Abstract
Septic shock patients who survive past the acute period are associated with an increased risk of long-term mortality. However, factors for predicting late death remain unclear. We aimed to investigate the prognostic factors associated with late mortality in septic shock patients with 28-day survival after admission. This retrospective observational study used a prospective, multi-center registry of septic shock patients between October 2015 and December 2019 involving 12 emergency departments (EDs) from the Korean Shock Society. Adult septic shock patients visiting the ED with 28-day survival after admission were included. Among 4624 septic shock patients, 3588 (77.6%) who survived past day 28 were analyzed. The 90-day mortality rate was 14.2%. Non-survivors were older (66.8 vs. 68.9 years; p = 0.032) and had higher lactate levels (3.7 vs. 4.0 mmol/L; p = 0.028) than survivors. Pulmonary and hepatobiliary infections and a history of malignancy (27.7 vs. 57.5%; p < 0.001) were more frequent in the non-survivor group than in the survivor group. Independent risk factors for late death on multivariate regression analysis were age; malignancy; and hemoglobin, blood urea nitrogen, and albumin levels. The length of intensive care unit stay and Sequential Organ Failure Assessment score were independently associated with late death. Approximately, one-seventh of septic shock patients who survived past day 28 of admission died by day 90. Physicians must pay attention to survivors with these risk factors during the post-acute period as they have an increased mortality risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis is a leading cause of mortality worldwide, contributing to an estimated 11 million deaths in 2017 or approximately 20% of all global deaths [1, 2]. Present guidelines for sepsis primarily focus on early recognition and prompt initiation of treatment [3]. However, patients who survive an episode of septic shock may have a significantly increased risk of mortality for a prolonged time period [4]. The cause for this increase in the risk of late mortality in critically ill patients has long been disputed [5,6,7]. Previous studies have aimed to examine whether this risk is associated with pre-existing comorbidities or the sepsis episode itself [8, 9]. However, Shankar-Hari et al. observed an inconsistent relationship between sepsis and 1-year post-acute mortality, which called for additional studies to better control for potential confounding factors [6]. It might be reasonable to separately identify the factors that determine long-term and short-term survival after initial episodes of a critical illness. However, most studies about septic shock reported a crude mortality rate that does not distinguish between early and late deaths [10]. Moreover, few studies have separately reported long-term survival from each time point of disease, for example, survival from the onset of critical illness and survival after the initial episode. Furthermore, accurate prognostication of the chance of survival is helpful for deciding the treatment plans for patients and their families.
We hypothesized that the primary determinants of short-term mortality from the onset of septic shock were distinct from the main determinants of late death among those who survive in the short term. This study aimed to investigate the prognostic factors associated with late mortality in septic shock patients who survived past day 28 of admission.
Methods
Study design and population
This study was a retrospective analysis of data from a prospectively collected, multi-center registry of the Korean Shock Society (KoSS) between October 2015 and December 2019. The KoSS is a collaborative research network that investigates and works to improve the quality of diagnosis and management of sepsis. Since October 2015, KoSS investigators have been prospectively collecting data from septic shock patients at the emergency departments (EDs) of 12 university-affiliated hospitals throughout South Korea [11,12,13,14]. In this registry, patients aged 19 years or older who had a suspected or confirmed infection and evidence of refractory hypotension or hypoperfusion were enrolled [15]. Hypotension was defined as systolic blood pressure (SBP) < 90 mmHg, mean arterial pressure < 70 mmHg, or a decrease of > 40 mmHg in SBP from the baseline value [16]. Refractory hypotension was defined as persistent hypotension despite fluid challenge (20–30 mL/kg or at least 1 L of crystalloid solution administered over 30 min). Initial resuscitation, including fluid therapy and vasopressor administration, was conducted following the Surviving Sepsis Campaign guidelines. Hypoperfusion was defined as a serum lactate level ≥ 4 mmol/L. We excluded patients who signed a “do not attempt resuscitation” order, who did not meet the inclusion criteria within 6 h of ED arrival. We also excluded patients who were transferred from another hospital and did not meet the inclusion criteria on ED arrival, and who were directly transferred from the ED to other hospital due to lack of information about initial management, diagnosis, and prognosis. Among the patients who survived 28 days past ED admission, we excluded those who were lost to follow-up at day 90. The institutional review board of each institution approved the study protocol. Informed consent, which allows multi-research using a registry, was obtained from the patients before data collection.
Data collection
The case report form from the KoSS septic shock registry includes standard definitions of 200 variables, including clinical characteristics, therapeutic interventions, and patient outcomes. All data were collected in a web-based electronic database by each hospital’s coordinator and anonymized through a standardized registry form. Outliers or incorrect values were primarily filtered by this data-entry system. To regularly monitor and review data quality, a quality control committee was established, consisting of emergency physicians, regional research coordinators, and investigators from all EDs. The committee members provided feedback to the research coordinators and investigators on the results of the quality control process, and questions concerning data were clarified using the system’s query function or directly through a telephone call.
Demographic and clinical data, including data on age, sex, medical history, suspected infection site, initial vital signs, laboratory values on admission, severity scores, and ED interventions, were retrieved from the septic shock registry. Among the medical history, the definition of malignancy refers to active cancer within 6 months including solid tumors, hematologic disease, and metastatic disease. The initial Sequential Organ Failure Assessment (SOFA) and APACHE II scores were calculated using the worst parameter values within 24 h of ED arrival [17, 18]. The primary outcome of this study was the 90-day mortality rate.
Statistical analysis
Continuous variables were analyzed as median with the interquartile range (IQR). Categorical variables were expressed as numbers and percentages. The Student’s t-test or Mann–Whitney U test was used to compare continuous variables, and the chi-square test or Fisher’s exact test was used for categorical variables. Univariate and multivariate analyses were performed using logistic regression to determine the factors associated with 90-day mortality. Variables with p values < 0.1 in univariate analysis were entered into the multivariate analyses. The goodness of fit of the logistic model was evaluated using the Hosmer–Lemeshow test. The results of multivariate logistic regression analysis were reported as odds ratios (ORs) and 95% confidence intervals (CIs). A two-sided p value of < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS for Windows version 21.0 (SPSS Inc., Chicago, IL, USA).
Results
Baseline characteristics and laboratory findings
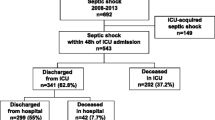
From October 2015 to December 2019, 4625 patients were enrolled in the KoSS septic shock registry. Of these, 1037 (22.4%) patients died by day 28 of admission. Among the 3,588 patients who survived past day 28, 219 patients were excluded due to loss to follow-up. Finally, 3369 patients were included in the study (Fig. 1). The overall 90-day mortality rate was 14.2% (478 patients).
Table 1 presents the baseline characteristics of the study population. Non-survivors were older than survivors and were predominantly male. Malignancy was more common in the non-survivor group than in the survivor group. Genitourinary infections were more common in the survivor group than in the non-survivor group. However, pulmonary and hepatobiliary infections were more common in the non-survivor group than in the survivor group. There were no significant differences in initial vital signs between the two groups. Laboratory findings are presented in Table 2. Hemoglobin and platelet levels were higher in the survivor group than in the non-survivor group. Blood urea nitrogen (BUN), C-reactive protein, and initial lactate levels were significantly higher in the non-survivor group than in the survival group. Prothrombin time was significantly longer in the non-survivor group than in the survivor group.
Clinical characteristics and management of septic shock
Clinical characteristics and details of septic shock management are presented in Table 3. The initial SOFA score within 24 h of admission and APACHE II scores were significantly higher in the non-survivor group than in the survivor group. The length of intensive care unit (ICU) stay (days) was significantly longer in the non-survivor group than in the survivor group.
There were no significant differences between the groups in source control or interventions within 1 h, including blood culture, antibiotic administration, vasopressor administration, and fluid infusion. The use of vasopressin and dual vasopressor therapy was more common in the non-survivor group than in the survivor group. The initiation of mechanical ventilation and renal replacement therapy was more common in the non-survivor group than in the survivor group.
Factors associated with 90-day mortality
Multivariate analysis was performed to identify potential risk factors associated with 90-day mortality, including variables with significant differences between the survivor and non-survivor groups in univariate analysis (Table 4). In multivariate regression analysis, the following clinical factors were found to be independent risk factors for 90-day mortality: age, malignancy, hemoglobin levels, albumin levels, BUN levels, initial SOFA score, and length of ICU stay.
Discussion
The main finding of this study was that an additional one-seventh of patients died by day 90 after the initial sepsis episode, and the independent risk factors for 90-day mortality included age, underlying medical conditions, length of ICU stay, and SOFA scores but not the infection site or initial management for septic shock.
In our study, the 90-day mortality rate among septic shock survivors was 14.2%. This result is similar to that of a previous study that evaluated late mortality in sepsis survivors 30 days after admission and reported that 17.4% of patients died by day 183[19]. A previous systematic review on post-acute mortality in sepsis patients reported that 16.1% of patients died after 1 year [6]. Consider our study period, the 90-day mortality rate of our study could be interpreted slightly higher than those reported by previous studies. Moreover, Prescott et al. reported that the highest ORs between the sepsis and other cohorts were observed during 31–90 days [9]; hence, physicians should pay attention to sepsis survivors during this post-acute period, as there is an increased risk of mortality. However, data about the risk factors of late death are limited and debatable. Although sepsis is associated with increased long-term mortality, there is a debate about which factors act as the main determinants of mortality after the initial episode. Some studies have shown that late mortality after sepsis may be associated with predisposing factors that initially put a patient at risk for sepsis [20]. Conversely, other studies have reported that the lasting damage from the sepsis episode itself is associated with increased mortality [21]. Our study showed that age, malignancy, laboratory parameters (albumin, BUN, and hemoglobin), initial SOFA scores, and length of ICU stay were independently associated with 90-day mortality in sepsis survivors. Albumin and hemoglobin are laboratory parameters that reflect the underlying medical condition of patients and are known to be associated with outcomes [22, 23]. The SOFA score is commonly used for assessing organ dysfunction and predicting the outcome but may be affected by the baseline status of patients [24]. Nevertheless, it is not clear whether laboratory markers were associated with the underlying medical condition or the damage from the sepsis episode. However, factors associated with the initial episode, such as inflammatory markers, infection site, and initial management, were not independent risk factors of late death. Therefore, it might suggest that underlying medical conditions were more related to late death than the characteristics of sepsis itself, such as infection site and initial management for septic shock.
There is a growing need for post-acute care after hospitalization in sepsis patients. Septic shock survivors frequently report new functional abilities and severe cognitive impairment [25,26,27,28]. “Post-sepsis syndrome” is a term used to describe the persistent adverse effects occurring after the treatment of sepsis [29]. To treat these impairments in sepsis survivors, several efforts have been made in post-acute care, including rehabilitation, medication optimization, and outpatient clinic follow-up after ICU discharge [30]. However, Taylor et al. recently reported that only 11% of patients received all the necessary elements of post-acute care within 90 days [31]. This suggests that more attention and further efforts are needed by physicians treating sepsis patients during initial hospitalization and thereafter. Further research to identify who may benefit from post-acute care and on how to provide after the initial sepsis episode is needed.
This study has several limitations. First, although this study aimed to identify risk factors associated with late death at admission, it would be more reasonable to use data at 28-day or discharge rather than data at admission. However, our septic shock registry has an inherent limitation that only includes mortality evaluated at 28 and 90 days. So, data associated with the status at 28 days were missing. Second, as this was a multi-center study, the enrollment periods and case volumes varied according to the hospital. Third, data that support the nutritional status of the patients before sepsis and 28 days after were missing. These data could be important for reflecting underlying medical conditions and predicting long-term mortality in sepsis survivors. Fourth, data on factors that could affect outcomes, such as socioeconomic status, were missing. As patients who survive often need extensive care, socioeconomic status could be a confounding factor. Fifth, data about the detailed information of malignancy such as stage of the disease, kind of malignancy and kind of treatment. If more information would be provided, findings would be changed. Sixth, there is a possibility that patients who experienced sepsis before were included in this study. Lastly, 6.1% of patients who survived past day 28 were excluded as they were lost to follow-up. This could introduce a selection bias toward patients with favorable outcomes.
Conclusion
Septic shock patients are at risk of mortality after the acute phase of the disease. This study found that among septic shock patients who survived past day 28, about one-seventh of the patients died by day 90. Physicians must pay attention to sepsis survivors during the post-acute period as they have an increased risk of mortality.
Availability of data and materials
The dataset used in the study is available from the corresponding author on reasonable request.
References
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR et al (2020) Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. Lancet 395(10219):200–211. https://doi.org/10.1016/s0140-6736(19)32989-7
Ryoo SM, Kang GH, Shin TG, Hwang SY, Kim K, Jo YH et al (2018) Clinical outcome comparison of patients with septic shock defined by the new sepsis-3 criteria and by previous criteria. J Thorac Dis 10(2):845–853. https://doi.org/10.21037/jtd.2018.01.96
Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R et al (2017) Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Intensive Care Med 43(3):304–377. https://doi.org/10.1007/s00134-017-4683-6
Winters BD, Eberlein M, Leung J, Needham DM, Pronovost PJ, Sevransky JE (2010) Long-term mortality and quality of life in sepsis: a systematic review. Crit Care Med 38(5):1276–1283. https://doi.org/10.1097/CCM.0b013e3181d8cc1d
Quartin AA, Schein RM, Kett DH, Peduzzi PN (1997) Magnitude and duration of the effect of sepsis on survival. Department of Veterans Affairs Systemic Sepsis Cooperative Studies Group. JAMA. 277(13):1058–63
Shankar-Hari M, Ambler M, Mahalingasivam V, Jones A, Rowan K, Rubenfeld GD (2016) Evidence for a causal link between sepsis and long-term mortality: a systematic review of epidemiologic studies. Crit Care 20:101. https://doi.org/10.1186/s13054-016-1276-7
Kennelly PJ, Martin-Loeches I (2016) Long term mortality following sepsis. Ann Transl Med 4(19):387. https://doi.org/10.21037/atm.2016.08.31
Garland A, Olafson K, Ramsey CD, Yogendran M, Fransoo R (2014) Distinct determinants of long-term and short-term survival in critical illness. Intensive Care Med 40(8):1097–1105. https://doi.org/10.1007/s00134-014-3348-y
Prescott HC, Osterholzer JJ, Langa KM, Angus DC, Iwashyna TJ (2016) Late mortality after sepsis: propensity matched cohort study. BMJ 353:i2375. https://doi.org/10.1136/bmj.i2375
Ou SM, Chu H, Chao PW, Lee YJ, Kuo SC, Chen TJ et al (2016) Long-term mortality and major adverse cardiovascular events in sepsis survivors. A Nationwide population-based Study. Am J Respir Crit Care Med. 194(2):209–17. https://doi.org/10.1164/rccm.201510-2023OC
Ko BS, Choi SH, Kang GH, Shin TG, Kim K, Jo YH et al (2020) Time to antibiotics and the outcome of patients with septic shock: a propensity score analysis. Am J Med 133(4):485–91.e4. https://doi.org/10.1016/j.amjmed.2019.09.012
Kim YJ, Kang J, Kim MJ, Ryoo SM, Kang GH, Shin TG et al (2020) Development and validation of the VitaL CLASS score to predict mortality in stage IV solid cancer patients with septic shock in the emergency department: a multi-center, prospective cohort study. BMC Med 18(1):390. https://doi.org/10.1186/s12916-020-01875-5
Ryoo SM, Han KS, Ahn S, Shin TG, Hwang SY, Chung SP et al (2019) The usefulness of C-reactive protein and procalcitonin to predict prognosis in septic shock patients: a multicenter prospective registry-based observational study. Sci Rep 9(1):6579. https://doi.org/10.1038/s41598-019-42972-7
Ko BS, Kim K, Choi SH, Kang GH, Shin TG, Jo YH et al (2018) Prognosis of patients excluded by the definition of septic shock based on their lactate levels after initial fluid resuscitation: a prospective multi-center observational study. Crit Care 22(1):47. https://doi.org/10.1186/s13054-017-1935-3
Peake SL, Delaney A, Bailey M, Bellomo R, Cameron PA, Cooper DJ et al (2014) Goal-directed resuscitation for patients with early septic shock. N Engl J Med 371(16):1496–1506. https://doi.org/10.1056/NEJMoa1404380
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM et al (2013) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 41(2):580–637. https://doi.org/10.1097/CCM.0b013e31827e83af
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H et al (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 22(7):707–10. https://doi.org/10.1007/bf01709751
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13(10):818–829
Thompson K, Taylor C, Jan S, Li Q, Hammond N, Myburgh J et al (2018) Health-related outcomes of critically ill patients with and without sepsis. Intensive Care Med 44(8):1249–1257. https://doi.org/10.1007/s00134-018-5274-x
Clermont G, Angus DC, Linde-Zwirble WT, Griffin MF, Fine MJ, Pinsky MR (2002) Does acute organ dysfunction predict patient-centered outcomes? Chest 121(6):1963–1971. https://doi.org/10.1378/chest.121.6.1963
Abu-Kaf H, Mizrakli Y, Novack V, Dreiher J (2018) Long-term survival of young patients surviving ICU admission with severe sepsis. Crit Care Med 46(8):1269–1275. https://doi.org/10.1097/ccm.0000000000003205
Finfer S, Bellomo R, McEvoy S, Lo SK, Myburgh J, Neal B et al (2006) Effect of baseline serum albumin concentration on outcome of resuscitation with albumin or saline in patients in intensive care units: analysis of data from the saline versus albumin fluid evaluation (SAFE) study. BMJ 333(7577):1044. https://doi.org/10.1136/bmj.38985.398704.7C
Qi D, Peng M (2021) Early hemoglobin status as a predictor of long-term mortality for sepsis patients in intensive care units. Shock 55(2):215–223. https://doi.org/10.1097/shk.0000000000001612
Ferreira FL, Bota DP, Bross A, Mélot C, Vincent JL (2001) Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 286(14):1754–1758. https://doi.org/10.1001/jama.286.14.1754
Borges RC, Carvalho CR, Colombo AS, da Silva Borges MP, Soriano FG (2015) Physical activity, muscle strength, and exercise capacity 3 months after severe sepsis and septic shock. Intensive Care Med 41(8):1433–1444. https://doi.org/10.1007/s00134-015-3914-y
Annane D, Sharshar T (2015) Cognitive decline after sepsis. Lancet Respir Med 3(1):61–69. https://doi.org/10.1016/s2213-2600(14)70246-2
Iwashyna TJ, Ely EW, Smith DM, Langa KM (2010) Long-term cognitive impairment and functional disability among survivors of severe sepsis. JAMA 304(16):1787–1794. https://doi.org/10.1001/jama.2010.1553
Hensley MK, Prescott HC (2018) Bad brains, bad outcomes: acute neurologic dysfunction and late death after sepsis. Crit Care Med 46(6):1001–1002. https://doi.org/10.1097/ccm.0000000000003097
Prescott HC, Costa DK (2018) Improving long-term outcomes after sepsis. Crit Care Clin 34(1):175–188. https://doi.org/10.1016/j.ccc.2017.08.013
Brown SM, Bose S, Banner-Goodspeed V, Beesley SJ, Dinglas VD, Hopkins RO et al (2019) Approaches to addressing post-intensive care syndrome among intensive care unit survivors. A narrative review. Ann Am Thorac Soc 16(8):947–56. https://doi.org/10.1513/AnnalsATS.201812-913FR
Taylor SP, Chou SH, Sierra MF, Shuman TP, McWilliams AD, Taylor BT et al (2020) Association between adherence to recommended care and outcomes for adult survivors of sepsis. Ann Am Thorac Soc 17(1):89–97. https://doi.org/10.1513/AnnalsATS.201907-514OC
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The institutional review board of each institution approved the study protocol, and informed consent was obtained from the patients before data collection (Asan Medical Center Institutional Review Board No. 2015-1283). All procedures performed in this study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, SM., Ryoo, S.M., Shin, T.G. et al. Prognostic factors for late death in septic shock survivors: a multi-center, prospective, registry-based observational study. Intern Emerg Med 17, 865–871 (2022). https://doi.org/10.1007/s11739-021-02847-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-021-02847-0