Abstract
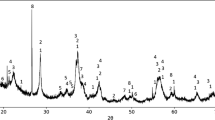
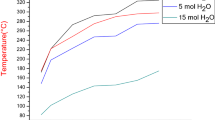
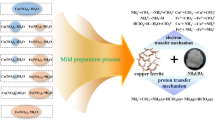
To catalytically decompose the greenhouse gas, CO2, spinel structure M-ferrites (M=Co, Ni, Cu, Zn) were synthesized by chemical co-precipitation using metal salts and sodium hydroxide as starting materials. The crystallite size of the newly-prepared M-ferrites increased and the BET surface area decreased with increasing calcination temperature. A thermal analysis of the reduction and reoxidation of M-ferrites indicated that substitution of divalent transition metals (i.e., Cu, Ni and Co) into Fe3O4 improved the reduction kinetics in the order of Cu>Ni>Co. ZnFe2O4 was the most difficult compound to completely reduce due to its stable structure. Commercial samples of the reduced Fe3O4, CoFe2O4 and ZnFe2O4 showed an increase in mass through the reoxidation process, but it was much more difficult for oxygen atoms to enter the structure of the reduced samples of NiFe2O4 and CuFe2O4. The M-ferrites in a batch type reactor showed better efficiency than the commercial Fe3O4. Also found was that CoFe2O4 showed a high regeneration potential, although it required a higher critical reaction temperature. NiFe2O4 and CuFe2O4 were excellent candidate materials for CO2 decomposition at lower temperatures.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
A. Sacco. and R. C. Reid, Carbon, 17, 459 (1979).
Y. Tamaura and M. Tahata, Nature, 346, 255 (1990).
T. Kodama, M. Tabata, K. Tominaga, T. Yoshida and Y. Tamaura, J. Mater. Sci., 28, 547 (1993).
M. Tabata, Y. Nishida, T. Kodama, K. Mimori, T. Yoshida and Y. Tamaura, J. Mater. Sci., 28, 971 (1993).
H. Kato, T. Kodama, M. Tsuji, Y. Tamaura and S. G. Chang, J. Mater. Sci., 29, 5689 (1994).
L. S. Chen, S. Chen and G. Lu, J. Mater. Sci., 41, 6465 (2006).
L. J. Ma, L. S. Chen and S. Y. Chen, J. Phys. Chem. Solids, 68, 6459 (2007).
J. Shen, Y. H. Lim and Y. M. Jo, Korean J. Ind. Chem., 22, 185 (2011).
M. H. Khedr, A. A. Omar and S. A. Abdel-Moaty, Colloids Surf., A: Physicochem. Eng. Aspects, 281, 8 (2006).
W. Z. Lv, B. Liu, Z. K. Luo, X. Z. Ren and P. X. Zhang, J. Alloys Compounds, 465, 261 (2008).
J. H. Ko, R. S. Park, J. K. Jeon, D. H. Kim, S. C. Jung, S. C. Kim and Y. K. Park, J. Ind. Eng. Chem., 32, 109 (2015).
E. H. Lee, R. S. Park, H. Kim, S. H. Park, S. C. Jung, J. K. Jeon, S. C. Kim and Y. K. Park, J. Ind. Eng. Chem., 37, 18 (2016).
C. S. Hwang and N. C. Wang, Mat. Chem. Phys., 88, 258 (2004).
M. Kobayashi, H. Shirai and M. Nunokawa, Ind. Eng. Chem. Res., 39, 1934 (2000).
M. H. Khedr, A. A. Omar and S. A. Abdel-Moaty, Colloids Surf., A: Physicochem. Eng. Aspects, 281, 8 (2006).
L. J. Ma, L. S. Chen and S. Y. Chen, Sol. State Sci., 11, 176 (2009).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated to Prof. Sung Hyun Kim on the occasion of his retirement from Korea University.
Rights and permissions
About this article
Cite this article
Jiaowen, S., Kim, D.W., Kim, S.B. et al. CO2 decomposition using metal ferrites prepared by co-precipitation method. Korean J. Chem. Eng. 33, 3162–3168 (2016). https://doi.org/10.1007/s11814-016-0192-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0192-5