Abstract
Few plants are habitat-indifferent halophytes (i.e., grow well in both saline and non-saline soils). These plants offer a good opportunity to study drought and salinity tolerances during germination for seeds developed and matured in soils differ in salinity. Here, we assessed drought tolerance during germination, as simulated with PEG, and response of germination to light and temperature for Suaeda vermiculata, a habitat-indifferent shrub. Seeds matured in saline and non-saline soils were germinated in six PEG concentrations (0 to − 1.0 MPa) and put in three incubators adjusted at different temperatures in both light and dark regimes. Drought tolerance was greater for seeds of the non-saline than those of saline soils, especially at higher temperatures. Seeds of the saline soils germinated in the lowest osmotic potentials (− 0.8 and − 1.0 MPa) only at lower temperatures, but seeds of the non-saline soils germinated to higher levels at all temperatures. Tolerances to drought and high temperatures were greater in light for seeds of saline soils, but in darkness for seeds of non-saline soils. Germination rate index did not differ significantly between seeds of the two soil types in higher osmotic potentials, but was significantly greater in seeds of non-saline at lower osmotic potentials. Most seeds that failed to germinate in the PEG concentrations recovered their germination when transferred to distilled water. Germination recovery levels and speeds increased with the decrease in osmotic potentials. Seeds of the saline soil postpone their germination until arrival of suitable temperatures and effective rainfalls that ensure seedling survival in salty habitats of the arid unpredictable deserts.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Drought is defined as a severe water deficit that affects development, growth and productivity of plants, especially under arid deserts (Battipaglia et al. 2014). Aridity is expected to increase with the increase in global climate changes in many areas of the world. This requires more research on plant response to water deficit (Petit et al. 1999). Drought stress can affect sensitive life-history traits, such as seed germination and seedling recruitments (Okçu et al. 2005). In arid and hyper-arid deserts, soil moisture condition is a critical factor that control germination and seedling establishment. Seed germination occurs when soil water potential is above certain thresholds, which usually happens after effective rainfalls (El-Keblawy 2004; Lewandrowski et al. 2017). In arid unpredictable deserts, however, several years receive very little rainfalls. For example, out of 71 years of rainfall records in Sharjah Airport, United Arab Emirates (UAE), 27 years received 50 mm or less; 8 years received less than 10 mm (Feulner 2006). To germinate in such dry years, seeds should tolerate drought stress. In such arid deserts, successful germination and plant recruitment depend on both speed of germination and seed capacity to germinate under low osmotic potentials caused by drought (Windauer et al. 2007). In dry years, seeds of halophytes in saline habitats are exposed to both drought and salinity stresses and consequently would not germinate until receive enough rainfalls that increase water potential and could ensure successful recruitments.
Seedling emergence and survival are important processes that affect the maintenance of plant density, which in turn affect population dynamics. These processes are mainly regulated by water potential and the other microenvironments at soil surface (Bertiller et al. 1996; Lauenroth et al. 1994; Novoplansky and Goldberg 2001). Light, temperature and water potential are important environmental signals at soil surface that can regulate time of seed germination, which in turn control seedling emergence and survival (El-Keblawy 2017). For example, higher tolerance to drought at earlier life history stages has been considered as an evolutionary strategy for xerophytes in arid hot deserts and is regulated with temperatures (Zeng et al. 2010). In arid deserts that receive erratic rainfalls, seed germination could be inhibited if a little rainfall is coincided with higher temperatures. In addition, amount and time of rainfalls would determine the soil salinity level, which in turn control time of germination. For example, Noe and Zedler (2001) monitored the density of seedlings in salt marshes of southern California and found that temporal variation in salinity and moisture content of soil can explain a significant portion of germination time. Moreover, the positions of seedling emerge in soil exposed to different water deficit and/or salinity stresses are controlled by both light and temperature conditions (El-Keblawy 2017). There is extensive knowledge on response of germination to temperature, light and water potential and their interactions on germination of both glycophytes and halophytes of arid deserts (Qu et al. 2008; Gul et al. 2013; Hameed et al. 2014). However, fewer studies assessed these environmental factors and their interactions on germination of seeds developed and matured in saline and non-saline soils of habitat-indifferent halophytes (El-Keblawy et al. 2016a, 2017a).
Halophytes can be classified as obligate (true), facultative, and habitat-indifferent (Von Sengbusch 2003). Obligate halophytes grow only in saline habitats. However, plants that are classified as facultative halophytes can be established on salty soils, but grow better in non-saline or in less salt-affected soils. Habitat-indifferent halophytes are still able to cope with salty soils and grow equally well in both salt-free and salty soils (Cushman 2001). To survive well in the salt enriched as well as in non-saline soils, habitat-indifferent halophytes would have a set of genes that would be turned on/off, based on salinity level of the soils. These genes would be turn on to enable habitat-indifferent halophytes to adapt physiologically the high salinity levels, but turn on when they grow in the non-saline soils (Parida and Jha 2010; Sabovljevic and Sabovljevic 2007). Several species of the arid/hyper-arid Arabian deserts, such as Salsola drummondii, Suaeda aegyptiaca, S. vermiculata and Zygophyllum qatarense, could be classified as habitat-indifferent halophytes (Jongbloed 2003).
Both drought and salinity stresses reduce soil water potential by reducing the free energy of water available to the plant to be below the water potential of pure free water. Polyethylene glycol (PEG) can be dissolved in water to create solutions with various negative water potentials (Money 1989). Drought effect is usually through affecting soil water potential; i.e., osmotic effect. However, the effect of salinity could be through ion toxicity, especially Na+ and/or reducing soil osmotic potential (Kranner and Seal 2013). PEG was used to simulate water deficit stress during germination and plant growth (Kołodziejek and Patykowski 2015). Germination inhibition of seeds treated with PEG is attributed merely to osmotic effects. However, the inhibition or marked reduction in germination observed in NaCl could be attributed either to osmotic effect and/or toxicity of specific ions (Sidari et al. 2008). Under higher levels of PEG, the ability of osmotic adjustment of seeds increases by the synthesis of organic osmotica, which enable them to extract water from such lower water potential solutes (Munns and Tester 2008).
The change in offspring phenotype (i.e., transgenerational plasticity) is stimulated by a certain environmental signals in the parental generation (Holeski et al. 2010). The adaptive transgenerational effects could be transmitted through maternal effects and/or epigenetic modifications that can persist across multiple generations (Herman and Sultan 2011; Soliman et al. 2018). Transgenerational plasticity provides offspring phenotypic variations that help in adaption to environmental stresses (Vu et al. 2015). For example, maternal and epigenetic effects could determine dormancy and timing of seed germination in plants, which are important traits for offspring survival in stressful environments (Vu et al. 2015). Environmental maternal effects are usually diminished in the first generation, but epigenetic effects transmit heritable plastic responses to environmental cues (Uller et al. 2008).
The effects of maternal environmental factors (e.g., temperature, day length, light intensity and quality and nitrogen level) on seed dormancy and germination response have been extensively studied (e.g., Huang et al. 2003; El-Keblawy and Al-Rawai 2005; El-Keblawy et al. 2017b). However, fewer studies compared germination behaviour for seeds of habitat-indifferent halophytes that are developed and matured in saline and non-saline soils (Bazzaz 1973; Bülow-Olsen 1983; Wang et al. 2012; El-Keblawy et al. 2016a, 2017a, 2018). According to our knowledge, no study compared germination tolerance to drought for plants of habitat-indifferent halophytes. It is important to assess seed dormancy, germination responses to environmental conditions (e.g., light and temperature) and tolerance to drought during germination for seeds developed and matured in soils differ in salinity levels. The occurrence of such differences indicates the need for conducting further studies that differentiate between the individual role of maternal and epigenetic effects on dormancy and germination traits.
Suaeda vermiculata Forssk. ex J.F.Gmel. (Amaranthaceae) is a dominant perennial shrub in the arid lands of South Europe, North Africa, Canary Islands and Southwest Asia (Creager 1988; Guma et al. 2010). It grows well in both salty and salt-free soils of the Arabian Desert. S. vermiculata is a C4 evergreen shrub that grows nicely in summer, flowers, and fruits in early falls. It produces large number of tiny, ready-to-germinate seeds that have no dispersal structures to carry them away from maternal plants. We assume that seeds that have limited abilities for dispersal are generally placed in surface soils around maternal plants. The limited dispersal of S. vermiculata limits the dispersion of seeds produced in saline habitats to non-saline habitats, indicating that they have to germinate under low water potential stress of the salts and water deficit in dry years. In an earlier study, El-Keblawy et al. (2018) reported that germination of seeds matured in salty soils was completely inhibited in higher salinities (400–500 mM NaCl), but those matured in non-saline soils attained high germination levels at the same salinity levels, especially when germinated in light. Our study aimed at evaluating the response of drought tolerance during germination, as simulated with PEG, to light and temperature of incubation for seeds matured in saline and non-saline soils of the habitat-indifferent Suaeda vermiculata. We hypothesize that germination of seeds matured in salty soils differ from those matured in salt-free soils in response to drought and other environmental signals, such as light and temperatures that could determine conditions for successful seeding survival and establishment. This is especially important as seeds developed in non-saline habitat are exposed to water deficit (i.e., drought) in years receive little rainfall, but those of saline habitats are exposed to lower water potential resulted from both salinity and water-deficit stresses.
Materials and methods
Study species and seed collection
Suaeda vermiculata is a common leaf succulent habitat-indifferent perennial shrub. It grows well in both salty and salt-free soils of the Arabian deserts. Both soil salinity and electric conductivity are significantly greater in saline than in non-saline soils. The saline soils support obligate halophytes, but the non-saline soils have glycophytes (El-Keblawy et al. 2018). S. vermiculata is valuable palatable forage for animals and considered as a useful shrub for the restoration of degraded rangelands (Osman et al. 2006). In addition, it has been considered as a good shrub for combating desertification (Osman and Ghassali 1997).
Mature seeds of S. vermiculata were collected from two sites differ in soil salinity (i.e., saline and non-saline) during January and February 2015. The sites are close to Kalba city, which is located on the northeast coast of the UAE. The descriptions of the two sites and method of collection, cleaning and storage are outlined in El-Keblawy et al. (2018). The average seed weight was determined by weighing three batches, each with 100 seeds, from both habitat types.
Effect of maternal habitat on germination response to PEG, light and temperature
To evaluate drought tolerance during germination for seeds matured in saline and non-saline soils, and the dependence of the tolerance on light and temperature of germination, S. vermiculata seeds of the two sites, were germinated in six PEG 6000 (Sigma–Aldrich) levels (0, − 0.2, − 0.4, − 0.6, − 0.8 and − 1.0 MPa).The different osmotic potentials were verified with Wescor Vapro 5520 (Wescor Inc., UT, USA). We selected the different PEG levels based on a preliminary test assessed the drought tolerance of S. vermiculata during germination. Germination was carried out in three CONVIRON plant growth chamber (model E-15) adjusted at three temperatures, which were the daily dark/light of 15/25, 20/30 and 25/35 °C, and two light regimes, which were 12 h light/12 h darkness and complete darkness (hereafter referred as light and dark, respectively). The white light (1400 µmol m−2 s−1 of photosynthetically active radiation) was provided by five (400 W) metal halide and five (400 W) high pressure sodium lamps. Germination was conducted in 9-cm Petri dishes on two layers of filter paper, moistened with 10 ml of the PEG solutions. As precaution for minimizing evaporation, the plates were wrapped with parafilm. Dark treatment was achieved by wrapping the dishes in aluminum foil. For each treatment, four replicate dishes, each with 25 seeds, were used. Visible radicle protrusion was used as an indication for germination. In light regime, germinated seeds were counted every two days for 20. Seed viability in each habitat type was assessed for three batches, each of 100 seeds, using 1% (w/v) 2,3,5-triphenyle-tetrazolium chloride solution (Bradbeer 1998).
Germination recovery
To assess germination recovery and calculate total germination, ungerminated seeds in the different PEG concentrations were washed and moved to distilled water. The seeds were incubated again in light at the three temperature regimes mentioned above. Germinated seeds were counted and removed daily for 8 days.
Data calculation and statistical analysis
The rate of germination was calculated using a modified Timson’s index of germination velocity = ΣG/t, where G is the percentage of seed germination at 2-day intervals and t is the total germination period (20 days). A higher the GRI value indicates a more rapid seed germination.
The germination recovery was calculated using the following formula:
where a is the total number of seeds germinated after being transferred to distilled water, b is the total number of seeds germinated in saline solution, and c is the total number of seeds. GRI of germination recovery was also calculated as above.
ANOVA tests were carried out to test the impact of the studied factors (maternal habitat, PEG, temperature and light) and their interactions on final germination, germination recovery and total germination (i.e., final germination in PEG solution plus germination recovery). ANOVA was also carried out to evaluate the effect of maternal habitat, PEG and temperature and their interactions on the GRI. Pearson correlation coefficients were calculated to evaluate the significance of the relationships between osmotic potentials of PEG and different germination attributes (i.e., final germination, GRI, recovery germination, total germination and GRI of germination recovery). Post hoc comparisons were performed to examine significant difference between means setting α = 0.05.
Results
Germination in PEG solutions
ANOVA test indicated significant effects for the main factors (i.e., maternal habitat, PEG, and temperature and light) and most of their interactions significantly on final germination of S. vermiculata (P < 0.05, Table 1). Almost no dormancy was observed; seeds matured in saline and non-saline soils attained more than 95% germination in both light and dark in distilled water and lower concentrations of PEG (Fig. 1). The relationships between water potentials and final germination were significantly positive at all temperatures for seeds matured in both saline and non-saline soils, indicating that germination decreased linearly with the decrease of water potential (Table 2).
The interactive effect of maternal habitat, PEG treatment and temperature was significant (P < 0.001). This indicates that the response of the saline and non-saline seeds to the different concentrations of PEG differ according to the temperature of the incubation. Insignificant differences were observed between seeds matured in saline and those matured in non-saline soils in PEG ≥ − 0.6 MPa at 15/25, in ≥ − 0.4 MPa at 20/30 °C, but only in ≥ − 0.2 MPa at 15/25. In lower water potentials, however, seeds matured in the non-saline soils germinated significantly greater than those matured in the saline soils. This indicates that tolerance to drought was higher for seeds matured in the non-saline than those in the saline soils and this tolerance was more obvious at higher temperatures. In addition, seeds matured in saline soils were able to germinate in lower water potentials (− 0.8 and − 1.0 MPa) at only the lower temperatures, but those matured in non-saline soils were able to germinate to higher levels at higher temperatures, indicating that seeds matured in the non-saline habitats can tolerate drought and temperatures stresses during seed germination.
The significant interaction between maternal habitat, PEG treatment, light and temperature (Table 1) indicates that the drought tolerance of the two seed lots depended on the incubation conditions. At 15/25 and 20/30 °C, germination did not differ significantly from between light and dark at most lower PEG concentrations. At the lowest osmotic potential (− 1.0 MPa), seeds from the two soil types attained significantly greater germination in dark, as compared to light. At the highest temperatures (25/35 °C), seeds from the saline soils tolerated up to − 0.6 MPa. At this osmotic potential, germination in light was significantly greater than in darkness for seeds matured in saline soils, but there was no significant difference between light and dark germination for seeds matured in non-saline soils. At higher levels of PEG, seeds from the non-saline soils germinated greater in dark than in light. This indicates that tolerances to drought and high temperature were greater in light for seeds from the saline soils, but in darkness for seeds from the non-saline soils (Fig. 1).
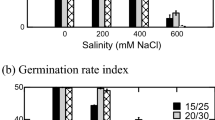
Maternal habitat, PEG and temperature and all of their interactions significantly affected GRI (Table 3). Whereas GRI did not differ significantly between seeds matured in saline and those in non-saline soils in higher osmotic potentials ≥ − 0.6 MPa PEG), it was significantly greater in seeds from non-saline than in seeds from saline soils at the lowest water potentials (− 0.8 and − 1.0 PMa PEG, Fig. 2). There were insignificant relationships between PEG osmotic potentials and GRI (Table 2).
Germination recovery
Maternal habitat, PEG and temperature significantly affected germination recovery of seeds failed to germinate in the different PEG solutions (Table 1). The trend of germination recovery was almost opposite to that of germination in PEG solutions. This result indicates that germination recovery occurred for most seeds that failed to germinate in the different PEG concentrations. Opposite to germination in PEG, both germination recovery and GRI of the recovery showed significant negative relationships with osmotic potentials of PEG at all temperatures, indicating that germination recovery levels and speeds significantly increased with the decrease in PEG osmotic potentials at all temperatures (Table 2).
Seeds of the saline soils attained significantly greater germination recovery than those of the non-saline soils. In addition, recovery from − 1.0 to − 0.8 MPa PEG was significantly greater than that of the higher PEG osmotic potentials. Furthermore, recovery was significantly greater at higher than at moderate and lower temperatures (Fig. 3).
The interaction between maternal habitat, PEG and temperature was significant. In higher concentrations of PEG (≤ − 0.6 MPa), whereas germination recovery did not differ significantly between seeds matured in saline and non-saline soils at 25/35 °C, it was significantly greater in seeds matured in saline than those matured in non-saline soils at 15/25 and 20/30 °C. In lower PEG concentrations, (− 0.2 and − 0.4 MPa), seeds matured in saline soils attained significantly greater recovery than seeds matured in non-saline soils at 25/35 °C (Fig. 3). Seeds of both habitat types recovered their germination within 24 h following their transfer to distilled water (data not shown).
Total germination
Similar to germination in PEG solutions, all the main factors (maternal habitat, PEG treatment, and temperature and light) significantly affected total germination of S. vermiculata seeds (P < 0.001). However, only four of the interactions of these factors had significant effects, compared to eight significant interactions for final germination in PEG solutions (Table 1). In different PEG levels up to − 0.8 MPa, either there was no significant difference between total germination of seeds matured in the two soil types or seeds matured in the non-saline soils germinated greater. At − 1.0 MPa, however, seeds matured in saline soils attained greater germination than those matured in non-saline soils (Fig. 4).
Effects of maternal habitat and temperature of incubation on total germination (i.e., germination in PEG solutions and recovery of non-germinated seeds after their transfer to distilled water, mean ± SE) of Suaeda vermiculata seeds in different concentrations of polyethylene glycol. Dark and light bars are for seeds matured in non-saline and saline soils, respectively
Discussion
Desert halophytes generally have very low innate dormancy (Gul et al. 2013). Our results showed that seeds of S. vermiculata had little dormancy, regardless of maternal habitat. Seeds of both habitat types attained around 95% germination after two days of imbibition in higher osmotic potentials (0, − 0.2 and − 0.4) at all temperatures. The study also showed that germination was very fast; most of the germination happened within two days in lower concentrations of PEG at all temperatures. Several other studies showed also lower dormancy with fast germination in other halophytes of the Arabian deserts. These include, for example, Halopeplis perfoliata and Halocnemum strobilaceum (El-Keblawy and Bhatt 2015), Haloxylon salicornicum (El-Keblawy and Al-Shamsi 2008) and Anabasis setifera (El-Keblawy et al. 2016a, b). In addition, low level of dormancy has been reported for fresh harvested seeds of several other Salsola species, including S. imbricata, S. affinis, S. nitraria, S. ferganica and S. iberica (Khan et al. 2002; El-Keblawy et al. 2007; Wei et al. 2008; Parsons 2012; Wang et al. 2013). The ability of S. vermiculata seeds to germinate to very high percentage and speed in higher osmotic pressures at all temperatures indicates the ability of immediate seed germination after effective rainfalls that could be happened at any time during the growing seasons.
Several studies have reported a very fast germination (i.e., within 24 h) in several species (Parsons 2012). Most of these species are from habitats face high stresses, such as arid and saline habitats, where they can quickly exploit temporarily favourable conditions for germination (Parsons 2012). Among 28 species that can germinate within 24 h, Parsons (2012) indicated that 15 species are from the subfamily Salsoloideae. Several species of the genus Salsola can also germinate very fast (i.e., within 24 h, Parsons 2012). Our study indicated that seeds of S. vermiculata can germinate within 2 days; the GRI was close to 50. Guma et al. (2010) indicated that the morphological structure of S. vermiculata seeds could help them to germinate rapidly under favourable conditions. The fast germination could be an ecological adaptation in reducing mortality of seedlings in a range of stressful habitats (El-Keblawy et al. 2016b, 2017a). In addition, seedlings established early in the season could have greater competitive ability, compared to those appear late in the season (Gul et al. 2013). For example, in several subtropical perennial halophytes, fast germination has been proposed as a common adaptation for young seedlings to benefit from brief availability of water after light rains (Gul et al. 2013). Under such conditions, fast germination can allow seedlings to grow faster and reach moist subsurface soils before surface soils dry out. This is especially important in sandy habitats that dry faster after receiving rainfalls (Gutterman 2003).
Drought affects seed germination by reducing osmotic potential, but the effect of salinity could be through ion toxicity and/or reducing osmotic soil potential (Kranner and Seal 2013). Guma et al. (2010) indicated that seeds of S. vermiculata were able to germinate in 600 mM NaCl (− 3.0 MPa) at a range of temperatures. In the present study, however, seeds of this species germinated only in up to − 1.0 MPa PEG. Other studies have also showed that the inhibitory effect of NaCl was greater than that of iso-osmotic PEG on seed germination of several desert plants, such as Ceratonia silique (Cavallaro et al. 2016), Henophyton deserti (Gorai et al. 2014), three pea cultivars (Okçu et al. 2005), Salsola imbricata (Zaman et al. 2010), Ephedra alata (Al-Taisan et al. 2010), two Atriplex species (Katembe et al. 1998), and Aristida adscensionis and Artemisia ordosica (Tobe et al. 1999). This indicates that osmotic, but not specific-ion toxicity, effect is responsible for germination inhibition in S. vermiculata. In addition, the high germination recovery from both saline solutions (Guma et al. 2010) and iso-osmotic solutions of PEG (the present study) further support the hypothesis that the failure of S. vermiculata seed germination in saline solutions could be attributed to osmotic, rather than specific-ion toxicity effect.
The results showed that S. vermiculata seeds matured in the saline soils attained significantly lower level of germination at lower osmotic potentials of PEG (− 0.8 and − 1.0 MPa), compared to seeds matured in the non-saline soils. Such result could be attributed to a maternal salinity effect. Seeds of S. vermiculata are usually developed and matured in September–November; i.e., before the onset of the first effective rainfall. Effective rainfalls are usually received in December–March (Feulner 2006). This indicates that S. vermiculata relies mainly on atmospheric moisture, rather than on the conventional soil water, especially it has a shallow root system. This further indicates that seeds matured in non-saline soils are independent on soil water potential, but those matured in saline habitats experienced the highest possible salinity (i.e., lowest osmotic potential) that is usually reached before the onset of rainy season. Several reports indicated that lower osmotic potential stress during seed maturation produced seeds with greater dormancy. For example, Dornbos et al. (1989) indicated that soybean plants exposed to lower osmotic potential stress during seed filling period produced seeds with significant low germination. Similarly, out of three populations of Impatiens capensis, drought resulted in enforced seed dormancy (i.e., low germination) in two populations (Maruyama et al. 2016).
In arid ecosystems, seed germination is restricted to a narrow recruitment window limited to short period of moisture availability. Germination usually happens after large rainfall events (Lewandrowski et al. 2017). Arid deserts of the Gulf regions are characterized by very harsh climate with erratic unpredictable rainfalls (Feulner 2006). Large rainfall events usually raise soil water potentials that stimulate germination and increase successful recruitment. Our results indicated that S. vermiculata germinated to very high levels in higher osmotic potential solutes when incubating in both light and dark. However, decreasing the osmotic potential to − 0.8 MPa reducted the final germination, especially for seeds matured in saline soils at higher temperatures. This indicates that the germination window is narrower for seeds of the saline soils. Their failure to germinate in the two light regimes at higher temperatures and lower osmotic potentials would be an ecological adaptation for surviving in the saline soils; little rainfalls would not encourage seed germination at the end of the growing season, when high temperatures evaporate water and increase soil salinity. However, germination window is wider for seeds matured in non-saline soils; they germinated in different temperatures and light regimes in the lower water osmotic potentials.
Seed light sensitivity is dependent on phytochrome action. In some species, this sensitivity is controlled by temperature and soil osmotic potential (Battla and Benech-Arnold 2014; Juroszek et al. 2017). In their study of the effect of soil moisture content on photoinduction of seed germination, Botto et al. (2000) concluded that dark germination was less sensitive to a reduction in soil moisture than light-induced germination in both Datura ferox and Chenopodium album. In the shallow soils, which are exposed to light, germination was significantly lower in non-irrigated (desiccated) soil than in irrigated plots (Botto et al. 2000). The results of our study indicate that germination of seeds matured in the two soil types was greater in darkness than in light in the lowest water osmotic potential (− 1.0 MPa) at lower and moderate temperatures. At higher temperatures, however, germination in darkness was greater than in light in − 0.8 MPa for seeds matured in the non-saline soils. Seeds of saline soils failed to germinate at higher temperatures at − 0.8 and − 1.0 MPa PEG. Such results indicate that some seeds could germinate in the non-saline, but not in the saline soils, when little rainfalls received by the end of the season, especially if seeds are covered with litters or present in dark cracks. As seeds of S. vermiculata are very small, (average seed weight is 0.153 mg and 0.447 mg for non-saline and saline habitats, respectively), it would be hard for them to emerge from deeper soils; seedlings from small seeds have little metabolic reserves available for seedling emergence from deep soil (Green and Juniper 2004).
Erratic patterns and limited quantities of effective rainfalls in arid ecosystems usually limit the frequency of plant recruitment events (Lewandrowski et al. 2017). In several studies, higher germination tolerance to water stress was regarded as an evolutionary strategy of xerophytes to survive the arid hot deserts (e.g., Zeng et al. 2010). Our results showed limited tolerance to lower osmotic potential during germination of S. vermiculata seeds, compared to many other desert plants; few seeds germinated in − 0.8 and − 1.0 MPa PEG. This indicates that tolerance of S. vermiculata to drought is less than other desert plants. For example, seed germination of Allenrolfea occidentalis happened at more negative soil water potentials (− 4 MPa) (Blank et al. 1994). In addition, Salsola imbricata, a leaf-succulent shrub, tolerated 2.4 MPa mannitol (Zaman et al. 2010). However, other species tolerated a moderate water potential, such as the desert shrub Ephedra alata that tolerated − 1.5 MPa PEG (Al-Taisan et al. 2010). The lower ability of S. vermiculata seeds to germinate in lower osmotic potentials, compared to other desert plants, indicates that they would not germinate below certain water threshold, above which successful seedlings recruitments would occur (Lewandrowski et al. 2017). Similarly, seeds of Artemisia sphaerocephala which produce mucilage to retain water, tolerated only − 1.0 MPa (Zheng et al. 2005). Exposing seeds to water deficiency limits the mobilization of the available reserves (Lin and Kao 1995). In addition, lower water potential during germination reduces activities of hydrolysis enzymes (Ahmad and Bano 1992).
Our results indicated that seeds matured in saline soils had greater dormancy, especially at the moderate and higher temperatures. In addition, seeds of saline soils attained significantly lower germination, compared to those of non-saline soils, in the lower water potentials (− 0.8 and − 1.0 MPa PEG). Around 80% of the seeds that failed to germinate under these PEG concentrations recovered their germination in distilled water, indicating that they were viable. The higher dormancy at the lower water potentials indicates that seeds matured in saline soils postpone germination till the arrival of suitable conditions for seed germination and subsequent seedling survival. Such condition usually happens after effective rainfalls that dilute soil salinity and increase soil water potential. Our result indicates that if environmental salinity and drought stresses occur repeatedly, it is advantageous for the produced seeds to be able to remember these stresses and to use this stored knowledge to adapt to new stress challenges (Kinoshita and Seki 2014). As seeds of S. vermiculata do not have any adaptation for dispersal, their multiple existence and exposures to both drought and salinity stresses in the saline soils could enable them to adapt to a new negative water stress by postponing the germination till the arrival of conditions suitable for seedling establishment. In addition, the result indicates that transgenerational phenotypic plasticity may probably be adaptive, in a sense that it could increase stress tolerance among offspring and consequently increase offspring reproductive success (Donohue and Schmitt 1998; Boyko and Kovalchuk 2011).
Conclusion
Seeds matured non-saline soils germinated to higher percentages in higher osmotic potentials in both light and darkness at the different temperatures, indicating that they can germinate at any time in the season, even after moderate rainfalls. Conversely, the saline habitat seeds showed higher dormancy at the lower osmotic potentials when germinated at higher temperatures and recovered their germination when transferred to distilled water. The total germination (i.e., in PEG solutions plus recovery in distilled water) was significantly greater in the lowest osmotic potential in the saline seeds, as compared to the non-saline habitat seeds, indicating that dormancy in seeds of the saline habitat is a strategy to cope with the erratic unpredictable rainfalls of the arid deserts. This emphasis the adaptive role of transgenerational effect for plants of the saline habitat that are exposed to both drought and salinity stresses. As transgenerational effect could be the results of maternal and epigenetic effects, it is important to conduct further studies that separate the individual role of maternal and epigenetic effects on dormancy and germination traits, especially for seeds matured in saline soils. This could be conducted by growing seeds from both habitat types under different salinity levels in a common garden or growth chambers.
Author contribution statement
NA-S: collected the data and helped in the manuscript preparation. AE-K: proposed the idea of the work, participated in the experimental design, helped in the statistical analyses and preparation of the manuscript. KAM: participated in the experimental design and preparation of the manuscript. TN: participated in the experimental design, helped in the statistical analyses and revised the manuscript.
References
Ahmed J, Bano M (1992) The effect of sodium chloride on physiology of cotyledons and mobilization of reserved food in Cicer arietinum. Pak J Bot 24:40–48
Al-Taisan W, Al-Qarawi AA, Alsubiee MS (2010) Effect of water stress by polyethylene glycol 8000 and sodium chloride on germination of Ephedra alata Decne seeds. Saudi J Biol Sci 17:253–257
Batlla D, Benech-Arnold RL (2014) Weed seed germination and the light environment: implications for weed management. Weed Biol Managt 14:77–87
Battipaglia G, De Micco V, Brand WA, Saurer M, Aronne G, Linke P, Cherubini P (2014) Drought impact on water use efficiency and intra-annual density fluctuations in Erica arborea on Elba (Italy). Plant Cell Environ 37:382–391
Bazzaz FA (1973) Seed germination in relation to salt concentration in three populations of Prosopis farcta. Oecologia 13:73–80
Bertiller MB, Zaixso P, Irisarri MP, Brevedan ER (1996) The establishment of Festuca pallescens in arid grasslands in Patagonia (Argentina): the effect of soil water stress. J Arid Environ 32:161–171
Blank RR, Young JA, Martens E, Palmquist DE (1994) Influence of temperature and osmotic potential on germination of Allenrolfea occidentalis seeds. J Arid Environ 26:339–347
Botto JF, Scopel AL, Sánchez RA (2000) Water constraints on the photoinduction of weed seed germination during tillage. Funct Plant Biol 27:463–471
Boyko A, Kovalchuk I (2011) Genome instability and epigenetic modification—heritable responses to environmental stress? Curr Opin Plant Biol 14:260–266
Bradbeer JW (1998) Seed Dormancy and Germination. Chapman & Hall, New York
Bülow-Olsen A (1983) Germination response to salt in Festuca rubra in a population from a salt marsh. Ecography 6:194–198
Cavallaro V, Barbera AC, Maucieri C, Gimma G, Scalisi C, Patanè C (2016) Evaluation of variability to drought and saline stress through the germination of different ecotypes of carob (Ceratonia siliqua L.) using a hydrotime model. Ecol Eng 95:557–566
Creager RA (1988) The biology of Mediterranean saltwort, Salsola vermiculata. Weed Sci 2:369–374
Cushman JC (2001) Osmoregulation in plants: implications for agriculture. Am Zool 4:758–769
Donohue K, Schmitt J (1998) Maternal environmental effects in plants: adaptive plasticity? In: Mousseau TA, Fox CW (eds) Maternal effects as adaptations? Oxford University Press, New York, pp 137–158
Dornbos DL, Mullen RE, Shibles RE (1989) Drought stress effects during seed fill on soybean seed germination and vigor. Crop Sci 29:476–480
El-Keblawy A (2004) Salinity effects on seed germination of the common desert range grass, Panicum turgidum. Seed Sci Technol 32:943–948
El-Keblawy A (2017) Light and temperature requirements during germination of potential perennial grasses for rehabilitation of degraded sandy Arabian Deserts. Land Degrad Dev 28:1687–1695
El-Keblawy A, Al-Rawai A (2005) Effects of salinity, temperature and light on germination of invasive Prosopis juliflora (Sw.) DC. J Arid Environ 61:555–565
El-Keblawy A, Al-Shamsi N (2008) Salinity, temperature and light affect seed germination of Haloxylon salicornicum, a common perennial shrub of the Arabian deserts. Seed Sci Technol 36:679–688
El-Keblawy A, Bhatt A (2015) Aerial seed bank affects germination behaviour of two small seeded halophytes in the Arabian deserts. J Arid Environ 115:10–17
El-Keblawy A, Al-Ansari F, Hassan N, Al-Shamsi N (2007) Salinity, temperature and light affect germination of Salsola imbricata. Seed Sci Technol 35:272–281
El-Keblawy A, Gairola S, Bhatt A (2016a) Maternal habitat affects germination requirements of Anabasis setifera, a succulent shrub of the Arabian deserts. Acta Bot Bras 30:35–40
El-Keblawy A, Gairola S, Bhatt A (2016b) Maternal salinity environment affects salt tolerance during germination in Anabasis setifera: a facultative desert halophyte. J Arid Environ 8:254–263
El-Keblawy A, Gairola S, Bhatt A, Mahmoud T (2017a) Effects of maternal salinity on salt tolerance during germination of Suaeda aegyptiaca: a facultative halophyte in the Arab Gulf desert. Plant Species Biol 32:45–53
El-Keblawy A, Shabana HA, Navarro T, Soliman S (2017b) Effect of maturation time on dormancy and germination of Citrullus colocynthis (Cucurbitaceae) seeds from the Arabian hyper-arid deserts. BMC Plant Biol 17(1):263
El-Keblawy A, Al-Shamsi N, Mosa K (2018) Effect of maternal habitat, temperature and light on germination and salt tolerance of Suaeda vermiculata, a habitat-indifferent halophyte of arid Arabian deserts. Seed Sci Res 28:140–147
Feulner GR (2006) Rainfall and climate records from Sharjah Airport: Historical data for the study of recent climatic periodicity in the UAE. Tribulus 16:3–9
Gorai M, El Aloui W, Yang X, Neffati M (2014) Toward understanding the ecological role of mucilage in seed germination of a desert shrub Henophyton deserti: interactive effects of temperature, salinity and osmotic stress. Plant Soil 374:727–738
Green PT, Juniper PA (2004) Seed mass, seedling herbivory and the reserve effect in tropical rainforest seedlings. Funct Ecol 18:539–547
Gul B, Ansari R, Flowers TJ, Khan MA (2013) Germination strategies of halophyte seeds under salinity. Environ Exp Bot 92:4–18
Guma IR, Padrón-Mederos MA, Santos-Guerra A, Reyes-Betancort JA (2010) Effect of temperature and salinity on germination of Salsola vermiculata L. (Chenopodiaceae) from Canary Islands. J Arid Environ 74(6):708–711
Gutterman Y (2003) The inhibitory effect of Negev Desert loess soil on the fast germinating caryopses of Schismus arabicus (Poaceae). J Arid Environ 54(4):619–631
Hameed A, Rasheed A, Gul B, Khan MA (2014) Salinity inhibits seed germination of perennial halophytes Limonium stocksii and Suaeda fruticosa by reducing water uptake and ascorbate dependent antioxidant system. Environ Exp Bot 107:32–38
Herman JJ, Sultan SE (2011) Adaptive transgenerational plasticity in plants: case studies, mechanisms, and implications for natural populations. Front plant sci 2:102
Holeski LM, Chase-Alone R, Kelly JK (2010) The genetics of phenotypic plasticity in plant defense: trichome production in Mimulus guttatus. Am Nat 175:391–400
Huang Z, Zhang X, Zheng G, Gutterman Y (2003) Influence of light, temperature, salinity and storage on seed germination of Haloxylon ammodendron. J Arid Environ 55(3):453–464
Jongbloed M (2003) The comprehensive guide to the wild flowers of the United Arab Emirates. Environmental Research and Wildlife Development Agency, Abu Dhabi, p 567
Juroszek P, Neuhoff D, Köpke U (2017) Night-time tillage revisited: the delayed soil desiccation process in night-time tilled plots may promote unexpected weed germination. Weed Res 5:213–217
Katembe WJ, Ungar IA, Mitchell JP (1998) Effect of salinity on germination and seedling growth of two Atriplex species (Chenopodiaceae). Ann Bot 82:167–175
Khan MA, Gul B, Weber DJ (2002) Seed germination in the Great Basin halophyte Salsola iberica. Can J Bot 80:650–655
Kinoshita T, Seki M (2014) Epigenetic memory for stress response and adaptation in plants. Plant Cell Physiol 5:1859–1863
Kołodziejek J, Patykowski J (2015) Effect of environmental factors on germination and emergence of invasive Rumex confertus in Central Europe. Sci World J 170176
Kranner I, Seal CE (2013) Salt stress, signalling and redox control in seeds. Funct Plant Biol 40:848–859
Lauenroth WK, Sala OE, Coffin DP, Kirchner TB (1994) The importance of soil water in the recruitment of Bouteloua gracilis in the shortgrass steppe. Ecol Appl 4:741–749
Lewandrowski W, Erickson TE, Dixon KW, Stevens JC (2017) Increasing the germination envelope under water stress improves seedling emergence in two dominant grass species across different pulse rainfall events. J Appl Ecol 54:997–1007
Lin CC, Kao CH (1995) NaCl stress in rice seedlings: starch mobilization and the influence of gibberellic acid on seedling growth. Bot Bull Acad Sin 36:169–173
Maruyama C, Goepfert Z, Squires K, Maclay T, Teal-Sullivan Q, Heschel MS (2016) Effects of population site and maternal drought on establishment physiology in Impatiens capensis Meerb. (Balsaminaceae) Rhodora 118:32–45
Money NP (1989) Osmotic pressure of aqueous polyethylene glycols. Plant Physiol 91:766–769
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Noe GB, Zedler JB (2001) Spatio-temporal variation of salt marsh seedling establishment in relation to the abiotic and biotic environment. J Veg Sci 12:61–74
Novoplansky A, Goldberg DE (2001) Effects of water pulsing on individual performance and competitive hierarchies in plants. J Veg Sci 12:199–208
Okçu G, Kaya MD, Atak M (2005) Effects of salt and drought stresses on germination and seedling growth of pea (Pisum sativum L.). Turk J Agric For 29:237–242
Osman EA, Ghassali F (1997) Effect of storage conditions and presence of fruiting bracts on the germination of Atriplex halimus and Salsola vermiculata. Exper Agric 33:149–155
Osman AE, Bahhady F, Hassan N, Ghassali F, Al Ibrahim T (2006) Livestock production and economic implications from augmenting degraded rangeland with Atriplex halimus and Salsola vermiculata in northwest Syria. J Arid Environ 65:474–490
Parida AK, Jha B (2010) Salt tolerance mechanisms in mangroves: a review. Trees 24:199–217
Parsons RF (2012) Incidence and ecology of very fast germination. Seed Sci Res 22:161–167
Petit JR, Jouzel J, Raynaud D, Barkov NI, Barnola JM et al (1999) Climate and atmospheric history of the past 420,000 years from the Vostok ice core. Antarctica Nature 399:429–436
Qu X, Baskin JM, Wang L, Huang Z (2008) Effects of cold stratification, temperature, light and salinity on seed germination and radicle growth of the desert halophyte shrub, Kalidium caspicum (Chenopodiaceae). Plant Growth Regul 54:241–248
Sabovljevic M, Sabovljevic A (2007) Contribution to the coastal bryophytes of the Northern Mediterranean: are there halophytes among bryophytes. Phytol Balc 13:131–135
Sidari M, Mallamaci C, Muscolo A (2008) Drought, salinity and heat differently affect seed germination of Pinuspinea. J For Res 13:326–330
Soliman S, El-Keblawy A, Mosa KA, Helmy M, Wani SH (2018) Understanding the phytohormones biosynthetic pathways for developing engineered environmental stress-tolerant crops. In: Gosal S, Wani S (eds) Biotechnologies of Crop Improvement, vol 2. Springer, Cham, pp 417–450
Tobe K, Zhang L, Omasa K (1999) Effects of NaCl on seed germination of five nonhalophytic species from a Chinese desert environment. Seed Sci Technol 27:851–863
Uller T (2008) Developmental plasticity and the evolution of parental effects. Trends Ecol Evol 23:432–438
Von Sengbusch P (2003) Halophytes. Botanik Online. Springer, University of Hamburg
Vu WT, Chang PL, Moriuchi KS, Friesen ML (2015) Genetic variation of transgenerational plasticity of offspring germination in response to salinity stress and the seed transcriptome of Medicago truncatula. BMC Evol Biol 15:59
Wang L, Baskin JM, Baskin CC, Cornelissen JHC, Dong M, Huang Z (2012) Seed dimorphism, nutrients and salinity differentially affect seed traits of the desert halophyte Suaeda aralocaspicavia multiple maternal effects. BMC Plant Biol 12:170
Wang Y, Jiang GQ, Han YN, Liu MM (2013) Effects of salt, alkali and salt–alkali mixed stresses on seed germination of the halophyte Salsola ferganica (Chenopodiaceae). Acta Ecol Sinica 33:354–360
Wei Y, Dong M, Huang ZY, Tan DY (2008) Factors influencing seed germination of Salsola affinis (Chenopodiaceae), a dominant annual halophyte inhabiting the deserts of Xinjiang, China. Flora 203:134–140
Windauer L, Altuna A, Benech-Arnold R (2007) Hydrotime analysis of Lesquerella fendleri seed germination responses to priming treatments. Ind Crop Prod 25:70–74
Zaman S, Padmesh S, Tawfiq H (2010) Seed germination and viability of Salsola imbricata Forssk. Int J Biod Cons 2:388–394
Zeng YJ, Wang YR, Zhang JM (2010) Is reduced seed germination due to water limitation a special survival strategy used by xerophytes in arid dunes? J Arid Environ 74(4):508–511
Zheng Y, Xie Z, Gao Y, Jiang L, Xing X, Shimizu H, Rimmington GM (2005) Effects of light, temperature and water stress on germination of Artemisia sphaerocephala. Ann App Biol 146:327–335
Acknowledgements
The authors would like to thank the Research Office of University of Sharjah for the partial support of this work through a grant to the Environmental and Chemical Biology Research Group (Grant # 150404).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Wojtaszek.
Rights and permissions
About this article
Cite this article
Al-Shamsi, N., El-Keblawy, A., Mosa, K.A. et al. Drought tolerance and germination response to light and temperature for seeds of saline and non-saline habitats of the habitat-indifferent desert halophyte Suaeda vermiculata. Acta Physiol Plant 40, 200 (2018). https://doi.org/10.1007/s11738-018-2771-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-018-2771-z