Abstract
Carabid species richness and phylogenetic diversity were evaluated in the four biogeographic provinces present in the state of Hidalgo, Mexico. Using a list of species and incorporating records obtained from a review of the Coleoptera Collection of the Autonomous University of the State of Hidalgo, 752 records were obtained, which included 138 species classified into six taxonomic categories. The provinces with the highest species richness are the Sierra Madre Oriental and the Chihuahuan Desert; those with the lowest species richness are the Transmexican Volcanic Belt and the Veracruzan province. The Chihuahuan Desert was the province with the greatest phylogenetic diversity, and the Sierra Madre Oriental the least diverse. This highlights the usefulness of complementary estimates in diversity analyses in regions with a variety of disparate biogeographical histories.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The number of species in an area has traditionally been the main factor defining its diversity at different spatial scales (Magurran 2004). However, the concept of diversity in living organisms can be understood as the variety, variability and ecological dynamics among them; that is, diversity within species, between species, and in ecosystems (Gaston 1996; Chao et al. 2014). Thus, the metrics for estimating diversity in different disciplines with distinct attributes are essential for understanding the structure, function, and patterns and processes of biodiversity under a complementary approach (Moreno et al. 2007).
In this context, phylogenetic information is one of the fundamental components in the various ecological disciplines, since it allows evolutionary differences between lineages to be recognized, which can serve to explain certain biological processes (Tucker et al. 2017). This is one of the reasons that phylogenetic metrics are increasingly used to estimate diversity by incorporating information from an evolutionary approach (Pielou 1975; Chao et al. 2010, 2014). However, it is difficult to make these estimates because there is little information on phylogenies in specific biological groups, limiting the usefulness of the method (Tucker et al. 2017; Pérez-Hernández 2019; Cruz-Elizalde et al. 2022), therefore, in this work it is used in this way.
Diversity measurements that incorporate information on the number of species and their evolutionary history can facilitate the understanding of the mechanisms that shape the structure of biological communities (Cruz-Elizalde et al. 2022). One such index is the average taxonomic distance index proposed by Warwick and Clarke (1995), which uses Linnaean hierarchies to represent practical approximations of phylogenies through taxonomic relatedness and species richness (Campbell and Novelo 2007). This index takes into account the taxonomic structure of the species present in a community for determining the relationship among them; that is, each site is characterized according to the taxonomic category of each organism (Pérez-Hernández 2019). This has proven to be a practical way to estimate phylogenetic diversity (Campbell and Novelo 2007; Moreno et al. 2009; Tolimieri and Anderson 2010; García de Jesús et al. 2016; Miller et al. 2017; Tucker et al. 2017; Peña-Joya et al. 2018).
Diversity can be estimated at different spatial scales: one of these is at the level of biogeographic provinces, which constitute the basic biogeographic categories in a regionalization supported by the concept of endemism. This method of regionalization is a hierarchical system that categorizes geographic areas in terms of their endemic biota (Escalante 2009). Four of the 14 biogeographic provinces of Mexico are present in the state of Hidalgo (Morrone et al. 2017); the Sierra Madre Oriental (SMO), the Chihuahuan Desert (CD), the Transmexican Volcanic Belt (TVB) and the Veracruzan province (VZ). These provinces represent natural biogeographic units, which are ideal reference frameworks for various types of biological analyses (Morrone 2018), such as estimates of diversity employing a complementary context that recognizes not only species richness but also the evolutionary differences between species.
The family Carabidae is one of the most diverse groups of the order Coleoptera, with 40,000 species described worldwide (Bouchard et al. 2017). Although 2,012 species are known for Mexico (Cerón-Gómez et al. 2022), there are few studies focused on estimating diversity using different metrics and in areas with different vegetation types, elevations, and geological histories, such as the different biogeographic provinces.
This study therefore sets out to estimate carabid diversity in the four biogeographic provinces of the state of Hidalgo, estimating species richness under a constant sample coverage, and determining phylogenetic diversity using the index of mean taxonomic distance as an approximation of evolutionary distance. It is expected that diversity will differ between the four biogeographic provinces because of their different characteristics and evolutionary histories. It is also expected that the values obtained for standardized richness and phylogenetic diversity will not reflect the same trends in all provinces because taxonomic distance index is independent of sampling effort and of species richness and abundance (Clarke and Warwick 1998).
Materials and methods
Study area
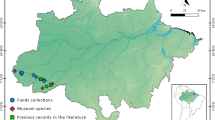
The state of Hidalgo is located in the central part of Mexico, with an area of 20,905 km2, which is 1.1% of the area of the entire country (Sánchez-Rojas and Bravo-Cadena 2017). Four biogeographic provinces are represented in the state (Fig. 1). The Sierra Madre Oriental (SMO) province crosses the state from northeast to southeast and is characterized by pine-oak and pine forest, cloud forest, and oak and arid tropical scrub, with medium and high elevations (above 1,000 m). The Chihuahuan Desert (CD) is located in the center-west of the state, where arid tropical scrub predominates, as well as grasslands and small fragments of pine-oak forest, with elevations of 1,000–2,400 m. The Transmexican Volcanic Belt (TVB) is in the south-southeast area of the state, with fragments of pine, pine-oak, táscate (Juniperus deppeana) and oyamel (Abies religiosa) forest, and with large cultivated areas and a considerable urban area (INEGI 2017). The province is located at medium and high elevations, ranging between 1,000 and 2,500 m. The Veracruzan (VZ) province has the lowest elevations, not exceeding 500 m, and the main cover is tropical evergreen forest (Montiel-Canales and Goyenechea 2021).
Map of biogeographic provinces present in the state of Hidalgo. Prepared from digital data in Morrone et al. (2017)
Data collection
The list of Carabidae species of the state of Hidalgo by biogeographic province was obtained from Cerón-Gómez et al. (2022), derived from the review of specialized literature, databases and entomological collections. In addition, some records not included in the latter study were incorporated in the present work; these were obtained from a review of additional specimens deposited in the Coleoptera Collection of the Autonomous University of the State of Hidalgo (CC-UAEH). This information is detailed in Online Resource 1 and 2.
Data analysis
Inventory completeness. Species accumulation curves by biogeographic province were calculated with the EstimateS program (Colwell 2013) based on the bootstrap non-parametric estimator using presence/absence data, which does not assume a predetermined mathematical model for estimating the total number of species (Pineda-López and Verdú-Faraco 2013).
Estimation of species richness (α diversity). For each biogeographic province, species richness (diversity of order 0; q = 0) was calculated with the same sample coverage (0.877). This method, according to Chao and Jost (2012), leads to a more integrated estimate that is more equitable between incomplete samples. The estimate is based on a replication principle that reflects the most significant information for different values of diversity between the communities being compared rather than the sizes of the samples.
Analysis of phylogenetic diversity. Using the concept of taxonomic distance proposed by Warwick and Clarke (1995), which is based not only on a taxonomic hierarchy, but also on the presence of carabid species in the four biogeographic provinces, the taxonomic distance index was calculated (average taxonomic distinctness, Δ+) between pairs of species from each site using the following formula (Clarke and Warwick 1998):
where S is the number of species and ωij is the taxonomic distance between species i and species j. This measure takes into account the taxonomic categories in which any pair of species are related; that is, the distance that separates pairs of species in the taxonomic classification.
The estimate of Δ+ was based on an aggregation matrix of six taxonomic categories (subfamily, tribe, subtribe, genus, subgenus and species) considered as “branches” with a standardized and cumulative differential weight up to 100 (Clarke and Warwick 1999), corresponding to the distance between two species of carabids. The analysis was carried out with the Primer 5 program from Primer–e for Windows using 1000 randomizations from each site.
Results
A total of 752 records of 138 carabid species were obtained for the state of Hidalgo in six taxonomic categories (10 subfamilies, 28 tribes, 24 subtribes, 49 subgenera, and 54 genera). The greatest number of species records was from the SMO province with 348 records (46.2%), followed by the CD province with 235 (31.2%), the TVB province with 99 (13.1%), and the smallest number from the VZ province with 70 (9.3%).
According to the bootstrap estimator, the species accumulation curves show higher values in all four biogeographic provinces, predicting 102 species for the SMO province, 78 species for the CD, 22 species for the TVB and 21 species for the VZ province (Fig. 2). This reflects percentages of inventory completeness of 77% in three of the provinces, and 76% for VZ.
Species accumulation curves for the four biogeographic provinces. The blue dots represent species richness, the red dots the number of species estimated by the bootstrap estimator. The X axis represents the number of sites per province; the Y axis the number of species (SMO = Sierra Madre Oriental; CD = Chihuahuan Desert; TVB = Transmexican Volcanic Belt; VZ = Veracruzana)
When species richness (q = 0) was analyzed using a standardized coverage value (0.877), different values of species richness were observed for each of the biogeographic provinces: SMO = 66.1, CD = 60, TVB = 12.06, VZ = 14.44 (Fig. 3). Based on these estimates and their associated confidence intervals, it is observed that there are no significant differences in richness between the SMO and CD provinces, or between the TVB and VZ provinces, but there are differences between SMO vs. TVB and SMO vs. VZ, and also between CD vs. TVB and CD vs. VZ.
The estimates of phylogenetic diversity (Δ+) show that two biogeographic provinces have values close to the average value expected by chance; namely, the TVB (Δ+ = 75.49) and VZ (Δ+ = 70.28) provinces. For the other two provinces, significant differences in phylogenetic diversity are observed. Phylogenetic diversity in the SMO (Δ+ = 69.86) is lower than expected, while it is higher than expected (Δ+ = 82.24) in the CD (Fig. 4).
Relationship between average taxonomic distance values (Δ+) and species richness in the four biogeographic provinces. The dotted line indicates the randomly determined average value of taxonomic distance and the solid lines are the confidence intervals. The abbreviations of the provinces are indicated in Fig. 3
When comparing the values obtained for phylogenetic diversity with species richness by biogeographic provinces, it can be seen that the SMO province presents a high richness of species, but is the least diverse according to the values of phylogenetic diversity. In contrast, the TVB province presents a low species richness, and a phylogenetic diversity above the mean value that would be expected by chance, although not significantly high, as indicated by the confidence intervals. The VZ province has phylogenetic diversity values similar to those of the SMO, but in terms of species richness, it is notably less diverse than the SMO province. The CD province shows a high phylogenetic diversity, despite not presenting significant differences in species richness with respect to the SMO province.
Discussion
The results obtained for species richness and phylogenetic diversity are different in most of the biogeographical provinces. Standardized species richness was significantly different between provinces compared by pairs; SMO plus CD compared to TVB plus VZ. One reason for this could be the close historical relationship between the SMO and the southern part (within the state of Hidalgo) of the CD province, where the two provinces overlap (Morrone et al. 1999). Moreover, the lower species richness found in the TVB and VZ provinces may be influenced by the various anthropocentric activities carried out in these regions. For example, the TVB contains the most populated cities in the state, including the capital Pachuca de Soto and its metropolitan area (consisting of six more municipalities), which has one of the highest urban growth in Mexico, more than doubling since 1990 and until 2020 (Franco-Sánchez and Contreras-Cerón 2021). Some species, such as Harpalus laevipes, Agonum placidum and Panagaeus sallei, with data from before 1990, have not been currently recorded in this area of the state of Hidalgo. This could be an indication of the negative effect caused by urbanization on the loss of diversity in some species of carabids and other groups of insects (Wagner et al. 2021). On the other hand, since the 70’s there has been a significant loss of humid forests in the country, the predominant ecosystem in the VZ province, due to different anthropocentric causes (Sánchez-Colón et al. 2009). In addition, this province is constantly threatened by oil extraction and the expansion of farmland and cattle ranching, among other activities (Dinerstein et al. 1995). Although some protected natural areas are located in the VZ (CONANP 2018), none are located in the part of the province present in the state of Hidalgo (Morrone 2019). All these factors could have an influence on the low representation of species in this biogeographic province.
The species accumulation curves as calculated by the bootstrap estimator show higher species numbers in each of the biogeographic provinces. Therefore, the species richness estimate is based on the same sample coverage for each province in order that non-sampled species may constitute the total set of species in a community (Chao and Jost 2012). However, the lower number of records in the TVB and VZ provinces is evident, suggesting that the numbers of species found are still far from 100% complete, even in the SMO and CD provinces. Ramírez-Hernández et al. (2022) report a similar trend when they estimated the species richness of tiger beetle species (Cicindelinae) in the 14 biogeographic provinces of Mexico, finding the highest richness in the CD province. This highlights the importance of this province as an area of endemism in the Nearctic region (Morrone et al. 2017); it has indeed been noted for its richness of genera and species of this group of carabids.
Phylogenetic diversity values are different from those of species richness in two of the four biogeographic provinces studied. The SMO province is particularly interesting due to its evident contrast between the two measures, since it showed the highest species richness and the lowest phylogenetic diversity. This may be related to the elevation ranges above one thousand meters present in the province. For example, it is known that high and mountainous areas in Mexico in general are notable for their high diversity of carabids at the genus level (Ball and Shpeley 2000), which is reflected in this province since it is where the largest number of genera (36) occur. These high elevation areas are important in terms of endemism. In particular, a high richness of endemic species has been reported for the SMO in other groups of Coleoptera with low locomotor capacity, such as the pasalids (Gutiérrez-Velázquez et al. 2013). The same pattern can be seen with carabids in the SMO province, since 67% of the 55 species registered in Hidalgo and which are endemic to Mexico are found in this biogeographic province.
However, the SMO presents a low phylogenetic diversity, which is an unexpected and important result considering that this province is part of the Mexican Transition Zone (MTZ) (Morrone 2015), so its species composition is a mixture of Nearctic and Neotropical elements (Fig. 5) that exhibit some of the distribution patterns proposed by Halffter (2017). For example, the northern genus Agonum underwent its diversification in the Eocene, when Europe and North America were connected, so its expansion towards the MTZ would have been during the early Cenozoic (Liebherr and Schmidt 2004); this would suggest a Paleoamerican distribution pattern (Mesoamerican montane subpattern). According to Liebherr (1994a) the holarctic genera Loricera, Notiobia, and Calathus present a Nearctic distribution pattern, which suggests a more recent expansion to the MTZ, probably between the Miocene and Pliocene. Taxa with a Mesoamerican origin are also recognized, such as the genus Onypterygia, which could fit the Mesoamerican montane pattern (Whitehead and Ball 1997).
Despite this, the low phylogenetic diversity in the SMO can be explained by the fact that 63% of the species belong to the subfamily Harpalinae, which implies that these species are more related to each other than to species of other subfamilies, and that they are related even at supraspecific taxonomic categories. For example, despite the different patterns exhibited by the genera Agonum, Onypterygia, and Platynus (Liebherr 1994a, b; Liebherr and Kipling 1996; Whitehead and Ball 1997), all are part of Platynini, belonging to the Harpalinae. This tribe is well represented in the mountain ranges of both the Sierra Madre Oriental and the Sierra Madre Occidental, provinces that are part of the Mexican montane component, which implies certain historical relationships and close affinities for which different biological groups in addition to carabids have provided evidence (Marshall and Liebherr 2000; Morrone and Márquez 2001; Morrone 2019), so the phylogenetic diversity appears more homogeneous.
In contrast to what was observed in the SMO, the CD is a province where a high species richness and the greatest phylogenetic diversity is obtained, which makes the same trend evident in both metrics. This pattern of similarity between different measures of diversity has been previously reported with Melolonthidae beetles in an area of the Transmexican Volcanic Belt (García de Jesús et al. 2016) and with beetles of the Scarabaeoidae superfamily, in a study also incorporating functional diversity (Moreno et al. 2007). This consistency in the trends of the estimates provides more robust information on diversity in the CD province, since the values are supported not only by the greater number of species but also by their phylogenetic variability, considering the taxonomic structure of their species.
The great variety in lineages can be explained because the CD province has a mixture of species with different biogeographical affinities (Fig. 5). Species of Neotropical affinity may have, according to Halffter (1964, 2017), expanded from South America towards the CD in the Miocene and diversified in the Pliocene when they were already in the CD. The presence of Nearctic affinity taxa (Morrone 2019) also contributes to the coexistence of different lineages, since they could have been part of the CD since Pangea separated into Laurasia and Gondwana, or they could have expanded from Canada and the United States as Halffter (2017) postulates for the Paleoamerican distribution pattern (Altiplano subpattern). This favors the coexistence of much less related elements at various different taxonomic levels; for example, the presence of species such as bombardier beetles (Brachininae) and tiger beetles (Cicindelinae), which are separated at the subfamily level.
Map showing the four biogeographic provinces and their convergence in the state of Hidalgo. Prepared from digital data in Morrone et al. (2017)
On the other hand, the TVB province corresponds to the mountain system considered to be the core of the Mexican Transition Zone (Halffter and Morrone 2017) which is where the overlap between the Nearctic and Neotropical regions is the greatest. This could imply a high phylogenetic diversity, since the characteristics of the transition zone determine diversity not only in terms of number of species, but also of phylogenetic lineages (Halffter 2017). It is known that there are lineages characteristic of the Nearctic region that come into contact with typical lineages of the Neotropical region, in addition to the endemic elements of the province. An example from another group of beetles is the genus Thanatophilus, belonging to the Silphidae family, which presents a Nearctic distribution pattern (Halffter and Morrone 2017). In the carabids, groups of species that exemplify this pattern in the TVB can also be recognized, such as the genera Elliptoleus and Sericoda (Liebherr 1994a). Lineages with a possible South American origin can be exemplified by species of the genus Pasimachus (Erwin 1985). In addition, there are other groups of Coleoptera, such as Canthon indigaceus and its three subspecies, which present a Neotropical distribution pattern with a broad penetration that reaches the TVB province and even further north (Halftter 2017; Halffter and Morrone 2017). This suggests that a large phylogenetic diversity of carabids should be expected in this biogeographic province, but it was not found in the present study. This could be due to the considerable fragmentation of the province, due to its heterogeneous climate and elevation, which are a result of its complex geological origin (Gámez et al. 2012). In addition, the large amount of anthropocentric activity present in the part of the state where the TVB province is located could also contribute to the results found in this study.
The VZ province is the only one of the four present in the state that is part of the Neotropical region (Morrone 2017, 2019). Evidence of this is the presence of species of the genera Agra, Morion, Galerita, and Phloeoxena (Reichardt 1967; Allen 1968; Shpeley and Ball 2000; Erwin 2017). The few species registered in this province belong to the tribes Lebiini and Cicindelini, which implies that they are more related at the tribe level, so the phylogenetic diversity values obtained are low. However, there is no statistically significant difference in phylogenetic diversity, due to the few species present in VZ.
Hypothetically, with an increase in the number of species in the TVB and VZ provinces, a greater phylogenetic diversity could be expected. However, this would depend entirely on the composition of the species present in each province, so a change in the trends found so far could not be guaranteed, since the average taxonomic distance index is independent of sampling effort and species richness; that is, greater species richness does not necessarily lead to greater phylogenetic diversity (Clarke and Warwick 1998).
When using the biogeographic provinces as a scale of analysis, a correspondence can be observed with the geological provinces that occur in Hidalgo made up of formations and lithological units (Hernández-Avelino 2008). With this, a greater species richness can be seen in a sedimentary sequence that includes limestone, lower Cretaceous limestone-shale and upper Cretaceous shale-limestone corresponding to the SMO province, and to a lesser extent, in Tertiary to Neogene substrates as polymictic-sandstone, rhyolite-rhyolitic tuff and basalt conglomerates, which correspond to the CD provinces and a part of the TVB, and in Paleogene sandstone-shale and upper Cretaceous limestone shale, corresponding to the VZ province (Yta et al. 1999). This could help to understand the structure of the communities according to the trophicity of the geological substrate, since it is known that this is an important factor in the abundance of carabid species (Šustek 2013).
The complementary approach to estimating diversity using two different metrics can support a more complete understanding of diversity in a given area. For example, the CD province was found to be highly diverse using both diversity indices. However, it must be taken into account that this does not mean that there is an ideal or preferred metric for the estimation of diversity, since the different estimation measures are not based on the same biological attributes. The use of the average taxonomic distance index is thus a way of incorporating information to obtain an estimate of diversity that can allow for other scenarios different from traditional metrics such as species richness and abundance, and spatial and ecological patterns, considered by some authors as zoogeographic patterns (Juárez-Barrera et al. 2018).
It is important to highlight the implementation of diversity measurement strategies under a complementary approach in regions with heterogeneous conditions, such as biogeographic provinces, since the taxa present in them are determined by ecological conditions and biogeographic history and a particular evolutionary history, which is used by the phylogenetic diversity as a characteristic between the lineages, showing information that is not reflected only with the number of species (Barragán et al. 2014; Hernández-Ruedas et al. 2019). With this, a relationship is established in the evolutionary history and current ecological conditions, which can be estimated using different dimensions of biodiversity such as taxonomy and phylogenetics (Moreno 2019). This serves as a reference for future studies of ecological diversity using different comparable metrics and a biogeographic scale such as the biogeographic provinces in the state of Hidalgo. Having this referential framework shows how diversity is distributed in space, which is useful when carrying out biological conservation measures (Tucker and Cadotte 2013).
It should be taken into account that the results of both estimates of diversity are not definitive for each part of the biogeographic provinces that converge in the state of Hidalgo, because the data obtained are not the product of systematic sampling, but of a variety of studies carried out during the last five decades, and in many cases, through casual collections efforts with very different sampling methods and objectives, contrary to the methods used in other carabid inventories in Mexico, in larger areas and with different ecological conditions (Zaragoza-Caballero et al. 2019; Shpeley and Van Devender 2022). Although, in Mexico it is assumed that there is a rich fauna of carabids and with a marked level of endemism (Ball and Shpeley 2000), studies with taxonomic, ecological and conservation approaches are still necessary to help to have a better knowledge of the biology of carabids, as has happened in Europe for more than half a century of studies dedicated to carabids (Kotze et al. 2011).
In this work, each of the two metrics reflects different information because they represent different aspects of biological diversity and its distribution in space. This can provide a way to explore why diversity patterns vary between biogeographic regions (Tucker and Cadotte 2013).
References
Allen RT (1968) A synopsis of the tribe Morionini in the western hemisphere with descriptions of two new species (Coleoptera: Carabidae). Caribb J Sci 8(3–4):141–163
Ball GE, Shpeley D (2000) Carabidae (Coleoptera). In: Llorente–Bousquets J, González–Soriano E, Papavero N (eds) Biodiversidad, taxonomía y biogeográfica de artrópodos de México: Hacia una síntesis de su conocimiento. Facultad de Ciencias, Universidad Nacional Autónoma de México, Ciudad de México, pp 363–399
Barragán F, Moreno CE, Escobar F, Bueno-Villegas J, Halffter G (2014) The impact of grazing on dung beetle diversity depends on both biogeographical and ecological context. J Biogeogr 41:1991–2002
Bouchard P, Smith ABT, Douglas H, Gimmel ML, Brunke AJ, Kanda K (2017) Biodiversity of Coleoptera. In: Foottit RG, Adler PH Insect Biodiversity: Science and Society Vol I., Willey-Blackwell Hoboken, New Jersey, pp 337–417
Bousquet Y (2012) Catalogue of Geadephaga (Coleoptera, Adephaga) of America, north of Mexico. Zookeys 245:1–1722. https://doi.org/10.3897/zookeys.245.3416
Campbell WB, Novelo-Gutiérrez R (2007) Reduction in odonate phylogenetic diversity associated with dam impoundment is revealed using taxonomic distinctness. Fundam Appl Limnol 168:83–92. https://doi.org/10.1127/1863-9135/2007/0168-0083
Cerón-Gómez R, Márquez J, Ramírez-Ponce A (2022) Fauna de Carabidae (Insecta: Coleoptera) del estado de Hidalgo, México: Lista de especies, primeros registros y datos de distribución geográfica. Acta Zool Mex 38:1–38. https://doi.org/10.21829/azm.2022.3812495
Chao A, Jost L (2012) Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533–2547. https://doi.org/10.1890/11-1952.1
Chao A, Chiu C-H, Jost L (2010) Phylogenetic diversity measures based on Hill numbers. Phil Trans R Soc 365:3599–3609. https://doi.org/10.1098/rstb.2010.0272
Chao A, Chiu C-H, Jost L (2014) Unifying species diversity, phylogenetic diversity, functional diversity, and related similarity and differentiation measures through hill numbers. Annu Rev Ecol Evol Syst 45:297–324. https://doi.org/10.1146/annurev-ecolsys-120213-091540
Clarke KR, Warwick RM (1998) A taxonomic distinctness index and its statistical properties. J Appl Ecol 35:523–531. https://doi.org/10.1046/j.1365-2664.1998.3540523.x
Clarke KR, Warwick RM (1999) The taxonomic distinctness measure of biodiversity: weighting of step lengths between hierarchical levels. Mar Ecol Prog Ser 184:21–29. https://doi.org/10.3354/meps184021
Colwell RK (2013) EstimateS: statistical estimation of species richness and shared species from samples. Version 9. User´s Guide and Application
CONANP (Comisión Nacional de Áreas Naturales Protegidas) (2018) https://www.gob.mx/conanp
Cruz-Elizalde R, Ochoa-Ochoa LM, Flores-Villela OA, Velasco JA (2022) Taxonomic distinctiveness and phylogenetic variability of amphibians and reptiles in the cloud forest of Mexico. Commun Ecol 23:87–102. https://doi.org/10.1007/s42974-022-00075-w
Dinerstein E, Olson DM, Graham DJ, Webster AL, Primm SA, Bookbinder MP, Ledec G (1995) Una evaluación del estado de conservación de las ecorregiones terrestres de América Latina Y El Caribe. World Bank, Washington D.C.
Erwin TL (1985) The taxon pulse: a general pattern of lineage radiation and extinction among carabid beetles. In: Ball GE (ed) Taxonomy, phylogeny and zoogeography of Beetles and ants. A volume dedicated to the memory of Philip Jackson Darlingon, Jr. 1904–1983. Dr W. Junk Dordrecht, The Hague, pp 437–472
Erwin TL (2017) Agra Fabricius (Coleoptera: Carabidae: Harpalinae: Lebiini: Agrina), arboreal beetles of neotropical forest: the rare, non-neotropical Texas species at the generic norther limit, with notes on their way of life. Coleopt Bull 7(4):639–651. https://doi.org/10.1649/0010-065X-71.4.639
Escalante T (2009) Un ensayo sobre la regionalización biogeográfica. Rev Mex Biodivers 80:551–560. https://doi.org/10.22201/ib.20078706e.2009.002.627
Franco-Sánchez LM, Contreras-Cerón LF (2021) El proceso de la urbanización en la zona metropolitana de Pachuca, en un contexto globalizador. In: Wong-González P, Isaac-Egurrola JE, Morales-García de Alba ER, Treviño-Aldape A (eds) La dimensión global de las regiones y sus reconfiguraciones económicas y urbanas. (Vol. II). Universidad Nacional Autónoma de México, Instituto de Investigaciones Económicas y Asociación Mexicana de Ciencias para el Desarrollo Regional, pp 1–20
Gámez N, Escalante T, Rodríguez G, Linaje M, Morrone JJ (2012) Caracterización biogeográfica de la Faja Volcánica Transmexicana y análisis de los patrones de distribución de su mastofauna. Rev Mex Biodivers 83:258–272. https://doi.org/10.22201/ib.20078706e.2012.1.786.
García de Jesús S, Moreno CE, Morón MA, Castellanos I, Pavón NP (2016) Integrando la estructura taxonómica en el análisis de la diversidad alfa y beta de los escarabajos Melolonthidae en la Faja Volcánica Transmexicana. Rev Mex Biodivers 87:1033–1044. https://doi.org/10.1016/j.rmb.2016.06.003
Gaston KJ (1996) Species richness: measure and measurement. In: Gaston KJ (ed) Biodiversity: a biology of numbers and difference. Blackwell Science, pp 77–113
Gutiérrez-Velázquez A, Rojas-Soto O, Reyes-Castillo P, Halffter G (2013) The classic theory of Mexican transition zone revisited: the distributional congruence patterns of Passalidae (Coleoptera). Invertebr Syst 27:282–293. https://doi.org/10.1071/IS12056
Halffter G (1964) Las regiones Neártica Y Neotropical desde El Punto De vista de su entomofauna. Anais do II Congreso Latinoamericano De Zoologia Sao Paulo 1:51–61
Halffter G (2017) La zona de transición mexicana y la megadiversidad de México: del marco histórico a la riqueza actual. Dugesiana 24(2):77–89. https://doi.org/10.32870/dugesiana.v24i2.6572
Halffter G, Morrone JJ (2017) An analytical review of Halffter’s Mexican transition zone, and its relevance for evolutionary biogeography, ecology and biogeographical regionalization. Zootaxa 4226(1):1–46. https://doi.org/10.11646/zootaxa.4226.1.1
Hernández-Avelino I (2008) Caracterización geológica y petrológica de la estructura circular mezquital (Estado de Hidalgo) y su posible riesgo geológico. Master´s degree Thesis. Instituto Politécnico Nacional. Mexico City
Hernández-Ruedas MA, Gómez-Ortiz Y, Herrera-Alsina L, Pérez-Hernández CX (2019) La diversidad filogenética y su utilidad para la conservación de la biodiversidad. In: Moreno CE (ed) La biodiversidad en un mundo cambiante: fundamentos teóricos y metodológicos para su estudio. Universidad Autónoma del Estado de Hidalgo/Libermex, Ciudad de México, pp 307–324
INEGI (Instituto Nacional de Estadística y Geografía) (2017) Anuario estadístico y geográfico de Hidalgo 2017. Instituto Nacional de Estadística y Geografía, México: INEGI, c2017
Juárez-Barrera F, Bueno-Hernández J, Morrone JJ, Barahona-Echeverria A (2018) Recognizing spatial patterns of biodiversity during the nineteenth century: the roots of contemporary biogeography. J Biogeogr 45(5):995–1002. https://doi.org/10.1111/jbi.13218
Kotze DJ, Brandmayr P, Casale A, Dauffy-Richard E, Dekoninck W, Koivula MJ, Lövei GL, Mossakowski D, Noordijk J, Paarmann W, Pizzolotto R, Saska P, Schwerk A, Serrano J, Szyszko J, Taboada A, Turin H, Venn S, Vermeulen R, Zetto T (2011) Forty years of carabid beetle research in Europe – from taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. Zookeys 100:55–148. https://doi.org/10.3897/zookeys.100.1523
Liebherr JK (1994a) Biogeographic patterns of montane Mexican and central American Carabidae (Coleoptera). Can Entomol 126:841–860. https://doi.org/10.4039/Ent126841-3
Liebherr JK (1994b) Identification of new world Agonum, review of the Mexican fauna, and description of Incagonum, new genus, from South America (Coleoptera: Carabidae: Platynini). J N Y Entomol Soc 102(1):1–55
Liebherr JK, Kipling WW (1996) New North America Platynus Bonelli (Coleoptera: Carabidae), a key to species north of Mexico, and notes on species from the southwestern United States. Coleopt Bull 50(4):301–320
Liebherr JK, Schmidt J (2004) Phylogeny and biogeography of the laurasian genus Agonum Bonelli (Coleoptera, Carabidae, Platynini). Dtsch Entomol Z 2:151–206. https://doi.org/10.1002/mmnd.20040510202
Magurran A (2004) Measuring biological diversity. Wiley-Blackwell, Hoboken, New Jersey
Marshall CJ, Liebherr JK (2000) Cladistic biogeography of the Mexican transition zone. J Biogeogr 27:203–213. https://doi.org/10.1046/j.1365-2699.2000.00388.x
Miller ET, Farine DR, Trisos CH (2017) Phylogenetic community structure metrics and null models: a review with new methods and software. Ecography 40:461–477. https://doi.org/10.1111/ecog.02070
Montiel-Canales G, Goyenechea I (2021) Provincias biogeográficas. In: La biodiversidad en Hidalgo. Estudio de estado. CONABIO, México, pp 67–74
Moreno CE (2019) La biodiversidad en un mundo cambiante: fundamentos teóricos y metodológicos para su estudio. Universidad Autónoma del Estado de Hidalgo/Libermex, Ciudad de México
Moreno CE, Verdú JR, Arita HT (2007) Elementos ecológicos e históricos como determinantes de la diversidad de especies en comunidades. In: Zunino M, Melic A (eds) Escarabajos, diversidad y conservación biológica. Ensayos en homenaje a Gonzalo Halffter. Monografías 3ercer Milenio, Aragonesa (S.E.A.) pp 179–192
Moreno CE, Castillo-Campos G, Verdú JR (2009) Taxonomic diversity as complementary information to assess plant species diversity in secondary vegetation and primary tropical deciduous forest. J Veg Sci 20:935–943. https://doi.org/10.1111/j.1654-1103.2009.01094.x
Morrone JJ (2015) Halffter’s Mexican transition zone (1962–2014), cenocrons and evolutionary biogeography. J Zool Syst Evol Res 53(3):249–257. https://doi.org/10.1111/jzs.12098
Morrone JJ (2018) The spectre of biogeographical regionalization. J Biogeogr 45:282–288. https://doi.org/10.1111/jbi.13135
Morrone JJ (2019) Regionalización biogeográfica y evolución biótica de México: encrucijada de la biodiversidad del nuevo mundo. Rev Mex Biodivers 90:1–68. https://doi.org/10.22201/ib.20078706e.2019.90.2980
Morrone JJ, Márquez J (2001) Halffter’s Mexican transition zone, beetle generalized tracks, and geographical homology. J Biogeogr 28:635–650. https://doi.org/10.1046/j.1365-2699.2001.00571.x
Morrone JJ, Espinosa-Organista D, Aguilar-Zúñiga C, Llorente-Bousquets J (1999) Preliminary classification of the Mexican biogeographic provinces: a parsimony analysis of endemicity based on plant, insect, and bird taxa. Southwest Nat 44:508–515. https://doi.org/10.2307/3672351
Morrone JJ, Escalante T, Rodríguez-Tapia G (2017) Mexican biogeographic provinces: map and shapefiles. Zootaxa 4277(2):277–279. https://doi.org/10.11646/zootaxa.4277.2.8
Peña-Joya KE, Téllez-López J, Rodríguez-Zaragoza FA, Rodríguez-Troncoso AP, Quijas S, Cupul-Magaña FG (2018) Diversidad taxonómica de Lagartijas (Squamata: Lacertilia) asociada a cuatro tipos de vegetación de la Sierra del Cule, Jalisco, México. Acta Zool Mex 34:1–12. https://doi.org/10.21829/azm.2018.3412129
Pérez-Hernández CX (2019) Distintividad taxonómica: Evaluación de la diversidad en la estructura taxonómica en los ensambles. In: Moreno CE (ed) La biodiversidad en un mundo cambiante: fundamentos teóricos y metodológicos para su estudio. Universidad Autónoma del Estado de Hidalgo/Libermex, Ciudad de México, pp 285–306
Pielou EC (1975) Ecological diversity. Wiley, New York
Pineda-López R, Verdú-Faraco JR (2013) Cuaderno de prácticas. medición de la biodiversidad: diversidades alfa, beta y gamma. Universidad Autónoma de Querétaro, Universidad de Alicante, Editorial Universitaria, Colección Académica
Ramírez-Hernández RJ, Moreno CE, Pérez-Hernández CX, Martínez-Falcón AP, Castellanos I (2022) Diversidad de Escarabajos tigre (Carabidae: Cicindelinae) en México. Rev Mex Biodivers 93:1–15. https://doi.org/10.22201/ib.20078706e.2022.93.4162
Reichardt H (1967) A monographic revision of the American Galeritini (Coleoptera: Carabidae). Arq Zool 15:1–176. https://doi.org/10.11606/issn.2176-7793.v15i1-2p1-176
Sánchez-Colón S, Flores-Martínez A, Cruz-Leyva IA, Velázquez A (2009) Estado y transformación de los ecosistemas terrestres por causas humanas. Capital natural de México. Estado de conservación y tendencias de cambio, vol II. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México City, pp 75–129
Sánchez-Rojas G, Bravo-Cadena J (2017) Medio físico del estado de Hidalgo. In: Ramírez-Bautista A, Sánchez-González A, Sánchez-Rojas G, Cuevas-Cardona C (eds) Biodiversidad del estado de Hidalgo. Tomo I. Universidad Autónoma del Estado de Hidalgo/Consejo Nacional De Ciencia Y Tecnología. Pachuca de Soto, Hidalgo, México, pp 29–42
Shpeley D, Ball GE (2000) A taxonomic review of the subtribe Pericalina (Coleoptera: Carabidae: Lebiini) in the western hemisphere, with descriptions of new species and notes about classification and zoogeography. Insecta Mundi 14:1–185
Shpeley D, Van Devender TR (2022) An annotated checklist of ground beetles (Insecta: Coleoptera: Carabidae) occurring in the state of Sonora, Mexico with descriptions of new species and notes about some collection localities. Dugesiana 29(1):41–137. https://doi.org/10.32870/dugesiana.v29i1.7158
Šustek Z (2013) Carabid communities (Coleoptera: Carabidae) in spruse forest in Central Europe. Oltenia J Nat Sci 29(2):140–154
Tolimieri N, Anderson MJ (2010) Taxonomic distinctness of demersal fishes of the California current: moving beyond simple measures of diversity for marine ecosystem-based management. PLoS ONE 5(5):e10653. https://doi.org/10.1371/journal.pone.0010653
Tucker CM, Cadotte MW (2013) Unifying measures of biodiversity: understanding when richness and phylogenetic diversity should be congruent. Divers Distrib 19:845–854. https://doi.org/10.1111/ddi.12087
Tucker CM, Cadotte MW, Carvalho SB, Davies TJ, Ferrier S, Fritz SA, Grenyer R, Helmus MR, Jin LS, Mooers AO, Pavoine S, Purschke O, Redding DW, Rosauer DF, Winter M, Mazel F (2017) A guide to phylogenetic metrics for conservation, community ecology and macroecology. Biol Rev 92:698–715. https://doi.org/10.1111/brv.12252
Wagner DL, Grames EM, Forister ML, Berenbaum MR, Stopak D (2021) Insect decline in the anthropocene: Death by a thousand cuts. Proc Natl Acad Sci USA 118(2):1–10. https://doi.org/10.1073/pnas.202398911
Warwick RM, Clarke KR (1995) New biodiversity measures reveal a decrease in taxonomic distinctness with increasing stress. Mar Ecol Prog 129:301–305. https://doi.org/10.3354/meps129301
Whitehead DR, Ball GE (1997) The Middle American genus Onypterygia Dejean (Insecta: Coleoptera: Carabidae: Platynini): a taxonomic revision of the species, with notes about their way of life and geographical distribution. Ann Carnegie Mus 66(3):289–409. https://doi.org/10.5962/p.215140
Yta M, Galván-Chávez J, Esparza-Contrera R (1999) Carta metalogénica geológico-estructural [Map] 1:500,000. Hidalgo. Servicio Geológico Mexicano
Zaragoza-Caballero S, Rodríguez-Mirón GM, Vega-Badillo V, Gonzáles-Ramírez M, Zurita-García M, Domínguez-León DE, López-Pérez S, Gutiérrez-Carranza IG, Cifuentes-Ruiz P, Pérez-Hernández CX, Ramírez del Valle E, Gutiérrez N (2019) A checklist of the Coleoptera (Insecta) from Morelos, Mexico. Zootaxa 4580(1):1–122. https://doi.org/10.11646/zootaxa.4580.1.1
Acknowledgements
The first author thanks CONACyT for the scholarship (771314) granted to carry out his doctorate in Sciences in Biodiversity and Conservation at the Autonomous University of the State of Hidalgo. Thanks are also extended to this institution for facilitating access to this grant. Thank you to Claudia Moreno for her help and advice on diversity analyses. Thank you to Margaret Schroeder for her help with the translation of the manuscript. We thank two anonymous reviewers for their valuable help in revising and improving this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cerón-Gómez, R., Márquez, J., Ramírez-Ponce, A. et al. Diversity of carabids (Coleoptera: Carabidae) from the state of Hidalgo, Mexico, determined using two complementary approaches. Biologia 79, 1367–1377 (2024). https://doi.org/10.1007/s11756-024-01625-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-024-01625-5