Abstract
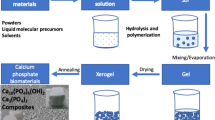
We report the synthesis and characterization of crystalline calcium phosphate (CaP) nanostructures from calcium inositol hexakisphosphate (CaIP6) precursor in water-ethanol mixed solutions. We show how these CaPs can be prepared by a solvo-hydrothermal reaction and determined their compositions and structures using a battery of material characterization techniques. Our results show that only the hydroxyapatite (HAP) and dicalcium phosphate anhydrous (DCPA) phases of CaP were present in the nanostructures produced in water-ethanol mixtures, and that HAP/DCPA ratio of the rod- and plate-shaped CaP nanostructures produced were affected by the amount of ethanol present in these mixtures. The described method can be used to improve morphological control of CaP-based biomate-rials and has potential use in bone regenerative medicine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
C. Combes, S. Cazalbou and C. Rey, Minerals, 6, 34 (2016).
S. V. Dorozhkin, Materials, 6, 3840 (2013).
M. Epple, K. Ganesan, R. Heumann, J. Klesing, A. Kovtun, S. Neumann and V. Sokolova, J. Mater. Chem., 20, 18 (2010).
W. Habraken, P. Habibovic, M. Epple and M. Bohner, Mater. Today, 19, 69 (2016).
Y. Q. Shen, Y. J. Zhu, F. F. Chen, Y. Y. Jiang, Z. C. Xiongand F. Chen, J. Mater. Chem. B, 6, 4985 (2018).
J. Zhao, Y. Liu, W. B. Sun and H. Zhang, Chem. Cent. J., 5, 1 (2011).
M. Nagano, T. Nakamura, T. Kokubo, M. Tanahashi and M. Ogawa, Biomaterials, 17, 1771 (1996).
R. A. Surmenev, M. A. Surmeneva and A. A. Ivanova, Acta Biomater., 10, 557 (2014).
R. Mi, Y. Liu, X. Chen and Z. Shao, Nanoscale, 8, 20096 (2016).
S. Bose and S. Tarafder, Acta Biomater., 8, 1401 (2012).
K. W. Wang, Y. J. Zhu, X. Y. Chen, W. Y. Zhai, Q. Wang, F. Chen, J. A. Chang and Y. R. Duan, Chem. Asian J., 5, 2477 (2010).
L. X. Yang, J. J. Yin, L. L. Wang, G. X. Xing, P. Yin and Q. W. Liu, Ceram. Int., 38, 495 (2012).
J. M. Delgado-Lopez, M. Iafisco, I. Rodriguez-Ruiz and J. Gomez-Morales, J. Inorg. Biochem., 127, 261 (2013).
K. L. Lin, C. T. Wu and J. Chang, Acta Biomater., 10, 4071 (2014).
M. Sadat-Shojai, M. T. Khorasani and A. Jamshidi, J. Cryst. Growth, 361, 73 (2012).
H. Ito, Y. Oaki and H. Imai, Cryst. Growth Des., 8, 1055 (2008).
Y. Y. Jiang, Y. J. Zhu, F. Chen and J. Wu, Ceram. Int., 41, 6098 (2015).
N. Eliaz and N. Metoki, Materials, 10, 334 (2017).
A. Haider, S. Haider, S. S. Han and I. K. Kang, RSC Adv., 7, 7442 (2017).
A. Fihri, C. Len, R. S. Varma and A. Solhy, Coord. Chem. Rev., 347, 48 (2017).
A. C. Tas, J. Am. Ceram. Soc., 92, 2907 (2009).
Y. R. Cai and R. K. Tang, J. Mater. Chem., 18, 3775 (2008).
D. Q. Xiao, Z. Tan, Y. K. Fu, K. Duan, X. T. Zheng, X. Lu and J. Weng, Ceram. Int., 40, 10183 (2014).
A. Shamsuddin and J. von Fraunhofer, US Pattern Application Publication, US 2007/0212449 A1 (2007).
F. Grases, M. Ramis and A. Costa-Bauzá, Urol. Res., 28, 136 (2000).
D. Q. Xiao, F. Yang, X. Zhou, Z. Chen, K. Duan, J. Weng and G. Feng, RSC Adv., 7, 44371 (2017).
Z. Q. He, C. W. Honeycutt, T. Q. Zhang and P. M. Bertsch, J. Environ. Qual., 35, 1319 (2006).
T. Goto, I. Y. Kim, K. Kikuta and C. Ohtsuki, Ceram. Int., 38, 1003 (2012).
L. J. Hao, H. Yang, S. L. Du, N. R. Zhao and Y. J. Wang, Mater. Lett., 131, 252 (2014).
M. Dardouri, J. P. Borges and A. D. Omrani, Ceram. Int., 43, 3784 (2017).
R. X. Sun, L. L. Yang, Y. X. Zhang, F. Chu, G. Y. Wang, Y. P. Lv and K. Z. Chen, CrystEngComm, 18, 8030 (2016).
K. Ganesan and M. Epple, New J. Chem., 32, 1326 (2008).
J. H. Han and S. Chung, Appl. Chem. Eng., 29, 740 (2018).
M. Yoshimura, P. Sujaridworakun, F. Koh, T. Fujiwara, D. Pongkao and A. Ahniyaz, Mater. Sci. Eng. C, 24, 521 (2004).
M. J. Larsen, A. Thorsen and S. J. Jensen, Calcified Tissue Int., 37, 189 (1985).
W. Gelsema, C. De Ligny, A. Remijnse and H. Blijleven, Recueil. des. Travaux. Chimiques. Des. Pays., 85, 647 (1966).
X. Y. Liu, Bioinspiration: from nano to micro scales, Springer, New York (2012).
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Korea (NRF-2015R1D1A1 A01059580 and NRF-2019R1F1A1060060), by the Nano-Material Technology Development Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (2009–0082580), and by the Basic Core TechTechnology Development Program for the Oceans and the Polar Regions of the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (NRF-2015M1A5A1037054).
Author information
Authors and Affiliations
Corresponding author
Supporting Information
11814_2020_496_MOESM1_ESM.pdf
Solvo-hydrothermal synthesis of calcium phosphate nanostructures from calcium inositol hexakisphosphate precursor in water-ethanol mixed solutions
Rights and permissions
About this article
Cite this article
Han, JH., Pack, S.P. & Chung, S. Solvo-hydrothermal synthesis of calcium phosphate nanostructures from calcium inositol hexakisphosphate precursor in water-ethanol mixed solutions. Korean J. Chem. Eng. 37, 891–897 (2020). https://doi.org/10.1007/s11814-020-0496-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0496-3