Abstract
Abiotic factors play important roles on the habitat preferences and dispersal decisions of species. The objective of this study was to estimate the groups of abiotic variables best explaining the variation of ostracods species taken from 31 water bodies (27 streams and 4 lakes) sampled twice (October 2020 and April 2021) in the Eastern Mediterranean region of Turkey. In this study, a total of 34 ostracod taxa (24 recent and 10 sub-fossil) were reported, and Ilyocypris bradyi and Prionocypris zenkeri were the most common species occurred more than 10 times. Based upon to the variation partitioning analysis, dissolved oxygen+elevation+magnesium (DO+Elev+Mg2+) was the best model with 12.66% explanation power in the variations of ostracod species in the present study. The distance-based redundancy analysis elucidated 14.1% of the total variation in the species distribution matrix that was significantly affected by Mg2+ and Elev. The effectiveness of variables on the occurrence of species was tested by Generalized Linear Models resulted in positive roles of Elev for Psychrodromus olivaceus, Neglecandona neglecta and Pseudocandona albicans, Ca2+ for Cyprideis torosa and P. albicans, and DO for P. olivaceus, but negative roles of Tw for N. neglecta and P. albicans, Ca2+ for Herpetocypris helenae, and DO for N. neglecta. A positively significant association was found between high Mg2+ values and the abundance of P. zenkeri. Results suggest that effectiveness rates of environmental factors on the occurrence of species can change according to ecoregional differences when the variables are evaluated in the analyses together. Therefore, especially ecoregion-based ecological information of species should be determined for better inferences about the ecological preferences of species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A high level of biodiversity, approximately 100,000 out of 1.8 million species, is found in freshwaters constituting about 0.8% of the Earth’s surface area (Dudgeon et al. 2006). Freshwaters are one of the best ecosystems to study the metacommunity dynamics because they are the systems bearing high environmental heterogeneities in the sense of connectivity and spatial extension (Heino 2011). Through time and space, the structure of ecological communities is shaped by the association of biotic (e.g., competition, predation) and abiotic (e.g., local environmental conditions) factors, historical and dispersal process (Leibold et al. 2004). Distribution of aquatic invertebrates is affected by environmental factors, and the determination of the responses of organisms to the environmental variables and climate is an important issue to estimate the ecological results of regional and global changes (Heino et al. 2009).
Of the aquatic invertebrates, ostracods are the small (0.3–5 mm long) bivalved crustaceans living in all aquatic bodies from fresh to marine waters, underground waters and even in semiterrestrial habitats (Moore 1961; Meisch 2000). A pair of chitinous valves consisting of low magnesium calcite documenting the host water’s chemical and isotopic data or carapaces of ostracods allow the fossilization of them, and they have been recorded from Ordovician to recent (Holmes and Chivas 2002; Oakley et al. 2012; Siveter et al. 2014). They are commonly used to reconstruct past environmental conditions (Griffiths and Holmes 2000). Their distribution and abundance are controlled by ecoregional biotic and abiotic factors, which is also the subject of this study. For example, water ion composition (Baltanás et al. 1990), nutrient levels (Danielopol et al. 1993), habitat type and nature of substrate type (Benzie 1989), elevation, water temperature, salinity, calcium, alkalinity, and nutrients (Van der Meeren et al. 2010), pH, lake area and magnesium (Viehberg 2006) and dissolved oxygen and moisture (Uçak et al. 2014) are some of the abiotic factors affecting the diversity of ostracod assemblages. The sensitivity of ostracods to a wide range of chemical compounds is much more than other invertebrates (e.g., Odonata) (Shuhaimi-Othman et al. 2011; Ruiz et al. 2013; César dos Santos Lima et al. 2019). They also show species-specific respond to organic pollutions and so they can be used as the indicator of habitat disturbance and water quality (Mezquita et al. 1999a).
Above mentioned abiotic factors affecting the distribution of ostracods allow one to consider that whether the ecoregional differences (Loucks 1962; Omernik 1987) change the effects of variables on the distribution of ostracods. Most recently, Çelekli et al. (2021) underlined the importance of ecoregion (differences in soil structure, land uses, climate, altitude, geology, and hydrology data) on the trophic weight and ecological preferences of benthic diatoms. This is also the case for ostracods. A large-scale survey research emphasized the importance of precipitation, temperature, and elevation on the distribution of ostracods in South America (de Oliveira da Conceição et al. 2019). A recent study conducted in another region pinpointed that the ostracods found in 77% of the 243 Patagonian freshwater bodies (in Argentina) with climatic heterogeneity are mainly controlled by the dissolved oxygen, water temperature, precipitation, and air temperature (Ramos et al. 2022). The authors also suggested that further studies need to be done in the different regions with respect to the spatial scale and environmental heterogeneity. In addition, Cusminsky et al. (2020) reported the associations of non-marine ostracods (28 taxa) collecting from 69 environments in Argentinian ecoregions with electrical conductivity, altitude, pH and water temperature. Therefore, the determination of the ecoregion-based abiotic factors affecting the species distribution will give more accurate result about the ecological preferences of species (e.g., ostracods).
Abiotic factors play important roles on the habitat preferences and dispersal decisions of species (Katz et al. 2017). Therefore, choosing of the correct abiotic variable/s to evaluate the distribution of species allow us to make strong and accurate inferences about the ecological preferences of species. In the present study, estimation of the efficient abiotic variable group/groups explaining the variation of ostracod species was aimed. To reach this aim, 31 water bodies (27 streams and 4 lakes) were sampled twice in the Eastern Mediterranean region of Turkey.
Material methods
Study area
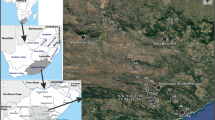
The study was carried out within the borders of Antalya, Mersin, Karaman, and Konya provinces in the Eastern Mediterranean region of Turkey (does not cover the whole borders of these provinces). It is noteworthy to mention that this region is one of the hottest regions in Turkey (Fig. 1). In 2016, the monthly general temperature average of the region was reported as 17.78 °C, while the monthly lowest (−0.64 °C) and highest (30.57 °C) average temperatures were recorded in Konya (in December) and Mersin (in July), respectively. The annual average precipitation rate of the region is about 745 mm, and lower precipitation values are seen from west to east and inland. From the coastline to inland, terrestrialization is evident and the difference in annual average precipitation between the coastal and inland parts is about 500 mm (RTMFWM 2018). According to MGM (2022), the average data for the last 70–90 years show that the average temperature (Atemp) and precipitation (Apre) values of the provinces located on the coastline (Atemp = 19.2 °C and Apre = 615.5 mm for Mersin from 1940 to 2020, and Atemp = 18.8 °C and Apre =1061.7 mm for Antalya from 1930 to 2020) were higher than the provinces located in the interior (Atemp = 11.7 °C and Apre = 329.2 mm for Konya from 1929 to 2020, and Atemp = 12 °C and Apre = 339.8 mm for Karaman from 1951 to 2020). The region generally has a karstic structure, and its geology consists of different aged limestone formations. Therefore, the soils are generally calcareous clay and loamy clay types with neutral or alkaline reactions, and the iron in the soil has caused the soil to turn red because of chemical reactions with the effect of climatic heat (RTMFWM 2018).
Sampling and laboratory analyses
Samples were taken from 31 water bodies (4 lakes and 27 streams) twice, in October of 2020 and April of 2021 (Fig. 1 and Table 1). Before ostracod samplings, 100 ml of water samples were picked up from each sampling site and stored in a cooler container for the analyses of total phosphate (Tot-P), total nitrogen (Tot-N), magnesium (Mg2+), and calcium (Ca2+) according to APHA (1998). Total hardness (Tot-Hard) was calculated using the values of Ca2+ and Mg2+ for each site (Tot-Hard (mg L−1 CaCO3) = (Ca2+ × 2.5) + (Mg2+ × 4.12)) (Boyd et al. 2016). Of the abiotic variables dissolved oxygen concentration (DO, mg L−1), water temperature (Tw, °C), electrical conductivity (EC, μS cm−1), pH, salinity (‰), and total dissolved solids (TDS, mg L−1) were measured by the aid of a YSI Professional Plus multimeter. Water samples taken from each site were read 10 times with a turbidimeter (WPA Turbidity Meter TU1100) to determine the average turbidity value of water. Geographical data (elevation and coordinates) was gained by a GARMIN Etrex Vista H global positioning system (Garmin Ltd., Kansas, USA).
Sediments including ostracod specimens were collected from the littoral regions of lakes (up to 1 m depth) and slow-flowing parts of the streams (up to 0.5 m in depth) with a hand net (200 mm mesh size). Subsequently, collected samples were fixed with 70% ethanol in 250 ml plastic bottles in situ. In the laboratory, all samples were washed under tap water through standard-sized sieves with 0.5, 1.0, 1.5 and 2.0 mm mesh sizes. Afterwards, ostracods were sorted from the sediment using fine needles under a stereo microscope (Olympus ACH 1X) and put into small glass vials with 70% ethanol for further research. Soft body parts of adult specimens with complete carapaces were dissected in lactophenol solution for taxonomic description following Meisch (2000) and Karanovic (2012) under an Olympus BX-51 light microscope. Each sample was deposited in the Limnology Laboratory of Bolu Abant İzzet Baysal University, and can be available upon request.
Statistical analyses
Ecological Community Analysis II Software was used to test the multicollinearity among the environmental variables (Seaby and Henderson 2007). Accordingly, variables having an inflation factor larger than 10 indicating a possibility of multicollinearity were removed from the analyses. Of the abiotic variables, total phosphate and total nitrogen were not used in this analysis because their values were below detectable limits for many sites (Online Resource 1). Consequently, six abiotic variables (water temperature (Tw), pH, dissolved oxygen concentration (DO), calcium (Ca2+), magnesium (Mg2+), and elevation (Elev)) did not display multicollinearity. Then after, the triple combination of these six environmental variables (C(6, 3) = 6!/(6–3)!*3!) = 20) were used in most of the analyses given below to estimate the best environmental variables group/groups explaining the variation in the ostracod species. Variation partitioning (VP) (Borcard et al. 1992) was applied to determine the relative contribution of each predictor variable to elucidate the variation of ostracod species in the triple combination (or model) of abiotic variables using the adjusted R2 (Peres-Neto et al. 2006). The importance of each model was tested by aid of 999 random permutations. The sum of all the explained variations and residual variance may exceed “1” or “100%” when looking at the VP results because of the presence of the negative explained variances and certain relationships in the data. Distribution of ostracod species among the abiotic variables used in VP analysis was displayed in ternary plots for better visualization. Generalized Linear Models (GLM) were performed for the presence-absence of ostracod species data using binomial family and logit link functions (Zuur et al. 2009) to see the effect of each predictor in the six abiotic variables and in the triple combination of them. Level of variation explained by the predictor variables in GLM was calculated with aid of the formula ((Null deviance - Residual deviance)/Null deviance)*100, and this is termed as the Pseudo-R2 throughout the manuscript. Null and Residual deviances represent how well response variable forecasting by a model only with intercept term and by a specific model with predictors, respectively. The significance of each model was tested by the Chi-square test (Zuur et al. 2009). The relationships between the abiotic variables (Tw, pH, DO, Ca+2, Mg+2, and Elev) and the ostracod species distribution matrix were examined by a distance-based linear model (DISTLM) / a distance-based redundancy analysis ordination (dbRDA) using Bray Curtis similarities and Akaike Information decision Criterion (AICc) in PRIMER v7 with PERMANOVA+ (Clarke and Gorley 2015). Species data were Hellinger transformed because of including many zeros (Peres-Neto et al. 2006; Legendre and Gallagher 2001) for Variation partitioning and DISTLM / dbRDA analyses, while log transformation was applied to environmental variables to get the near-normal distribution except pH in PAST 3.26 software (Hammer et al. 2001) for the last analysis. A weighted averaging regression was used to estimate the optimum (Opt) and tolerance (Tol) levels of species for explanatory variables using C2 Software (Juggins 2003). A non-parametric Spearman Rank Correlation analysis was used to test the meaningful correlations between species and environmental variables, and among environmental variables (IBM-SPSS Statistics Version 21). The packages Vegan 1.5–7 (Oksanen et al. 2020) and glmm (Knudson et al. 2018) in R Version 3.6.3 (R Core Team 2020) were used to perform Variation partitioning and Generalized Linear Model analyses, respectively. For ternary plots, ggtern package (Hamilton and Ferry 2018) in R Version 4.1.2 (R Core Team 2021) were utilized. Above mentioned packages in different versions of R-statistics were runned with aid of RStudio software v.1.4.1103 (R studio Team 2021). In all statistical analyses, adult individuals occurring at least two or more times with complete soft body parts and carapaces were used.
Results
The descriptive statistics of abiotic variables measured in the present study are given in Table 2. A total of 34 ostracod taxa (24 recent and 10 sub-fossil) were found in the present study (Tables 1 and 3). High ostracod taxa diversity was found in the coastline sampling site S7 (9 taxa (6 recent), followed by the inland parts sampling sites as S15 (7 recent), S17 (7 taxa (5 recent)) and S20 (7 taxa (6 recent)) (Table 1 and Online Resource 2). S7 was the hottest (mean water temperature = 21.4 °C) site among the above-mentioned sites. The mean pH values of these sites were ranged from 7.9 to 8.2 which indicates the slightly alkaline conditions. S7 had the lowest mean of calcium (Ca2+ = 62.1 mg L−1) and the highest mean of magnesium (Mg2+ = 46.8 mg L−1), when S17 was of the lowest mean Mg2+ (6.8 mg L−1) and highest mean Ca2+ (77.1 mg L−1) values, compared with S15 and S20. They were ranked from low to high as S17, S20, S15, and S7 in the sense of elevation (Online Resource 1).
Of the 24 recent species, ten were collected only in October of 2020 while four species (Eucypris pigra (Fischer, 1851), Herpetocypris intermedia Kaufmann 1900, H. reptans (Baird, 1835) and Potamocypris fulva (Brady, 1868)) in April of 2021, and the rest species were common among both sampling periods (Table 3). Ilyocypris bradyi Sars, 1890 was the most common species with an occurrence frequency (Ocfr) of 17 times, followed by Prionocypris zenkeri (Chyzer & Toth, 1858) with a frequency of 12 times (Online Resource 2). In the sense of abundance, I. bradyi was the last one among first five species with 210 individuals (ind.) while the first four are Cyprideis torosa (Jones, 1850) (560 ind. from three Ocfr), P. zenkeri (415 ind. from 12 Ocfr), Heterocypris salina (Brady, 1868) (318 ind. from five Ocfr) and Stenocypris bolieki Ferguson, 1962 (157 ind. from two Ocfr) (see Table 3 and Online Resource 2).
The explained percentage fractions of variation in the ostracod species by abiotic variables in different models based on the triple combination of water temperature (Tw, °C), pH, dissolved oxygen concentration (DO, mg L−1), calcium (Ca2+, mg L−1), magnesium (Mg2+, mg L−1) and elevation (Elev, m asl.) were given in Fig. 2. While all models were statistically significant (p < 0.05), models presented in Figs. 2r (Tw + Ca2++pH), 2 s (pH + DO+Ca2+), 2 t (Tw + pH + DO) and 2u (Tw + pH + Elev) were not. Among the statistically important models, the highest explanation power was reported for DO+Elev+Mg2+ with 12.66% (Fig. 2a) whereas Ca2++Tw + DO was the lowest with 4.11% (Fig. 2o). Overall models, Mg2+ was the single abiotic variable delineating the most fractions in the Tw + pH + Mg2+ model with 6.77% (Fig. 2h) that is followed by Elev in the DO+Elev+pH model with 4.61% (Fig. 2m). Highest elucidation fractions in the double (3.20%) and triple (0.72%) intersections were observed between Ca2+ and Mg2+ in Ca2++Mg2++Elev model (Fig. 2e) and among Ca2++DO+Mg2+ (Fig. 2g), respectively. Relatively high unexplained fractions from 88.62% to 99.13% were found in all models (Fig. 2). The distributions of the species among the triple combinations of abiotic variables were displayed in the ternary plots in Fig. 2. These distributions showed changes from partially homogeneous (e.g., Fig. 2a, b) to highly clustered structures (e.g., Fig. 2o-t) when the explained variations in ostracod species going from high to low.
Venn diagram indicating the fraction of variation explained by the individual variables and combination of them in each model constructed by the triple combination of water temperature (Tw, °C), pH, dissolved oxygen concentration (DO, mg L−1), calcium (Ca2+, mg L−1), magnesium (Mg2+, mg L−1) and elevation (Elev, m asl.) according to variation partitioning of ostracod communities, and ternary plots showing the distribution of ostracod species among the triple combination of abiotic variables. Residuals show the unexplained fractions in each model and species codes were given in Table 3. Lower case letters from a to u indicate the models and ternary plots constructed by the triple combination of used six environmental variables
The ordination of ostracod species according to the effect of predictor variables on their distribution was displayed in Fig. 3 after the application of distance-based linear model (DISTLM) and distance-based redundancy analysis (dbRDA). The dbRDA elucidated only 14.1% of the total variation in the species distribution matrix. The first two axes of dbRDA explained 67.1% of the fitted variations that is meaning a high correlation between predictor environmental variables and species distribution matrix. According to the correlation’s coefficients between predictor variables and axes, axis 1 was constructed by magnesium (Mg2+, 46.4%) and elevation (Elev, 78.1%) when axis 2 by calcium (Ca2+, 74.4%) and water temperature (Tw, 54.1%). The distribution matrix of ostracod species was significantly affected by Mg2+ (Pseudo-F = 2.92, p = 0.004) and Elev (Pseudo-F = 3.55, p = 0.002) but not by DO (Pseudo-F = 1.49, p = 0.152), Ca2+ (Pseudo-F = 1.47, p = 0.164), Tw (Pseudo-F = 1.21, p = 0.272) and pH (Pseudo-F = 0.26, p = 0.989).
Distance-based redundancy analysis (dbRDA) of the ostracod species distribution matrix and environmental variables (arrows). Abbreviations of environmental variables were provided in Table 2, and the number (2) after the sampling site code (e.g., S24–2) means the sampling in the April of 2021. Distribution of species in each sampling site (circle) was showed using triangles with different colors
The Generalized Linear Model (GLM) revealed the effects of predictor variables on the presence-absence probability of only P. olivaceus (Brady & Norman, 1889), C. torosa, H. helenae G.W. Müller, 1908, N. neglecta (Sars, 1887) and P. albicans (Brady, 1864) (Table 4). Dissolved oxygen (DO) showed a positive efficacy on the occurrence probability of P. olivaceus in all triple combination groups of abiotic variables, and elevation (Elev) also displayed a similar effect for this species in Ca2+ (calcium) + DO+Elev and pH + DO+Elev groups. Calcium indicated positive impact on the occurrence probability of C. torosa in all triple groups, but negative for H. helenae in Ca2++pH + Mg2+ (magnesium) group. The occurrence probability of P. albicans was positively affected by Ca+2 in two groups. The GLM results showed the negative influence of DO and water temperature (Tw) on the occurrence probabilities of N. neglecta and P. albicans, while another regional factor, Elev, indicated a positive effect on the occurrence probabilities of both species (see Table 4).
Spearman Rank correlation analysis pointed out the negative and positive correlations of water temperature with elevation (rs = −0.408, p < 0.01) and pH (rs = 0.465, p < 0.01), respectively. Negatively important correlations were observed between dissolved oxygen and calcium (rs = −0.393, p < 0.01), and between elevation and magnesium (rs = −0.49, p < 0.01). Prionocypris zenkeri, as the only species showing an important association with one of the abiotic variables, exhibited a positively strong correlation with magnesium (rs = 0.689, p < 0.05).
Optimum and tolerance levels of species encountered at least two times in the present study for water temperature, pH, calcium, magnesium, elevation and dissolved oxygen were dedicated in Table 5. Accordingly, H. helenae and P. olivaceus were of lowest and highest optimum levels for pH (7.76) and dissolved oxygen (8.73 mg L−1), respectively. Cyprideis torosa was the only species having highest optimum levels for both of calcium (129.66 mg L−1) and magnesium (180.35 mg L−1) (see Table 5).
Discussion
Seasonal species diversity
Relatively high species diversity was found in the present study. The ostracod taxa per sampling sites (or ratio = 1.1) herein was approximately 2.5 times higher than the ratio (0.43) obtained from the study including 117 sampling sites in the Mersin province in October 2015 (Dalgakıran et al. 2020). The low ratios were also reported in studies handling in the close or neighbor regions, e.g., a ratio of 0.44 getting from the 63 samplings in Osmaniye province in May 2015 (Külköylüoğlu et al. 2021), 0.3 ratio in Hatay province after 70 samplings in summer of 2012 (Akdemir and Külköylüoğlu 2021), ratios of 0.81, 0.68 and 0.66 recorded from the 26 sites in Konya closed, 22 sites in Antalya and 32 sites in West Mediterranean basins sampled in August and October of 2017, respectively (Yavuzatmaca 2019) and a ratio of 0.41 acquired from the 68 sites sampled in July of 2014 in Muğla province (Akdemir et al. 2020). Of them, Yavuzatmaca (2019) announced the highest diversity of ostracods in October for Konya closed and West Mediterranean basins like the present study (22 taxa in April and 30 taxa in October, Table 3), while it was the highest in August for Antalya River basin (13 species in August, eight species in October, and six species common). Most recently, the ratios 0.63 and 0.73 were noted from the 41 sites sampled in three seasons (spring, summer and autumn) of 2018 in the Eastern Black Sea and from the 40 sites sampled in three seasons of 2019 in the Eastern Anatolian regions of Turkey by Yavuzatmaca (2020a) and Yavuzatmaca (2021), respectively. In both studies, the highest species diversity was recorded in autumn (Yavuzatmaca 2020a) and summer (Yavuzatmaca 2021) seasons. Like the above given studies including more than one habitat types from Turkey, lower ratios from the present study were found in the studies outside of Turkey, e.g., a ratio of 0.44 from 106 sampling sites sampled in summer of 2004 and 2005 in Western Mongolia (Van der Meeren et al. 2010) and 0.83 ratio from 49 sites visited from 2006 to 2008 in different months and seasons in subarctic and temperate Europe (Iglikowska and Namiotko 2012). As stated before (Külköylüoğlu et al. 2016; Yavuzatmaca 2020a), species diversity does not increase with the number of sampling sites up to a critical level because seasonality appears play important role more than the number of sampling sites. In addition, although it seemed that the autumn season (from September to November in Turkey) can be rich in species diversity, higher diversity can also be found in different seasons even in regions close to the study area. Thereby, regional (or local) differences should be considered, and then regional biodiversity studies should be conducted by investigating which season or seasons can be better in terms of species richness.
Species diversity and stream orders
Streams (S7, S15, S17 and S20) with high taxa diversity in the present study were at least 2nd order streams according to Strahler (1957) classification system, which supports the finding of Yavuzatmaca (2020a) as low ostracod species diversity in 1st order streams. The reason of it has been linked to the streams’ size increasing from head (or 1st order) to following streams (2nd, 3rd order streams) (Vander Vorste et al. 2017) because an ascending trend is observed in the species diversity with that (Vannote et al. 1980). Examination of this issue has not been widely discussed for ostracods in literature so studies revealing species distributions and characteristics according to streams’ size are needed to provide ecologically important information for estimation of habitat characteristics (lotic in this sense) using ostracods.
Explanation power of environmental variables on the species composition
The changing of rates of variation explained by each of the individual variables combined with other variables effective on the ostracod species composition can be seen in Fig. 2. Even if the percentage of explanation powers of models were low, the best model might be the dissolved oxygen+elevation+magnesium (DO+Elev+Mg2+) with 12.66% (Fig. 2a) followed by pH + DO+Mg2+ with 10.6% (Fig. 2i). The importance of local factors (e.g., DO, Mg2+, Tw) and the secondary effect of Elev (as a regional factor) were commonly discussed in the literature for ostracods (e.g., Viehberg 2006; Uçak et al. 2014; Yavuzatmaca et al. 2018). However, changing of one variable in the models caused a decrease of important percentage in the explained variation, e.g., 2.38, 2.86 and 2.87% reduction were observed when the accompanying variable of Mg2+ and Elev was calcium (Ca2+, Fig. 2e), pH (Fig. 2c) and Tw (Fig. 2b) instead of DO (Fig. 2a), respectively. The situation was the same when checking the whole models in Fig. 2. These results emphasized the importance of understanding the intertwined and complex relationships among abiotic variables to determine whether which variable increases or decreases the effectiveness of the accompanying variable/s. These findings are also enabled to estimate the best environmental variable groups elucidating the species composition variation.
Elevation displays a negative association with the rate of dissolved gases (e.g., DO, CO2) because of the effect of barometric pressure on their solubility (Goldman and Horne 1983). In addition, ionic salinity because of decreasing the available intermolecular space (Goldman and Horne 1983) and temperature (Wetzel 2001) have negative correlations with the solubility or occurrences of gases (e.g., DO) in waters. Similarly, a negatively significant correlation was reported between DO and Ca2+ in the present study. Main rock structure in the studied area, limestone, is a mixture of calcite (CaCO3) and dolomite (MgCO3) but mostly CaCO3 in nature that is dissolved by the disintegration of CO2 in waters to form carbonic acid (Boyd et al. 2016). Then, this weak acid solubilizes limestone and resulted in an increase in the amount of ionized Ca2+ and HCO31− in water (Wetzel 2001). Using of HCO31− for photosynthesis increase the CO32− and OH1− causing pH to rise. The presence of Ca2+ limits the increase of pH by precipitating CO32− as CaCO3. This implies the relationships among pH, DO and the availability of Ca2+ in water. Considering the limestone dominancy in the studied area and the pH range (7.40–8.80 (Table 2)), the availability of Ca2+ in water seems a non-limiting factor for the ostracods to calcify their valves. Although Mg2+ is found in the limestone, its main source is dolomite rock, and Mg2+ is indicated as a counterpart of Ca2+ because of their similar chemistry (Goldman and Horne 1983). However, Mg2+ compounds are more soluble than Ca2+. Therefore, important amount of MgCO3 and magnesium hydroxide start to precipitate when the pH of waters increases to very high levels (>10) (Wetzel 2001).
After the information given above, the explanatory power of Mg2+ mostly increased with water temperature (Fig. 2b–h) in the present study but the level of this power changed according to the variable added as the 3rd in the model. For instance, elevation is a factor indirectly affecting the concentration of Mg2+ in waters, and a significantly negative relationship was also found between them in the present study (p < 0.01). Also, the distribution of species found in waters at high elevations corresponds to low Mg2+ content in the ternary plot when looking at Fig. 2b. The explanatory power of Mg+2 was the highest alone (Fig. 2h) when the 3rd factor was pH but decreased partially when DO used (Fig. 2d). On the other hand, Ca2+ (Fig. 2j) was the 3rd factor, power of Mg2+ was almost halved, while the joint explanatory power rises. This may be due to the importance of Mg2+ and Ca2+ for the calcification of ostracod shells and their similar chemistry (see above). The increasing power of Mg2+ with Tw may be explained by the correlation between Tw and ostracod shells’ Mg2+ content (Palacios-Fest and Dettman 2001) because Tw has the management roles on the minor element composition of ostracod shells (see Dettman et al. 2002). Water temperature also affects the development, body size and life cycle of ostracods (Ruiz et al. 2013; Aguilar-Alberola and Mesquita-Joanes 2014) that may elucidate the reason why 2.87% of variations were less explained when using Tw instead of DO with Elev+Mg2+ (Fig. 2a, b). The effects of water temperature on the ostracods can be more effective on the growth period and its molting stage, in other words, it is more effective on juveniles rather than adults. Considering the used adults in the last stage of molting in all analyses, it can be understood why dissolved oxygen-bearing metabolic importance for aerobic organisms displayed the higher explanation power than Tw in the present study. Furthermore, DO was also effective against pH, when used together with Ca2+ and Mg2+ because the variation explained by the model with DO was 8.92% (Fig. 2g), while the model with pH explained only 7.03% (Fig. 2l). An increase of the activity of Ca2+ intersection Mg2+ along with pH was observed since they elucidated 2.68% (Fig. 2l) and 1.90% (Fig. 2g) of variations when the accompanying variable was pH and DO, respectively. The activity of Mg2+ was not significantly changed when using the DO (Fig. 2g) and pH (Fig. 2l) with Ca2+. When the efficiency of Ca2+ and DO was examined, the activity of DO was higher than that of Ca2+ checking the models made with Elev (Fig. 2f) and Mg+2 (Fig. 2g). Looking the models constructed with Ca2+, Tw and pH (Fig. 2r), and others (see Fig. 2), a ranking like Ca2+, Tw and pH can be seen according to their explanation powers on the variation of ostracod species. In all, the importance or explanation power of environmental variables on the ostacod species composition in the present study can be listed as Mg2+, Elev, DO, Ca2+, Tw and pH.
The answer to question “Why is Mg2+ more effective than other variables?” lie in the geology of Turkey. This is because Turkey is located on a dense tectonic activity consisting of approximately 40% of carbonate and evaporitic rocks suitable for dissolution, and this ratio can also reach to a value of 60% when caves as the characteristics of ground karstification are considered (Nazik and Poyraz 2017). Therefore, it is expected that the dissolved Ca2+ ratio in waters will be higher than Mg2+, and the results of the studies conducted in different regions of Turkey also strengthen this argument. Of them, Yavuzatmaca et al. (2017a) reported a higher mean Ca2+ value (71.26 mg L−1) than Mg2+ (15.25 mg L−1) in the Sinop province in the Black Sea region of Turkey, and similarly Külköylüoğlu et al. (2020) shared mean Ca2+ = 46.9 mg L−1 and Mg2+ = 9.63 mg L−1 values for the ten sampling sites in Artvin province located in the same region of Turkey. The similar results were also published in the Kütahya province (Ca2+ = 69.6 mg L−1, Mg2+ = 30.7 mg L−1) in Aegean (Külköylüoğlu et al. 2018), Muğla province (Ca2+ = 55.17 mg L−1, Mg2+ = 17.54 mg L−1) in the Southwest (Akdemir et al. 2020), Malatya province (Ca2+ = 85.2 mg L−1, Mg2+ = 31.3 mg L−1) in East Anatolia (Batmaz et al. 2020) and in Mersin province (Ca2+ = 58.11 mg L−1, Mg2+ = 11.53 mg L−1) in Mediterranean (Dalgakıran et al. 2020) regions of Turkey. The Ca2+ and Mg2+ ratios in water are of great importance for ostracods because they get the cation and anions (e.g., Ca2+, Mg2+, CO32−) required to calcify their low magnesium calcite carapaces from the waters where they live (Turpen and Angell 1971). Accordingly, Mg2+ may be shown as a limiting factor for the ostracods in the current study when considering the rock formation of the studied area, and the mean Ca2+ (60.36 mg L−1) and Mg2+ (24.19 mg L−1) values in the present study (Table 2).
The results of the dbRDA also supported the importance of Mg2+ and Elev (p < 0.05) among other variables for the distribution and abundance of ostracod species in the present study (Fig. 3). Similarly, the significant influence of Mg2+ (e.g., Viehberg 2006; Yavuzatmaca et al. 2017a) and Elev (e.g., Külköylüoğlu et al. 2019; Yavuzatmaca 2019, 2020b) on the occurrence and distribution of species were reported. Notwithstanding, this does not mean that other variables (DO, Ca2+, Tw and pH) are not important for the distribution of ostracods. This is because their importance was emphasized for many times before in and out of Turkey (e.g., Van der Meeren et al. 2010; Iglikowska and Namiotko 2012; Akdemir et al. 2020; Külköylüoğlu et al. 2020, 2021) and even in a study where the studied area overlapping with some of the studied area here in the present study (Dalgakıran et al. 2020). Most recently, Cusminsky et al. (2020) highlighted the effects of EC, elevation, and pH for the ostracod assemblages in Patagonian (Argentinian) ecoregions and stated that they are followed by Mg2+ and Tw. All these suggest that the effects of especially local factors on the distribution of ostracods may vary from region to region, in the sampling season or times, and even in the sampled habitat differences. Therefore, revealing the ecoregion-based effective factors should be the topic of future studies to efficiently use indicator species to estimate past and present environmental conditions.
Environmental variables and individual species
GLM results showed the positive effect of Ca2+ on the presence of (Table 4) euryhaline widespread species, C. torosa, that is mostly occur in the brackish water of coastal areas (Meisch 2000). This is the case in the present study because the living and subfossil forms of species were encountered in the coastal sampling sites S1, S7, S24 and L1 (Fig. 1, Table 1, Online Resource 2). The positively strong correlations of species with conductivity (Yavuzatmaca 2019), and Ca2+ and Mg2+ (Akdemir et al. 2020) and the effect of Mg2+ level on its occurrence (Viehberg 2006) were announced. Recently, Gusakov et al. (2021) collected species from the polyhaline Chernavka River having high Ca2+ (0.92–1.44 g L−1) and Mg2+ (0.68–0.89 g L−1) levels of the Lake Elton Basin in the European territory of the Russian Federation, and they declared a very high upper limit of salinity tolerances (96–150 g L−1) for C. torosa. The range of Ca2+ (88.41–181.81 mg L−1) and Mg2+ (165.53–190.73 mg L−1) of water where living form of species found (Online Resource 1) and the highest optimum levels for both Ca2+ (129.66 mg L−1) and Mg2+ (180.35 mg L−1) (Table 5) in the present study reinforced these previous statements about the C. torosa. The previously reported close relationship of the species with conductivity (see above) and the strong association between conductivity and Ca2+ (Iglikowska and Namiotko 2012) are considered, the answer to the question “why did the Ca2+ has a positive action on the occurrence of C. torosa in the present study?” has been given.
Mezquita et al. (1999b) pinpointed the occurrence of H. helenae in Mg2+ enriched waters concerning Ca2+ and its preference for high dissolved oxygen and pH level. It was found in waters with high pH mean (8.04) but low Ca2+ (46.04 mg L−1) and Mg2+ (10.05 mg L−1) mean values (Online Resource 1), and it displayed low Ca2+ (47.55 mg L−1) and Mg2+ (9.56 mg L−1) optimum levels when compared with other species (Table 5) in the present study. Also, mean DO value (7.84 mg L−1) of water where species present (Online Resource 1) showed conformity with the DO range (5.4–19.1 mg L−1) (Uçak et al. 2014; Mezquita et al. 1999b) getting from the literature. All these findings support that H. helenae prefers waters with low Ca2+ level because escalating of DO and pH cause the depletion of Ca2+ and so the Mg2+ and Ca2+ levels of water begin approach to each other as emphasized by Mezquita et al. (1999b). Thereby, the negatively significant effect Ca2+ on the occurrence of H. helenae in the present study was a promising finding (Table 4) due to the geologic rock structure of the studied area.
Neglecandona neglecta is a well-known cosmoecious species (Külköylüoğlu 2013). Tolerance of species to hypoxic condition (below 3 mg L−1 DO) (Meisch 2000), its presence in anoxic environment (= 0.32 mg L−1 DO) (Külköylüoğlu 2009) and an important negative correlation of it with DO (Külköylüoğlu et al. 2014) were previously documented. After all, Yavuzatmaca (2020b) underlining statistically important indicator potential of N. neglecta for DO level equals 9.10 mg L−1 showed conformably the close relationship of the species with DO in streams (Yavuzatmaca 2021). Rieradevall and Roca (1995) indicated the contradictory effect of high Tw on the abundance of N. neglecta, while low Tw having positive outcome on its development in Lake Banyoles, Spain. Yavuzatmaca (2020b) found the separation of a group of sampling sites possessing mean Tw value corresponds to 19.7 °C from other by N. neglecta, when the negatively significant relationships of species with Tw were presented in literature (e.g., Yılmaz and Külköylüoğlu 2006; Yavuzatmaca 2021). Both DO and Tw have negative relationships with Elev (see mentioned above) but N. neglecta formerly displayed positive correlation with Elev (Pieri et al. 2009; Yavuzatmaca 2019). In the present study, elevation revealed a positive result on the occurrences of N. neglecta, while Tw and DO negatively affect the occurrence of the species (Table 4). Also, N. neglecta had high (1144 m asl.) and low (14.25 °C) optimum level for Elev and Tw, respectively, after P. albicans (Table 5). Models constructed with Tw, DO, Elev, pH, Ca2+ and Mg2+ explained a range from 26.07% to 34.13% in the variation of the occurrence probability of N. neglecta (Table 4). The causation of these low percentage ratios may be the wide tolerance of the species to those variables (e.g., Tw, pH, DO, Elev). The validity of this view is consistent with the ranges of variables significantly affecting the occurrence probability of N. neglecta provided by Yavuzatmaca et al. (2017b), e.g., Tw (2.13–28.9 °C), DO (0.32–15.4 mg L−1) and Elev (0–3194 m asl.). Like N. neglecta, the cosmopolitan species (Külköylüoğlu et al. 2012a), P. albicans, had been found in the wide ranges of DO (0.75–15.8 mg L−1), Tw (2.9–29.2 °C) and Elev (61–2290 m asl.) (Yavuzatmaca et al. 2017b). Among the importantly effective variables, DO and Tw showed higher negative coefficients than variables (Elev and Ca2+) that had positive coefficients on the probability of P. albicans (Table 4), while its positive association with Tw (Yavuzatmaca 2021) but negative with Tw and Elev (Külköylüoğlu et al. 2012b) were shown. Although the percentages explained (31.93–55.77%) by the models in Table 4 were higher than N. neglecta, the activities of the variables were similar to N. neglecta. The range of Ca+2 (41.18–98.35 mg L−1 (Online Resources 1 and 2)) for waters where P. albicans gathered exhibited conformity with the range (10–160 mg L−1) given in Iglikowska and Namiotko (2012). The optimum (78.89 mg L−1) and tolerance (25.52 mg L−1) levels of Ca2+ for species (Table 5) were higher than the optimum (66.1 mg L−1) but lower than tolerance (35.04 mg L−1) levels in Batmaz et al. (2020). Also, Van der Meeren et al. (2010) encountered species in waters with %Ca2+ mean equals to 55.9 in Western Mongolia. All these suggest that species prefers a high Ca2+ level that is also supported by the positive role of Ca2+ on the occurrence probability of P. albicans in the present study (Table 4).
Psychrodromus olivaceus is another well-known cosmoecious species (Külköylüoğlu 2013) and it was found in a wide range of DO (1.74–20 mg L−1) and Elev (0.5–1700 m asl.) (Yavuzatmaca et al. 2017b). Even though Külköylüoğlu et al. (2013) write up the negative correlation of species with DO, the indicative potential of P. olivaceus for two groups of sampling sites having high DO mean values (8.62 and 9.64 mg L−1) were estimated in Yavuzatmaca (2020b). A strong positive correlation of species with elevation (rs = 0.88) was demonstrated by Yavuzatmaca et al. (2018), and then after, Dalgakıran et al. (2020) find the changing of length and height of P. olivaceus across elevational ranges and reported the high tolerance of species to elevation. Highest optimum for DO (8.73 mg L−1) and tolerance for Elev (511.4 m asl.) (Table 5) and the positive effect of DO and Elev on the occurrence of species (Table 4) reinforce these previous findings. Although the species has wide tolerance to environmental variables, it can be said that DO has a positive effect on the presence of species. The cosmopolitan species, P. zenkeri, showed a very strong correlation with the Mg2+ but it was encountered in a limited range of Mg2+ (5.69–25.4 mg L−1) in the present study (Online Resources 1 and 2). The optimum (22.27 mg L−1) level of species for Mg2+ in the present study (Table 5) was higher than the levels equal to 16.65 mg L−1 and 12.29 mg L−1 given in Batmaz et al. (2020) and Dalgakıran et al. (2020), respectively. For the abundance of species (Online Resources 1 and 2), it could be seen that the highest abundance of species are found in sites having high Mg2+ concentrations as S15 (Mg2+ = 25.4 mg L−1; 220 individuals (ind.)) and S21 (Mg2+ = 20.1 mg L−1, 160 ind.) and both were sampled in October of 2020 but only 3 ind. were collected in S9 sampled in April of 2021 with 23.5 mg L−1 Mg2+ concentration (Online Resources 1 and 2). In terms of the species colonization, the low abundance in S9 with a high Mg2+ value in the April sampling seems normal since the S9 station dried at all in October. These results show that high Mg2+ values favor the abundance of P. zenkeri.
Conclusion
A relatively high ostracod taxon diversity (34 taxa) was detected in four lakes and 27 streams located in the Eastern Mediterranean region of Turkey. The model constructed with DO+Elev+Mg2+ was found as the best model to declare the variation in the species composition that is followed by pH + DO+Mg2+. Among environmental variables, Mg2+ and Elev showed statistically important direct effects on species composition in the present study. The variation in activities of environmental variables on the species has been observed when comparing the results in the present study with the finding reported in different geographical regions even if the species (e.g., cosmopolitan species) have wide tolerance levels to ecological variables. This pinpoints the importance of ecoregion-based studies because they will be better to reveal the environmental variable preferences of the species and the activities of these variables. Using of findings as presented in this study may result in more accurate data as compared to general ecological data in terms of estimation of current or past environmental conditions by using bioindicator species (e.g., ostracods). This deduction supports the statement of Willis and Whittaker (2002) as variables having important roles for the local and/or recent time species richness may not be such factors for the richness at regional spatial scale or longer time scale. Therefore, the increase in the number of studies to determine the region-based ecological preferences of species in the future will allow us to use species more effectively as bioindicators.
References
Aguilar-Alberola JA, Mesquita-Joanes F (2014) Breaking the temperature-size rule: thermal effects on growth, development and fecundity of a crustacean from temporary waters. J Therm Biol 42:15–24. https://doi.org/10.1016/j.jtherbio.2014.02.016
Akdemir D, Külköylüoğlu O (2021) Effects of temperature changes on the spatial distribution and ecology of ostracod (Crustacea) species. LimnoFish 7(1):1–13. https://doi.org/10.17216/LimnoFish.765049
Akdemir D, Külköylüoğlu O, Yavuzatmaca M, Tanyeri M, Gürer M, Alper A, Dere Ş, Çelen E, Yılmaz O, Özcan G (2020) Ecological characteristics and habitat preferences of Ostracoda (Crustacea) with a new bisexual population record (Muğla, Turkey). Appl Ecol Env Res 18(1):1471–1487. https://doi.org/10.15666/aeer/1801_14711487
APHA (1998) Standard methods for the examination of water and wastewater, 20th Edn. American Public Health Association, American Water Works Association, Water Environment Federation, Washington D.C
Baltanás A, Montes C, Martino P (1990) Distribution patterns of ostracods in the Iberian saline lakes. Influence of ecological factors. Hydrobiologia 197:207–220. https://doi.org/10.1007/978-94-009-0603-7_18
Batmaz F, Külköylüoğlu O, Akdemir D, Yavuzatmaca M (2020) Effective roles of ecological factors on nonmarine Ostracoda (Crustacea) in shallow waters of Malatya (Turkey). Ecol Res 35:511–523. https://doi.org/10.1111/1440-1703.12120
Benzie JAH (1989) The distribution and habitat preference of ostracods (Crustacea: Ostracoda) in a coastal sand-dune lake, loch of Strathbeg, north-East Scotland. Freshw Biol 22:309–321. https://doi.org/10.1111/j.1365-2427.1989.tb01104.x
Borcard D, Legendre P, Drapeau P (1992) Partialling out the spatial component of ecological variation. Ecology 73:1045–1055. https://doi.org/10.2307/1940179
Boyd CE, Tucker CS, Somridhivej B (2016) Alkalinity and hardness: critical but elusive concepts in aquaculture. J World Aquacult Soc 47(1):6–41. https://doi.org/10.1111/jwas.12241
Çelekli A, Lekesiz Ö, Yavuzatmaca M (2021) Bioassessment of water quality of surface waters using diatom metrics. Turk J Bot 45:379–396. https://doi.org/10.3906/bot-2101-16
Clarke KR, Gorley RN (2015) Primer V7 with Permanova+. Primer v7: User Manual/Tutorial PRIMER-E: Plymouth
Cusminsky G, Coviaga C, Ramos L, Pérez AP, Schwalb A, Markgraf V, Ariztegui D, Viehberg F, Alperin M (2020) Characterization ecoregions in Argentinian Patagonia using extant continental ostracods. An Acad Bras Ciênc 92(Suppl 2):e20190459. https://doi.org/10.1590/0001-3765202020190459
Dalgakıran E, Külköylüoğlu O, Yavuzatmaca M, Akdemir D (2020) Correlational analyses of the relationships between altitude and carapace size of Ostracoda (Crustacea). Ann Limnol Int J Limnol 56:2. https://doi.org/10.1051/limn/2019025
Danielopol DL, Handl M, Yin Y (1993) Benthic ostracods in the prealpine deep Lake Mondsee. Notes on their origin and distribution. In: McKenzie KG, Jones JP (eds) Ostracoda in the earth and life science. Balkema, Rotterdam, pp 465–479
de Oliveira da Conceição E, Mantovano T, de Campos R, Rangel TF, Martens K, Bailly D, Higuti J (2019) Mapping the observed and modelled intracontinental distribution of non-marine ostracods from South America. Hydrobiologia 847:1663–1687. https://doi.org/10.1007/s10750-019-04136-6
Dettman DL, Palacios-Fest M, Cohen AS (2002) Comment on G. Wansard & F. Mezquita, the response of ostracode shell chemistry to seasonal change in a Mediterranean freshwater spring environment. J Paleolimnol 27:487–491. https://doi.org/10.1023/A:1020535820345
César dos Santos Lima J, Gazonato Neto AJ, de Pádua AD, Freitas EC, Moreira RA, Miguel M, Daam MA, Rocha O (2019) Acute toxicity of four metals to three tropical aquatic invertebrates: the dragonfly Tramea cophysa and the ostracods Chlamydotheca sp. and Strandesia trispinosa. Ecotoxicol Environ 30(180):535–541. https://doi.org/10.1016/j.ecoenv.2019.05.018
Dudgeon D, Arthington AH, Gessner MO, Kawabata Z-I, Knowler DJ, Lévêque C, Naiman RJ, Prieur-Richard A-H, Soto D, Stiassny MLJ, Sullivan CA (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev 81:163–182. https://doi.org/10.1017/S1464793105006950
Goldman CR, Horne AJ (1983) Limnology. McGraw-Hill Book Co., New York, p 464
Griffiths HI, Holmes JA (2000) Non-marine ostracods and quaternary palaeoenvironments. Quaternary Research Association, Technical Guide, London, p 8
Gusakov VA, Makhutova ON, Gladyshev MI, Golovatyuk LV, Zinchenko TD (2021) Ecological role of Cyprideis torosa and Heterocypris salina (Crustacea, Ostracoda) in saline rivers of the Lake Elton basin: abundance, biomass, production, fatty acids. Zool Stud 60:53. https://doi.org/10.6620/ZS.2021.60-53
Hamilton NE, Ferry M (2018) Ggtern: ternary diagrams using ggplot2. J Stat Softw 87(3):1–17. https://doi.org/10.18637/jss.v087.c03
Hammer Ø, Harper DA, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):9
Heino J (2011) A macroecological perspective of diversity patterns in the freshwater realm. Freshw Biol 56:1703–1722. https://doi.org/10.1111/j.1365-2427.2011.02610.x
Heino J, Virkkala R, Toivonen H (2009) Climate change and freshwater biodiversity: detected patterns, future trends and adaptations in northern regions. Biol Rev 84:39–54. https://doi.org/10.1111/j.1469-185X.2008.00060.x
Holmes JA, Chivas AR (eds) (2002) The Ostracoda. Applications in quaternary research. Geoph Monog 131:301–313. https://doi.org/10.1029/GM131
Iglikowska A, Namiotko T (2012) The impact of environmental factors on diversity of Ostracoda in freshwater habitats of subarctic and temperate Europe. Ann Zool Fenn 49:193–218. https://doi.org/10.5735/086.049.0401
Juggins S (2003) Software for ecological and palaeoecological data analysis and visualization – C2 user guide. University of Newcastle, Newcastle-upon-Tyne
Karanovic I (2012) Recent freshwater ostracods of the world. Springer-Verlag, Heidelberg (Berlin), p 608
Katz N, Shavit R, Pruitt JN, Scharf I (2017) Group dynamics and relocation decisions of a trap-building predator are differentially affected by biotic and abiotic factors. Curr Zool 63(6):647–655. https://doi.org/10.1093/cz/zow120
Knudson CP, Geyer CJ, Benson S (2018) Glmm: generalized linear mixed models via Monte Carlo likelihood approximation (R software package). https://cran.r-project.org/package=glmm. Accessed 5 Jan 2022
Külköylüoğlu O (2009) Ecological succession of freshwater Ostracoda (Crustacea) in a newly developed rheocrene spring (Bolu, Turkey). Turk J Zool 33:115–123. https://doi.org/10.3906/zoo-0712-12
Külköylüoğlu O (2013) Diversity, distribution and ecology of non-marine Ostracoda (Crustacea) in Turkey: application of pseudorichness and cosmoecious species concepts. Rec Res Devel Ecol 4:1–18
Külköylüoğlu O, Sarı N, Akdemir D (2012a) Distribution and ecological requirements of ostracods (Crustacea) at high altitudinal ranges in northeastern Van (Turkey). Ann Limnol-Int J Limnol 48:39–51. https://doi.org/10.1051/limn/2011060
Külköylüoğlu O, Yavuzatmaca M, Akdemir D, Sarı N (2012b) Distribution and local species diversity of freshwater Ostracoda in relation to habitat in the Kahramanmaraş province of Turkey. Int Rev Hydrobiol 97(4):247–261. https://doi.org/10.1002/iroh.201111490
Külköylüoğlu O, Akdemir D, Sarı N, Yavuzatmaca M, Oral C, Başak E (2013) Distribution and ecology of Ostracoda (Crustacea) from troughs in Turkey. Turk J Zool 37:277–287. https://doi.org/10.3906/zoo-1205-17
Külköylüoğlu O, Sarı N, Dügel M, Dere Ş, Dalkıran N, Aygen C, Çapar Dinçer S (2014) Effects of limnoecological changes on the Ostracoda (Crustacea) community in a shallow Lake (lake Çubuk, Turkey). Limnologica 46:99–108. https://doi.org/10.1016/j.limno.2014.01.001
Külköylüoğlu O, Yavuzatmaca M, Sarı N, Akdemir D (2016) Elevational distribution and species diversity of freshwater Ostracoda (Crustacea) in Çankırı region (Turkey). J Freshw Ecol 31(2):219–230. https://doi.org/10.1080/02705060.2015.1050467
Külköylüoğlu O, Yavuzatmaca M, Çelen E, Akdemir D, Dalkıran N (2018) Ecological classification of the freshwater Ostracoda (Crustacea) based on physicochemical properties of waters and habitat preferences. Ann Limnol-Int J Limnol 54:26. https://doi.org/10.1051/limn/2018017
Külköylüoğlu O, Yavuzatmaca M, Akdemir D, Yılmaz O, Çelen E, Dere Ş, Dalkıran N (2019) Correlational patterns of species diversity, swimming ability and ecological tolerance of non-marine ostracoda (Crustacea) with different reproductive modes in shallow water bodies of ağrı region (Turkey). J Freshw Ecol 34(1):151–165. https://doi.org/10.1080/02705060.2019.1576551
Külköylüoğlu O, Akdemir D, Yavuzatmaca M (2020) Non-marine Ostracoda (Crustacea) as indicator species group of habitat types. Aquat Ecol 54:519–533. https://doi.org/10.1007/s10452-020-09757-x
Külköylüoğlu O, Yavuzatmaca M, Akdemir D (2021) Occurrence patterns, photoperiod and dispersion ability of the non-marine Ostracoda (Crustacea) in shallow waters. Turk J Fish Aquat Sc 21(2):73–85. https://doi.org/10.4194/1303-2712-v21_2_03
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280. https://doi.org/10.1007/s004420100716
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D, Loreau M, Gonzalez A (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7:601–613. https://doi.org/10.1111/j.1461-0248.2004.00608.x
Loucks OL (1962) A forest classification for the maritime provinces. Proc Nova Scot Inst Sci 25(2):1958–1962
Meisch C (2000) Freshwater Ostracoda of western and central Europe Heidelberg: spektrum akademischer verlag. Süßwasserfauna von Mittele 8:i–xii
Mezquita F, Griffiths HI, Sanz SJ, Soria M, Pinon A (1999a) Ecology and distribution of ostracods associated with flowing waters in the eastern Iberian Peninsula. J Crustacean Biol 19:344–354. https://doi.org/10.1163/193724099X00150
Mezquita F, Tapia G, Roca JR (1999b) Ostracoda from springs on the eastern Iberian Peninsula: ecology, biogeography and palaeolimnological implications. Palaeogeogr Palaeoecol 148:65–85. https://doi.org/10.1016/S0031-0182(98)00176-X
MGM (2022) General Directorate of Meteorology. https://www.mgm.gov.tr/veridegerlendirme/il-ve-ilceler-istatistik.aspx?m=ANTALYA. (Accessed 20 January 2022)
Moore RC (Ed.) (1961) Treatise on invertebrate paleontology, part Q, Arthropoda 3, Crustacea, Ostracoda. Geological Society of America and University of Kansas Press, New York and Lawrence, pp 442, 334 figs
Nazik L, Poyraz M (2017) A region that characterise the general karst geomorphology of Turkey: Central Anatolia Plateau karst zone (in Turkish). Turk Geogr Rev 68:43–56. https://doi.org/10.17211/tcd.300414
Oakley TH, Wolfe JM, Lindgren AR, Zaharoff AK (2012) Phylotranscriptomics to bring the understudied into the fold: monophyletic Ostracoda, fossil placement, and Pancrustacean phylogeny. Mol Biol Evol 30:215–133. https://doi.org/10.1093/molbev/mss216
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, et al. (2020) Vegan Community Ecology Package Version 2.5–7. https://cran.r-project.org/web/packages/vegan/index.html. (Accessed 5 Jan 2022)
Omernik JM (1987) Ecoregions of the conterminous United States. Ann Assoc Am Geogr 77(1):118–125. https://doi.org/10.1111/j.1467-8306.1987.tb00149.x
Palacios-Fest MR, Dettman DL (2001) Temperature controls monthly variation in ostracode valve mg/ca: Cypridopsis vidua from a small lake in Sonora, Mexico. Geochim Cosmochim Ac 65:2499–2508. https://doi.org/10.1016/S0016-7037(01)00602-0
Peres-Neto PR, Legendre P, Dray S, Borcard DA (2006) Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87:2614–2625. https://doi.org/10.1890/0012-9658(2006)87[2614:VPOSDM]2.0.CO;2
Pieri V, Martens K, Stoch F, Rossetti G (2009) Distribution and ecology of non-marine ostracods (Crustacea, Ostracoda) from Friuli Venezia Giulia (NE Italy). J Limnol 68(1):1–15. https://doi.org/10.4081/jlimnol.2009.1
R Core Team (2020) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.r-project.org/index.html. (Accessed 5 Jan 2022)
R Core Team (2021) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/. (Accessed 5 Jan 2022)
Ramos L, Epele LB, Grech MG, Manzo LM, Macchi PA, Cusminsky GC (2022) Modelling influences of local and climatic factors on the occurrence and abundance of non-marine ostracods (Crustacea: Ostracoda) across Patagonia (Argentina). Hydrobiologia 849:229–244. https://doi.org/10.1007/s10750-021-04722-7
Rieradevall M, Roca JR (1995) Distribution and population dynamics of ostracodes (Crustacea, Ostracoda) in a karstic lake: Lake Banyoles (Catalonia, Spain). Hydrobiologia 310:189–196. https://doi.org/10.1007/BF00006830
RStudio Team (2021) RStudio: Integrated development environment for R. RStudio, PBC, Boston, MA. http://www.rstudio.com/. (Accessed 5 Jan 2022)
RTMFWM (2018) Republic of Turkey Ministry of Forestry and Water Management, Water Management General Directorate, Flood and Drought Management Department. Eastern Mediterranean basin drought management plan, Volume I: General description of the basin and drought analysis. Ankara, Turkey (in Turkish), pp 202
Ruiz F, Abad M, Bodergat AM, Carbonel P, Rodríguez-Lázaro J, González-Regalado ML, Toscano A, García EX, Prenda J (2013) Freshwater ostracods as environmental tracers. Int J Environ Sci Technol 10:1115–1128. https://doi.org/10.1007/s13762-013-0249-5
Seaby RM, Henderson PA (2007) Ecological Community Analysis II (ECOM II) Version 2.1.3.137. Pisces Conservation Ltd., Lymington
Shuhaimi-Othman M, Yakub N, Ramle N-A, Abas A (2011) Toxicity of metals to a freshwater ostracod: Stenocypris major. J Toxic 2011:1–8. https://doi.org/10.1155/2011/136104
Siveter DJ, Tanaka G, Farrell UC, Martin MJ, Siveter DJ, Briggs DEG (2014) Exceptionally preserved 450-million-year-old Ordovician ostracods with brood care. Curr Biol 24:801–806. https://doi.org/10.1016/j.cub.2014.02.040
Strahler AN (1957) Quantitative analysis of watershed geomorphology: Transactions of the American Geophysical Union 38:913–920
Turpen JB, Angell RW (1971) Aspects of molting and calcification in the ostracod Heterocypris. Biol Bull 140:331–338
Uçak S, Külköylüoğlu O, Akdemir D, Başak E (2014) Distribution, diversity and ecological characteristics of freshwater Ostracoda (Crustacea) in shallow aquatic bodies of the Ankara region, Turkey. Wetlands 34:309–324. https://doi.org/10.1007/s13157-013-0499-5
Van der Meeren T, Almendinger JE, Ito E, Martens K (2010) The ecology of ostracodes (Ostracoda, Crustacea) in western Mongolia. Hydrobiologia 641:253–273. https://doi.org/10.1007/s10750-010-0089-y
Vander Vorste R, McElmurray P, Bell S, Eliason KM, Brown BL (2017) Does stream size really explain biodiversity patterns in lotic systems? A call for mechanistic explanations. Diversity 9(3):26. https://doi.org/10.3390/d9030026
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing DH (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137. https://doi.org/10.1139/f80-017
Viehberg FA (2006) Freshwater ostracod assemblages and their relationship to environmental variables in waters from Northeast Germany. Hydrobiologia 571(1):213–224. https://doi.org/10.1007/s10750-006-0241-x
Wetzel RG (2001) Limnology: lake and river ecosystems, 3rd edn. Academic Press, San Diego, California, USA, pp 1006
Willis KJ, Whittaker RJ (2002) Species diversity: scale matters. American Association for the Advancement of Science. Am Assoc Advanc Sci 295(5558):1245–1248. https://doi.org/10.1126/science.1067335
Yavuzatmaca M (2019) Comparative analyses of non-marine ostracods (Crustacea) among water basins in Turkey. Acta Zool Acad Sci Hung 65(3):269–297. https://doi.org/10.17109/AZH.65.3.269.2019
Yavuzatmaca M (2020a) Diversity analyses of nonmarine ostracods (Crustacea, Ostracoda) in streams and lakes in Turkey. Turk J Zool 44:519–530. https://doi.org/10.3906/zoo-2005-20
Yavuzatmaca M (2020b) Species assemblages of Ostracoda (Crustacea) from west-site of Turkey: their indicator potential for lotic and lentic habitats. Biologia 75:2301–2314. https://doi.org/10.2478/s11756-020-00494-y
Yavuzatmaca M (2021) Comparison of Ostracoda (Crustacea) species composition between lakes and streams at high elevations in Turkey. Acta Zool Acad Sci Hung 67(4):377–401. https://doi.org/10.17109/AZH.67.4.377.2021
Yavuzatmaca M, Külköylüoğlu O, Yılmaz O, Akdemir D (2017a) On the relationship of ostracod species (Crustacea) to shallow water ion and sediment phosphate concentration across different elevational range (Sinop, Turkey). Turk J Fish Aquat Sc 17:1333–1346. https://doi.org/10.4194/1303-2712-v17_6_40
Yavuzatmaca M, Külköylüoğlu O, Yılmaz O (2017b) Estimating distributional patterns of non-marine Ostracoda (Crustacea) and habitat suitability in the Burdur province (Turkey). Limnologica 62:19–33. https://doi.org/10.1016/j.limno.2016.09.006
Yavuzatmaca M, Külköylüoğlu O, Akdemir D, Çelen E (2018) On the relationship between the occurrence of ostracod species and elevation in Sakarya province, Turkey. Acta Zool Acad Sci Hung 64(4):329–354. https://doi.org/10.17109/AZH.64.4.329.2018
Yılmaz F, Külköylüoğlu O (2006) Tolerance, optimum ranges, and ecological requirements of freshwater Ostracoda (Crustacea) in Lake Aladağ (Bolu, Turkey). Ecol Res 21:165–173. https://doi.org/10.1007/s11284-005-0121-2
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgments
My special thanks go to Prof. Dr. Okan Külköylüoğlu (Bolu Abant İzzet Baysal University, Turkey), Prof. Dr. Abuzer Çelekli (Gaziantep University, Turkey) and Mrs. Filiz Batmaz (Bolu Abant İzzet Baysal University, Turkey) for their comments on the first draft. I also thank to Mrs. Mary Theresa Dorothy Williams (North Carolina State University) for her help with English. I would like to thank Mr. Ömer Lekesiz (Gaziantep University, Turkey) for his help to construct the map.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Online Resource 1
(PDF 329 kb)
Online Resource 2
(PDF 894 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yavuzatmaca, M. Determination of environmental variables groups affecting the occurrence of non-marine ostracods (Crustacea) in the Eastern Mediterranean region of Turkey. Biologia 77, 3185–3202 (2022). https://doi.org/10.1007/s11756-022-01208-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01208-2