Abstract
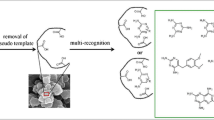
Synthesis and characterization of magnetic letrozole imprinted polymer nanoparticles is described herein for the first time. Magnetic molecularly imprinted polymers (MMIPs) were synthesized by precipitation polymerization using methacrylic acid (MAA) as functional monomer and trimethylolpropane trimethacrylate (TRIM) as crosslinker in the presence of letrozole as template and MAA-modified magnetite nanoparticles as magnetic component. The nanoparticles were characterized by scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray powder diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), thermogravimetric analysis (TGA), and a vibrating sample magnetometer (VSM). The synthesized MMIP nanoparticles, with particle size of about 100 nm, showed superparamagnetic features with a saturation magnetization of 12.5 emu·g−1 and had thermal stability below 240 °C. The adsorption experiments indicated better template recognition of MMIP than magnetic non-imprinted polymer (MNIP) nanoparticles. Moreover, the release profile of letrozole from MMIP and MNIP revealed the controlled release ability of MMIP nanoparticles for the letrozole anticancer drug. We also found that applying an external alternative magnetic field results in increasing the rate of the drug release.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
X. Xu, W. Ho, X. Zhang, N. Bertrand and O. Farokhzad, Trends Mol Med., 21, 223 (2015).
A. Wickia, D. Witzigmannb, V. Balasubramanianc and J. Huwylerb, J. Control. Release, 200, 138 (2015).
S. Ummadi, B. Shravani, N. G. R. Rao, M. S. Reddy and B. Nayak, Int J. Pharm. Sci., 3, 258 (2013).
D. Bhowmik, H. Gopinath, B. Pragati Kumar, S. Duraivel and K. P. Sampath Kumar, Pharma Innovation., 1, 24 (2012).
C. Alvarez-Lorenzo and A. Concheiro, J. Chromatogr. B., 804, 231 (2004).
H. Kempe, A. Parareda Pujolràs and M. Kempe, Pharm. Res., 32, 375 (2015).
M. Ali and M. E. Byrne, Pharm. Res., 26, 714 (2009).
F. Azizi Ishkuh, M. Javanbakht, M. Esfandyari-Manesh, R. Dinarvand and F. Atyabi, J. Mater. Sci., 49, 6343 (2014).
M. E. Byrne, J. Zachary Hilt and N. A. Peppas, J. Biomed. Mater. Res., 84, 137 (2008).
P. Luliski, Acta Pol. Pharm., 70, 601 (2013).
O. Ramstrom and R. J. Ansell, Chirality, 10, 195 (1998).
M. Komiyama, T. Takeuchi, T. Mukawa and H. Asanuma, Molecular Imprinting: From Fundamentals to Applications, Wiley-VCH, Weinheim (2003).
G. Vasapollo, R. Del Sole, L. Mergola, M. R. Lazzoi, A. Scardino, S. Scorrano and G. Mele, Int. J. Mol. Sci., 12, 5908 (2011).
F. Omidi, M. Behbahani, H. Sadeghi Abandansari, A. Sedighi and S. J. Shahtaheri, J. Environ. Health Sci. Eng., 12, 137 (2014).
T. Takeuchia and J. Haginakab, J. Chromatogr. B., 72, 1 (1999).
M. Ulbricht, J. Chromatogr. B., 804, 113 (2004).
L. Uzun and A. Turner, Biosens. Bioelectron., 76, 131 (2016).
J. Xia, X. Cao, Z. Wang, M. Yang, F. Zhang, B. Lu, F. Li, L. Xia, Y. Li and Y. Xia, Sens. Actuators, B: Chemical., 225, 305 (2016).
W. J. Cheong, F. Ali, J. H. Choi, J. O. Lee and K. Y. Sung, Talanta, 106, 45 (2013).
J. Lee, S. Bernard and X. Liu, React. Funct. Polym., 69, 650 (2009).
K. Ohkubo, Y. Urata, S. Hirota, Y. Honda, Y. Fujishita and T. Sagawa, J. Mol. Catal, 93, 189 (1994).
D. L. Rathbone, Adv. Drug Deliv. Rev., 57, 1854 (2005).
F. Puoci, G. Cirillo, M. Curcio, O. I. Parisi, F. Iemma and N. Picci, Expert Opin. Drug Deliv., 8, 1379 (2011).
B. Sellergren and C. J. Allender, Adv. Drug Deliv. Rev., 57, 1733 (2005).
C. F. van Nostrum, Drug Discov. Today Technol., 2, 119 (2005).
M. S. da Silva, F. L. Nobrega, A. A. Ricardo, E. J. Cabrita and T. Casimiro, J. Supercrit. Fluids, 58, 150 (2011).
G. Cirillo, F. Iemma, F. Puoci, O. I. Parisi, M. Curcio, U. G. Spizzirri and N. Picci, J. Drug Target., 17, 72 (2009).
W. Chen, Y. Ma, J. Pan, Z. Meng, G. Pan and B. Sellergren, Polymers, 7, 1689 (2015).
D. R. Kryscio and N. A. Peppas, AIChE J., 55, 1311 (2009).
A. Shkilnyy, E. Munnier, K. Herve, M. Souce, R. Benoit, S. Cohen-Jonathan, P. Limelette, M. L. Saboungi, P. Dubois and I. Chourpa, J. Phys. Chem. C., 114, 5850 (2010).
M. Mahmoudi, A. Simchi, M. Imani and U. O. Hafeli, J. Phys. Chem. C., 113, 8124 (2009).
T. Chen, M. I. Shukoor, R. Wang, Z. Zhao, Q. Yuan, S. Bamrungsap, X. Xiong and W. Tan, ACS Nano., 5, 7866 (2011).
P. Dramou, P. Zuo, H. He, L. A. Pham-Huy, w. Zou, D. Xiao, C. Pham-Huyc and T. Ndorbora, J. Mater. Chem. B., 1, 4099 (2013).
O. L. Parisi, C. Morelli, F. Puoci, C. Saturnino, A. Caruso, D. Sisci, G. E. Trombino, N. Piccia and M. S. Sinicropia, J. Mater. Chem. B., 2, 6619 (2014).
X. Kan, Z. Geng, Y. Zhao, Z. Wang and J. J. Zhu, Nanotechnology, 20, 1 (2009).
X. Xia, E. P. C. Lai and B. Ormeci, Polym. Eng. Sci., 52, 1775 (2012).
C. Dunn and S. J. Keam, PharmacoEconomics, 24, 495 (2006).
D. Simpson, M. P. Curran and C. M. Perry, Drugs, 64, 1213 (2004).
J. M. Dixon, C. D. B. Love, C. O. C. Bellamy, D. A. Cameron1, R. C. F. Leonard, H. Smith and W. R. Miller, Breast Cancer Res Treat., 66, 191 (2001).
C. Fontaine, A. Meulemans, M. Huizing, C. Collen, L. Kaufman, J. De Mey, C. Bourgain, G. Verfaillie, J. Lamote, R. Sacre, D. Schallier, B. Neyns, J. Vermorken and J. De Greve, Breast., 17, 376 (2008).
A. Lipton, L. M. Demers, H. A. Harvey, K. B. Kambic, H. Grossberg, C. Brady, H. Adlercruefz, P. F. Trunet and R. J. Sanfen, Cancer., 75, 2132 (1995).
Q. J. Khan, P. S. Reddy, B. F. Kimler, P. Sharma, S. E. Baxa, A. P. O’Dea, J. R. Klemp and C. J. Fabian, Breast Cancer Res Treat., 119, 111 (2010).
S. K. Dey, B. Mandal, M, Bhowmik and L. K. Ghosh, Braz. J. Pharm. Sci., 45, 585 (2009).
A. J. Siddiqa, K. Chaudhury and B. Adhikari, Colloids Surf., B., 116, 169 (2014).
M. R. Saboktakin, R. M. Tabatabaie1, A. Maharramov and M. A. Ramazanov, J. Pharm. Educ. Res., 1, 62 (2010).
S. Kazemi, A. A. Sarabi Daryani, M. Abdouss and Z. Shariatinia, J. Theor. Comput. Chem., 15, 1650015 (2016).
Y. Shi, H. Lv, X. Lu, Y. Huang, Y. Zhanga and W. Xue, J. Mater. Chem., 22, 3889 (2012).
T. Jing, H. Du, Q. Dai, H. Xia, J. Niu, Q. Hao, S. Mei and Y, Zhou, Biosens. Bioelectron., 26, 301 (2010).
Y. Ding, Y. Hu, L. Zhang, Y. Chen and X. Jiang, Biomacromolecules, 7, 1766 (2006).
B. Liu, M. Han, G. Guan, S. Wang, R. Liu and Z. Zhang, J. Phys. Chem. C., 115, 17320 (2011).
D. Niu, Z. Zhou, W. Yang, Y. Li, L. Xia, B. Jiang, W. Xu, W. Huang and T. Zhu, J. Appl. Polym. Sci., 130, 2859 (2013).
O. Karaagac, H. Kockar, S. Beyaz and T. Tanrisever, IEEE Trans. Magn., 46, 3987 (2010).
H. Surikumaran, S. Mohamad and N. Muhamad Sarih, Int. J. Mol. Sci., 15, 6111 (2014).
H. G. Schild, J. Polym. Sci. Part A Polym. Chem., 31, 2403 (1993).
Y. Zhang, R. Liu, Y. Hu and G. Li, Anal. Chem., 81, 967 (2009).
A. H. Lu, E. L. Salabas and F. Schuth, Angew. Chem., 46, 1222 (2007).
S. Beyaz, H. Kockar and T. Tanrisever, J. Optoelectron. Adv. M., 1, 447 (2009).
M. Mahdavi, M. Bin Ahmad, J. Haron, F. Namvar, B. Nadi, M. Z. Ab Rahman and J. Amin, Molecules, 18, 7533 (2013).
X. X. Li, L. F. Hao, Y. P. Huang, H. Q. Duan and Z. S. Liu, Polym. Eng. Sci., 52, 1440 (2012).
S. Likhitkar and A. K. Bajpai, Carbohydr. Polym., 87, 300 (2012).
N. Griffete, J. Fresnais, A. Espinosa, C. Wilhelm, A. Bée and C. Ménager, Nanoscale, 7, 18891 (2015).
G. R. Mahdavinia, H. Etemadi and F. Soleymani, Carbohydr. Polym., 128, 112 (2015).
M. Uva, D. Pasqui, L. Mencuccini, S. Fedi and R. Barbucci, J. Biomater. Nanobiotechnol., 5, 116 (2014).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kazemi, S., Sarabi, A.A. & Abdouss, M. Synthesis and characterization of magnetic molecularly imprinted polymer nanoparticles for controlled release of letrozole. Korean J. Chem. Eng. 33, 3289–3297 (2016). https://doi.org/10.1007/s11814-016-0171-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0171-x