (2013) Asymmetric and frequency-dependent pollinator-mediated interactions may influence competitive displacement in two vernal pool plants. Ecol Lett 16:183–190
Staggemeier VG, Morellato LPC (2011) Reproductive phenology of coastal plain Atlantic forest vegetation: comparisons from seashore to foothills. Int J Biometeorol 55:843–854
Staggemeier VG, Diniz-Filho JAF, Morellato LPC (2010) The shared influence of phylogeny and ecology on the reproductive patterns of Myrteae (Myrtaceae). J Ecol 98:1409–1421
Takakura K, Nishida T, Matsumoto T, Nishida S (2009) Alien dandelion reduces the seed-set of a native congener through frequency-dependent and one-sided effects. Biol Inv 11:973–981
Thomson JD (2010) Flowering phenology, fruiting success and progressive deterioration of pollination in an early-flowering geophyte. Phil Trans R Soc B 365:3187–3199
Torezan-Silingardi HM (2007) A influência dos herbívoros florais, dos polinizadores e das características fenológicas sobre a frutificação das espécies da família Malpighiaceae em um cerrado de Minas Gerais Doc Thesis. Universidade de São Paulo
Torezan-Silingardi HM (2011) Predatory behavior of Pachodynerus brevithorax (Hymenoptera: Vespidae, Eumeninae) on endophytic herbivore beetles in the Brazilian Tropical Savanna. Sociobiology 57:181–189
Torezan-Silingardi HM, Oliveira PEAM (2004) Phenology and reproductive ecology of Myrcia rostrata and M. tomentosa (Myrtaceae) in Central Brazil. Phyton 44:23–44
Torres C, Galetto L (2011) Flowering phenology of co-occurring Asteraceae: a matter of climate, ecological interactions, plant attributes or of evolutionary relationships among species? Org Divers Evol 11:9–19
Van Asch M, Van Tienderen PH, Holleman LJM, Visser ME (2007) Predicting adaptation of phenology in response to climate change, an insect herbivore example. Glob Change Biol 13:1596–1604
Van Schaik CP, Terborgh JW, Wright SJ (1993) The phenology of tropical forests: adaptive significance and consequences for primary consumers. Annu Rev Ecol Syst 24:353–377
Vanderplaats GN (2005) Numerical optimization techniques for engineering design. Vanderplaats Research and Development Inc, Colorado Springs
Velasque M, Del-Claro K (2016) Host plant phenology may determine the abundance of an ecosystem engineering herbivore in a tropical savanna. Ecol Entomol 41:421–430
Vilela AA, Torezan-Silingardi HM, Del-Claro K (2014) Conditional outcomes in ant-plant-herbivore interactions influenced by sequential flowering. Flora 209:359–366
Zar JH (1996) Biostatistical analysis, 3rd edn. Prentice-Hall International Editions, New Jersey
Acknowledgements
We thank the three anonymous reviewers for reviewing the manuscript. We also thank CNPq and FAPEMIG for funding this research (KDC/HMTS/VTSDC) and FAPEMIG for awarding fellowships to AAV.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Heikki Hokkanen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (AVI 57599 kb)
AVI movie produced in CSVM program (Claro 2015) showing the influence of climatic parameters in real field data of leaf, bud, flower, and fruit production over 3 years to the Brazilian savanna Malpighiaceae: Banisteriopsis laevifolia, Peixotoa tomentosa, Banisteriopsis malifolia and Banisteriopsis campestris
Rights and permissions
About this article
Cite this article
Vilela, A.A., Del Claro, V.T.S., Torezan-Silingardi, H.M. et al. Climate changes affecting biotic interactions, phenology, and reproductive success in a savanna community over a 10-year period. Arthropod-Plant Interactions 12, 215–227 (2018). https://doi.org/10.1007/s11829-017-9572-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-017-9572-y