Abstract
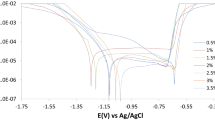
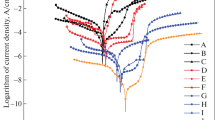
As a material with good corrosion resistance, 5083 aluminum alloy has a great application prospect in marine environment. In this work, the corrosion characteristics of 5083 aluminum alloy in seawater containing phosphate were investigated with Potentiodynamic Polarization, Electrochemical Impedance Spectroscopy (EIS), Scanning Electron Microscope (SEM), Energy Dispersive Spectroscopy Analysis (EDSA), X-ray Photoelectron Spectroscopy (XPS) and Laser Confocal Microscope. The results indicated that the effects of phosphate in seawater were two-fold. Firstly, phosphate slightly accelerated the corrosion of 5083 in seawater in the early stage of corrosion. HPO42− competed with OH− in the adsorption process on the alloy surface, which weakened the contact between OH− and Al3+ near the interface of the alloy, and inhibited the formation as well as the self-repair of the passive film, thus accelerating the activation dissolution process. Compared with the natural seawater, the charge transfer resistance of 5083 in the seawater containing phosphate decreased faster during the early stage of corrosion, and the corrosion current density icorr was higher in seawater containing phosphate. On the other hand, the addition of phosphate would not affect the cluster distribution of the second phase of 5083 in seawater, but it changed the composition of the corrosion product layer and had an obvious inhibitory effect on the local corrosion of 5083 in seawater. After 16-day exposure, shallower and more sparsely distributed pits could be observed on the derusted surface of 5083 in the seawater containing phosphate, and the pitting coefficient in the seawater containing phosphate was significantly lower than that in natural seawater. The reduction of pitting tendency could be realized mainly through two ways. First, the HPO42− adsorbed on the surface of the passive film in the early stage of corrosion and repeled the corrosive anions such as Cl−. Second, phosphate participated in the construction of the CaHPO4 precipitation film, which acted as a barrier and protection.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Agnès, D. P., William, P., Theophil, L., and Ginette, A., 2005. Detection or quantitative analysis of a corrosion inhibitor, the sodium monofluorophosphate, in concrete. Cement and Concrete Composites, 27(6): 679–687, DOI: https://doi.org/10.1016/j.cemconcomp.2004.11.002.
Andrade, C., Alonso, C., Acha, M., and Malric, B., 1992. Preliminary testing of Na2PO3F as a curative corrosion inhibitor for steel reinforcements in concrete. Cement and Concrete Research, 22(5): 869–881.
Bach, L. X., Son, D. L., Phong, M. T., Thang, L. V., Bian, M. Z., and Nam, N. D., 2019. A study on Mg and ALN composite in microstructural and electrochemical characterizations of extruded aluminum alloy. Composites Part B Engineering, 156: 332–343, DOI: https://doi.org/10.1016/j.compositesb.2018.08.139.
Basame, S. B., and White, H. S., 2000. Pitting corrosion of titanium: The relationship between pitting potential and competitive anion adsorption at the oxide fifilm/electrolyte interface. Journal of the Electrochemical Society, 147(4): 1376–1381.
Conde, A., and De Damborenea, J., 1996. Effect of surface depletion of lithium on corrosion behaviour of aluminium alloy 8090 in a marine atmosphere. Journal of Materials Science, 31: 4921–4926, DOI: https://doi.org/10.1007/BF00355881.
Conde, A., and De Damborenea, J., 1997. An electrochemical impedance study of a natural aged Al-Cu-Mg alloy in NaCl. Corrosion Science, 39(2): 295–303, DOI: https://doi.org/10.1016/S0010-938X(97)83348-3.
Conde, A., De Damborenea, J., Durán, A., and Menning, M., 2006. Protective properties of a sol-gel coating on zinc coated steel. Journal of Sol-Gel Science and Technology, 37(1): 79–85, DOI: https://doi.org/10.1007/s10971-005-5357-3.
Dhouibi, L., Triki, E., Salta, M., Rodrigues, P., and Raharinaivo, A., 2003. Studies on corrosion inhibition of steel reinforcement by phosphate and nitrite. Materials and Structures, 36(8): 530–540.
Huang, P., Somers, A., Howlett, P. C., and Forsyth, M., 2016. Film formation in trihexyl(tetradecyl)phosphonium diphenylphosphate ([P6,6,6,14][dpp]) ionic liquid on AA5083 aluminium alloy. Surface & Coatings Technology, 303: 385–395, DOI: https://doi.org/10.1016/j.surfcoat.2015.12.060.
Jafarzadeh, K., Shahrabi, T., and Hosseini, M. G., 2008. EIS study on pitting corrosion of AA5083-H321 aluminum-magnesium alloy in stagnant 3.5% NaCl solution. Journal of Materials Science and Technology, 24(2): 215–219.
Jafarzadeh, K., Shahrabi, T., Hadavi, S. M. M., and Hosseini, M. G., 2007. Role of chloride ion and dissolved oxygen in electrochemical corrosion of AA5083-H321 aluminum-magnesium alloy in NaCl solutions under flow conditions. Journal of Materials Science and Technology, 23(5): 623–628.
Jaya, P. V., Rao, N. M., Kamaluddin, S., and Surya, K., 2018. A study of microstructure and tribological properties of Al 5083 MMC processed by direct extrusion. Materials Today: Proceedings, 5(2): 8232–8240.
Jones, R. H., Baer, D. R., Danielson, M. J., and Vetrano, J. S., 2001. Role of Mg in the stress corrosion cracking of an Al-Mg alloy. A, Physical Metallurgy and Materials Science, 32(7): 1699–1711, DOI: https://doi.org/10.1007/s11661-001-0148-0.
Kaoru, M., Anders, N., and Ingemar, O., 2001. Surface reactions during pickling of an aluminium-magnesium-silicon alloy in phosphoric acid. Corrosion Science, 43(2): 381–396, DOI: https://doi.org/10.1016/S0010-938X(00)00069-X.
Liu, H. X., Du, Q. L., Duan, J. Z., Zhai, X. F., and Hou, B. R., 2016. Effects of natural seawater microorganisms on initial corrosion behavior of 5083 aluminum alloy. Corrosion Science and Protection Technology, 28(1): 51–57.
Lytle, D. A., and Nadagouda, M. N., 2010. A comprehensive investigation of copper pitting corrosion in a drinking water distribution system. Corrosion Science, 52(6): 1927–1938, DOI: https://doi.org/10.1016/j.corsci.2010.02.013.
Nam, N. D., Hung, T. V., Ngan, D. T., Hung, N. L. T., and Hoi, T. K. N., 2016. Film formation in Y(4NO2Cin)3 compound on 6061 aluminum alloy to protect against corrosion in chloride ion media. Journal of the Taiwan Institute of Chemical Engineers, 67: 495–504, DOI: https://doi.org/10.1016/j.jtice.2016.08.005.
Nam, N. D., Phung, V. D., Thuy, P. T. P., Dao, V. A., Kim, S. H., and Yi, J. S., 2019. Corrosion behaviours of hot-extruded Al-xMg alloys. Journal of Materials Research and Technology, 8(6): 5246–5253, DOI: https://doi.org/10.1016/j.jmrt.2019.08.047.
Natishan, P. M., Yu, S. Y., O’Grady, W. E., and Ramaker, D. E., 2002. X-ray absorption near edge structure and X-ray photo electron spectroscopy studies of chloride in passive oxide films. Electrochimica Acta, 47(19): 3131–3136, DOI: https://doi.org/10.1016/S0013-4686(02)00232-3.
Rudd, W. J., and Scully, J. R., 1980. The function of the repassivation process in the inhibition of pitting corrosion on ahuminium. Corrosion Science, 20(5): 611–631.
Soltis, J., 2015. Passivity breakdown, pit initiation and propagation of pits in metallic materials — Review. Corrosion Science, 90: 5–22, DOI: https://doi.org/10.1016/j.corsci.2014.10.006.
Theivaprakasam, S., Girard, G., Howlett, P., Forsyth, M., Mitra, S., and MacFarlane, D., 2018. Passivation behaviour of aluminium current collector in ionic liquid alkyl carbonate (hybrid) electrolytes. NPJ Materials Degradation, 2(1): 1–9, DOI: https://doi.org/10.1038/s41529-018-0033-6.
Wagner, M. F., 2018. Light-weight aluminum-based alloys — From fundamental science to engineering applications. Metals, 8(4): 260, DOI: https://doi.org/10.3390/met8040260.
Yang, S. H., Zhang, D. C., and Tian, Y. B., 2008. Study on the corrosion of 5083 Al-Mg alloy in different concentrations of NaCl solution. Science and Engineering of Nonferrous Metals, 9(2): 1–5 (in Chinese with English abstract).
Yohai, L., Schreiner, W. H., Vázquez, M., and Valcarce, M. B., 2011. Surface characterization of copper, zinc and brass in contact with tap water inhibited with phosphate ions. Applied Surface Science, 257(23): 10089–10095, DOI: https://doi.org/10.1016/j.apsusc.2011.07.002.
Zaid, B., Saidi, D., Benzaid, A., and Hadji, S., 2008. Effects of pH and chloride concentration on pitting corrosion of AA6061 aluminum alloy. Corrosion Science, 50(7): 18411847, DOI: https://doi.org/10.1016/j.corsci.2008.03.006.
Zazi, N., and Chopart, J., 2017. Dissolution of Ag/AgCl reference electrode and deposition of silver onto the surface of 5083 H321 aluminum alloy, during corrosion in 3wt% NaCl solution at rest potential. Protection of Metals and Physical Chemistry of Surfaces, 53(6): 1114–1119, DOI: https://doi.org/10.1134/S2070205117060235.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (No. U1706221).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, J., Yang, H., Du, M. et al. Corrosion Mechanism of 5083 Aluminum Alloy in Seawater Containing Phosphate. J. Ocean Univ. China 20, 372–382 (2021). https://doi.org/10.1007/s11802-021-4545-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-021-4545-2