Abstract
In patients with systemic sclerosis, a ratio between forced vital capacity (FVC) and diffusing capacity of carbon monoxide (DLCO, FCV%/DLCO%) >1.5 might be a predictor of pulmonary hypertension (PH). The aim of this study is, therefore, to evaluate whether this index can be used in patients with PH, regardless of etiology. 83 consecutive outpatients with suspected PH at non-invasive work-up underwent spirometry and DLCO test before right heart catheterization (RHC); FVC%/DLCO% ratio was then calculated and compared with mean pulmonary-artery-pressure (mPAP) and mortality at 5-year follow-up. Significant correlations between FVC%/DLCO% and PAsP and mPAP levels were found (p < 0.05). After ROC curve analysis and definition of best cut-off values for PAsP and FVC%/DLCO%, increased mPAP values at RHC were observed comparing subjects with both PAsP and FVC%/DLCO% values below cut off values (−/−), either PAsP or FVC%/DLCO% above cut off values (±), or both above (+/+) (p < 0.05). Poorer survival rates are observed at follow-up with higher FVC%/DLCO% values (0% for <1, 17.4% for 1–3, 33.3% for >3, p < 0.05), when comparing subjects with either increased PAsP and FVC%/DLCO% values or both with those with lower (log-rank p < 0.05). Even in subjects with mPAP at RHC >25 mmHg, increased FVC%/DLCO% values predicted a worse outcome (p < 0.05). FVC%/DLCO% values are related to mPAP in subjects with suspected PH, and may further stratify the risk of mortality in addition to PAP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pulmonary hypertension (PH) is a clinical disorder characterized by an increased pulmonary arterial pressure due to several clinical conditions including several cardiovascular and respiratory disorders. By definition, mean pulmonary arterial pressure (mPAP) measured at right heart catheterization (RHC) ≥25 mmHg is required for a diagnosis of PH.
According to international guidelines [1], patients who have symptoms, signs and history suggestive for PH should undergo a multi-step work-up which includes echocardiography, high resolution computed tomography, ECG, pulmonary function test, and pulmonary scintigraphy before RHC.
Nowadays, the supposition of PH is based on a few non-invasive findings, such as the estimation of systolic pulmonary artery pressure (PAsP) and other parameters at echocardiography, or the evaluation of the ratio between aortic and pulmonary trunk diameter measured at CT scan [2]; however, such examinations are expensive, and further tools are required to improve the efficacy of screening tests and reduce the costs.
Some authors suggest that a forced vital capacity/diffusing capacity of the lungs for carbon monoxide FVC%/DLCO% (percentage predicted) ratio >1.5 may be useful in the diagnosis of pulmonary vasculopathy in systemic sclerosis (SSc) patients with or without interstitial lung disease [3]. In fact, disproportionate reduction in diffusing capacity in terms of abnormal FVC/DLCO ratio (F/D) has been previously described as a marker of PH in patients with scleroderma [4].
F/D ratio has been used in the large, prospective, PHAROS study, as a clinical marker for the detection of patients with scleroderma and a higher risk of developing PH [5]. Modification of DLCO can be explained by changes in gas exchange area, alteration of the alveolar capillary membrane, and ventilation/perfusion relationship in the lung and pulmonary capillaries involvement [6]. Therefore, DLCO may be reduced in patients with PH [7]. An algorithm described in the DETECT study has also been proposed for the early identification of patients in SSc potentially suffering from PH [8].
Furthermore, there is evidence that DLCO may stratify the risk of adverse events in subjects with PH. DLCO is an independent predictor of death in patients with WHO Group I PAH [9]. In patients with PH and preserved ejection fraction, a low DLCO is strongly associated with mortality [10].
In prior studies, F/D was widely used for the detection of patients with SSc and suspected PH, who should be, therefore, assessed by echocardiography and RHC to confirm the diagnosis [3, 5, 8]; however, this marker has not been used for the detection of PH in all the other groups included in international classifications on PH.
The purpose of this study, therefore, is to assess whether an F/D ratio can provide useful information for screening and risk stratification in unselected patients with suspected PH.
Methods
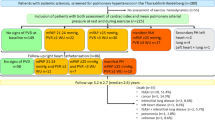
This was a prospective observational study. Between 2010 and 2015, consecutive patients referred to PH services at University Hospital (Ospedali Riuniti) in Foggia and General Hospital F. Miulli, Acquaviva delle Fonti (BA) for suspected PH were enrolled in the study. Patients were referred by general practitioner or other specialist usually in the presence of severe dyspnea. In this case, patients who showed at echo-color-Doppler, a non-invasive finding of PAsP >25 mmHg combined with other echocardiography features such as increased dimensions of right heart chambers, abnormal shape and function of the interventricular septum, increased right ventricular wall thickness, and dilated main pulmonary artery [1], or an intermediate or high risk of PH, as suggested by international guidelines ([1], Table 8a, b), and 160 patients were screened; only 83 (52%) were selected for study and underwent RHC.
All involved patients showed stable dyspnea for at least 3 months [and from the last chronic obstructive pulmonary disease (COPD) exacerbation or acute heart failure]. Patients with suspected PH underwent pulmonary and cardiology assessment by clinical examination, spirometry and DLCO test (and calculation of FVC%/DLCO% ratio), 6MWT, echocardiography, performed by a cardiologist experienced in PH and then assessed by RHC. At the time of enrollment no patients received a specific treatment for PH. Chest CT, pulmonary scintigraphy and other examinations included within PH diagnostic algorithm were performed when necessary. Patients with confirmed PAH diagnosis were treated according to ESC/ERS guidelines [1]. During follow-up, all patients were evaluated every 6 months, or less if necessary, by clinical examination, 6MWT and echocardiography. Data about mortality were then collected during a 5-year follow-up.
The study was approved by the local ethics committee, and the all procedures were performed after written informed consent.
Spirometry and DLCO measurement
Pulmonary function tests were performed in the pulmonary function laboratory using a spirometer (Sensormedics, USA) according to GOLD guidelines [11], the subjects were classified as COPD if a reversibility test was negative [12]. Single-breath DLCO measurements were made according to ATS criteria with a 10-s breath-hold [13]; correction for hemoglobin was performed.
Echocardiography
Conventional echocardiography was used to assess LV dimensions and ejection fraction (LVEF), peak velocities of trans-mitral early (E) and late diastolic (A) LV filling, the ratio of trans-mitral early to late (E/A ratio) LV filling velocity.
Transthoracic echocardiography was performed with the use of iE33 (Philips Medical Systems, Andover, MA, USA). LV dimensions and LVEF were calculated as recommendations in the joint ASE/ESC guidelines. LVEF was calculated according to the Simpson’s rule.
Peak PA systolic pressures were estimated by calculating the systolic pressure gradient between the right ventricle and right atrium by the maximum velocity of the tricuspid regurgitant jet using the modified Bernoulli equation, and then adding to this gradient an estimated right atrial pressure, based on the size of the inferior vena cava and its variation with respiration.
Right heart catheterization
Hemodynamic assessment was performed by RHC. Pulmonary arterial (systolic, diastolic and mean), right atrial, and pulmonary capillary wedge pressures were recorded at the end of a quiet respiratory cycle. Pulmonary and systemic flows were obtained by the Fick principle using table-derived oxygen consumption values and calculated oxygen content at the correspondent different sites. The trans-pulmonary pressure gradient was defined as the difference between the mean pulmonary arterial pressure and the mean pulmonary capillary wedge. Pulmonary and systemic vascular resistance indices were calculated using the standard formula. In PAH vaso-reactivity testing has been performed at the time of diagnostic RHC.
Statistical analysis
Data are presented as mean ± standard deviation. Differences between groups were assessed by the T-Student or Mann–Whitney according to data distribution. Linear correlations were determined by measuring the Pearson’s correlation coefficient. Pearson’s χ2 test and log-rank test were used for trend analysis.
The association between time to mortality and F/D ratio was assessed using log-rank tests, and Kaplan–Meier plots used to describe the likelihood of the outcome during the follow-up period.
Logistic regression analysis was used for the calculation of hazard ratio. Linear regression analysis was used for estimating linear trends.
Multivariable Cox analysis and multiple regression analysis were used for correction for principal bias.
A p value <0.05 was considered significant. Statistical Software (GraphPad 6.01, GraphPad Software Inc. 2012) was used to analyze the data.
Results
83 consecutive outpatients matching the inclusion criteria were enrolled. After RHC, the patients were divided in two groups, 61 patients with PH (mPAP >25 mmHg at RHC, PH+) and 22 without PH (PH−). Among PH+ patients, 28 belonged to ESC guidelines group 1 (46%) and 13 to group 3 (21%), while the remaining were distributed into the others groups (Table 1). Just four subjects among 13 in the group 3 had pulmonary fibrosis, and none with emphysema. General characteristics of the patients are shown in Table 2. PH+ patients were older than the control group (67.68 ± 12.06 vs 60.53 ± 12.35, p = 0.04). No significant difference was found among principal spirometry parameters, while at echocardiography, PAsP was higher in PH + group (72.65 ± 23.39 vs 57.54 ± 21.27 mmHg, p = 0.04). At RHC, several differences were observed between two groups: the cardiac index was lower in PH+ patients, as well as NHYA class, and pulmonary capillary wedge pressures were higher. 6MWT results were lower, although not significantly, in PH+ group.
A significant correlation between FVC/DLCO and PAsP values at echo-color Doppler evaluation was found (r 0.26, p < 0.05, Fig. 1). Increased mPAP levels were found with increasing values of FVC%/DLCO% (28.14 ± 9.86, 38.04 ± 16.64, 43.13 ± 16.68 mmHg, p for trend <0.05, Fig. 2).
After ROC curve analysis, best cut-off value for PAsP is identified at 57 mmHg (sensitivity 93%, specificity 44%, positive predictive power 28%, negative predictive power 96%, area under the curve 0.681, 95% CI 0.561–0.785), and 1.56 for FVC%/DLCO% (sensitivity 79%, specificity 51%, positive predictive power 24%, negative predictive power 92%, area under the curve 0.608, 95% CI 0.487–0.721, p 0.5479 vs PAsP). Increased mPAP values at RHC are found comparing subjects with both PAsP <57 mmHg and FVC%/DLCO% <1.56 (−/−), either PAsP >57 mmHg or FVC/DLCO >1.56 (±), or both PAsP >57 mmHg and FVC%/DLCO% >1.56 (+/+) (p for trend <0.05, Fig. 3). The trend remained significant even after correction for age and gender at multivariable linear regression analysis (p < 0.001, Table 2).
Poorer survival rates are observed at follow-up with higher FVC%/DLCO% levels (0% for FVC%/DLCO% <1, 17.4% for FVC%/DLCO% between 1-3, 33.3% for FVC%/DLCO% >3, log-rank p < 0.05, Fig. 4), and comparing subjects with both PAsP <57 mmHg and FVC%/DLCO% <1.56 (−/−), either PAsP >57 mmHg or FVC%/DLCO% >1.56 (±), or both PAsP >57 mmHg and FVC%/DLCO% >1.56 (+/+) (0%, 13.8%, 33.3%, respectively, p < 0.05, Fig. 5), even after multivariable Cox’ correction for age and gender (p < 0.05, Table 3).
Even considering just subjects with mPAP >25 mmHg at RHC, those with FCV%/DLCO% levels above a cut off level were characterized by a worse prognosis (Fig. 6, p for trend <0.05, significant even after correction for age and gender at multivariable analysis, hazard ratio 1.68, 95% CI 1.00–2.82, Table 3).
Discussion
This is one of the first reports on FVC%/DLCO% as a prognostic marker for PH and mortality in an unselected population of consecutive outpatients with suspected PH at non-invasive assessment. FVC%/DLCO% values were also related to PAsP non-invasively assessed at echo-color Doppler and mPAP at right heart catheterization. Such data may support a possible use of the FVC%/DLCO% ratio for the non-invasive screening of subjects with suspected PH to be referred for RHC, and as an additional marker of poor prognosis in subjects with PH. We also found an additional prognostic value of FVC%/DLCO% beyond mPAP values.
The diagnosis and confirmation of PH is usually based on RHC, that, however, in patients with PH can be technically demanding [1], and associated with severe complications. Thus, invasive diagnostics should be performed in high volume third level centers [14]. So, the aim of this study is to evaluate the efficacy of FVC/DLCO ratio in identifying patients with suspected PH to be submitted to RHC and its role as a prognostic index.
Moreover, despite increasing awareness of PAH, a considerable lag between the onset of symptoms and diagnosis of PAH may be often observed. Indeed, the REVEAL (Registry to Evaluate Early and Long-Term PAH Disease Management) registry shows that 21% of patients have symptoms for more than 2 years before diagnosis [15, 16]. Therefore, early detection of PH should be pursued for an early treatment and the prevention of RH failure [17].
In our study we aimed to assess the performance of FVC/DLCO ratio in a group of PH patients with mixed etiology.
A low DLCO is a typical finding in PAH, and reflects the pathological mechanisms of this disease such as muscularization of smaller, more peripheral pulmonary arteries, medial thickening of the muscular arteries, intimal thickening, and reduction in peripheral vascular bed [18].
In contrast, the FVC and other lung function tests, like FEV1, are not proportionately reduced compared to DLCO [19], and, therefore, FVC/DLCO ratio can be clinically useful in suspecting (but not excluding) the diagnosis of PAH.
Already in 1992, Steen et al. [20] demonstrate that patients with systemic sclerosis (SSc) which had a FVC/DLCO ratio >1.4 could more frequently develop pulmonary hypertension. In 2003 Coghlan et al. [8] showed in the DETECT study, which tested a two-step decision algorithm for the detection of PH in SSc patients, that, among the non-echocardiographic variables used for the identification of PAH, FVC/DLCO ratio with a threshold of 1.5 performs a 71.5% area under the curve at ROC curve analysis. Also Hsu et al. [5], in a recent study, consider, along with other parameters, a FVC/DLCO ratio >1.6 to predict future PH in high-risk scleroderma patient.
Few data, however, are available about other populations and other groups of PH. Nathan and al. [21] sought to assess whether FVC/DLCO ratio might be a good predictor of PH in idiopathic pulmonary fibrosis; the authors found that a FVC/DLCO ratio >1.5 is associated with a nearly twofold increased risk of PH.
Again in PH Group III, COPD patients show a PH disproportionately severe when compared to their obstructive pulmonary disease, thus being possible candidates for the testing of vasodilatator agents [22]. In fact, a resting mPAP of 35–40 mmHg is quite unusual in COPD patients, except for episodes of acute exacerbation or associated cardiopulmonary disease, such as left heart disease, collagen vascular disease, or obesity hypoventilation syndrome [23]. However, Weitzenblum et al. describe an “atypical” subgroup of patients who, despite a moderate airflow obstruction (FEV1 50%), have a severe PH defined as mPAP >40 mmHg [24]. FEV1 is, therefore, used to assess the severity of obstruction, but also the FVC is reduced in obstructive lung diseases although proportionately less than FEV1, while they are both reduced in restrictive lung diseases. This could allow the use of FVC/DLCO ratio assessment to all causes of PH group III. This may have clinically relevant implications since 66% of COPD patients develop some degree of PH [25], and COPD patients with PH have a worse prognosis [26], and they could be potential candidates for the testing of vasodilatator agents [22]. So FVC/DLCO-PAsP combined assessment might be useful for the screening of patients to be submitted to RHC.
We also evaluated whether the FVC%/DLCO% ratio might be a prognostic index of all forms of PH. PAH has a worse prognosis: the median survival is 2.8 years, with 1-, 3-, and 5-year survival rates of 68, 48, and 34%, respectively, before the advent of specific therapy [27].
Several risk markers have been proposed for the risk stratification of subjects with PH, and are recommended by guidelines: functional class, 6MWT, BNP, cardiopulmonary exercise testing, echocardiographic parameters as TAPSE and hemodynamics [1, 28]. In addition to such well established parameters, FCV/DLCO may further improve risk stratification in PH by identifying those subjects with a poorer lung function [29], although its role has been validated only in PH group I patients. In previous studies, DLCO is an independent predictor of survival; a value of DLCO <7.2 mL/mmHg/min is associated with a hazard ratio of 5.3 [30]. In isolated scleroderma-related PAH, DLCO are useful for screening and to predict mortality at 72-month follow-up, even at multivariable analysis [31].
The capacity of stratifying the risk of death in long term follow-up with a closer view to pulmonary impairment may represent the original contribution of FVC/DLCO besides other more ‘cardiovascular’ risk markers in PH. Moreover, FVC/DLCO may also be assessed in not able to perform a 6MWT or a cardiopulmonary exercise test, or rejecting invasive examinations (RHC).
Conclusions
FVC%/DLCO% values are related to mPAP in subjects with suspected PH at non-invasive assessment, and may further stratify the risk of mortality in addition to PAsP and mPAP.
Limitations
This is a small observational study in an unselected population of outpatients: these preliminary results should be confirmed in larger cohorts of patients, allowing a multivariable analysis including other potential confounders (drug therapy, functional class).
Differences in terms of FVC%/DLCO% values between PH+ and PH− subjects after RHC are not statistically significant, given the relatively small number of subjects in the PH− group: larger groups would be required to find such difference as significant.
The low number of patients enrolled in the study does not allow for a detailed analysis of survival according to PH etiology, and classification or specific drug therapy.
Despite 5-year follow-up and given the dyshomogeneity of PH’s etiologies, this study’s conclusions cannot be automatically extended to every PH group and any type of secondary PH; FVC/DLCO is not validated in any PH subgroup by this study. Possibly, a larger patient population and separated analysis of PH subgroups according to etiology might have given different results. Moreover, in some PH groups, DLCO alone might have the same or better predictive value than the FVC/DLCO ratio.
References
Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, Simonneau G, Peacock A, Vonk Noordegraaf A, Beghetti M, Ghofrani A, Gomez Sanchez MA, Hansmann G, Klepetko W, Lancellotti P, Matucci M, McDonagh T, Pierard LA, Trindade PT, Zompatori M, Hoeper M (2015) ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J (pii ERJ-01032-2015)
Iyer AS, Wells JM, Vishin S, Bhatt SP, Wille KM, Dransfield MT (2014) CT scan-measured pulmonary artery to aorta ratio and echocardiography for detecting pulmonary hypertension in severe COPD. Chest 145:824–832
Sivova N, Launay D, Wémeau-Stervinou L, De Groote P, Remy-Jardin M, Denis G, Lambert M, Lamblin N, Morell-Dubois S, Fertin M, Lefevre G, Sobanski V, Le Rouzic O, Hatron PY, Wallaert B, Hachulla E, Perez T (2013) Relevance of partitioning DLCO to detect pulmonary hypertension in systemic sclerosis. PLoS One 8:e78001
Sidhu B, Hashemi N, Rashidian A, Sharma S, Gill N, Mills P, Balasubramanian V (2015) Utility of pulmonary function testing in detection of pulmonary arterial hypertension. A58. Clinical evaluation of pulmonary hypertension. p A1875 (First published online 17 Feb 2015)
Hsu VM, Chung L, Hummers LK, Wigley F, Simms R, Bolster M, Silver R, Fischer A, Hinchcliff ME, Varga J, Goldberg AZ, Derk CT, Schiopu E, Khanna D, Shapiro LS, Domsic RT, Medsger T, Mayes MD, Furst D, Csuka ME, Molitor JA, Alkassab F, Steen VD (2014) Development of pulmonary hypertension in a high-risk population with systemic sclerosis in the pulmonary hypertension assessment and recognition of outcomes in scleroderma (PHAROS) cohort study. Semin Arthr Rheum 44:55–62
Al Plummer (2008) The carbon monoxide diffusing capacity: clinical implications, coding, and documentation. Chest 134:663–667
Sun XG, Hansen JE, Oudiz RJ, Wasserman K (2003) Pulmonary function in primary pulmonary hypertension. J Am Coll Cardiol 41:1028–1035
Coghlan JG, Denton CP, Grünig E, Bonderman D, Distler O, Khanna D, Müller-Ladner U, Pope JE, Vonk MC, Doelberg M, Chadha-Boreham H, Heinzl H, Rosenberg DM, McLaughlin VV, Seibold JR, DETECT study group (2014) Evidence-based detection of pulmonary arterial hypertension in systemic sclerosis: the DETECT study. Ann Rheum Dis 73:1340–1349
Chandra S, Shah SJ, Thenappan T, Archer SL, Rich S, Gomberg-Maitland M (2010) Carbon monoxide diffusing capacity and mortality in pulmonary arterial hypertension. J Heart Lung Transplant 29:181–187
Hoeper MM, Meyer K, Rademacher J, Fuge J, Welte T, Olsson KM (2016) Diffusion capacity and mortality in patients with pulmonary hypertension due to heart failure with preserved ejection fraction. JACC Heart Fail 4:441–449
Pauwels RA, Buist AS, Calverley PM, Jenkins CR, Hurd SS (2001) Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease [GOLD] Workshop summary. Am J Resp Crit Care Med 163:1256–1276
Pellegrino R, Viegi G, Brusasco V, Crapo RO, Burgos F, Casaburi R, Coates A, van der Grinten CPM, Gustafsson P, Hankinson J, Jensen R, Johnson DC, MacIntyre M, McKay R, Miller MR, Navajas D, Pedersen OF, Wanger J (2005) Interpretative strategies for lung function tests. Eur Respir J 26:948–968
Macintyre N, Crapo RO, Viegi G, Johnson DC, van der Grinten CP, Brusasco V, Burgos F, Casaburi R, Coates A, Enright P, Gustafsson P, Hankinson J, Jensen R, McKay R, Miller MR, Navajas D, Pedersen OF, Pellegrino R, Wanger J (2005) Standardisation of the single-breath determination of carbon monoxide uptake in the lung. Eur Respir J 26:720–735
Hoeper MM, Lee SH, Voswinckel R, Palazzini M, Jais X, Marinelli A, Barst RJ, Ghofrani HA, Jing Z, Opitz C, Seyfarth H, Halank M, McLaughlin V, Oudiz RJ, Ewert R, Wilkens H, Kluge S, Bremer H, Baroke E, Rubin LJ (2006) Complications of right heart catheterization procedures in patients with pulmonary hypertension in experienced centers. J Am Coll Cardiol 48:2546–2552
Humbert M, Sitbon O, Chaouat A, Bertocchi M, Habib G, Gressin V, Yaici A, Weitzenblum E, Cordier JF, Chabot F, Dromer C, Pison C, Reynaud-Gaubert M, Haloun A, Laurent M, Hachulla E, Simonneau G (2006) Pulmonary arterial hypertension in France: results from a national registry. Am J Respir Crit Care Med 173:1023–1030
Brown LM, Chen H, Halpern S, Taichman D, McGoon MD, Farber HW, Frost AE, Liou TG, Turner M, Feldkircher K, Miller DP, Elliott CG (2011) Delay in recognition of pulmonary arterial hypertension: factors identified from the REVEAL registry. Chest 140:19–26
Galiè N, Rubin LJ, Hoeper MM, Jansa P, Al-Hiti H, Meyer GMB, Chiossig E, Kusic-Pajic A, Simonneau G (2008) Treatment of patients with mildly symptomatic pulmonary arterial hypertension with bosentan (EARLY study): a double-blind, randomised controlled trial. Lancet 371:2093–2100
Meyrick B, Reid L (1983) Pulmonary hypertension. Anatomic and physiologic correlates. Clin Chest Med 4:199–217
Sun XG, Hansen JE, Oudiz RJ, Wasserman K (2003) Pulmonary function in primary pulmonary hypertension. J Am Coll Cardiol 41:1028–1035
Steen VD, Graham G, Conte C, Owens G, Medsger TA Jr (1992) Isolated diffusing capacity reduction in systemic sclerosis. Arthr Rheum 35:765–770
Nathan SD, Shlobin OA, Ahmad S, Urbanek S, Barnett SD (2007) Pulmonary hypertension and pulmonary function testing in idiopathic pulmonary fibrosis. Chest 131:657–663
Thabut G, Dauriat G, Stern JB, Logeart D, Levy A, Marrash-Chahla R, Mal H (2005) Pulmonary hemodynamics in advanced COPD candidates for lung volume reduction surgery or lung transplantation. Chest 127:1531–1536
Chaouat A, Bugnet AS, Kadaoui N, Schott R, Enache I, Ducoloné A, Ehrhart M, Kessler R, Weitzenblum E (2005) Severe pulmonary hypertension in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 172:189–194
Weitzenblum E, Chaouat A (2005) Severe pulmonary hypertension in COPD: is it a distinct disease? Chest 127:1480–1482
Scharf SM, Iqbal M, Keller C, Criner G, Lee S, Fessler HE (2002) Hemodynamic characterization of patients with severe emphysema. Am J Respir Crit Care Med 166:314–322
Oswald-Mammosser M, Weitzenblum E, Quoix E, Moser G, Chaouat A, Carpentier C, Kessler R (1995) Prognostic factors in COPD patients receiving long-term oxygen therapy: importance of pulmonary artery pressure. Chest 107:1193–1198
D’Alonzo GE, Barst RJ, Ayres SM, Bergofsky EH, Brundage BH, Detre KM, Fishman AP, Goldring RM, Groves BM, Kernis JT (1991) Survival in patients with primary pulmonary hypertension: results from a national prospective registry. Ann Intern Med 115:343–349
McLaughlin VV, McGoon MD (2006) Pulmonary arterial hypertension. Circulation 114:1417–1431
Chandra S, Shah SJ, Thenappan T, Archer SL, Rich S, Gomberg-Maitland M (2010) Carbon monoxide diffusing capacity and mortality in pulmonary arterial hypertension. J Heart Lung Transplant 29:181–187
Marini C, Formichi B, Bauleo C, Michelassi C, Pancani R, Prediletto R, Miniati M, Catapano G, Monti S, Mannucci F, Tavoni A, D’Ascanio A, Pastormerlo LE, Giannoni A, Giuntini C (2014) Improved survival in limited scleroderma-related pulmonary artery hypertension. Intern Emerg Med 9:385–396
Marini C, Formichi B, Bauleo C, Michelassi C, Airò E, Rossi G, Giuntini C (2016) Survival protection by bodyweight in isolated scleroderma-related pulmonary artery hypertension. Intern Emerg Med 11:941–952
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical standard
All procedures performed in our studies were in accordance with ethical standards of the 1964 Helsinki declaration and its later amendments.
Statement of human and animal rights
This study does not contain any studies with human and animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Donato, L., Giovanna Elisiana, C., Giuseppe, G. et al. Utility of FVC/DLCO ratio to stratify the risk of mortality in unselected subjects with pulmonary hypertension. Intern Emerg Med 12, 319–326 (2017). https://doi.org/10.1007/s11739-016-1573-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-016-1573-9