Abstract
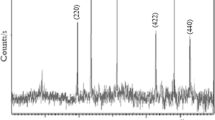
The research results of poly(1-naphthylamine)/Fe3O4 (PNA/Fe3O4) nanocomposites synthesized by a chemical method for As(III) wastewater treatment are presented in this paper. XRD patterns and TEM images showed that the Fe3O4 grain size varied from 13 to 20 nm. The results of Raman spectral analysis showed that PNA participated in part of the PNA/Fe3O4 composite samples. The grain size of PNA/Fe3O4 composite samples is about 25–30 nm measured by SEM. The results of vibrating sample magnetometer measurements at room temperature showed that the saturation magnetic moment of PNA/Fe3O4 samples decreased from 63.13 to 43.43 emu/g, while the PNA concentration increased from 5% to 15%. The nitrogen adsorption–desorption isotherm of samples at 77 K at a relative pressure P/P 0 of about 1 was studied in order to investigate the surface and porous structure of nanoparticles by the BET method. Although the saturation magnetic moments of samples decreased with the polymer concentration increase, the arsenic adsorption capacity of the PNA/Fe3O4 sample with the PNA concentration of 5% is better than that of Fe3O4 in a solution with pH = 7. In the solution with pH > 14, the arsenic adsorption of magnetic nanoparticles is insignificant.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Shah P, Sohma M, Kawaguchi K, et al. Growth conditions, structural and magnetic properties of M/Fe3O4/I (M = Al, Ag and I = Al2O3, MgO) multilayers. Journal of Magnetic and Materials, 2002, 247(1): 1–5
Liu J, Bin Y, Matsuo M. Magnetic behavior of Zn-doped Fe3O4 nanoparticles estimated in terms of crystal domain size. Journal of Physical Chemistry C, 2012, 116(1): 134–143
Bertone J F, Cizeron J, Wahi R K, et al. Hydrothermal synthesis of quartz nanocrystal. Nano Letters, 2003, 3(5): 655–659
Rusanov A I. Surface thermodynamic revisited. Surface Science Reports, 2005, 58(5–8): 111–239
Gu H, Huang Y, Zhang X, et al. Magnetoresistive polyaniline-magnetite nanocomposites with negative dielectrical properties. Polymer, 2012, 53(3): 801–809
Khodabakhshi A, Amin M M, Mozaffari M. Synthesis of magnetic nanoparticles and evaluation of its efficiency for arsenic removal from simulated industrial wastewater. Iranian Journal of Environmental Health Sciences & Engineering, 2011, 8(3): 189–200
Auffan M, Rose J, Proux O, et al. Enhanced adsorption of arsenic onto magnetic nanoparticles: As(III) as a probe of surface structure and heterogeneity. Langmuir, 2008, 24(7): 3215–3222
Zouboulis A I, Katsoyiannis I A. Recent advances in the bioremediation of arsenic-contaminated groundwaters. Environment International, 2005, 31(2): 213–219
Chaudhary G R, Saharan P, Kumar A, et al. Adsorption studies of cationic, anionic and azo-dyes via monodispersed Fe3O4 nanoparticles. Journal of Nanoscience and Nanotechnology, 2013, 13(5): 3240–3245
Liu R, Lu Y, Shen X, et al. Adsorption kinetics and isotherms of arsenic(V) from aqueous solution onto Ni0.5Zn0.5Fe2O4 nanoparticles. Journal of Nanoscience and Nanotechnology, 2013, 13(4): 2835–2841
Fang X B, Fang Z Q, Tsang P K E, et al. Selective adsorption of Cr (VI) from aqueous solution by EDA-Fe3O4 nanoparticles prepared from steel pickling waste liquor. Applied Surface Science, 2014, 314: 655–662
Hao T, Yang C, Rao X, et al. Facile additive-free synthesis of iron oxide nanoparticles for efficient adsorptive removal of Congo red and Cr(VI). Applied Surface Science, 2014, 292: 174–180
Yang G, Tang L, Lei X, et al. Cd(II) removal from aqueous solution by adsorption on a-ketoglutaric acid-modified magnetic chitosan. Applied Surface Science, 2014, 292: 710–716
Chen Q, He Q, Lv M, et al. The vital role of PANI for the enhanced photocatalytic activity of magnetically recyclable N–K2Ti4O9/MnFe2O4/PANI composites. Applied Surface Science, 2014, 311: 230–238
Jiang Q L, Zheng S W, Hong R Y, et al. Folic acid-conjugated Fe3O4 magnetic nanoparticles for hyperthermia and MRI in vitro and in vivo. Applied Surface Science, 2014, 307: 24–233
Chen M J, Shen H, Li X, et al. Facile synthesis of oil-soluble Fe3O4 nanoparticles based on a phase transfer mechanism. Applied Surface Science, 2014, 307: 306–310
Babu C M, Palanisamy B, Sundaravel B, et al. A novel magnetic Fe3O4/SiO2 core–shell nanorods for the removal of arsenic. Journal of Nanoscience and Nanotechnology, 2013, 13(4): 2517–2527
Chen L, Xin H, Fang Y, et al. Application of metal oxide heterostructures in arsenic removal from contaminated water. Journal of Nanomaterials, 2014, 793610 (10 pages)
Park J W, Jang A N, Song J H, et al. Electronic structure of Zn doped Fe3O4 thin films. Journal of Nanoscience and Nanotechnology, 2013, 13(3): 1895–1898
Li X, Zhang F, Ma C, et al. Green synthesis of uniform magnetite (Fe3O4) nanoparticles and micron cubes. Journal of Nanoscience and Nanotechnology, 2012, 12(3): 2939–2942
Zapotoczny B, Dudek M R, Guskos N, et al. FMR study of the porous silicate glasses with Fe3O4 magnetic nanoparticles fillers. Journal of Nanomaterials, 2012, 341073 (7 pages)
Méndez-Rodríguez L, Zenteno-Savín T, Acosta-Vargas B, et al. Differences in arsenic, molybdenum, barium, and other physicochemical relationships in groundwater between sites with and without mining activities. Natural Science, 2013, 5(2): 238–243
Lin K S, Dehvari K, Liu Y J, et al. Synthesis and characterization of porous zero-valent iron nanoparticles for remediation of chromium-contaminated wastewater. Journal of Nanoscience and Nanotechnology, 2013, 13(4): 2675–2681
Zaki H M, Al-Heniti S, Umar A, et al. Magnesium-zinc ferrite nanoparticles: effect of copper doping on the structural, electrical and magnetic properties. Journal of Nanoscience and Nanotechnology, 2013, 13(6): 4056–4065
Larumbe S, Gómez-Polo C, Pérez-Landazábal J I, et al. Ni doped Fe3O4 magnetic nanoparticles. Journal of Nanoscience and Nanotechnology, 2012, 12(3): 2652–2660
Rathore D, Kurchania R, Pandey R K. Structural, magnetic and dielectric properties of Ni1–x ZnxFe2O4 (x = 0, 0.5 and 1) nanoparticles synthesized by chemical co-precipitation method. Journal of Nanoscience and Nanotechnology, 2013, 13(3): 1812–1819
Liu X, Zhong Z, Tang Y, et al. Review on the synthesis and applications of Fe3O4 nanomaterials. Journal of Nanomaterials, 2013, 902538 (7 pages)
Abdallah H M, Moyo T. Evidence of superparamagnetism in Mg0.5Mn0.5Fe2O4 nanosized ferrite. Journal of Superconductivity and Novel Magnetism, 2015, 28(3): 955–960
Genç F, Turhan E, Kavas H, et al. Magnetic and microwave absorption properties of NixZn0.9–x Mn0.1Fe2O4 prepared by boron addition. Journal of Superconductivity and Novel Magnetism, 2015, 28(3): 1047–1050
Uwamariya V, Petrusevski B, Slokar Y M, et al. Effect of fulvic acid on adsorptive removal of Cr(VI) and As(V) from groundwater by iron oxide-based adsorbents. Water, Air, and Soil Pollution, 2015, 226(6): 184
Fakour H, Pan Y F, Lin T F. Effect of humic acid on arsenic adsorption and pore. blockage on iron-based adsorbent. Water, Air, and Soil Pollution, 2015, 226(2): 14
Ameen S, AkhtarMS, Umar A, et al. Effective modified electrode of poly(1-naphthylamine) nanoglobules for ultra-high sensitive ethanol chemical sensor. Chemical Engineering Journal, 2013, 229: 267–275
Ameen S, Akhtar M S, Kim Y S, et al. Synthesis and characterization of novel poly(1-naphthylamine)/zinc oxide nanocomposites: Application in catalytic degradation of methylene blue dye. Colloid & Polymer Science, 2010, 288(16–17): 1633–1638
Webb P A, Orr C, Camp R W, et al. Analytical Methods in Fine Particle Technology. Norcross, GA, USA: Micromeritics Instrument Corporation, 1997, 60–62
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tran, M.T., Nguyen, T.H.T., Vu, Q.T. et al. Properties of poly(1-naphthylamine)/Fe3O4 composites and arsenic adsorption capacity in wastewater. Front. Mater. Sci. 10, 56–65 (2016). https://doi.org/10.1007/s11706-016-0320-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11706-016-0320-5