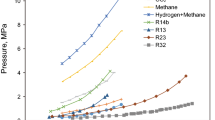
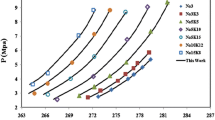
Abstract
Several models have been proposed to investigate the kinetics of gas hydrate formation. The main differences between the proposed models are the definition of the driving force, thermodynamics approach and the number of resistances to study the gas consumption by the hydrate phase. This paper concentrates on gas hydrate formation from multicomponent mixture, which has not been much studied before. In the present research, chemical potential has been considered as the driving force and, consequently, a new resistance coefficient was introduced. A complete discussion and reasonable assumptions has been provided to support this modelling.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
E.D. Sloan, J. Chem. Therm., 35, 41 (2003).
H.P. Veluswamy, P. S.R. Prasad and P. Linga, Korean J. Chem. Eng., 33, 1 (2015).
A. K. Sum, C. Koh and E.D. Sloan, Ind. Eng. Chem. Res., 48, 7457 (2009).
I. Chatti, Energy Convers. Manage., 46, 1333 (2005).
K. Nazridoust and G. Ahmadi, Chem. Eng. Sci., 62, 6155 (2007).
A. Vysniauskas and P. Bishnoi, Chem. Eng. Sci., 38, 1061 (1983).
P. Englezos and P. Bishnoi, Chem. Eng. Sci., 42, 2647 (1987).
C. Gaillard, J. Monfort and J. Peytavy, International Conference on Natural Gas Hydrates (1996).
J. Monfort, Annals of the New York Academy of Sciences, 912, 753 (2000).
M.A. Clarke and P. Bishnoi, Chem. Eng. Sci., 60, 695 (2005).
J. Zhang, S. Lee and J.W. Lee, Ind. Eng. Chem. Res., 46, 6353 (2007).
P. Skovborg and P. Rasmussen, Chem. Eng. Sci., 49, 1131 (1994).
D. Kashchiev and A. Firoozabadi, J. Crystal Growth, 241, 220 (2002).
C. P. Ribeiro and P.L. Lage, Chem. Eng. Sci., 63, 2007 (2008).
V. Mohebbi, A. Naderifar, R.M. Behbahani and M. Moshfeghian, Chem. Eng. Sci., 76, 580 (2012).
S. Lee, J. Zhang, R. Mehta, T. Woo and W. Lee, J. Phys. Chem., 111, 4734 (2007).
M.K. Chun and H. Lee, Korean J. Chem. Eng., 13, 620 (1996).
M. B. Malegaonkar, P.D. Dholabhai and P.R. Bishnoi, Canadian J. Chem. Eng., 75, 1090 (1997).
S. Bergeron and P. Servio, Fluid Phase Equilib., 265, 30 (2008).
S. Bergeron, J. G. Beltrán and P. Servio, Fuel, 89, 294 (2010).
J. Zhang and J.W. Lee, Ind. Eng. Chem. Res., 48, 5934 (2008).
M. Naseh, V. Mohebbi and R. Behbahani, J. Chem. Eng. Data, 59, 3710 (2014).
S. Bergeron and P. Servio, Fluid phase Equilib., 276, 150 (2009).
M. Najafi and V. Mohebbi, J. Nat. Gas Sci. Eng., 21, 738 (2014).
A. Izadpand, M. Vafaie and F. Varaminian, Iranian J. Chem. Chem. Eng., 26, 61 (2007).
B. Peng, J. Phys. Chem., 111, 12485 (2007).
S. Babaee, J. Chem. Therm., 81, 52 (2015).
H. Hashemi, J. Chem. Therm., 82, 47 (2015).
R. Christiansen and E.D. Sloan, Gas Processors Association, Tulsa (1995).
V. Mohebbi, A. Naderifar, R. M. Behbahani and M. Moshfeghian, J. Chem. Therm., 51, 8 (2012).
G. Soave, Chem. Eng. Sci., 27, 1197 (1972).
A. Danesh, PVT and phase behavior of petroleum reservoir fluids, Elsevier (1998).
V. Mohebbi and R. Behbahani, J. Nat. Gas Sci. Eng., 18, 47 (2014).
R. E. Treybal, Mass transfer operations, McGraw Hill (1980).
H. S. Fogler, Elements of chemical reaction engineering, Prentice Hall (1999).
W.R. Parrish and J. M. Prausnitz, Ind. Eng. Chem. Process Des. Dev., 11, 26 (1972).
Gas Processors and Suppliers Association Engineering Data Book, Tulsa, Oklahoma, U.S.A. (2004).
J. Munck, S. Skjold-Jørgensen and P. Rasmussen, Chem. Eng. Sci., 43, 2661 (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohebbi, V., Behbahani, R.M. & Naderifar, A. A new approach for modeling of multicomponent gas hydrate formation. Korean J. Chem. Eng. 34, 706–716 (2017). https://doi.org/10.1007/s11814-016-0318-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0318-9