Abstract
We aimed to estimate the diagnostic latency of patients with visceral leishmaniasis (VL). A monocentric retrospective observational study was conducted including all confirmed cases of VL diagnosed from January 2005 to March 2022. Epidemiological and clinical characteristics of patients with VL were collected. The diagnostic latency was defined as the number of days between the first contact with a health-care provider for signs and/or symptoms referable to VL and the laboratory diagnosis of leishmaniasis. Twenty-four cases of VL were included in the study, mostly male (75%) and Italians (79.2%), with a median age of 40 years [Inter Quartile Range (IQR 30–48)]. Fourteen (58.3%) VL cases were people living with HIV (PLWH) and 4 (16.6%) subjects were on immunosuppressive therapy. For VL the median diagnostic latency was 54 days (IQR 28–162). The shorter diagnostic latency was observed in PLWH [31 days (IQR 20–47)] followed by immunocompetent patients [160 days (IQR 133–247)] and those on immunosuppressive therapy [329 days (IQR 200–678)]. Twelve patients (50%) reported at least one medical encounter before the diagnosis of VL and 6 patients received a wrong therapy. Diagnostic delay in VL was significant in patients under immune suppressive treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Leishmaniasis is a vector-borne protozoan disease included by the World Health Organization (WHO) in the list of neglected tropical diseases which is endemic not only in tropical and subtropical areas but also in the Mediterranean basin [1]. Eight countries (Brazil, Eritrea, Ethiopia, India, Kenya, Somalia, South Sudan, and Sudan) report globally 89% of visceral leishmaniasis (VL) cases, while 88% of all cases of cutaneous leishmaniasis (CL) are reported by 9 countries (Afghanistan, Algeria, Brazil, Colombia, Iraq, Iran (Islamic Republic of), Pakistan, Peru, and the Syrian Arab Republic) [2]. Italy is considered a low endemic country for leishmaniasis by WHO with the last available data from 2021 reporting 75 autochthonous cases of VL and 25 autochthonous cases of CL [2]. However, the above-mentioned data probably reflect an under-reporting of the disease and do not consider the changing epidemiology of leishmaniasis in our country with the description of new foci occurring in northern areas of the country previously considered Leishmania free [3,4,5].
VL has a broad spectrum of clinical presentation and still represents a diagnostic challenge due to multiple factors such as signs and symptoms that can overlap with other infections among immunocompromised subjects, atypical manifestations, and low awareness by physicians [6, 7]. Moreover, in the case of VL, a delayed diagnosis might be responsible of a potentially life-threatening evolution.
The aim of our study was to estimate the time of diagnostic latency of patients finally diagnosed with VL and characterize the medical encounters with a missed diagnostic opportunity before the correct diagnosis of leishmaniasis was reached at a reference center in Milan, Italy.
Methods
Study design and setting
We performed a monocentric retrospective observational study including all subjects with a diagnosis of VL managed at the Infectious Diseases Department of Luigi Sacco Hospital (ASST-FBF-Sacco, Milan, Italy) from January 2005 to March 2022 (end of observation).
Study population
We included all patients with VL diagnosis based on clinical features and on positivity of serologic and/or parasitological tests (direct histological demonstration of Leishmania spp. amastigotes on bone marrow or other tissues, positivity by means of PCR on blood, bone marrow, or other tissues, or culture positivity).
Definitions
VL cases were classified in accordance with WHO classification: a case of VL is a person showing clinical signs (mainly prolonged irregular fever, splenomegaly and weight loss) with serological (at geographical area level) and/or parasitological confirmation (when feasible at central level) of the diagnosis [8].
The diagnostic latency was defined as the number of days elapsing between the first contact with a health-care provider for signs and/or symptoms referable to VL, and the laboratory diagnosis of leishmaniasis. A wrong or incongruent treatment for leishmaniasis before the diagnosis was defined as a treatment not recommended for the disease in accordance with the current Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH) [9].
For laboratory parameters, the following definitions were applied: leukopenia (white blood cells < 4190/µl), anemia (hemoglobin < 14.2 for male, hemoglobin < 12.5 g/dL for female; severe anemia was defined as hemoglobin < 8 g/dL), low platelets count (platelets < 155,000/µL for male and < 169,000/µL for female; severe thrombocytopenia was defined as a platelet count < 50,000/ µL), hypergammaglobulinemia (gamma globulins > 19.6%), and high ferritin levels (ferritin > 200 ng/mL for male and > 120 ng/mL for female).
Data collection and laboratory procedures
We collected anonymized data from the clinical records from both our inpatient and outpatient wards. Epidemiological and clinical characteristics of the subjects were recorded such as laboratory examination and all the diagnostic procedures performed to reach the diagnosis of leishmaniasis.
The serologic, molecular, and culture laboratory procedures adopted at our center to attempt a leishmaniasis’ diagnosis have been extensively described elsewhere [10, 11]. In brief, until 2013, the molecular species typing for Leishmania spp. was performed by the Infectious and Tropical Diseases laboratory of the University of Milan with an In-House method [10]. After 2013, the procedure was performed by Istituto Superiore di Sanità (ISS) in Rome. The species typing from culture isolates was obtained by MultiLocus Enzyme Electrophoresis (MLEE), complying the nomenclature through the characterization of zymodemes (zymodeme Montpellier, MON-; and zymodeme Rome, ZROM-). The results were analyzed looking for the association between exoenzymatic markers and geographical areas and in accordance with the unpublished data gathered by ISS’ laboratories. The molecular identification of Leishmania spp. was obtained by polymerase chain reaction–restriction fragment length polymorphism (PCR–RFLP) of Leishmania target sequences. The WHO reference Leishmania DNA stumps were used as controls. All PCR–RFLP samples were analyzed with MethaPhor gel 4% electrophoresis (EuroClone) or with capillary electrophoresis Qiaxcel (Qiagen GmbH, Hilden, Germany). The microscopy test was performed by the Pathological Anatomy Institute of Hospital L. Sacco, Milan. The histological preparations were colored with hematoxylin–eosin and Giemsa methods. The culture medium was EMTM (Evans Modified Tobie Medium), consisting in a solid-phase NNN (Novy-McNeal-Nicolle) and a liquid phase. The search for anti-Leishmania antibodies was carried out using a commercial kit for the IFAT and Western Blot (WB) IgG method, routinely used at the Microbiology Laboratory of the L. Sacco Hospital.
Statistical analysis
Data from the medical records were identified in continuous and categorical variables where appropriate and categorized accordingly. Quantitative variables were reported using median and interquartile range (IQR). Considering the descriptive nature of the study, neither a priori hypothesis was tested nor a formal sample size calculation was run.
Ethical statement
The study was approved by our Comitato Etico Interaziendale Area 1 (Protocol N° 0037058). Considering the retrospective nature of the study, no written informed consent was required but when available patients were contacted to obtain written informed consent.
Results
Characteristics of subjects with VL
Twenty-four cases of VL were included in the study, mostly male (75%) and Italians (79.2%), with a median age of 40 years (IQR 30–48). VL patients’ epidemiological, clinical, and laboratory features are reported in Table 1 according to being diagnosed during the hospitalization without previous medical encounters or after more than one medical encounter with a missed diagnostic opportunity. Fourteen VL cases were people living with HIV (PLWH) and 4 subjects were on immunosuppressive therapy (two patients were on chronic steroid therapy, one patient was on prednisone plus azathioprine plus hydroxychloroquine, and the last one on methotrexate plus abatacept plus rituximab). Characteristics of patients under immunosuppressive treatment are reported in Table 2. At the time of hospitalization for VL, 9 out of 14 PLWH were on effective antiretroviral treatment and 8 subjects had a detectable (> 50 copies/mL) HIV-RNA viral load. Eleven out of 14 PLWH had a CD4 + count < 200 cell/µL at hospital admission, with a median value of 111 CD4 + /µL (IQR 54–141). VL was the first manifestation of AIDS in 4 patients with a newly diagnosis of HIV. Only 5 patients reported residence in a recognized endemic area of Italy (Liguria, Campania, and Sicily). Eighteen patients lived in Lombardy and among them 9 were PLWH, one was on immunosuppressive therapy and 6 were intravenous drug users. In 7 cases, the patients reported traveling outside Italy (Portugal, Libya, Egypt and Croatia (one case), India, Nigeria, Corsica, and Eritrea).
Most patients presented with hepatosplenomegaly (87.5%). Fever (66.7%) and weight loss (41.7%) were also common findings. Twenty-two patients (91.7%) had more than one symptom at the time of presentation and 22 (91.7%) had more than one laboratory abnormalities. In particular, pancytopenia was observed in 91% of the patients, while leukopenia and thrombocytopenia were seen in all the cases. Five (20.8%) patients presented with severe anemia and 6 (25%) patients with severe thrombocytopenia. Hypergammaglobulinemia and high ferritin levels were observed in 89% and 93% of the cases, respectively. VL diagnosis was confirmed with a direct method in all patients. In particular, molecular methods turned positive in the majority of cases (18/19 on peripheral blood, 5/6 on bone marrow biopsy, and 2/2 histological samples one from liver and the other from stomach). A pathological demonstration of Leishmania spp. was obtained in 10 out of 15 specimens exanimated (in one case on a stomach sample; in one case, amastigotes were documented on peripheral blood smear; in one case a histologic test was positive both on cutaneous lesions and on bone marrow; and in the remaining, the histology was positive on bone marrow samples). Most of VL were caused by Leishmania infantum (20, 83.3%) followed by L.donovani (2, 8.3%), and for 2 (8.3%) patients, the Leishmania species was unknown.
Diagnostic delay
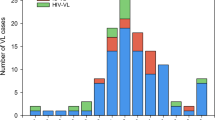
For VL, the median time of diagnostic delay was 54 days (IQR 28–162) with a double time for non-Italians when compared to the Italians with VL [47 days (IQR 28–140) vs 82 days (IQR 64–269)]. The shorter diagnostic latency was observed in PLWH [31 days (IQR 20–47)] followed by immunocompetent patients [160 days (IQR 133–247)] and those on immunosuppressive therapy [329 days (IQR 200–678)]. Twelve patients (50%) reported at least one medical encounter with different specialists before the diagnosis of VL (Fig. 1). In 6 cases, the medical encounter was with a physician in charge for the management of a chronic underlying condition (2 infectious diseases specialists, 2 rheumatologists, 1 oncologist, and 1 hepatologist). Six patients received a wrong therapy before the diagnosis (Table 3); in particular, one patient received a wrong diagnosis of multiple myeloma and a consequent treatment.
A number of medical encounter since signs and symptoms onset before the diagnosis of visceral leishmaniasis was made; B type of medical encounter since signs and symptoms onset before the diagnosis of visceral leishmaniasis was made according to being or not in charge of the same physician for a chronic underlying medical condition
Discussion
In our study, the observed diagnostic delay for VL (median 54 days) is similar to the median 40 days reported by Varani et al. in an Italian study conducted in Bologna province (Italy) during an outbreak of VL occurred in 2012–13 [3] and to the 42 days reported in a study conducted in Belgium between 2010 and 2018 [12]. On the contrary, shorter diagnostic delay for VL was reported by Franceschi et al. (22 days)] during an outbreak of VL in the Modena province (Italy) between 2012 and 2015 [5] and in another study conducted during a VL outbreak in Madrid (Spain) between 2009 and 2018 (median delay of 30 days and 16 days before and after the implementation of the RDT (rK39-ICT) test, respectively [13]. These observations suggest how the awareness of an ongoing outbreak is likely to reduce the diagnostic time for VL which could be on the contrary delayed in sporadic cases both in endemic [3] and non-endemic countries [12]. It is worth mentioning that the time for VL diagnosis was almost doubled in non-Italians suggesting barrier faced to reach an appropriate medical management as already observed in Germany for CL cases [14]. The higher awareness and better knowledge of leishmaniasis by infectious diseases physicians, which in Italy are also in charge for the management of PLWH, could partially explain the shorter time for VL diagnosis in PLWH. This seems not to be the case of other medical providers especially when sign and symptoms compatible with VL (i.e., prolonged irregular fever, splenomegaly, and weight loss) overlap with that of the underlying chronic condition they are managing. Nevertheless, the laboratory examinations performed in VL cases at diagnosis presented the classical hallmarks of VL (i.e., pancytopenia, hypergammaglobulinemia) in the majority of subjects underlying the poor awareness of the condition by the physicians. Thus, intervention looking at an increased awareness of this neglected tropical disease should be pursued on a wide range of medical providers especially in the case of management of immunosuppressed subjects. This could avoid the use of improper or wrong medication that not only are ineffective for VL but as in the case of immunosuppressant could also determine a progression of VL.
Italy is considered a low endemic country for leishmaniasis with southern regions (Sicily, Calabria, Puglia, Campania), Sardinia, some shoreline areas of Tuscany and Lazio and the Ponente Ligure provinces (Savona and Imperia) reporting autochthonous foci of the disease [15]. Nevertheless, in the last 30–40 years, the epidemiology of leishmaniasis in Italy has substantially changed, due to several factors such as the HIV epidemic [15, 16]; the increasing travels to high endemic countries with an increase of imported cases due to species others than L. infantum [17, 18]; climate changes, causing the vector’ spread to northern Italian regions which were not considered endemic for the disease [19, 20]; the increased number of subjects exposed to immunosuppressive agents modified the population at risk of VL and increased the number of atypical presentation with mucocutaneous involvement. For those reasons, it is important to keep up the awareness of this neglected tropical disease also in countries considered at low endemicity in which a remarkable diagnostic delay could be observed.
In our study, one out of three patients with VL reported a travel outside Italy as a potential source of imported disease. This observation contrast with a study conducted by the Italian National Institute of Health (ISS) performed in the period from 1986 to 2012 [17] in which only 3.5% of the total number of cases notified in Italy were imported. The autochthonous leishmaniasis cases were observed almost exclusively in subjects who were resident or had an history of trips in Italian regions traditionally considered endemic for leishmaniasis (Sicily, Campania, Liguria, Calabria, and Sardinia). The cases of VL observed in subjects who were resident in Lombardy were all out one PLWH with a history of intravenous drug use. In these cases, it has been proved that a parenteral transmission happened years before the development of immunosuppression condition and the manifestation of VL [21, 22].
The predominance of male subjects (75%) observed in our study is in line with the literature and for VL is mainly driven by the epidemiological intersection with the Italian HIV epidemic initially involving mainly males intravenous drug users [16]. This also could explain the low median age observed in VL cases. In particular, the HIV epidemic in Italy turned leishmaniasis from a pediatric disease into and adult disease and this phenomenon is further evolving in recent year due to on the one hand the introduction of effective antiretroviral treatment and on the other by the increased number of patients with iatrogenic immunosuppression [15]. Thus, immunomodulatory and/or immunosuppressant therapy has to be considered as an emerging risk factor for developing leishmaniasis [23].
Study limitations
Our study has a number of limitations. In particular, as the date of symptoms onset, such as the number and type of medical encounters before the leishmaniasis diagnosis was made, relied on a backwards reviewing of patients’ clinical charts, we cannot exclude an underestimation of our estimates. On the other side, we cannot exclude possible circumstances in which the opportunity to testing for leishmaniasis was offered, especially when and invasive procedure was required, and the patients would have refused the opportunity, thus overestimating the diagnostic latency. In addition, the exact date of immunosuppressive treatment start was not available in most of cases. Finally, the single Italian center design in and endemic region might not reflect the experiences of different health-care systems or other centers especially in non-endemic settings. However, we believe that our experience regarding this neglected disease highlights the need of an improved awareness to reach a prompt diagnosis of VL.
Conclusions
In our experience, VL diagnosis is burdened by a significant diagnostic latency which is longer for subjects under immunosuppressive treatment. A not negligible number of patients received wrong or improper treatments before the correct diagnosis of VL was made exposing the subjects not only to the untoward effect of the improper treatments but also, in the case of immunosuppressive treatments, to a potential faster progression of the disease. In conclusion, our findings highlight the importance of interventions aimed at increasing the awareness of the disease to a wide range of medical providers and specialists also in countries considered endemic for this neglected tropical disease.
Data availability
The complete dataset will be provided in txt format upon reasonable request to the corresponding author.
References
Obwaller AG, Karakus M, Poeppl W et al (2016) Could Phlebotomus mascittii play a role as a natural vector for Leishmania infantum? New data Parasite Vectors 9:458
https://www.who.int/data/gho/data/themes/topics/gho-ntd-leishmaniasis. Accessed 16 Jan 2023
Varani S, Cagarelli R, Melchionda F et al (2013) Ongoing outbreak of visceral leishmaniasis in Bologna Province, Italy, November 2012 to May 2013. Euro Surveill 18(29):20530
Franceschini E, Puzzolante C, Menozzi M et al (2016) Clinical and microbiological characteristics of visceral leishmaniasis outbreak in a Northern Italian nonendemic area: a retrospective observational study. Biomed Res Int 2016:6481028
Gaspari V, Ortalli M, Foschini MP et al (2017) New evidence of cutaneous leishmaniasis in north-eastern Italy. J Eur Acad Dermatol Venereol 31(9):1534–1540
Rosenthal E, Marty P, del Giudice P et al (2000) HIV and leishmania coinfection: a review of 91 cases with focus on atypical locations of leishmania. Clin Infect Dis 31(4):1093–1109
Yadav P, Azam M, Ramesh V, Singh R (2023) Unusual observations in leishmaniasis—an overview. Pathogens 12(2):297
https://www.who.int/news-room/fact-sheets/detail/leishmaniasis. Accessed 16 Jan 2023
Aronson N, Herwaldt BL, Libman M et al (2016) Diagnosis and treatment of leishmaniasis: clinical practice guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Clin Infect Dis 63(12):e202–e264. https://doi.org/10.1093/cid/ciw670
Pizzuto M, Piazza M, Senese D et al (2001) Role of PCR in diagnosis and prognosis of visceral leishmaniasis in patients coinfected with human immunodeficiency virus type 1. J Clin Microbiol 39(1):357–361
Antinori S, Calattini S, Longhi E et al (2007) Clinical use of polymerase chain reaction performed on peripheral blood and bone marrow samples for the diagnosis and monitoring of visceral leishmaniasis in HIV-infected and HIV-uninfected patients: a single-center, 8-year experience in Italy and review of the literature. Clin Infect Dis 44(12):1602–1610
Vandeputte M, van Henten S, van Griensven J et al (2020) Epidemiology, clinical pattern and impact of species-specific molecular diagnosis on management of leishmaniasis in Belgium, 2010–2018: a retrospective study. Travel Med Infect Dis 38:101885
Horrillo L, Castro A, Matía B et al (2019) Clinical aspects of visceral leishmaniasis caused by L. infantum in adults. Ten years of experience of the largest outbreak in Europe: what have we learned? Parasit Vector 12:359
Lindner AK, Richter J, Gertler M et al (2020) Cutaneous leishmaniasis in refugees from Syria: complex cases in Berlin 2015–2020. J Travel Med 27(7):taaa161
Gramiccia M, Scalone A, Di Muccio T, et al (2013) The burden of visceral leishmaniasis in Italy from 1982 to 2012: a retrospective analysis of the multi-annual epidemic that occurred from 1989 to 2009. Euro Surveill. 2013;18(29):20535
Gradoni L, Scalone A, Gramiccia M, Troiani M (1996) Epidemiological surveillance of leishmaniasis in HIV-1-infected individuals in Italy. AIDS 10:785–791
Di Muccio T, Scalone A, Bruno A et al (2015) Epidemiology of imported leishmaniasis in Italy: implications for a European Endemic Country. PLoS ONE 10(6):e0129418
Antinori S, Gianelli E, Calattini S et al (2005) Cutaneous leishmaniasis: an increasing threat for travellers. Clin Microbiol Infect 11(5):343–346
Maroli M, Rossi L, Baldelli R et al (2008) The northward spread of leishmaniasis in Italy: evidence from retrospective and ongoing studies on the canine reservoir and phlebotomine vectors. Trop Med Intern Health 13(2):256–264
Biglino A, Bolla C, Concialdi E et al (2010) Asymptomatic Leishmania infantum Infection in an area of Northwestern Italy (Piedmont Region) where such infections are traditionally nonendemic. J Clin Microbiol 48(1):131–136
Gradoni L, Scalone A, Gramiccia M (1993) HIV-Leishmania co-infections in Italy: serological data as an indication of the sequence of acquisition of the two infections. Trans R Soc Trop Med Hyg 87:94–96
Amela C, López-Gay D, Alberdig JC, Castilla J (1996) Injecting drug use as risk factor for visceral leishmaniasis in AIDS patients. Eur J Epidemiol 12:91–92
van Griensven J, Carrillo E, López-Vélez R, Lynen L, Moreno J (2014) Leishmaniasis in immunosuppressed individuals. Clin Microbiol Infect 20(4):286–299
Acknowledgements
The authors would like to thank all the subjects involved in this study.
Funding
The study was not funded.
Author information
Authors and Affiliations
Contributions
AP, AG, and SA designed the study; AG and AP responsible for the statistical analysis; MC, RG and MN performed the laboratory examinations. All of the authors contributed to patient enrollment, and the collection and interpretation of the data. SA supervised the project. AP prepared a preliminary draft of the manuscript, which was critically reviewed by AG and SA. All of the authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest relating to this study. AG has received consultancy fees from Mylan and Jansen, and non-financial educational support and a research grant from Gilead sciences and ViiV Healthcare. GR has received grants and fees for speaker bureaus, advisory boards, and CME activities from BMS, ViiV, MSD, AbbVie, Gilead, Janssen, and Roche. SA has received support for research activities from Pfizer and Merck Sharp & Dome. The other authors have nothing to declare.
Ethics approval statement
This study was approved by our Ethics Committee (Comitato Etico Interaziendale Area 1, Milan, Italy: Protocol No. 0037058).
Patient consent statement
Informed consent was obtained directly from the patients capable of making informed decisions about their medical care and participation in the study; otherwise, informed consent was obtained from his/her legal guardian or representative.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Poloni, A., Giacomelli, A., Corbellino, M. et al. Delayed diagnosis among patients with visceral leishmaniasis. Intern Emerg Med 18, 2293–2300 (2023). https://doi.org/10.1007/s11739-023-03430-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-023-03430-5